Abstract
The Brazilian Organization for Crohn's Disease and Colitis (GEDIIB) established a national registry of inflammatory bowel disease (IBD). The aim of the study was to identify clinical factors associated with disease severity in IBD patients in Brazil. A population-based risk model aimed at stratifying the severity of IBD based on previous hospitalization, use of biologics, and need for surgery for ulcerative colitis (UC) and Crohn’s Disease (CD) and on previous complications for CD. A total of 1179 patients (34.4 ± 14.7y; females 59%) were included: 46.6% with UC, 44.2% with CD, and 0.9% with unclassified IBD (IBD-U). The time from the beginning of the symptoms to diagnosis was 3.85y. In CD, 41.2% of patients presented with ileocolic disease, 32% inflammatory behavior, and 15.5% perianal disease. In UC, 46.3% presented with extensive colitis. Regarding treatment, 68.1%, 67%, and 47.6% received biological therapy, salicylates and immunosuppressors, respectively. Severe disease was associated with the presence of extensive colitis, EIM, male, comorbidities, and familial history of colorectal cancer in patients with UC. The presence of Montreal B2 and B3 behaviors, colonic location, and EIM were associated with CD severity. In conclusion, disease severity was associated with younger age, greater disease extent, and the presence of rheumatic EIM.
Similar content being viewed by others
Introduction
The incidence and prevalence of inflammatory bowel disease (IBD), including Crohn´s disease (CD) and ulcerative colitis (UC), are changing over the decades1,2,3. Recent studies have described the global epidemiological trends in incidence and prevalence, which can be stratified into four epidemiological stages: emergence of new cases, acceleration in incidence, compounding prevalence, and prevalence equilibrium4.
Developing countries in Latin America, Asia, and Eastern Europe have shown an increase in IBD cases, which could be attributed to an occidental lifestyle due to increased exposure to ultra-processed food, tobacco, sedentarism, stress, and air pollution2,4,5,6. In 2020, Salgado et al.7 published an article that considered the most important risk factors of IBD in the Brazilian population. Predictive factors for CD and UC were female sex (odds ratio [OR] 1.31; OR 1.69), low monthly family income (OR 1.78; OR 1.57), fewer cohabitants (OR 1.70; OR 1.60), absence of vaccination (OR 3.11; OR 2.51), previous history of bowel infections (OR 1.78; OR 1.49), and family history of IBD (OR 5.26; OR 3.33). IBD societies worldwide are alert to the importance of preventive behaviors in changing IBD’s emergence and healthcare costs related to IBD8.
However, there is still a lack of high-quality national population-based epidemiological data from newly industrialized countries. To date, there is a lack of detailed national-level clinical information on IBD in Brazil, including disease factors associated with severity or poor outcomes of the disease, making it difficult to compare data with those of other countries and across diverse regions of Brazil. The early identification of factors associated with a worse prognosis allows the stratification of patients according to severity and the indication of an early effective treatment, avoiding disease complications and disability, thus contributing to better disease control and health-related quality of life9,10.
The IBD National Patient Registry is an initiative of the GEDIIB (Brazilian Organization for Crohn's Disease and Colitis) that aims to survey the epidemiological profile of patients with IBD by creating a centralized registry with data on patients monitored in public and private healthcare services. This study aimed to characterize the profile and identify the clinical factors associated with disease severity in patients with IBD in Brazil.
Materials and methods
Study design
This cohort study was conducted between August 2020 and August 2022. Data obtained from medical records and/or from patients during regular follow-up visits were registered on the REDCap data platform. Participants of IBD Public and Private Centers previously approved in their Ethical Committee, who fulfilled the inclusion criteria, were subsequently recruited (random sampling method). Sample size was calculated considering a Brazilian epidemiological study3 with an estimation of at least 1000 patients in this study. The inclusion criteria were confirmed diagnosis of IBD and agreement to participate in the study. IBD diagnosis was based on established clinical, endoscopic, radiological, and histological criteria according to the literature11,12,13. There were no age restrictions; the study included all age groups. The exclusion criteria were lack of main variables, such as any type of IBD (CD, UC, or unclassified IBD (IBD-U) and duplicate data.
Clinical and sociodemographic variables
The variables from the registration data included date of birth; current age; type of healthcare service utilized; ethnicity (as personally declared); sex; education; medical history of comorbidities; smoking status (never smoked, current smoker, or past smoker); city/state of outpatient care; type of IBD (CD, UC, or IBD-U); age at symptom onset, diagnosis, and registration; Montreal classification14; disease severity; initial manifestations; extraintestinal manifestations (EIMs); presence of fistulas; comorbidities; and body mass index (BMI). Surgeries related to IBD, such as intestinal resections, placement of setons, colectomy due to intractable disease activity or neoplasia, and surgical complications (including complications related to short bowel syndrome or fecal incontinence) were also included. Information about previous hospitalizations (related to disease activity or IBD complications such as infectious diseases) and previous or current use of medications were recorded. In addition, data on family history of IBD or other immune-mediated diseases/cancers, including the degree of kinship, were also collected.
Outcome variables
Disease severity was characterized by previous hospitalization, use of biologics, and need for surgery in patients with CD and UC. Complications such as disease activity, pancreatitis, or infectious diseases (herpes simplex or zoster, tuberculosis, upper airway infection, fungal infection, pneumonia, and urinary infection) were also considered for patients with CD. For CD, the independent variables for the final model were the Montreal Classification (L-disease location, B-disease behavior), age at diagnosis (1–20 y, 21–40 y, 41–60 y, and 61–88 y), sex (male vs. female), smoking status (current vs. past), comorbidities (yes vs. no), and rheumatic EIMs (yes vs. no). For UC, the independent variables were the extent of the disease, age at diagnosis (1–20 y, 21–40 y, 41–60 y, and 61–88 y), sex (male vs. female), smoking status (current vs. past), comorbidities (yes vs. no), rheumatic EIMs (yes vs. no), and presence of a familial history of colorectal colon cancer (yes vs. no).
Statistical analysis
Data are expressed as mean ± standard deviation or median (range) for continuous variables and as frequency (proportion) for qualitative variables. Statistical analysis was performed using the Poisson regression model to obtain the raw and adjusted frequency ratios15. For patients with UC, four models were constructed based on four independent dependent variables: history of colectomy, colorectal cancer, hospitalization, and use of biologics or small molecules. The initial regression model was theoretically conceived as having as independent variables such as extensive colitis, proctitis, age at diagnosis, EIMs, comorbidities, BMI, smoking status, sex, and familial history of colon cancer. However, BMI was removed because of numerical insufficiency. Four models were constructed for patients with CD, based on four separately dependent variables: previous surgery, presence of complications (as described above), use of biologics, and hospitalization. As initial independent variables, the models considered the behavior of the disease (B2-stricturing; B3-penetrating), ileal location, colonic location, age at diagnosis, EIMs, comorbidities, smoking status, and sex. For all models, in assessing the contribution of each independent variable to the respective regression model, the module of the percentage difference was calculated for each exponentiated Beta of the regression (frequency ratio) compared with the value 1 as a reference. Patients with IBD-U were excluded because of the small sample size. A value of 5% was set as the minimum permanence value of the variable in the model. The goodness of fit of the models was assessed using the Akaike Information Criterion16 and residual analysis. To assess the assumption of non-overdispersion, the ratio of the deviance residuals to their degrees of freedom17 was used. Because the sampling plan was non-probabilistic, P-values or confidence intervals were not calculated owing to the lack of stability in the standard error estimates18,19. The global variance inflation factor (GVIF) was used to evaluate the presence of collinearity, assuming a GVIF < 5% with its absence20. Finally, the profile-predictive models were calculated from the final Poisson model equation for models with at least one remaining independent variable. Analysis was performed using the statistical package R (version 4.2.2)21 in Linux Mint version 21. The statistical review of the study was performed by biomedical statistician.
Ethical considerations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. This study was approved by the Local Research Ethics Committee, (CAAE: 71343417.2.1001.5629) and by all respective boards from the participating centers (listed in the Declarations). All participants received explanations about the study aims and expected results, having been enrolled in the study only after signing the informed consent term.
Results
A total of 1179 patients were included: 600 (51%) with UC, 568 (48%) with CD, and 11 (0.9%) with IBD-U. Table 1 shows the clinical and demographic characteristics of the patients with IBD. A total of 108 patients were excluded due to inconsistence data.
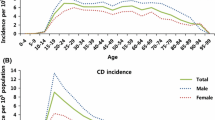
Regarding the initial symptoms, 42% presented with diarrhea, 38% with abdominal pain, and 20% with weight loss. The age at IBD symptom onset ranged from 1 to 87 years (32.3 ± 14.4) and the time from the beginning of the symptoms to diagnosis was 3.85 years. According to the Montreal Classification of CD, most patients were diagnosed between 17 and 40 years of age (63.1%), the main location was ileocolonic region (41.2%), and the main behavior was non-stricturing and non-penetrating (32%). Among patients with UC, 46.3% presented with extensive colitis, 30% had left-sided colitis, and 23.7% had proctitis.
According to BMI, 3.9% were malnourished, 30.9% were overweight, and 18% were obese. The main EIMs were rheumatic (21%), dermatological (4.2%), hepatic (2.6%), nephrological (2.1%), and ophthalmological (0.9%).
The Southeast region of Brazil, comprising the states of São Paulo, Rio de Janeiro, Minas Gerais, and Espírito Santo, had the highest percentage of patients included in the study (72.3%), followed by the Midwest (13.8%), Northeast (6.9%), South (4.9%), and North (1.9%).
Regarding medical treatment, 72.4% of patients were using or had previously received biologics (34.3% infliximab, 21.9% adalimumab, 7.2% vedolizumab, 6.5% ustekinumab, 1.7% certolizumab pegol, and 0.5% golimumab), 73.1% salicylates, 51.6% immunosuppressors, and 0.9% tofacitinib (Table 2).
Of those who underwent surgery (n = 312, 26.5%; elective vs. urgent/emergency: 54% vs. 46%), 240 (76.9%) had CD, 69 (22.1%) had UC, and 4 (1%) had U-IBD. Most procedures (80%) involved an open laparotomy, whereas 20% were laparoscopic. In total, 227 (72.7%) were abdominal (8.5% colectomy, 6.1% enterectomy, 1.2% diagnostic laparotomy, and 1.2% appendectomy, among others) and 76 (24%) were perianal (6% seton placement, 8% abscess drainage and curettage, and 4% fistulotomy, among others). Complications related to surgery were more prevalent in patients with CD (CD vs. UC: 45.3% vs. 19.3%). Almost 30% of the patients underwent more than one surgery.
At least one complication was reported in 62% of the patients; most of them were infectious disorders requiring prolonged hospitalization. Comorbidities were present in 72% of patients, and the most prevalent was high blood pressure 8.3%/3.3%, obesity 0.8%/0.5%, diabetes 0/0.3%, heart failure 0/0.3%, thrombosis 0.3%/0.6% and nephrolithiasis 2.3%/1.4%, in CD and UC patients, respectively.
Clinical factors associated with severity in patients with CD
Table 3 shows the adjusted frequency ratio (FR) for disease severity in patients with CD. The presence of rheumatic EIMs was associated with the use of biological therapy (FR > 1.1). Patients’ presenting behavior, according to the Montreal classification, of B2 and B3; colonic location; and presence of rheumatic EIMs demonstrated a higher risk of disease complications (FR > 1.1). Frequency ratios indicated the absence of a contribution of the variables to the model for hospitalization and surgery.
From the final adjusted model, it was possible to obtain the association between the different profiles of patients with CD and the need for biologics. Thus, the most associated patient profile had stricturing disease (B2 behavior), young age at diagnosis, presence of rheumatic EIMs, male sex, and no smoking/no history of smoking status, with an expected average value of 50.24% of the need for biological therapy. The profile most associated with the presence of complications was stricturing or penetrating behavior, not having an ileal location, having a colonic location, being young, having a rheumatic EIM, not having a comorbidity, not smoking, and being male. For such a profile, there is an expected 56% chance of disease complications.
Clinical factors associated with severity in patients with UC
Table 4 shows the adjusted FR for disease severity with respect to UC. The presence of extensive colitis, rheumatic EIMs, male sex, and a familial history of colorectal cancer were associated with the necessity of biological therapy (FR > 1.1). Hospitalization was not associated with any of the variables studied. Regarding the need for colectomy, the associated variables were the presence of extensive colitis, rheumatic EIMs, comorbidities, and male sex (FR > 1.1).
From the final adjusted model, we suggest that patients with extensive colitis, young age at diagnosis, history of colorectal cancer in the family, presence of rheumatic EIMs, male sex, absence of comorbidities, and nonsmoking status had an expected mean value of 55.64% for the need for biological therapy. We also suggest that patients with extensive colitis, young age at diagnosis, presence of rheumatic EIMs, male sex, presence of comorbidity, and non-smoking status presented an expected mean value of 20.4% for the need for colectomy.
Discussion
This study is the first cohort of patients with IBD from the Brazilian National Registry of Patients conducted by the GEDIIB in Brazil. Epidemiological studies are important knowledge tools for a given population aiming for future interventions. Studies with continuous inclusion of patients in their database can generate interesting results in the short, medium, and long terms and can help society, medical organizations, and the government in planning public policies to serve this specific population.
A recent increase in the incidence of IBD has been described in the literature, mostly in Asia22 and Latin America23, although a higher prevalence has also been reported in Africa24. In absolute numbers, the burden of IBD in developing countries may be greater than that of the combined burden in Europe and North America25, which increases costs for the healthcare system, as in most developing countries. In addition, early diagnosis is still lacking, with patients receiving a diagnosis at a later stage with complications. The number of preventable surgeries, outdated treatments (as 5-ASA for CD and corticosteroid use in maintenance therapy), and complications (such as neoplasia) compromise a patient's quality of life.
In Latin America, there are few epidemiological reports from national databases. Juliao-Banos et al.26 examined the epidemiological trends of IBD in Colombia using a national database of 33 million adults, encompassing 97.6% of the Colombian population. This study calculated the incidence and prevalence of UC and CD from 2010 to 2017 and examined the epidemiological trends according to urbanicity, demographics, and region. In addition, the IBD phenotype (Montreal Classification), prevalence of IBD-related surgeries, and types of IBD medications prescribed to adult patients attending a regional IBD clinic were assessed. Using a nationally representative sample and a regional clinic cohort, they found that UC is more common in Colombia and is increasing in urban regions. There were 649 patients with IBD in the clinical cohort: 73.7% with UC and 24.5% with CD. The mean ages at diagnosis of CD and UC were 41.0 years and 39.9 years, respectively. Most patients with UC developed extensive colitis (43%), whereas most patients with CD developed ileocolonic disease. A total of 16.7% of patients with CD had perianal disease. Patients with CD received more biologics than those with UC.
The National Registry from GEDIIB aims to quantify the real prevalence of IBD in the Brazilian territory as well as to identify the differences between the geographic regions27 of the country, given the territorial dimension of the same. Our sample demonstrated a predominance of extensive colitis in UC but had a higher percentage of patients with CD (48%). The mean age at the time of IBD diagnosis was 32 years in Brazil and 40 years in Colombia. The time from the beginning of the symptoms to diagnosis was approximately 3.8 years, almost the same as that found by Nobrega et al.28 and better than that reported by Fróes et al.27, who found a delay of 5 years between the reports of the first symptoms and the diagnosis of CD in Rio de Janeiro.
Long-term population studies may have a lower number of patients in their first publications, and it would be interesting to compare population profiles over time. The ECCO-EpiCom 2011 inception cohort analyzed the differences in disease phenotype, medical therapy, surgery, and hospitalization rates during the first year after diagnosis. A total of 258 patients with CD, 380 with UC, and 71 with unclassified IBD were included. Overall, 178 (25%), 460 (65%), and 71 (10%) patients were diagnosed in Eastern Europe, Western Europe, and Australia, respectively. During the first year after diagnosis, surgery and hospitalization rates were significantly higher in patients with CD in Eastern Europe than in those in Western Europe and Australia. In contrast, significantly more patients with CD were treated with biologics in Western European and Australian centers29. Our sample included 1179 cases, although modest, a larger number than in the ECCO-EpiCom 2011 inception and Colombia papers, with patients mostly found in the southeastern region of Brazil.
Our study included a representative sample of five regions of Brazil with retrospective and prospective data. It is important to note that the findings presented here reflect only initial data regarding the country's higher number of patients with IBD, as shown by Quaresma et al.3. According to their data, IBD has recently reached a prevalence of more than 60 cases per 100,000 inhabitants in Brazil, depending on the degree of urbanization in the region. Therefore, IBD is no longer considered rare. The results of regional studies also show a south-north gradient, with a higher incidence in urbanized areas in the South/Southeast region of the country2,3,5,30. In the states of São Paulo and Espírito Santo30,31, prevalence rates of IBD were 52.6/100,000 and 38.2/100,000, respectively in contrast with 12.8/100,000 in the state of Piaui32, localized in the northern part of the country, with lower human development index.
To date, most studies in Brazil have analyzed public health data from University Hospitals or the Unified Health System (DATASUS), which is an open-access population-based health and disease registry that contains information from the national unified health system (SUS) through records of hospital admissions, outpatient procedures, consultations, and IBD-related medication dispensing, searched according to the International Statistical Classification of Diseases and Related Health Problems, Tenth Revision (ICD-10), with codes K50 for CD and K51 for UC2,5,30,31,33. However, these studies lacked information on private services, which may have led to underestimation. As supplementary health is responsible for approximately 25% of the country's health coverage34, a National Registry can contribute to a more accurate epidemiological profile of IBD in the country. In this first analysis of the National Registry results, almost the same number of patients were followed up at private and public (academic or not) centers, suggesting that a relatively high proportion of patients can afford private health insurance. A National IBD Database with inputs from most gastroenterologists and proctologists of reference for IBD will greatly contribute to the establishment of a powerful, accurate, and dynamic tool for the study of IBD.
Our data are in line with those reported in the literature, considering the risk factors related to severe disease. After performing adjustments, we found that severe disease, defined as the need for hospitalization, surgery, and the use of biologics for UC, was associated with the presence of extensive colitis, rheumatic EIMs, male sex, comorbidities, and a familial history of colorectal colon cancer. Regarding CD, severe disease was associated with Montreal phenotypes B2 and B3, colonic location, and the presence of rheumatic EIMs. Younger age at initial diagnosis, the presence of perianal lesions, ileal involvement, smoking, and the need for corticosteroid therapy are the major predictors of a disabling disease or change of behavior to a more aggressive disease as reported by Blonski et al.35. Romberg-Camps et al.36 showed that in CD, small bowel location, stricturing disease, and young age predicted disease recurrence, whereas in UC, extensive colitis and older age at diagnosis were negative prognostic predictors. Recently, Sacramento et al.37 reported the variables associated with moderate-to-severe CD progression. These results are also in line with our findings, demonstrating perianal disease, stricturing or penetrating behavior, and ileocolonic localization. Age of < 40 years at diagnosis (81.3% vs. 62.0%, P = 0.004), upper gastrointestinal tract involvement (21.8% vs. 10.3%, P = 0.040), and perianal disease (35.9% vs. 16.3%, P < 0.001) were also associated with the use of immunobiological agents.
Almost half the patients in this study were overweight or obese. This is in line with studies linking environmental factors, especially diet, to an increased risk of immune-mediated diseases38,39,40. In Brazil, the increased consumption of ultra-processed foods over the last few decades has contributed to the burden of obesity41,42. Between 1974 and 2009, the prevalence of obesity in children aged 5–9 years increased from 2.9 to 16.5% among boys and from 1.8 to 11.8% among girls. For adolescents (aged 10–19 years), the prevalence varied from 0.4 to 5.9% in males and from 0.7 to 4.0% in females during the same period. In adults aged 20 years or older, the obesity prevalence increased by more than eightfold (2.8–22.8%) among men and threefold (8.0–30.2%) among women from 1974 to 201941,42. The gradual weakening of traditional food patterns, based on fresh or minimally processed foods, with the concomitant increase in the consumption of ultra-processed foods, contributes to immune dysregulation of the intestinal microbiome in those with a genetic propensity for IBD and development of the disease. This is an important point of discussion as many healthcare professionals do not recognize IBD in non-malnourished patients. However, it is well known that being overweight or obese is associated with sarcopenia and vitamin deficiencies, possibly contributing to the worst prognosis in IBD.
Until 2020, Brazil had unequal access to medication for CD and UC, as demonstrated by Vilela et al.43, with a higher number of physicians reporting difficulty in accessing or releasing medicines for UC than for CD. Martins et al.44 also observed that the complication rate among patients with UC undergoing therapy available in the National Health System from 2011 to 2020 was higher among those receiving only conventional therapy. No biological therapy was available for UC in the public system until 2020, and only anti-TNF therapy was available for moderate-to-severe CD. Among patients from private services, only those with moderate-to-severe CD had access to anti-TNFs, anti-integrins, and anti-interleukins. The publication of national epidemiological studies2,3,5,30,31,32,33,37,44,45,46,47,48, physicians´ perceptions of IBD in Brazil43, and studies on the socioeconomic impact of IBD, including absence from work27,49,50, served to alert the authorities and resulted in the incorporation of biological therapy for UC in the public sphere in 2020 and in the private system in 2021.
The National Patient Registry, organized by the GEDIIB, is a prospective study in its beginning. An important limitation of this study is that once the registry was initiated during the COVID-19 pandemic, it did not include a large number of patients. However, as it includes data from the entire national territory, and it comprises a data registry with continuous inclusion of patients in their database, which can generate interesting results in the short, medium, and long terms and help society, medical organizations, and the government in planning public policies to serve this specific population with IBD, a major gastrointestinal disease.
Conclusion
To date, no epidemiological study with public and private data has covered the entire Brazilian territory considering the clinical aspects associated with IBD severity. The results obtained from the ongoing registry will be fundamental for improving the information quality in a country with continental dimensions, such as Brazil. The greater the number of qualified participating researchers from different regions of the country, the greater the representativeness of the data, which may greatly help direct government actions on behalf of patients with IBD.
Data availability
Data, analytical methods, and study materials are available to other researchers upon specific request. Please contact the corresponding author.
References
Kaplan, G. G. & Ng, S. C. Understanding and Preventing the global increase of inflammatory bowel disease. Gastroenterology 152(2), 313-321.e. https://doi.org/10.1053/j.gastro.2016.10.020 (2017).
da Luz, M. A. et al. Geosocial features and loss of biodiversity underlie variable rates of inflammatory bowel disease in a large developing country: A population-based study. Inflamm. Bowel Dis. 28(11), 1696–1708. https://doi.org/10.1093/ibd/izab346 (2022).
Quaresma, A. B. et al. Temporal trends in the epidemiology of inflammatory bowel diseases in the public healthcare system in Brazil: A large population-based study. Lancet Reg. Health Am. 9(13), 100298. https://doi.org/10.1016/j.lana.2022.100298 (2022).
Kaplan, G. G. & Windsor, J. W. The four epidemiological stages in the global evolution of inflammatory bowel disease. Nat. Rev. Gastroenterol. Hepatol. 18(1), 56–66. https://doi.org/10.1038/s41575-020-00360-x (2021).
Quaresma, A. B., Kaplan, G. G. & Kotze, P. G. The globalization of inflammatory bowel disease: The incidence and prevalence of inflammatory bowel disease in Brazil. Curr. Opin. Gastroenterol. 35(4), 259–264. https://doi.org/10.1097/MOG.0000000000000534 (2019).
Torres, J. et al. Results of the Seventh Scientific Workshop of ECCO: Precision medicine in IBD-prediction and prevention of inflammatory bowel disease. J. Crohns Colitis 15(9), 1443–1454. https://doi.org/10.1093/ecco-jcc/jjab048 (2021).
Salgado, V. C. L. et al. Risk factors associated with inflammatory bowel disease: A multicenter case-control study in Brazil. World J. Gastroenterol. 26(25), 3611–3624. https://doi.org/10.3748/wjg.v26.i25.3611 (2020).
Torres, J., Burisch, J., Riddle, M., Dubinsky, M. & Colombel, J. F. Preclinical disease and preventive strategies in IBD: Perspectives, challenges and opportunities. Gut 65(7), 1061–1069. https://doi.org/10.1136/gutjnl-2016-311785 (2016).
Herrera-deGuise, C., Casellas, F., Robles, V., Navarro, E. & Borruel, N. Predictive value of early restoration of quality of life in Crohn’s disease patients receiving antitumor necrosis factor agents. J. Gastroenterol. Hepatol. 30(2), 286–291. https://doi.org/10.1111/jgh.12803 (2015).
Peyrin-Biroulet, L. et al. Defining disease severity in inflammatory bowel diseases: Current and future directions. Clin. Gastroenterol. Hepatol. 14(3), 348-354.e17. https://doi.org/10.1016/j.cgh.2015.06.001 (2016).
Gionchetti, P. et al. 3rd European evidence-based consensus on the diagnosis and management of Crohn’s disease 2016: Part 2: Surgical management and special situations. J. Crohns Colitis 11(2), 135–149. https://doi.org/10.1093/ecco-jcc/jjw169 (2017).
Gomollón, F. et al. 3rd European evidence-based consensus on the diagnosis and management of Crohn’s disease 2016: Part 1: Diagnosis and medical management. J. Crohns Colitis 11(1), 3–25. https://doi.org/10.1093/ecco-jcc/jjw168 (2017).
Magro, F. et al. Third European evidence-based consensus on diagnosis and management of ulcerative colitis. Part 1: definitions, diagnosis, extra-intestinal manifestations, pregnancy, cancer surveillance, surgery, and ileo-anal pouch disorders. J. Crohns Colitis 11(6), 649–670. https://doi.org/10.1093/ecco-jcc/jjx008 (2017).
Satsangi, J., Silverberg, M. S., Vermeire, S. & Colombel, J. F. The Montreal classification of inflammatory bowel disease: Controversies, consensus, and implications. Gut 55(6), 749–753. https://doi.org/10.1136/gut.2005.082909 (2006).
Coutinho, L. M. S., Scazufca, M. & Menezes, P. R. Métodos para estimar razão de prevalência em estudos de corte transversal. Rev. Saúde Públ. 42(6), 992–998. https://doi.org/10.1590/S0034-8910200800060000, (2008).
Bozdogan, H. Model selection and Akaike’s information criterion (AIC): The general theory and its analytical extensions. Psychometrika 52, 345–370. https://doi.org/10.1007/BF02294361 (1987).
Cameron, A. C. & Trivedi, P. K. Regression Analysis of Count Data 2nd edn. (Cambridge University Press, 2013).
Wasserstein, R. L. & Lazar, N. A. The ASA statement on p-values: Context, process, and purpose. Am. Stat. 70(2), 129–133. https://doi.org/10.1080/00031305.2016.1154108 (2016).
Ludwig, D. A. Use and misuse of p-values in designed and observational studies: Guide for researchers and reviewers. Aviat. Space Environ. Med. 76(7), 675–680 (2005).
Dodge Y. The Concise Encyclopedia of Statistics. 2010 a edição (Springer, Philadelphia, 2008). Google Search [Internet]. [cited 2023 Apr 18]. https://doi.org/10.1007/978-0-387-32833-1
R: The R Project for Statistical Computing [Internet]. [cited 2023 Apr 18]. Available from: https://www.r-project.org/
Park, J. & Cheon, J. H. Incidence and prevalence of inflammatory bowel disease across Asia. Yonsei Med. J. 62(2), 99–108. https://doi.org/10.3349/ymj.2021.62.2.99 (2021).
Kotze, P. G. et al. Progression of inflammatory bowel diseases throughout Latin America and the Caribbean: A systematic review. Clin. Gastroenterol. Hepatol. 18(2), 304–312. https://doi.org/10.1016/j.cgh.2019.06.030 (2020).
Watermeyer, G. et al. Inflammatory bowel disease in sub-Saharan Africa: Epidemiology, risk factors, and challenges in diagnosis. Lancet Gastroenterol. Hepatol. 7(10), 952–961. https://doi.org/10.1016/S2468-1253(22)00047-4 (2022).
Singh, P., Ananthakrishnan, A. & Ahuja, V. Pivot to Asia: Inflammatory bowel disease burden. Intest. Res. 15(1), 138–141. https://doi.org/10.5217/ir.2017.15.1.138 (2017).
Juliao-Baños, F. et al. Trends in the epidemiology of inflammatory bowel disease in Colombia by demographics and region using a nationally representative claims database and characterization of inflammatory bowel disease phenotype in a case series of Colombian patients. Medicine (Baltimore) 100(7), e24729. https://doi.org/10.1097/MD.0000000000024729 (2021).
de SB Fróes, R. et al. The socio-economic impact of work disability due to inflammatory bowel disease in Brazil. Eur. J. Health Econ. 19(3), 463–470. https://doi.org/10.1007/s10198-017-0896-4 (2018).
Nóbrega, V. G. et al. THE onset of clinical manifestations in inflammatory bowel disease patients. Arq. Gastroenterol. 55(3), 290–295. https://doi.org/10.1590/S0004-2803.201800000-73 (2018).
Vegh, Z. et al. Treatment steps, surgery, and hospitalization rates during the first year of follow-up in patients with inflammatory bowel diseases from the 2011 ECCO-Epicom inception cohort. J. Crohns Colitis 9(9), 747–753. https://doi.org/10.1093/ecco-jcc/jjv099 (2015).
Gasparini, R. G., Sassaki, L. Y. & Saad-Hossne, R. Inflammatory bowel disease epidemiology in São Paulo State, Brazil. Clin. Exp. Gastroenterol. 30(11), 423–429. https://doi.org/10.2147/CEG.S176583 (2018).
Lima Martins, A., Volpato, R. A. & Zago-Gomes, M. D. P. The prevalence and phenotype in Brazilian patients with inflammatory bowel disease. BMC Gastroenterol. 18(1), 87. https://doi.org/10.1186/s12876-018-0822-y (2018).
Parente, J. M. et al. Inflammatory bowel disease in an underdeveloped region of Northeastern Brazil. World J. Gastroenterol. 21(4), 1197–1206. https://doi.org/10.3748/wjg.v21.i4.1197 (2015).
Gomes, T. N. F., de Azevedo, F. S., Argollo, M., Miszputen, S. J. & Ambrogini, O. Jr. Clinical and demographic profile of inflammatory bowel disease patients in a reference center of São Paulo, Brazil. Clin. Exp. Gastroenterol. 17(14), 91–102. https://doi.org/10.2147/CEG.S288688 (2021).
Website Agência Nacional da Saúde (ANS) [Internet]. Available from: https://www.ans.gov.br/images/stories/Materiais_para_pesquisa/Perfil_setor/sala-de-situacao.html
Blonski, W., Buchner, A. M. & Lichtenstein, G. R. Clinical predictors of aggressive/disabling disease: Ulcerative colitis and Crohn disease. Gastroenterol. Clin. N. Am. 41(2), 443–462. https://doi.org/10.1016/j.gtc.2012.01.008 (2012).
Romberg-Camps, M. J. et al. Influence of phenotype at diagnosis and of other potential prognostic factors on the course of inflammatory bowel disease. Am. J. Gastroenterol. 104(2), 371–383. https://doi.org/10.1038/ajg.2008.38 (2009).
Sacramento, C. D. S. B. et al. Variables associated with progression of moderate-to-severe Crohn’s disease. BMJ Open Gastroenterol. 9(1), e001016. https://doi.org/10.1136/bmjgast-2022-001016 (2022).
Harper, J. W. & Zisman, T. L. Interaction of obesity and inflammatory bowel disease. World J. Gastroenterol. 22(35), 7868–7881. https://doi.org/10.3748/wjg.v22.i35.7868 (2016).
Kotze, P. G. Obesity and Crohn’s disease: What comes first, the egg or the chicken?. Arq. Gastroenterol. 54(3), 268. https://doi.org/10.1590/S0004-2803.201700000-37 (2017).
Martinez, K. B., Leone, V. & Chang, E. B. Western diets, gut dysbiosis, and metabolic diseases: Are they linked?. Gut Microbes 8(2), 130–142. https://doi.org/10.1080/19490976.2016.1270811 (2017).
Canhada, S. L. et al. Ultra-processed foods, incident overweight and obesity, and longitudinal changes in weight and waist circumference: The Brazilian longitudinal study of adult health (ELSA-Brasil). Public Health Nutr. 23(6), 1076–1086. https://doi.org/10.1017/S1368980019002854 (2020).
Louzada, M. L., Steele, E. M., Rezende, L. F. M., Levy, R. B. & Monteiro, C. A. Corrigendum: Changes in obesity prevalence attributable to ultra-processed food consumption in Brazil between 2002 and 2009. Int. J. Public Health 8(67), 1605178. https://doi.org/10.3389/ijph.2022.1605178 (2022).
Vilela, E. G. et al. inflammatory bowel disease care in Brazil: How it is performed, obstacles and demands from the physicians’ perspective. Arq. Gastroenterol. 57(4), 416–427. https://doi.org/10.1590/S0004-2803.202000000-77 (2020).
Martins, A. L. et al. Intestinal complications in Brazilian patients with ulcerative colitis treated with conventional therapy between 2011 and 2020. World J. Gastroenterol. 29(8), 1330–1343. https://doi.org/10.3748/wjg.v29.i8.1330 (2023).
da Silva, B. C. et al. The demographic and clinical characteristics of ulcerative colitis in a Northeast Brazilian population. Biomed. Res. Int. 2015, 359130. https://doi.org/10.1155/2015/359130 (2015).
Zaltman, C. et al. Real-world disease activity and sociodemographic, clinical and treatment characteristics of moderate-to-severe inflammatory bowel disease in Brazil. World J. Gastroenterol. 27(2), 208–223. https://doi.org/10.3748/wjg.v27.i2.208 (2021).
Andrade, A. R. et al. Risk of thrombosis and mortality in inflammatory bowel disease. Clin. Transl. Gastroenterol. 9(4), 142. https://doi.org/10.1038/s41424-018-0013-8 (2018).
Victoria, C. R., Sassak, L. Y. & Nunes, H. R. Incidence and prevalence rates of inflammatory bowel diseases, in midwestern of São Paulo State, Brazil. Arq. Gastroenterol. 46(1), 20–25. https://doi.org/10.1590/s0004-28032009000100009 (2009).
de Sá Brito Fróes, R. et al. Prevalence, indirect costs, and risk factors for work disability in patients with Crohn’s disease at a tertiary care center in Rio de Janeiro. Dig. Dis. Sci. 66(9), 2925–2934. https://doi.org/10.1007/s10620-020-06646-z (2021).
de Codes, L. M. G. et al. Anorectal function and clinical characteristics associated with fecal incontinence in patients with Crohn’s disease. J. Crohns Colitis https://doi.org/10.1093/ecco-jcc/jjad048 (2023).
Acknowledgements
We would like to thank to Prof. Maurício Cardeal statistician, Camila da Silveira Guimarães, and Kauyza Beatriz Martins Scanferla, research coordinators, Fatima Lombardi financial administrative manager of GEDIIB, and we also would like to express our gratitude to the investigators who collaborated with the inclusion of data of the National Registry Data of IBD Patients in Brazil: Anna Luiza Pereira Álvares, Bernardo Dias São José, Mary Carmem Santos Silveira, Francisco Guilherme Cancela Penna, Gilmara Pandolfo Zabot, José Miguel Luz Parente, Mariana de Oliveira Pantoja, Ornella Sari Cassol, Silvia Ferreira Araújo, Valéria Ferreira de Almeida e Borges, Vivian Menegassi.
Funding
Supported by GEDIIB—Brazilian Organization for Crohn's Disease and Colitis.
Author information
Authors and Affiliations
Contributions
RSP, CZ, CHMS, MB, ABQ, GOS, RLL, SFLJ, MMS, GSPH, RLKJ, CRN, OF, JRB participated in the acquisition, analysis, and interpretation of the data, and drafted the initial manuscript. RSBF, ARA, MAGF, HSPS, SJM, LYS, RSH had substantial contributions to the conception and design of the study, data acquisition, analysis, and interpretation of data; made critical reviews related to the important intellectual content of the manuscript; and reviewed the article critically for important intellectual content. All the authors revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
Renata de Sá Brito Fróes—Received lecture fee[s] from AbbVie, Janssen, Takeda, Ferring and Pfizer; is advisory committee member for Janssen and Takeda; Adriana Ribas Andrade—Received lecture fee[s] from AbbVie, Janssen; Mikaell Alexandre Gouvea Faria—Received lecture fee[s] from AbbVie, Janssen, Takeda, Nestlé, advisory committee member for Janssen; Rogério Serafim Parra—Received lecture fee[s] from AbbVie, Janssen, Takeda, and Pfizer; is advisory committee member for Janssen and AbbVie; Cyrla Zaltman—Received lecture fee[s] from Janssen, Takeda, Celltrion, Pfizer; Carlos Henrique Marques dos Santos—Received lecture fee[s] from AbbVie, Janssen, Takeda; Mauro Bafutto—Received lecture fee[s] from AbbVie, Janssen, Takeda; Abel Botelho Quaresma—Received lecture fee[s] from AbbVie, Janssen; Genoile Oliveira Santana—Received lecture fee[s] from AbbVie, Janssen, Takeda, Ferring and Pfizer; is advisory committee member for Janssen; Rafael Luís Luporini—Received lecture fee[s] from AbbVie, Janssen, Takeda; Mardem Machado de Souza—Received lecture fee[s] from AbbVie, Janssen, Takeda; Ligia Yukie Sassaki—Received lecture fee[s] from Janssen; Rogerio Saad-Hossne—Received lecture fee[s] and advisory committee member for AbbVie, Takeda, Janssen, Pfizer, Fresenius and Amgen. Heitor Siffert Pereira de Souza, Sérgio Figueiredo de Lima Junior, Sender Jankiel Miszputen, Giedre Soares Prates Herrerias, Roberto Luiz Kaiser Junior, Catiane Rios do Nascimento, Omar Féres, Jaqueline Ribeiro de Barros—No potential conflict of interest to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fróes, R.S.B., Andrade, A.R., Faria, M.A.G. et al. Clinical factors associated with severity in patients with inflammatory bowel disease in Brazil based on 2-year national registry data from GEDIIB. Sci Rep 14, 4314 (2024). https://doi.org/10.1038/s41598-024-54332-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-54332-1
- Springer Nature Limited