Abstract
People with substance use disorders (SUD) have a high prevalence of chronic hepatitis C virus (HCV) infection and mental health disorders. We aimed to assess the impact of integrated HCV treatment on psychological distress measured by Hopkins-symptom-checklist-10 (SCL-10). This multi-center randomized controlled trial evaluated psychological distress as a secondary outcome of integrated HCV treatment (INTRO-HCV trial). From 2017 to 2019, 289 participants were randomly assigned to receive either integrated or standard HCV treatment with direct-acting antiviral therapy. Integrated HCV treatment was delivered in eight decentralized outpatient opioid agonist therapy clinics and two community care centers; standard treatment was delivered in internal medicine outpatient clinics at centralized hospitals. Participants in the integrated treatment arm had a sustained virologic response of 93% compared to 73% for those in standard treatment arm. Psychological distress was assessed using SCL-10 prior to initiation of HCV treatment and 12 weeks after treatment completion. The mean SCL-10 score prior to HCV treatment was 2.2 (standard deviation [SD]: 0.7) for patients receiving integrated HCV treatment and 2.2 (SD: 0.8) for those receiving standard HCV treatment. Twelve weeks after the end of treatment, the mean SCL-10 score change was − 0.1 (− 0.3;0.0) in the integrated compared to the standard arm. Psychological distress did not substantially change during the treatment period and was not significantly different between the treatment arms.
Similar content being viewed by others
Introduction
People with substance use disorders (SUD) have a high prevalence of chronic hepatitis C virus (HCV) infection, and consequently liver-related complications1,2. In addition, more than half of people with SUD suffer from substantial mental health symptoms of psychological distress, like fearfulness, tension, worthlessness, sleeping problems, and hopelessness3. These symptoms have a significant impact on health-related quality of life and well-being4,5,6, where the prevalence is particularly associated with the utilization of benzodiazepines, opioids, and cannabis, as well as underlying mental and somatic disorders4,7,8,9. HCV infection by itself is also associated with certain extrahepatic events, such as neurological and psychiatric complications1. Therefore, it is important to explore this intricate interplay between HCV, substance use, and mental health symptoms in high-quality intervention studies. Possibly, ameliorating the burden of serious somatic conditions such as HCV infection might have an added positive effect on mental health.
The introduction of direct-acting antivirals (DAA) represented a paradigm shift, providing an effective and safe treatment for HCV infection despite co-occurring mental health disorders and SUD10. Along with highly effective DAA, decentralization of treatment from conventional HCV follow-up, as well as simplifying and integrating HCV care in harm-reduction sites such as opioid agonist therapy (OAT) clinics and community care centers (CCC), has been crucial to providing more widespread access to testing, linkage to care, and successful treatment for HCV in this population11.
It is estimated that up to 80% of patients receiving OAT or injecting substances have been infected with HCV, and up to 70% have at least one mental health disorder12,13,14. Challenges related to health and life situations, such as suicidal ideation, high fatigue levels, and unstable living conditions, may worsen psychological distress and impact HCV treatment adherence5,6. Some observational studies have suggested that HCV treatment may reduce mental health symptoms, but the studies are prone to confounding and biases limiting any causal interpretations11,15. Randomizing affected patients to receive HCV treatment or no treatment is not ethically acceptable, but the effects of HCV treatment can be evaluated in trials evaluating strategies to increase treatment initiation and sustained virologic response. Integrated treatment of hepatitis C in a randomized control trial (INTRO-HCV), involving multidisciplinary teams and close follow-ups in outpatients SUD clinics, improved sustained virological response (SVR) up to 93% compared to 73% in the standard arm2. The absolute difference between the arms in SVR was 20% (with 95% confidence interval 11–29%) in favor of integrated treatment. In addition, integrated HCV treatment showed a tendency to improve fatigue in this population16. These findings may suggest that integrated HCV treatment can also effectively relieve symptoms related to mental health and psychological distress among patients with SUD. Two potential mechanisms could be that knowing one is free of HCV could provide hope for a better future, but there could also be psychological effects from the virus itself.
This study investigated the effect of integrated as compared to standard HCV treatment on psychological distress measured by Hopkins Symptom Checklist-10 (SCL-10) among patients with co-occurring SUD.
Methods
This study presents a secondary outcome analysis of the INTRO-HCV trial17. We recruited people with SUD and chronic HCV infection who were eligible for HCV treatment with DAA in accordance with Norwegian HCV treatment guidelines (Supplementary file 1). Participants were recruited from eight outpatient clinics providing OAT in Bergen and Stavanger, and two CCC in Bergen providing primary healthcare to people with SUD. Data was collected from May 2017 to June 2019. For a more comprehensive description of the trial, a published protocol is available17 as is the main outcomes paper2.
A total of 298 participants were randomized to either receive integrated HCV treatment (n = 148) or standard HCV treatment (n = 150) (Fig. 1), and due to 4 deaths and 5 not having baseline assessment of psychological distress, 145 in the integrated arm and 144 in the standard arm were included the analyses.
Intervention – standard HCV treatment
After initial HCV assessment onsite at the OAT/CCC clinics, participants randomized to standard HCV treatment were referred to the centralized outpatient infectious disease clinic for HCV treatment, and an appointment was usually scheduled within a few weeks after the referral. The SCL-10 questionnaires were filled out by all the participants in both groups during separate visits to either OAT clinics or CCC.
Intervention – integrated HCV treatment
For participants in the integrated HCV treatment group, HCV assessments and follow-up were provided onsite at the OAT/CCC clinics. Those receiving OAT eligible for treatment were subsequently provided DAA along with their OAT medications, administrated by a nurse at the related OAT clinic. DAA medication and other HCV care were provided at the related CCC when the participant was not undergoing OAT. The differences between the delivery platforms in the two trial arms involve the following main aspects: No need to come for specific appointments at hospital and no need for travel for treatment in the integrated arm, less blood sampling in the integrated arm, integrated treatment provided by clinicians known the patients while standard care by hospital physicians not providing regular care to the patients, and less time used for follow-up of the treatment in the integrated arm. For a more detailed description of the methodology, see Supplementary files 2, 3.
Main outcome measure: Hopkins Symptom Checklist-10 (SCL-10)
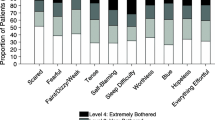
We used the SCL-10, which is designed to record responses on 10 items to measure symptoms of mental health disorders and psychological distress18. Each item is scored on four dimensions from “not bothered at all” (item score = 1) to “extremely bothered” (item score = 4). To derive the mean item score, the scores on each item were summarized and divided by the number of items answered.
Ethics approval and consent to participate
The present study was reviewed and approved by the Regional Ethical Committee for Health Research (REC) West, Norway (reference number: 2017/51/REK Vest, dated 29.03.2017/20.04.2017). All recruited participants were fully informed about the study, and their written informed consent was provided before their inclusion and randomization. All methods were carried out in accordance with relevant guidelines and regulations.
Results
Characteristics at baseline
The baseline sociodemographic characteristics were similar between the arms. No significant differences (p > 0.05 for all comparisons) emerged between the arms for the baseline characteristics (Table 1). Achieving SVR did not significantly reduce the SCL-10 score 12 weeks after DAA treatment compared to those not achieving SVR (Supplementary files 4, 5).
Mean SCL-10 scores at baseline and 12 weeks after the end of treatment
At baseline, the SCL-10 mean score for participants in the integrated HCV treatment arm was 2.2 (standard deviation [SD]: 0.7), and 2.2 (SD: 0.8) for those on standard HCV treatment. At 12 weeks after the end of treatment (EOT12), the mean SCL-10 score for participants in both groups was 2.1 (SD: 0.7) (Supplementary files 6, 7, 8).
The impact of integrated HCV treatment on psychological distress
Integrated HCV treatment did not significantly reduce the mean SCL-10 score from baseline to EOT12 compared with standard HCV treatment (ΔSCL-10 mean score: − 0.1, 95% confidence interval [CI] − 0.3; 0.0) (Table 2, Fig. 2). Substantial individual variations in mean SCL-10 score were observed over time across the treatment groups (Fig. 3). Per protocol analysis and sensitivity analyses showed similar results (Supplementary files 9, 10, 11, 12 and 13).
A linear prediction of changes in mean SCL-10 scores from baseline to EOT12 (intention-to-treat analysis) (n = 289). EOT12: 12 weeks after the end of HCV treatment; SCL-10: Hopkins symptom checklist-10; HCV: Hepatitis C virus. The figure displays the linear prediction (fixed portion) including 95% confidence intervals of the mean SCL-10 score (Δ mean SCL-10 score) at baseline and from baseline (prior to HCV treatment) to EOT12 for integrated and standard HCV treatment groups. A Δmean SCL-10 score < 0 indicates improvement, while a Δmean SCL-10 score > 0 indicates impairing of the SCL-10 score from baseline to EOT12. The mean SCL-10 score ranged from 1 “not bothered at all” to 4 “extremely bothered”.
Pen’s parades of the mean SCL-10 scores at baseline and EOT12 (n = 289). Legends: EOT12: 12 weeks after the end of HCV infection treatment; HCV: Hepatitis C virus; SCL-10: Hopkins symptom checklist-10. The figures display participants who received integrated and standard HCV treatment. The graphs demonstrate the mean SCL-10 scores at baseline/prior to the HCV treatment and EOT12. A total of 23% (n = 33) in the integrated HCV treatment group and 38% (n = 56) in the standard HCV treatment group had no SCL-10 measurement at EOT12. The red line represents the SCL-10 mean scores at baseline when the participants are in sorted order by mean SCL-10 scores (from highest (left) to lowest (right) score). The blue spikes demonstrate the mean SCL-10 score at EOT12. The length of the spikes mark the changes in the mean SCL-10 score from baseline to EOT12. Participants without spikes did not complete SCL-10 assessment at EOT12. The dotted line represents the cut-off value of 1.85 indicating substantial mental health distress. The mean SCL-10 score ranged from 1 (not bothered at all) to 4 (extremely bothered).
Discussion
This randomized controlled trial has showed that psychological distress was barely changed during the treatment of HCV and was not significantly different between integrated and standard treatment arms. Overall, and regardless of treatment approach, the symptom burden was substantial both at baseline and 12 weeks after DAA treatment. A small non-significant trend of − 0.1 (ΔSCL-10 mean score, p = 0.091) reduction in psychological distress was observed with integrated HCV treatment compared to the standard arm.
The symptom burden reported at EOT12 was substantial for participants in both the integrated and standard HCV treatment groups with mean SCL-10 scores of 2.2, which is far above the considered threshold for significant symptoms of psychological distress and symptoms of mental health disorder (mean SCL-10 score of 1.85). This is considerably higher compared to the general Norwegian population with mean SCL-10 score of 1.419. This finding on the level of psychological distress in this population is consistent with our previous research on a large cohort of SUD patients. Furthermore, in a retrospective study from Australia 30% of the HCV infected patients reported a pre-existing mental health disorder of which 25% had depression, 2.6% bipolar disorder, 3.5% schizophrenia and 0.4% schizoaffective disorder20. Compared to interferon-based therapies, treatment with DAA has been associated with lower incidence of depressive symptoms21. In contrast to our findings, one other study of patients with HCV and co-occurring mental health disorders and SUD, showed significantly reduced symptoms of depression at EOT12 with DAA treatment, although differences in study design and methodology should make such interpretations cautious22.
Substantial intra-individual variations in mean SCL-10 score were observed from baseline to EOT 12 in both the integrated and standard treatment groups. In addition, these variations seem to go in both positive and negative directions and correspond with the findings in our cohort study on SCL-10 among 707 participants with SUD3. This is likely attributable to a range of external factors such as unstable living conditions, worrying debt situation, educational attainment, and severity of substance use, which also seems to be independent predictors of life-time symptoms of mental health disorders23. This indicates that, despite a strong trial design, a large intra-individual variation may reduce the analytic power. Finally, achieving SVR did not impact the mean SCL-10 score when adjusting for sociodemographic factors, intravenous injecting use, and any legal and illegal substance use in sensitivity analyses.
A major strength of this study is its design of individual randomization with balanced groups, which minimizes potential confounding. A limitation of this study might be the selection of outpatient clinics, where most participants received OAT, affecting the generalizability of our results to non-OAT populations. The sample size may also be too low to observe a potentially significant difference between the arms. Moreover, both groups had a huge intraindividual variation in SCL-10 score from baseline to EOT12. This led to high variances and reduced statistical power in the analyses, which could hide significant differences between the groups. Another limitation is that 23% in the integrated treatment group and 38% in the standard treatment group were loss-to-follow-up of the SCL-10 assessments at EOT12, which may make the findings more uncertain. Sensitivity analyses without estimated data were performed to consider the impact of estimated values between the groups. Still, compared to the analyses with estimated data, no significant differences in SCL-10 scores between the groups were found. Furthermore, due to system and individual delays and changes in national guidelines for HCV treatment during the study period, the SCL-10 assessments were not conducted in exact concurrence with HCV treatment initiation and EOT12. This may have affected the results.
Conclusion
This randomized controlled trial showed that integrated HCV treatment is unlikely to have substantial co-benefits related to psychological distress within a 12-week timeframe of the study. Nevertheless, there may be subgroups who might benefit from an integrated care approach more than others, in addition to the benefits of facilitated HCV treatment initiation with implications for potential future somatic complications and improved quality of life, emphasizes the importance of integrated treatment approaches in people with substance use disorders.
Data availability
The datasets analyzed during the current study are not publicly available due data protection requirements but are available from the corresponding author on reasonable request.
Abbreviations
- CCC:
-
Community care centers
- CI:
-
Confidence interval
- DAA:
-
Direct-acting antiviral agent
- EOT12:
-
12 weeks after the end of treatment
- SCL-10:
-
Hopkins symptom checklist-10
- HCV:
-
Hepatitis C virus
- OAT:
-
Opioid agonist therapy
- SD:
-
Standard deviation
- SUD:
-
Substance use disorder
- SVR:
-
Sustained virological response
References
Degenhardt, L. et al. Global prevalence of injecting drug use and sociodemographic characteristics and prevalence of HIV, HBV, and HCV in people who inject drugs: A multistage systematic review. Lancet Global Health 5(12), e1192–e1207 (2017).
Fadnes, L. T. et al. Integrated treatment of hepatitis C virus infection among people who inject drugs: A multicenter randomized controlled trial (INTRO-HCV). PLoS Med. 18(6), e1003653 (2021).
Aas, C. F. et al. Substance use and symptoms of mental health disorders: A prospective cohort of patients with severe substance use disorders in Norway. Subst. Abuse Treat. Prev. Policy 16(1), 20 (2021).
Aas, C. F. et al. Health-related quality of life of long-term patients receiving opioid agonist therapy: A nested prospective cohort study in Norway. Subst. Abuse Treat. Prev. Policy 15(1), 68 (2020).
Vold, J. H. et al. Impact of clinical and sociodemographic factors on fatigue among patients with substance use disorder: A cohort study from Norway for the period 2016–2020. Subst. Abuse Treat. Prev. Policy 15(1), 93 (2020).
Vold, J. H. et al. Prevalence and correlates of suicide attempts in high-risk populations: A cross-sectional study among patients receiving opioid agonist therapy in Norway. BMC Psychiatry 22(1), 181 (2022).
Vold, J. H. et al. Potentially addictive drugs dispensing to patients receiving opioid agonist therapy: A register-based prospective cohort study in Norway and Sweden from 2015 to 2017. BMJ Open 10(8), e036860 (2020).
Aas, C. F. et al. Uptake and predictors of direct-acting antiviral treatment for hepatitis C among people receiving opioid agonist therapy in Sweden and Norway: A drug utilization study from 2014 to 2017. Subst. Abuse Treat. Prev. Policy 15(1), 44 (2020).
Vold, J. H. et al. Attention-deficit/hyperactivity disorder (ADHD) symptoms and their relation to diagnosed ADHD, sociodemographic characteristics, and substance use among patients receiving opioid agonist therapy: A Norwegian cohort study. BMC Psychiatry 23(1), 479 (2023).
Gutiérrez-Rojas, L. et al. Patients with severe mental illness and hepatitis C virus infection benefit from new pangenotypic direct-acting antivirals: Results of a literature review. Gastroenterol. Hepatol. 46(5), 382–396 (2023).
Oru, E., Trickey, A., Shirali, R., Kanters, S. & Easterbrook, P. Decentralisation, integration, and task-shifting in hepatitis C virus infection testing and treatment: A global systematic review and meta-analysis. Lancet Global Health 9(4), e431–e445 (2021).
Roncero, C. et al. Psychiatric comorbidities in opioid-dependent patients undergoing a replacement therapy programme in Spain: The PROTEUS study. Psychiatry Res. 243, 174–181 (2016).
Roncero, C. et al. HIV And HCV infection among opiate-dependent patients and methadone doses: The PROTEUS study. AIDS Care 29(12), 1551–1556 (2017).
Hepatitis C among drug users in Europe: Epidemiology, treatment and prevention. European Monitoring Centre for Drugs and Drug Addiction 2016, https://www.emcdda.europa.eu/publications/insights/hepatitis-c-among-drug-users-in-europe_en (downloaded: 7 Dec 2023).
Vold, J. H. et al. Integrated care of severe infectious diseases to people with substance use disorders; A systematic review. BMC Infect. Dis. 19(1), 306 (2019).
Vold, J. H. et al. The efficacy of integrated hepatitis C virus treatment in relieving fatigue in people who inject drugs: A randomized controlled trial. Subst. Abuse Treat. Prev. Policy 18(1), 25 (2023).
Fadnes, L. T. et al. Integrated treatment of hepatitis C virus infection among people who inject drugs: Study protocol for a randomised controlled trial (INTRO-HCV). BMC Infect Dis 19(1), 943 (2019).
Derogatis, L. R., Lipman, R. S., Rickels, K., Uhlenhuth, E. H. & Covi, L. The Hopkins Symptom Checklist (HSCL): A self-report symptom inventory. Behav. Sci. 19(1), 1–15 (1974).
Strand, B. H., Dalgard, O. S., Tambs, K. & Rognerud, M. Measuring the mental health status of the Norwegian population: A comparison of the instruments SCL-25, SCL-10, SCL-5 and MHI-5 (SF-36). Nordic J. Psychiatry 57(2), 113–118 (2003).
Wu, J. Y. et al. Influence of psychiatric diagnosis on treatment uptake and interferon side effects in patients with hepatitis C. J. Gastroenterol. Hepatol. 29(6), 1258–1264 (2014).
Egmond, E. et al. Incidence of depression in patients with hepatitis C treated with direct-acting antivirals. Braz. J. Psychiatry 42(1), 72–76 (2020).
Sundberg, I., Lannergård, A., Ramklint, M. & Cunningham, J. L. Direct-acting antiviral treatment in real world patients with hepatitis C not associated with psychiatric side effects: A prospective observational study. BMC Psychiatry 18(1), 157 (2018).
Bakken, K., Landheim, A. S. & Vaglum, P. Axis I and II disorders as long-term predictors of mental distress: A 6-year prospective follow-up of substance-dependent patients. BMC Psychiatry 7, 29 (2007).
Acknowledgements
We thank Nina Elisabeth Eltvik, Christer Kleppe, Rafael Alexander Leiva, and Christian Ohldieck for valuable help and input during the planning and preparation phases. We also thank the INTRO-HCV Study Group for important contribution relating to data collection. AGL and PV acknowledge support from the National Institute for Health and Care Research (NIHR) Health Protection Research Unit (HPRU) in Behavioural Science and Evaluation at the University of Bristol (NIHR200877). AGL and PV acknowledge support from the Well- come Trust (WT 220866/Z/20/Z) and UK NIHR (NIHR133208). INTRO-HCV Study Group participating investigators: Bergen: Christer Frode Aas, Vibeke Bråthen Buljovcic, Fatemeh Chalabianloo, Jan Tore Daltveit, Silvia Eiken Alpers, Lars T. Fadnes (principal investigator), Trude Fondenes Eriksen, Per Gundersen, Velinda Hille, Kristin Holmelid Håberg, Kjell Arne Johansson, Rafael Alexander Leiva, Siv-Elin Leirvåg Carlsen, Martine Lepsøy Bonnier, Lennart Lorås, Else-Marie Løberg, Mette Hegland Nordbotn, Cathrine Nygård, Maria Olsvold, Christian Ohldieck, Lillian Sivertsen, Hugo Torjussen, Jørn Henrik Vold, Jan-Magnus Økland. Stavanger: Tone Lise Eielsen, Nancy Laura Ortega Maldonado, Ewa Joanna Wilk. proLAR: Ronny Bjørnestad, Ole Jørgen Lygren, Marianne Cook Pierron. Oslo: Olav Dalgard, Håvard Midgard, Svetlana Skurtveit. Bristol: Aaron G. Lim, Peter Vickerman.
Funding
Open access funding provided by University of Bergen. This work was supported by The Norwegian Research Council (BEHANDLING, contract no 269855); and the Western Norway Regional Health Authority («Åpen prosjektstøtte») with Department of Addiction Medicine, Haukeland University Hospital, Bergen, Norway as responsible institution. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. The authors were funded by their respective affiliations.
Author information
Authors and Affiliations
Contributions
J.H.V. and C.F.A. have led the study design, analysis, and writing the article preparation. F.C., E.M.L., A.G.L., P.V., K.A.J., and L.T.F. have contributed to the study design, analysis, and article preparation. All authors have read and approved the final article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Aas, C.F., Vold, J.H., Chalabianloo, F. et al. Effect of integrated hepatitis C virus treatment on psychological distress in people with substance use disorders. Sci Rep 14, 816 (2024). https://doi.org/10.1038/s41598-024-51336-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-51336-9
- Springer Nature Limited