Abstract
To delineate the phenotype of erosive hand osteoarthritis (EHOA) in a Spanish population and assess its correlation with metabolic syndrome. We conducted a cross-sectional study using baseline data from the Prospective Cohort of Osteoarthritis from A Coruña (PROCOAC). Demographic and clinical variables, obtained through questionnaires, clinical examinations, and patient analytics, were compared among individuals with hand OA, with and without EHOA. We performed appropriate univariate and multivariate stepwise regression analyses using SPSS v28. Among 1039 subjects diagnosed with hand OA, 303 exhibited EHOA. Multivariate logistic regression analysis revealed associations with inflamed joints, nodular hand OA, and total AUSCAN. Furthermore, the association with a lower prevalence of knee OA remained significant. The influence of metabolic syndrome (MetS) on EHOA patients was analyzed by including MetS as a covariate in the model. It was observed that MetS does not significantly impact the presence of EHOA, maintaining the effect size of other factors. In conclusion, in the PROCOAC cohort, EHOA is associated with nodular hand OA, inflammatory hand OA, and a higher total AUSCAN. However, EHOA is linked to a lower prevalence of knee OA. Importantly, in our cohort, no relationship was found between EHOA and MetS.
Similar content being viewed by others
Introduction
Osteoarthritis (OA) of the hand is a highly prevalent disease, notably exhibiting a recognized female predominance1,2. The prevalence estimates vary depending on the definition employed. Radiographic hand OA, associated with the highest prevalence (ranging from 29 to 76%), contrasts with the symptomatic type, where frequencies are lower (ranging from 3 to 16%)1,2,3. Furthermore, compared with other forms of OA, such as knee or hip, hand OA generally has a higher prevalence4.
Hand OA encompasses various subsets5 and is acknowledged as a heterogeneous disease resulting in considerable disability with variable manifestations6,7,8,9,10. Erosive hand OA (EHOA) stands out among these subsets. The term “erosive” was first coined in 1966 by Peter and colleagues11. However, Crain previously used the term “Interphalangeal osteoarthritis” to describe a localized form characterized by destructive changes, intermittent inflammatory episodes, and eventual deformities and ankyloses12. While EULAR recommendations consider EHOA as a subset of hand OA, controversy arises due to its unclear etiopathogenesis. There’s a debate on whether to regard EHOA as a distinct disease entity or a severe phase within the continuum of hand OA13,14. Data from the Framingham Osteoarthritis study revealed EHOA prevalence ranging from 3.6% in men to 9.8% in women, increasing to 9.5% and 24.3%, respectively, over 60 years. Its development mainly occurs in patients with non-erosive hand OA (non-EHOA) at baseline15.
EHOA is radiographically defined by subchondral bone erosions in interphalangeal joints, cortical destruction, and subsequent reparative changes, including potential bony ankylosis5. Clinical features involve an abrupt onset of severe pain, varying degrees of stiffness, joint swelling, deformities, and erythema16,17. Additionally, EHOA is characterized by more severe and frequent synovitis, as well as radiographic progression18.
Hand OA commonly occurs, although not exclusively, in the context of generalized OA5, but findings in this regard are somewhat conflicting19,20,21,22,23. Controversy also exists regarding the varying degree of association between metabolic alterations—such as dyslipidemia, diabetes, hypertension, or overweight—and hand OA or EHOA24,25,26,27,28. Based on this evidence, further investigation is warranted to determine whether metabolic disturbances, specifically the metabolic syndrome, are associated with severe hand OA and whether this association differs for EHOA compared to non-EHOA.
With this background, the aim of this study is to describe the EHOA phenotype in the PROCOAC cohort (Prospective Cohort of Osteoarthritis from A Coruña) and its relationship with metabolic syndrome (MetS).
Methods
For this study, we utilized cross-sectional data at baseline (when the patient was diagnosed) from the PROCOAC cohort, a population-based study investigating the determinants and severity of osteoarthritis (OA) in the knees, hips, and hands29. The Galician ethics committee approved the study, and written informed consent was obtained from all participants. Patient recruitment commenced in 2006. Patients were included based on the following criteria: i) patients from Rheumatology consultations with hand pain and diagnosed with hand OA following ACR criteria; ii) patients with knee pain diagnosed with radiographic knee OA following ACR criteria; and iii) patients with hip pain diagnosed with radiographic hip OA following ACR criteria. The cohort comprised 1252 subjects, of which 1039 were diagnosed with hand OA following ACR criteria30,31.
Statement of ethics and consent
The Galician ethics committee, part of Xunta de Galicia, Spain, approved the study, and written informed consent was obtained from all participants. Research was conducted in accordance with relevant guidelines/regulations, and the manuscript includes a statement confirming that informed consent was obtained from all participants and/or their legal guardians. Research involving human participants must have been performed following the Declaration of Helsinki.
Measurements
All X-rays from patients with hand OA underwent rigorous review to confirm the erosive phenotype31, conducted by both a radiologist and a trained rheumatologist specialist in a blinded manner. Erosive hand OA (EHOA) was radiographically defined by subchondral erosion, cortical destruction, and subsequent reparative changes in interphalangeal (IP) joints, potentially involving bony ankylosis in at least one hand. Additionally, the total number of IP joints with erosions per patient was counted and recorded. The reliability of all X-rays was meticulously assessed. Evaluation of right and left-hand joints, including wrists, was conducted using anteroposterior images. Magnified views of the entire hand were employed to confirm the presence of typical erosions4.
For this study, baseline data included the following variables: age, gender, smoking habit (categorized as never smoker, current smoker, or former smoker), body mass index (BMI), abdominal perimeter, personal history of hypertension, type 2 diabetes, dyslipidemia, psoriasis, osteoporosis, and osteopenia. Definitions for conditions included systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mmHg for hypertension32, fasting plasma glucose ≥ 126 mg/dL, glycated hemoglobin ≥ 6.5%, or classic symptoms of hyperglycemia for diabetes33, total cholesterol ≥ 220 mg/dL, low-density lipoprotein (LDL) ≥ 150, high-density lipoprotein (HDL) < 40 mg/dL in men and < 50 mg/dL in women, and triglycerides > 150 mg/dL for dyslipidemia. Osteoporosis was defined as a bone mineral density (BMD) ≤ − 2.5 SD in the femoral neck or lumbar spine, and osteopenia as BMD between − 1.5 and − 2.5 SD34. Metabolic Syndrome status was assessed following ALAD (Asociación Latinoamericana de Diabetes) 2010 criteria35, considering criteria such as waist circumference ≥ 94 cm in men and ≥ 88 cm in women, and meeting at least 2 of 4 specified criteria. Nodular hand OA was defined as Heberden and/or Bouchard nodes plus underlying interphalangeal damage. Thumb base OA, inflamed IP joints (characterized by swelling, heat, pain, and redness), and fasting blood levels of glucose, LDL, HDL, triglycerides, erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), rheumatoid factor (RF), and anticitrulline antibodies (ACPA) were also collected at baseline. Additionally, the AUStralian CANadian index (AUSCAN Osteoarthritis Hand index NRS 3.1) questionnaire was assessed in a subset of 575 hand OA patients, providing a total score that considers pain, stiffness, and function, with each section evaluated on a scale from 0 to 10036,37.
Statistical analysis
The statistical analysis encompassed a descriptive examination of the studied variables. Continuous variables were presented as mean ± standard deviation (SD) and median, while qualitative variables were articulated in absolute values (n) and percentages.
Mean comparisons between two groups were conducted utilizing the Student’s t-test or Mann–Whitney test, depending on appropriateness, subsequent to normality verification through the Kolmogorov–Smirnov test. Associations of qualitative variables were scrutinized using the Pearson Chi-Square test.
Univariate and multivariate logistic regression models were employed to investigate factors linked to the presence of erosive osteoarthritis (OA) of the hand. Construction of the multivariable models adhered to the backward selection method, where the Wald statistic served as the elimination criterion in each step, retaining variables with a significance level of p < 0.05 or clinical relevance. The impact of MetS on the probability of presenting EHOA was assessed by adjusting for variables selected in the final model. The effect of each factor was expressed through estimation of the odds ratio (OR) and its 95% confidence interval (CI). The area under the receiver operating characteristic (ROC) curve (AUC) quantified the discrimination performance of the EHOA predictive models. Sensitivity, specificity, and predictive values were reported with their 95% confidence interval.
The relationship between age and the risk of EHOA was explored using multivariable-adjusted cubic splines curves, incorporating five knots in the model.
All analyses were conducted using statistical software, specifically SPSS v.28 and R v.4.2. The “pROC” package facilitated ROC analysis, while restricted cubic splines regression was implemented with the “rms” package. Significance levels reported were two-sided, and the threshold for statistical significance was defined as p < 0.05.
Results
A total of 1039 patients had hand OA with a mean age of 62.95 years and a predominance of women (82.58%). Table 1 illustrates the characteristics of our study population. Within this cohort, 303 patients (29.16%) had EHOA.
Patients with EHOA in the PROCOAC cohort were younger (59 ± 8.17 vs. 65 ± 8.97; p < 0.001) and had a lower abdominal perimeter (93 ± 12.71 vs. 96 ± 11.98; p = 0.002) than non-EHOA patients (Table 2). AUSCAN scores were significantly higher in the EHOA group than in the non-EHOA group, including total AUSCAN (54 ± 26.92 vs. 41 ± 29.50; p < 0.001), and all its components: pain (56 ± 27.81 vs. 43 ± 31.85; p < 0.001), stiffness (54 ± 32.88 vs. 39 ± 36; p < 0.001), and function (54 ± 28.44 vs. 40 ± 30.28; p < 0.001).
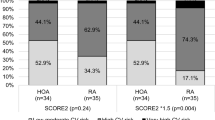
Comparing patients with EHOA to non-EHOA, there was a lower representation of hypertension (37.75 vs. 52.52%; p < 0.001), knee OA (53.26 vs. 77.5%; p < 0.001), hip OA (37.33 vs. 57.43%; p < 0.001), and thumb base OA (53.16 vs. 67.45%; p < 0.001) (Table 2). Conversely, the EHOA group exhibited a higher presence of clinically inflamed joints (26.49 vs. 5.08%; p < 0.001) and nodular hand OA (96.03 vs. 78.72%; p < 0.001).
The most significant factors independently associated with EHOA, after adjusting for variables (age, sex, hypertension, hip OA, and BMI), were the concurrent presence of inflamed joints (OR = 3.21; 95%CI 1.86–5.52; p < 0.001), nodular hand OA (OR = 7.47; 95%CI 2.89–19.31; p < 0.001), and total AUSCAN (OR = 1.01; 95%CI 1.01–1.21; p < 0.001). Additionally, the association with a lower prevalence of knee OA (OR = 0.57; 95%CI 0.35–0.92; p = 0.021) remained significant (Table 3). This EHOA predictive model exhibited an AUC of 0.75, sensitivity of 0.75, and specificity of 0.64 (Table 3 and Fig. 1).
ROC curve of EHOA predictive model without and with MetS The continuous line represents the data from the EHOA predictive model that includes the variables from Tables 3 and 4 (age at diagnosis, sex, hypertension, body mass index, knee OA, nodular hand OA, inflammatory hand OA and hip OA). This model has a sensitivity of 0.75, an specificity of 0.64, a negative predictive value of 0.83 and a positive predictive value of 0.53. The dashed line represents the model by adding metabolic syndrome (MetS). This model with MetS has a sensitivity of 0.75, an specificity of 0.63, a negative predictive value of 0.83 and a positive predictive value of 0.51.
Regarding MetS, only hypertension and abdominal circumference showed a statistically significant relationship in univariate analysis, but these results were not sustained in multivariate analysis (Table 3).
In analyzing the influence of MetS on EHOA patients by including MetS as a covariate in the proposed model, it was observed that MetS did not significantly influence the presence of EHOA, maintaining the size of the effect of other factors (Table 4). Although not an independent factor for predicting EHOA in the analyzed sample, a non-statistically significant trend of increased EHOA risk was observed among patients with MetS (OR = 1.54; 95%CI 0.89–2.68) (Table 4 and Fig. 1).
Moreover, multivariable-adjusted restricted cubic splines demonstrated a non-significant relationship between age and EHOA (p for overall trend = 0.335) (Supplementary Fig. 1). Upon thorough data review and considering a threshold at age 63 to explore a possible change in the association between age and EHOA risk and its interaction with MetS, a reversed trend was observed among those over 63, indicating a higher prevalence of MetS among EHOA (31.91 vs. 26.04%), although these differences were not significant in either group (Supplementary Table 1 and Fig. 2).
Analyzing the association between age and MetS independently revealed a significant association, with a higher prevalence of metabolic syndrome in those over 63 years of age (Fig. 2). Among patients ≥ 63 years, there were more with non-EHOA than with EHOA, while EHOA predominated among the youngest. Analyzing age and MetS within each erosive group (non-EHOA vs. EHOA) showed a significant association between age and MetS in both groups, with a greater presence of MetS among those ≥ 63 years of age (Fig. 2).
The number of erosions, total erosions on both hands, and the average number of erosions between the hands were recorded. The mean number of total erosions between both hands was around 3, both in patients with MetS and without MetS (p = 0.327) (Table 5).
Discussion
One objective of this study was to describe the Erosive Hand Osteoarthritis (EHOA) phenotype in the Spanish PROCOAC cohort. Characterizing this phenotype revealed that, compared to non-EHOA, patients with EHOA are younger, exhibit a lower presence of knee Osteoarthritis (OA), a higher concurrent presence of clinically inflamed joints, and an increased prevalence of nodular hand OA.
The heightened levels of pain, disability, and inflammation observed in EHOA patients, in addition to the increased presence of nodes and erosions, contribute to a greater clinical burden when compared to patients with non-erosive hand OA, aligning with prior findings7,22,38,39.
In our cohort, EHOA showed no association with Rheumatoid Factor (RF). These results are consistent with previous publications40,41,42, and similarly, there is no observed relationship between EHOA and Anti-Cyclic Citrullinated Peptide antibodies (ACPA), a novel finding not reported before in our knowledge. However, there are existing data about the connection between ACPA and inflammatory hand OA43.
The second aim was to elucidate the relationship between EHOA and Metabolic Syndrome (MetS). Univariate and multivariate analyses were conducted without revealing a significant association. Additionally, a predictive model for EHOA incorporating MetS showed no modification in the results. This is in concordance with findings from the Strand publication based on the Framingham cohort, which found no significant association between hand OA, including EHOA, and MetS44. Notably, there was a borderline association with hypertension, aligning with our cohort data.
The association of MetS with hand OA and EHOA has yielded conflicting results in the literature. While some studies suggest a potential link, others present contradictory evidence25,26,27. Our study adds to this complexity, emphasizing the need for further investigation. Notably, our analysis of EHOA patients did not demonstrate a significant association with MetS, consistent with findings from the Framingham cohort44.
The association of hand OA or EHOA with more generalized OA has been debated5,21,45. Our study indicates a lower prevalence of OA in other joints, specifically the knees, among patients with EHOA in the PROCOAC cohort. This could be attributed to the relatively lower age of EHOA patients and their lower BMI compared to non-EHOA patients. However, this finding contrasts with some studies showing a more common occurrence of hand OA in the context of generalized OA20. A recent publication shows the inverse associationof two loci between EHOA and knee OA46. In this paper they found 4 significant EHOA loci with high effect on EHOA risk. Two of them, rs1800801 and rs4496445 was associated with EHOA risk and with protection of knee OA. Apart from the manuscript of Haugen18 and Marshall13, to our knowledge, none of the abrove-mentioned studies focused their analysses on the erosive subtype specifically, but rahter on hand OA in general. The study of Marshall et al. consisted of the description of subsets of hand OA in a prospective cohort of 1076 older adult patients, of which only 52 (4.8%) developed the erosive phenotype and they concluded that radiographic knee OA was not increased in EHOA patients.
While the present study focuses on EHOA, the analysis of other studies is predominantly centered on hand OA in general. Future research with large, prospective cohorts is imperative to delve deeper into the association between EHOA and other joint involvements.
This study has potential limitations, including its cross-sectional approach to hand OA patients, limiting the establishment of causality. Unmeasured confounders, such as diet, alcohol consumption, or socioeconomic status, could influence the results. Nevertheless, the proportion of EHOA in our cohort, at 20.3%, exceeds the mean compared with other studies. Other studies patients.
Conclusion
Within the PROCOAC cohort, Erosive Hand Osteoarthritis (EHOA) demonstrates associations with nodular hand OA, inflammatory hand OA, and elevated total AUSCAN scores. Notably, EHOA exhibits a lower prevalence of knee Osteoarthritis (OA) in our cohort. Additionally, we observed no discernible relationship between EHOA and Metabolic Syndrome (MetS) in our study.
Data availability
The datasets used and/or analysed during the current study available from the corresponding author on reasonable request.
References
Dahaghin, S. et al. Prevalence and pattern of radiographic hand osteoarthritis and association with pain and disability (the Rotterdam study). Ann. Rheum. Dis. 64(5), 682–687 (2005).
Marshall, M., Watt, F. E., Vincent, T. L. & Dziedzic, K. Hand osteoarthritis: clinical phenotypes, molecular mechanisms and disease management. Nat. Rev. Rheumatol. 14(11), 641–656 (2018).
Blanco, F. J., Silva-Díaz, M., Quevedo Vila, V., Seoane-Mato, D., Pérez Ruiz, F., Juan-Mas, A. et al. Prevalence of symptomatic osteoarthritis in Spain: EPISER2016 study. Reumatol. Clin. 28, S1699–258X(20)30023-1 (2020).
Pereira, D. et al. The effect of osteoarthritis definition on prevalence and incidence estimates: A systematic review. Osteoarthr. Cartil. 19(11), 1270–1285 (2011).
Zhang, W. et al. EULAR evidence-based recommendations for the diagnosis of hand osteoarthritis: report of a task force of ESCISIT. Ann. Rheum. Dis. 68(1), 8–17 (2009).
Zhang, Y. et al. Prevalence of symptomatic hand osteoarthritis and its impact on functional status among the elderly: The Framingham study. Am. J. Epidemiol. 156(11), 1021–1027 (2002).
Kwok, W. Y., Vliet Vlieland, T. P. M., Rosendaal, F. R., Huizinga, T. W. J. & Kloppenburg, M. Limitations in daily activities are the major determinant of reduced health-related quality of life in patients with hand osteoarthritis. Ann. Rheum. Dis. 70(2), 334–336 (2011).
Tenti, S. et al. Impact of thumb osteoarthritis on pain, function, and quality of life: A comparative study between erosive and non-erosive hand osteoarthritis. Clin. Rheumatol. 39(7), 2195–2206 (2020).
Addimanda, O. et al. Clinical associations in patients with hand osteoarthritis. Scand. J. Rheumatol. 41(4), 310–313 (2012).
Addimanda, O. et al. Clinical and radiographic distribution of structural damage in erosive and nonerosive hand osteoarthritis. Arthritis Care Res. (Hoboken) 64(7), 1046–1053 (2012).
Peter, J. B., Pearson, C. M. & Marmor, L. Erosive osteoarthritis of the hands. Arthritis Rheum. junio de 9(3), 365–388 (1966).
Crain, D. C. Interphalangeal osteoarthritis. JAMA 175, 1049–1053 (1961).
Marshall, M. et al. Subsets of symptomatic hand osteoarthritis in community-dwelling older adults in the United Kingdom: Prevalence, inter-relationships, risk factor profiles and clinical characteristics at baseline and 3-years. Osteoarthr. Cartil. 21(11), 1674–1684 (2013).
Gazeley, D. J., Yeturi, S., Patel, P. J. & Rosenthal, A. K. Erosive osteoarthritis: A systematic analysis of definitions used in the literature. Semin. Arthritis Rheum. 46(4), 395–403 (2017).
Haugen, I. K. et al. Prevalence, incidence and progression of hand osteoarthritis in the general population: The Framingham Osteoarthritis Study. Ann. Rheum. Dis. 70(9), 1581–1586 (2011).
Ramonda, R., Frallonardo, P., Musacchio, E., Vio, S. & Punzi, L. Joint and bone assessment in hand osteoarthritis. Clin. Rheumatol. 33(1), 11–19 (2014).
Neuprez, A. et al. Aesthetic discomfort in hand osteoarthritis: Results from the LIège Hand Osteoarthritis Cohort (LIHOC). Arthritis Res. Ther. 17, 346 (2015).
Haugen, I. K. et al. Synovitis and radiographic progression in non-erosive and erosive hand osteoarthritis: Is erosive hand osteoarthritis a separate inflammatory phenotype?. Osteoarthritis Cartil. 24(4), 647–654 (2016).
Felson, D. T. et al. Risk factors for incident radiographic knee osteoarthritis in the elderly: The Framingham Study. Arthritis Rheum. 40(4), 728–733 (1997).
Dahaghin, S. et al. Does hand osteoarthritis predict future hip or knee osteoarthritis?. Arthritis Rheumatol. 52(11), 3520–3527 (2005).
Addimanda, O. et al. Clinical associations in patients with hand osteoarthritis. Scand. J. Rheumatol. 41(4), 310–313 (2012).
Haugen, I. K. et al. The association between erosive hand osteoarthritis and subchondral bone attrition of the knee: The Framingham Osteoarthritis Study. Ann. Rheum. Dis. 71(10), 1698–1701 (2012).
Visser, A. W. et al. The relative contribution of mechanical stress and systemic processes in different types of osteoarthritis: The NEO study. Ann. Rheum. Dis. 74(10), 1842–1847 (2015).
Dahaghin, S., Bierma-Zeinstra, S. M. A., Koes, B. W., Hazes, J. M. W. & Pols, Ha. P. Do metabolic factors add to the effect of overweight on hand osteoarthritis? The Rotterdam Study. Ann. Rheum. Dis. 66(7), 916–920 (2007).
Marshall, M. et al. Erosive osteoarthritis: A more severe form of radiographic hand osteoarthritis rather than a distinct entity?. Ann. Rheum. Dis. 74(1), 136–141 (2015).
Sanchez-Santos, M. T. et al. Association of metabolic syndrome with knee and hand osteoarthritis: A community-based study of women. Semin. Arthritis Rheum. 48(5), 791–798 (2019).
Marshall, M. et al. Metabolic risk factors and the incidence and progression of radiographic hand osteoarthritis: A population-based cohort study. Scand. J. Rheumatol. 48(1), 52–63 (2019).
Oreiro-Villar, N. et al. PROCOAC (PROspective COhort of A Coruña) description: Spanish prospective cohort to study osteoarthritis. Reumatol. Clin. 18(2), 100–104 (2022).
Altman, R. D. Criteria for classification of clinical osteoarthritis. J. Rheumatol. Suppl. 27, 10–12 (1991).
Altman, R. et al. The American College of Rheumatology criteria for the classification and reporting of osteoarthritis of the hand. Arthritis Rheum. 33(11), 1601–1610 (1990).
Verbruggen, G., Wittoek, R., Vander Cruyssen, B. & Elewaut, D. Morbid anatomy of «erosive osteoarthritis» of the interphalangeal finger joints: An optimised scoring system to monitor disease progression in affected joints. Ann. Rheum. Dis. 69(5), 862–867 (2010).
Williams, B. et al. 2018 ESC/ESH guidelines for the management of arterial hypertension: The task force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension. J. Hypertens. 36(10), 1953 (2018).
American Diabetes Association. 2. Classification and diagnosis of diabetes: Standards of medical care in diabetes-2021. Diabetes Care 44(Suppl 1), S15–S33 (2021).
Naranjo Hernández, A. et al. Recommendations by the Spanish Society of rheumatology on osteoporosis. Reumatol. Clin. (Engl. Ed.) 15(4), 188–210 (2019).
Rosas Guzmán, J., González Chávez, A., Aschner, P. & Bastarrachea, R. Consenso latinoamericano de la Asociación Latinoamericana de Diabetes (ALAD): Epidemiología, diagnóstico, control, prevención y tratamiento del síndrome metabólico en adultos. Rev. ALAD 18(1), 25–42 (2010).
Allen, K. D., Jordan, J. M., Renner, J. B. & Kraus, V. B. Validity, factor structure, and clinical relevance of the AUSCAN Osteoarthritis Hand Index. Arthritis Rheum. 54(2), 551–556 (2006).
Validación en español: Arreguín Reyes, R., López López, C. O., Alvarez Hernández, E., & Medrano Ramírez, G., Montes Castillo, M. D. la L, Vázquez-Mellado, J. Evaluation of hand function in rheumatic disease. Validation and usefulness of the Spanish version AUSCAN, m-SACRAH and Cochin questionnaires. Reumatol. Clin. 8(5), 250–254 (2012).
Vlychou, M., Koutroumpas, A., Alexiou, I., Fezoulidis, I. & Li, S. High-resolution ultrasonography and 3.0 T magnetic resonance imaging in erosive and nodal hand osteoarthritis: High frequency of erosions in nodal osteoarthritis. Clin. Rheumatol. 32(6), 755–762 (2013).
Bijsterbosch, J. et al. Clinical burden of erosive hand osteoarthritis and its relationship to nodes. Ann. Rheum. Dis. 69(10), 1784–1788 (2010).
Guidelli, G. M., Morozzi, G., Simpatico, A. & Fioravanti, A. Rheumatoid factor isotypes in patients with erosive osteoarthritis of the hand. Int. J. Rheum. Dis. 14(3), e49-50 (2011).
van Delft, M. A. M., van Beest, S., Kloppenburg, M., Trouw, L. A. & Ioan-Facsinay, A. Presence of autoantibodies in erosive hand osteoarthritis and association with clinical presentation. J. Rheumatol. 46(1), 101–105 (2019).
Lechtenboehmer, C. A., Jaeger, V. K., Kyburz, D., Walker, U. A. & Hügle, T. Brief report: Influence of disease activity in rheumatoid arthritis on radiographic progression of concomitant interphalangeal joint osteoarthritis. Arthritis Rheumatol. Hoboken NJ 71(1), 43–49 (2019).
Vannini, A. et al. A seroimmunological profile of erosive hand osteoarthritis. Acta Reumatol. Port 38(1), 39–43 (2013).
Strand, M. P., Neogi, T., Niu, J., Felson, D. T. & Haugen, I. K. Association between metabolic syndrome and radiographic hand osteoarthritis: Data from a community-based longitudinal cohort study. Arthritis Care Res. 70(3), 469–474 (2018).
Cicuttini, F. M., Baker, J., Hart, D. J. & Spector, T. D. Relation between Heberden’s nodes and distal interphalangeal joint osteophytes and their role as markers of generalised disease. Ann. Rheum. Dis. 57(4), 246–248 (1998).
Styrkarsdottir, U. et al. Meta-analysis of erosive hand osteoarthritis identifies four common variants that associate with relatively large effect. Ann. Rheum. Dis. 82, 873–880 (2023).
Acknowledgements
This study was supported by grants (PI19/01206, PI20/00614, PMP22/00101, PMPTA22/00115, RICORS RD21/0002/0009 and PRB3-ISCIII-PT17/0019/0014) from Fondo de Investigacion Sanitaria, ISCIII-General Subdirection of Assessment and Promotion of Research-European Regional Development Fund (FEDER) “A way of making Europe”/“Investing in your future”. This work was also funded by Grant IN607A 2021/07 from Axencia Galega de Innovación-Xunta de Galicia. The Biomedical Research Networking Center (CIBER) is an initiative from the Instituto de Salud Carlos III (ISCIII). The authors further acknowledge AE CICA-INIBIC (ED431E 2018/03). MSD is supported by Contrato Rio Hortega Fondo de Investigacion Sanitaria (CM17/00101).
Author information
Authors and Affiliations
Contributions
N.O., F.J.B. and I.R.P. contributed equally in the design and coordination of the study; they conceived the study, participated in its design and helped to draft the final version of the manuscript; N.O. is the main responsible for the collection of the different variables of the PROCOAC cohort and the medical supervisor of patients included in the cohort; M.S.D. and A.R.S. was involved in the supervision of patients and in the collection of clinical data; C.T.A. coordinated informatics support where all the variables of the cohort are recorded; S.P. and V.B.B. supervised the statistical analyses. All the authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Silva-Díaz, M., Pértega-Díaz, S., Balboa-Barreiro, V. et al. Metabolic syndrome is not associated with erosive hand osteoarthritis: a cross-sectional study using data from the PROCOAC cohort. Sci Rep 14, 5968 (2024). https://doi.org/10.1038/s41598-024-55374-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-55374-1
- Springer Nature Limited