Abstract
As a global health concern, cirrhosis contributes significantly to morbidity and mortality. This prospective cohort study aimed to investigate the association between dietary acid load (DAL) and cirrhosis-related mortality. Present study was conducted on 121 patients with newly diagnosed cirrhosis who were followed up for 48 months. Anthropometric measures, nutritional status and dietary intakes were assessed and DAL was estimated based on potential renal acid load (PRAL) and net endogenous acid production (NEAP) scores. Crude and multivariable-adjusted hazard ratios (HR) with 95% confidence intervals (CI) were estimated using Cox proportional hazard analyses. Participants in the high PRAL and NEAP scores had significantly higher intakes of grains and lower intakes of fruits and vegetables. Also, the intake of dairy products and legumes, nuts and seeds decreased significantly with increasing NEAP score. After adjustment of all the confounders, the risk of mortality in the second and third tertiles of PRAL was 5.9 times and 10.97 higher than those in the first tertile, respectively (P trend: 0.006). Similarly, comparing the risk of mortality in the second and third tertiles with the first tertile of NEAP showed a 4.46-fold and 12.3-fold increased risk, respectively (P trend: 0.010). Our findings suggested that DAL was significantly associated with cirrhosis-related mortality and highlight the need for further research to understand the underlying mechanisms and establish optimal DAL levels in cirrhotic patients.
Similar content being viewed by others
Introduction
Cirrhosis is a chronic and progressive liver disease that affects millions of people worldwide and causes substantial morbidity and mortality1. Cirrhosis can lead to serious complications, such as portal hypertension, variceal bleeding, ascites, hepatic encephalopathy, and hepatocellular carcinoma, that often require hospitalization and increase the risk of mortality2. According to the Global Burden of Disease (GBD) Study in 2019, cirrhosis was responsible for 1.47 million deaths worldwide in 2019, an increase of 9.7% compared to 20173,4. Likewise, based on the latest available statistics, in Iran, cirrhosis accounted for 1.42% of total deaths in 20175. Therefore, it is important to identify the risk factors and preventive strategies for cirrhosis and its complications. Diet plays an important role in the etiology and management of liver cirrhosis, and it can be considered a low-cost and low-risk treatment plan to be applied to the majority of patients6,7,8,9,10.
Diet can contain acidic or alkaline load. Indeed, the idea that diet-induced acidosis can cause chronic disease has been a topic of interest for years11,12,13,14. Some observational studies have investigated the possible link between diets that produce acid with an increased risk of hypertension (HTN)15,16, osteoporosis17,18, insulin resistance (IR), and diabetes19,20. Moreover, a recent comprehensive study revealed a significant link between metabolic syndrome and dietary acid load (DAL)21. Nonetheless, the effects of DAL on cirrhosis, especially cirrhosis-related mortality, have not been well elucidated. DAL refers to the balance between acid-forming and alkaline-forming components in the diet, which can affect the acid–base balance in the body and potentially affect health22. DAL can be estimated based on two indices: net endogenous acid production (NEAP) and potential renal acid load (PRAL) based on dietary intake of five nutrients (protein, potassium, phosphorous, calcium, and magnesium)23. In this concept, animal foods such as meat, fish, and cheese, which are rich in acid-forming amino acids, increase the acid load of the diet, whereas most fruits and vegetables buffer the acid load of the diet due to their potassium content13,22.
It has been shown that diets with a high acid load can cause low-grade metabolic acidosis, which is associated with metabolic abnormalities22. These metabolic abnormalities, especially insulin resistance (IR)24, dysglycemia19, high blood pressure (HTN)25 and metabolic syndrome21, are considered risk factors for liver diseases such as cirrhosis. On the other hand, high consumption of red meat and low consumption of fruits and vegetables are characteristics of a diet with a high acid load, each of which alone can contribute to the development and progression of cirrhosis26,27.
In the current study, we aimed to investigate the possible association between DAL (based on PRAL and NEAP) and cirrhosis-related mortality using data from a prospective cohort study in cirrhotic patients.
Methods and materials
Study design and population
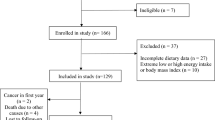
166 newly diagnosed cases (identified within 6 months of diagnose), aged over 18 years, willing to participate in the study were included in the present cohort study. Patients were recruited from two educational hospitals in Tehran, Iran. Exclusion criteria were: breastfeeding or pregnancy in women, history of renal failure, various types of cancer, diabetes mellitus, infectious diseases, cardiac disease, acquired immune deficiency syndrome, pancreatic insufficiency.
Patients were enrolled between 2016 and 2018 and followed up for 48 months until April 30, 2022. In order to track the occurrence of death or any other medical event, telephone calls were made to the participants annually. 45 patients were excluded for reasons including cancer diagnosis in the first year, missing or incomplete baseline data, high or low energy intake (< 500 or > 5000 kcal/day), and extreme body mass index (BMI) (< 15 or > 50 kg/m2). Finally, 121 patients (38 women and 83 men) were included in the final analysis.
The study protocol received approval from the National Nutrition and Food Technology Research Institute (NNFTRI) ethics committee (Ir.sbmu.nnftri.1396.186.) in accordance with the Declaration of Helsinki. Written informed consents were obtained from all participants, after providing explanations about the study protocol.
Dietary assessment and dietary acid load calculation
The usual dietary intakes of the participants were collected through a face-to-face interview, using a reliable and valid food frequency questionnaire (FFQ) consisting of 168 items28. The frequency of consumption of each food during the last year was recorded based on the daily, weekly monthly and consumption of each food and converted into grams based on household measurements. Dietary data were assessed and analyzed by a skilled nutritionist using Nutritionist IV software. The average daily intake of energy and nutrients was calculated using The United States Department of Agriculture (USDA) food composition table (FCT).
The dietary acid load was estimated by the method previously developed based on two measures (PRAL and NEAP), using the following algorithms:
PRAL (mEq/d) = 0.4888 × dietary protein (g/d) + 0.0366 × dietary phosphorus (mg/d) − 0.0205 × dietary potassium (mg/d) − 0.0125 × calcium (mg/d) − 0.0263 × magnesium (mg/d)29.
Negative values indicate an alkaline-forming potential, while positive values reflect an acid-forming potential.
NEAP (mEq/d) = 54.5 × protein intake (g/d)/potassium intake (mEq/d) − 10.230.
Anthropometric, disease and nutritional status assessment
Baseline data including age, sex, alcohol and tobacco use, and the etiology of cirrhosis were collected at the beginning of the study. Weight was measured to the nearest 100g using digital scales and height was measured to the nearest 0.5 cm using a tape meter, while the subjects were minimally clothed, in a standing position without shoes. Body mass index (BMI) was calculated as weight (kg) divided by square of the height (m2).
The subjective global assessment (SGA) score was determined according to Destky et al.31 study, based on which the participants were categorized into three groups: well-nourished (A), moderately malnourished (B), and severely malnourished (C). Using clinical and biochemical parameters and based on Child–Pugh and model for end-stage liver disease (MELD) scores, the severity and prognosis of liver cirrhosis were evaluated32.
Statistical analysis
Statistical analyses were performed using SPSS (SPSS Inc., Chicago, Illinois), P values < 0.05 were considered statistically significant. Means ± standard deviation for continuous variables and number (percentages) for categorical variables were compared using one-way analysis of variance (ANOVA) method and the chi-square test (χ2), respectively. Energy-adjusted PRAL and NEAP (score × 1000/energy intake) were applied in statistical analysis and were assigned as tertiles. Crude and multivariable-adjusted hazard ratios (HR) with 95% confidence intervals (CI) were estimated using Cox proportional hazard analyses. There models were applied to address potential confounders: Model 1: adjusted for sex (male, female) and age (continuous); Model 2: additionally adjusted for BMI (continuous), alcohol use (yes > 30 g/day, no < 30 g/day), and smoking (yes, no); and Model 3: further adjusted for Child–Pugh (A, B & C), MELD (continuous), and etiology (virus, autoimmune, other).
Ethics approval and consent to participate
National nutrition and Food Technology Research Institute (NNFTRI) ethics committee approved the study protocol (Ir.sbmu.nnftri.1396.186.) in accordance with the Declaration of Helsinki. All participants provided written informed consent and were informed about the study.
Results
General characteristics of study participants across the tertiles of PRAL and NEAP are provided in Table 1. During 3955 person-month of follow-up, we documented 43 deaths (7 women, 36 men). Liver failure was responsible for 47% of deaths, cardiovascular diseases 40%, carcinoma 3% and other causes for 10% of deaths. Regarding the etiology, 56% of participants' cirrhosis was attributed to the virus, 31% to autoimmunity, and 13% to other causes. Patients were more likely to be male (68.6%), although there was no significant difference between the PRAL and NEAP tertiles. The number of smokers and alcohol drinkers represent no significant differences. The average age and anthropometric parameters of the participants did not show any significant difference across tertiles of DAL. The severity of cirrhosis, according to MELD and Child, showed a significant difference between tertiles, except for Child in PRAL tertiles. Also, there was no significant difference in the severity of malnutrition based on SGA.
In Table 2 dietary intakes of participants are compared across the tertiles of PRAL and NEAP. The intake of energy and macronutrients showed no significant difference across the tertiles of PRAL and NEAP, except for dietary protein in PRAL. Intakes of micronutrients including potassium, phosphorus, calcium, and magnesium were significantly different across the tertiles of NEAP (P < 0.05), except for phosphorus (P = 0.054). While, except for phosphorus (P = 0.014), the intake of other micronutrients was not significantly different between the PRAL tertiles.
The comparison of intake of food groups also indicated significant differences in intake of grains, fruits, and vegetables among PRAL tertiles. There was an also significant difference in intake of food groups between the NEAP tertiles, except for grains and red meats. The ratio of total, animal and vegetable protein to potassium, showed a significant increase among both NEAP and PRAL tertiles, except for animal protein in NEAP tertiles.
Table 3 indicates multivariable-adjusted hazard ratios and 95% confidence intervals for cirrhosis-related mortality across tertiles of DAL. The number of deaths was substantially significantly increasing throughout the DALs tertiles. In model 1, after adjusting the results for age and sex, the risk of mortality increased significantly (P trend = 0.001) in those who were in the second (OR 6.6; 95% CI 0.8, 57.2) and third (OR 17.38; 95% CI 2.3, 130.3) tertiles of PRAL and NEAP (OR 4.7; 95% CI 1, 21.7 and OR 8.56; 95% CI 1.9, 38.8, respectively). Similar results were achieved in models 2 and 3. So that after adjustment of all confounders, comparing the risk of mortality in the second and third tertiles with the first tertile of PRAL and NEAP showed an increased risk.
Discussion
In the present study for the first time, we showed that higher DAL (based on PRAL and NEAP) was associated with a higher mortality risk in patients with cirrhosis. In comparing the highest and lowest tertiles of NEAP and PRAL scores in a fully adjusted model, we found that being in the highest tertiles of NEAP or PRAL was associated with a significant increase in mortality risk. Although there is no literature regarding DAL and cirrhosis-related mortality, several studies have investigated the association between DAL and disease-specific mortality, like cardiovascular diseases (CVDs) and cancer33,34,35,36,37.
According to a study by Hejazi et al.36 on a large population-based cohort study, being in the highest and lowest DAL scores was significantly associated with increased total and CVD-caused mortality. Similarly, Xu et al.37 found that both excess acid and alkali load in the diet may be associated with an increased mortality risk, especially CVD-caused mortality, among Swedish adults (a U-shaped association). Furthermore, the recent study conducted by Fereidouni et al.34 indicated that an increase in dietary acid load may lead to an increased risk of CVD-related mortality. Aside from that, several studies have suggested that DAL may be associated with non-alcoholic fatty liver disease (NAFLD) progression38,39,40. However, this association was not significant in all studies, and one study reported that NEAP and PRAL were not associated with advanced fibrosis39.
The exact mechanisms by which DAL may influence cirrhosis and its complications are currently unclear. However, several factors may contribute to this association, such as insulin resistance (IR)24, hypertension (HTN)25, high consumption of red meat26, and low consumption of fruits and vegetables27. It has been demonstrated that high acid-load diets can cause chronic low-grade metabolic acidosis (MA), which is associated with metabolic abnormalities22. One of the consequences of low-grade MA is increased cortisol secretion41. Hypercortisolism increases the risk of various metabolic disorders, such as sarcopenia42, IR43, HTN44,45, and CVDs46.
Hepatic steatosis and IR are closely related, as IR is both a cause and a consequence of NAFLD47,48. In fact, IR increases adipose tissue lipolysis, resulting in an elevated influx of free fatty acids (FFAs) to the liver49,50,51. FFAs and other lipid intermediates, such as diacylglycerol (DAG) and ceramides, can induce lipotoxicity in hepatocytes52,53. To counteract this, hepatocytes convert FFAs into triglycerides and store them in the liver. As a consequence of this protective mechanism, plasma levels of FFAs are reduced, and lipotoxicity related to FFAs is prohibited53,54. However, when the hepatocytes are overwhelmed by the lipotoxic effects of other lipid intermediates, such as ceramides and DAG, they trigger inflammation, necrosis, and fibrosis in the liver52. Specifically, this process leads to the progression of NAFLD to non-alcoholic steatohepatitis (NASH), characterized by inflammation and fibrogenesis mediated by hepatic stellate cells48,55. HTN is another possible mechanism that links DAL and NAFLD. The results of the study conducted by Fou et al.25 showed that HTN was associated with higher rates of liver steatosis and fibrosis. Similarly, animal models also revealed that HTN might affect the progression of NASH56 and fibrosis57. Moreover, clinical evidence has suggested that HTN may contribute to the onset of NAFLD and the advancement of liver fibrosis58,59.
In addition, reducing red meat consumption may slow the progression of NAFLD and fibrosis, as continued red meat consumption was linked to a higher risk of hepatic fibrosis60. Additionally, the study by Daftari et al.26 demonstrated that lower intakes of animal protein are associated with lower mortality risks in patients with cirrhosis. On the other hand, consuming potassium-rich foods, such as fruits and vegetables, may favor muscle preservation, while acidosis may impair protein synthesis, increase proteolysis, and enhance amino acid oxidation, leading to a greater loss of muscle mass61. The relation between DAL and skeletal muscle mass has also been observed previously62. Preserving skeletal muscle could improve survival in patients with cirrhosis63,64. Finally, it is important to note that 40% of the deaths in this study were due to CVD, which has previously been shown to be associated with DAL, and some similar mechanisms have been proposed to explain its association34,36,37.
To the best of our knowledge, this is the first prospective cohort study to examine the relationship between DAL and mortality in patients with cirrhosis. The 4-year follow-up duration and the adjustment of several possible confounding factors are among the strengths of the study. However, some limitations should be considered. First, the small sample size limited the precision of the effect estimates. Therefore, the findings should be confirmed by larger studies and interpreted cautiously. Second, the use of the FFQ may be subject to recall bias and measurement errors in dietary intake assessment. Third, about 15% of the enrolled patients were lost to follow-up. Finally, as with most observational studies, residual and unmeasured confounding may affect the results. Thus, clinical trials are needed to provide more robust evidence on the differences in complications and mortality between different levels of DAL in these patients.
Conclusion
In conclusion, our study provides valuable insights into the relationship between DAL and cirrhosis-related mortality, emphasizing the need for continued research to guide clinical interventions and preventive strategies. It was found that high DAL (based on PRAL and NEAP) is significantly associated with cirrhosis-related mortality. Further studies are warranted to elucidate the underlying mechanism(s) and optimal level of DAL in these patients.
Data availability
The datasets analyzed in the current study are available from the corresponding author on reasonable request.
References
Gines, P. et al. Liver cirrhosis. Lancet 398(10308), 1359–1376 (2021).
Nusrat, S., Khan, M. S., Fazili, J. & Madhoun, M. F. Cirrhosis and its complications: Evidence based treatment. World J. Gastroenterol. 20(18), 5442–5460 (2014).
Diseases, G. B. D. & Injuries, C. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the global burden of disease study 2019. Lancet 396(10258), 1204–1222 (2020).
Huang, D. Q. et al. Global epidemiology of cirrhosis—aetiology, trends and predictions. Nat. Rev. Gastroenterol. Hepatol. 20(6), 388–398 (2023).
Anushiravani, A. & Ghajarieh, S. S. Burden of liver diseases: A review from Iran. Middle East J. Dig. Dis. 11(4), 189–191 (2019).
Juakiem, W., Torres, D. M. & Harrison, S. A. Nutrition in cirrhosis and chronic liver disease. Clin. Liver Dis. 18(1), 179–190 (2014).
Pashayee-Khamene, F. et al. Food groups intake of cirrhotic patients, comparison with the nutritional status and disease stage. Gastroenterol. Hepatol. Bed Bench 12(3), 226–232 (2019).
Pashayee-Khamene, F. et al. Malnutrition and its association with the mortality in liver cirrhosis; a prospective nutritional assessment in two referral centers in Iran. Clin. Nutr. ESPEN 54, 453–458 (2023).
Pashayee-Khamene, F. et al. Dietary protein sources and disease severity, malnutrition and anthropometric measurements in cirrhotic patients. Gastroenterol. Hepatol. Bed Bench 12(2), 143–148 (2019).
Hariri, Z. et al. Dietary fiber intake and mortality among survivors of liver cirrhosis: A prospective cohort study. Heliyon 9(6), e16170 (2023).
Vormann, J. & Remer, T. Dietary, metabolic, physiologic, and disease-related aspects of acid-base balance: Foreword to the contributions of the second International acid-base symposium. J. Nutr. 138(2), 413s-s414 (2008).
Pizzorno, J., Frassetto, L. A. & Katzinger, J. Diet-induced acidosis: Is it real and clinically relevant?. Br. J. Nutr. 103(8), 1185–1194 (2010).
Adeva, M. M. & Souto, G. Diet-induced metabolic acidosis. Clin. Nutr. 30(4), 416–421 (2011).
Heidari, Z. et al. Diet-dependent acid load and the risk of breast cancer: A case-control study. Clin. Nutr. ESPEN 55, 97–102 (2023).
Chen, S.-W. et al. Association between dietary acid load and the risk of hypertension among adults from South China: Result from nutrition and health survey (2015–2017). BMC Public Health 19(1), 1599 (2019).
Krupp, D., Shi, L., Maser-Gluth, C., Pietzarka, M. & Remer, T. 11β Hydroxysteroid dehydrogenase type 2 and dietary acid load are independently associated with blood pressure in healthy children and adolescents. Am. J. Clin. Nutr. 97(3), 612–620 (2013).
García-Gavilán, J. F. et al. U-shaped association between dietary acid load and risk of osteoporotic fractures in 2 populations at high cardiovascular risk. J. Nutr. 151(1), 152–161 (2021).
Farshbaf-Khalili, A., Ostadrahimi, A., Heris, J. A., Sarrafi, S. & Mohammadisima, N. Dietary acid load is associated with primary osteoporosis in postmenopausal women aged 50–65 years: A cross-sectional study. Food Sci. Nutr. 11(2), 668–676 (2023).
Akter, S. et al. High dietary acid load score is associated with increased risk of type 2 diabetes in Japanese men: The Japan public health center-based prospective study. J. Nutr. 146(5), 1076–1083 (2016).
Fagherazzi, G. et al. Dietary acid load and risk of type 2 diabetes: The E3N-EPIC cohort study. Diabetologia 57(2), 313–320 (2014).
Al-Hawary, S. I. S. et al. Metabolic syndrome in relation to dietary acid load: A dose–response meta-analysis of observational studies. Front. Nutr. https://doi.org/10.3389/fnut.2023.1233746 (2023).
Osuna-Padilla, I. A., Leal-Escobar, G., Garza-García, C. A. & Rodríguez-Castellanos, F. E. Dietary acid load: Mechanisms and evidence of its health repercussions. Nefrologia 39(4), 343–354 (2019).
Remer, T. & Manz, F. Estimation of the renal net acid excretion by adults consuming diets containing variable amounts of protein. Am. J. Clin. Nutr. 59(6), 1356–1361 (1994).
Cetin, E. G., Demir, N. & Sen, I. The relationship between insulin resistance and liver damage in non-alcoholic fatty liver patients. Sisli Etfal Hastanesi tip bulteni 54(4), 411–415 (2020).
Fu, H., Yu, H., Zhao, Y., Chen, J. & Liu, Z. Association between hypertension and the prevalence of liver steatosis and fibrosis. BMC Endocr. Disord. 23(1), 85 (2023).
Daftari, G. et al. Dietary protein intake and mortality among survivors of liver cirrhosis: A prospective cohort study. BMC Gastroenterol. 23(1), 227 (2023).
Zhao, L. et al. Specific botanical groups of fruit and vegetable consumption and liver cancer and chronic liver disease mortality: A prospective cohort study. Am. J. Clin. Nutr. 117(2), 278–285 (2023).
Mirmiran, P., Esfahani, F. H., Mehrabi, Y., Hedayati, M. & Azizi, F. Reliability and relative validity of an FFQ for nutrients in the Tehran lipid and glucose study. Public Health Nutr. 13(5), 654–662 (2010).
Remer, T., Dimitriou, T. & Manz, F. Dietary potential renal acid load and renal net acid excretion in healthy, free-living children and adolescents. Am. J. Clin. Nutr. 77(5), 1255–1260 (2003).
Frassetto, L. A., Todd, K. M., Morris, R. C. Jr. & Sebastian, A. Estimation of net endogenous noncarbonic acid production in humans from diet potassium and protein contents. Am. J. Clin. Nutr. 68(3), 576–583 (1998).
Detsky, A. S. et al. What is subjective global assessment of nutritional status?. JPEN J. Parenter Enteral. Nutr. 11(1), 8–13 (1987).
Malinchoc, M. et al. A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts. Hepatology 31(4), 864–871 (2000).
Wu, T., Hsu, F. C. & Pierce, J. P. Increased acid-producing diet and past smoking intensity are associated with worse prognoses among breast cancer survivors: A prospective cohort study. J. Clin. Med. 9(6), 1817 (2020).
Fereidouni, S., Hejazi, N., Homayounfar, R. & Farjam, M. Diet quality and dietary acid load in relation to cardiovascular disease mortality: Results from Fasa PERSIAN cohort study. Food Sci. Nutr. 11(3), 1563–1571 (2023).
Chronister, B. N. C. et al. Dietary acid load, serum polychlorinated biphenyl levels, and mortality following breast cancer in the long Island breast cancer study project. Int. J. Environ. Res. Public Health 19(1), 374 (2021).
Hejazi, E. et al. Dietary acid load and mortality from all causes, CVD and cancer: Results from the Golestan Cohort Study. Br. J. Nutr. 128(2), 237–243 (2021).
Xu, H. et al. Modest U-shaped association between dietary acid load and risk of all-cause and cardiovascular mortality in adults. J. Nutr. 146(8), 1580–1585 (2016).
Krupp, D., Johner, S. A., Kalhoff, H., Buyken, A. E. & Remer, T. Long-term dietary potential renal acid load during adolescence is prospectively associated with indices of nonalcoholic fatty liver disease in young women. J. Nutr. 142(2), 313–319 (2012).
Chan, R. et al. Higher estimated net endogenous acid production may be associated with increased prevalence of nonalcoholic fatty liver disease in Chinese adults in Hong Kong. PloS one 10(4), e0122406 (2015).
Emamat, H. et al. The association between dietary acid load and odds of non-alcoholic fatty liver disease: A case-control study. Nutr. Health 29(4), 637–644 (2022).
Esche, J. et al. Higher diet-dependent renal acid load associates with higher glucocorticoid secretion and potentially bioactive free glucocorticoids in healthy children. Kidney Int. 90(2), 325–333 (2016).
Yanagita, I. et al. A high serum cortisol/DHEA-S ratio is a risk factor for Sarcopenia in elderly diabetic patients. J. Endocr. Soc. 3(4), 801–813 (2019).
Adam, T. C. et al. Cortisol is negatively associated with insulin sensitivity in overweight Latino youth. J. Clin. Endocrinol. Metab. 95(10), 4729–4735 (2010).
Fraser, R. et al. Cortisol effects on body mass, blood pressure, and cholesterol in the general population. Hypertension 33(6), 1364–1368 (1999).
Connell, J. M. C. et al. Effects of ACTH and cortisol administration on blood pressure, electrolyte metabolism, atrial natriuretic peptide and renal function in normal man. J. Hypertens. 5(4), 425–434 (1987).
Whitworth, J. A., Williamson, P. M., Mangos, G. & Kelly, J. J. Cardiovascular consequences of cortisol excess. Vasc. Health Risk Manag. 1(4), 291–299 (2005).
Marusic, M., Paic, M., Knobloch, M. & Liberati Prso, A. M. NAFLD, insulin resistance, and diabetes mellitus type 2. Can. J. Gastroenterol. Hepatol. 2021, 6613827 (2021).
Caturano, A. et al. Non-alcoholic fatty liver disease: From pathogenesis to clinical impact. Processes 9(1), 135 (2021).
Donnelly, K. L. et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. J. Clin. Investig. 115(5), 1343–1351 (2005).
Watt, M. J., Miotto, P. M., De Nardo, W. & Montgomery, M. K. The liver as an endocrine organ-linking NAFLD and insulin resistance. Endocr. Rev. 40(5), 1367–1393 (2019).
Bugianesi, E., Moscatiello, S., Ciaravella, M. F. & Marchesini, G. Insulin resistance in nonalcoholic fatty liver disease. Curr. Pharm. Des. 16(17), 1941–1951 (2010).
Neuschwander-Tetri, B. A. Hepatic lipotoxicity and the pathogenesis of nonalcoholic steatohepatitis: The central role of nontriglyceride fatty acid metabolites. Hepatology 52(2), 774–788 (2010).
Alkhouri, N., Dixon, L. J. & Feldstein, A. E. Lipotoxicity in nonalcoholic fatty liver disease: Not all lipids are created equal. Expert Rev. Gastroenterol. Hepatol. 3(4), 445–451 (2009).
Yamaguchi, K. et al. Inhibiting triglyceride synthesis improves hepatic steatosis but exacerbates liver damage and fibrosis in obese mice with nonalcoholic steatohepatitis. Hepatology 45(6), 1366–1374 (2007).
Jou, J., Choi, S. S. & Diehl, A. M. Mechanisms of disease progression in nonalcoholic fatty liver disease. Semin. Liver Dis. 28(4), 370–379 (2008).
Ikuta, T. et al. Spontaneously hypertensive rats develop pronounced hepatic steatosis induced by choline-deficient diet: Evidence for hypertension as a potential enhancer in non-alcoholic steatohepatitis. Hepatol. Res. 42(3), 310–320 (2012).
Hsu, C. T. Ultrastructural changes in liver damage induced by carbon tetrachloride in spontaneously hypertensive rats and Wistar-Kyoto rats. J. Auton. Nerv. Syst. 70(1–2), 79–83 (1998).
Zhang, T. et al. Metabolic syndrome and its components as predictors of nonalcoholic fatty liver disease in a northern urban Han Chinese population: A prospective cohort study. Atherosclerosis 240(1), 144–148 (2015).
Sorrentino, P. et al. Predicting fibrosis worsening in obese patients with NASH through parenchymal fibronectin, HOMA-IR, and hypertension. Am. J. Gastroenterol. 105(2), 336–344 (2010).
Ivancovsky-Wajcman, D. et al. High meat consumption is prospectively associated with the risk of non-alcoholic fatty liver disease and presumed significant fibrosis. Nutrients 14(17), 3533 (2022).
Dawson-Hughes, B., Harris, S. S. & Ceglia, L. Alkaline diets favor lean tissue mass in older adults. Am. J. Clin. Nutr. 87(3), 662–665 (2008).
Gholami, F. et al. The association of dietary acid load (DAL) with estimated skeletal muscle mass and bone mineral content: A cross-sectional study. BMC Nutr. 9(1), 31 (2023).
Montano-Loza, A. J. et al. Sarcopenic obesity and myosteatosis are associated with higher mortality in patients with cirrhosis. J. Cachexia, Sarcopenia Muscle 7(2), 126–135 (2016).
Hanai, T. et al. Rapid skeletal muscle wasting predicts worse survival in patients with liver cirrhosis. Hepatol. Res. Off. J. Jpn. Soc. Hepatol. 46(8), 743–751 (2016).
Author information
Authors and Affiliations
Contributions
Conceptualization, F.P. and Z.H.; Formal analysis, Z.Y.; Methodology, M.S., B.H., S.A., D.F., F.P. and S.K.; Project administration, Z.Y. and A.H.; Writing–original draft, D.F., F.P. and Z.H. and Z.Y.; Writing–review & editing, Z.Y. and A.H. All authors read and approved.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Pashayee-Khamene, F., Heidari, Z., Fotros, D. et al. Dietary acid load and cirrhosis-related mortality: a prospective cohort study. Sci Rep 14, 3675 (2024). https://doi.org/10.1038/s41598-024-53882-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-53882-8
- Springer Nature Limited