Abstract
The potential association between endurance exercise and myocardial fibrosis is controversial. Data on exercise exposure and diffuse myocardial fibrosis in endurance athletes are scarce and conflicting. We aimed to investigate the association between exercise exposure and markers of diffuse myocardial fibrosis by cardiovascular magnetic resonance imaging (CMR) in endurance athletes. We examined 27 healthy adult male competitive endurance athletes aged 41 ± 9 years and 16 healthy controls in a cross sectional study using 3 Tesla CMR including late gadolinium enhancement and T1 mapping. Athletes reported detailed exercise history from 12 years of age. Left ventricular total mass, cellular mass and extracellular mass were higher in athletes than controls (86 vs. 58 g/m2, 67 vs. 44 g/m2 and 19 vs. 13 g/m2, all p < 0.01). Extracellular volume (ECV) was lower (21.5% vs. 23.8%, p = 0.03) and native T1 time was shorter (1214 ms vs. 1268 ms, p < 0.01) in the athletes. Increasing exercise dose was independently associated with shorter native T1 time (regression coefficient − 24.1, p < 0.05), but expressed no association with ECV. Our results indicate that diffuse myocardial fibrosis has a low prevalence in healthy male endurance athletes and do not indicate an adverse dose–response relationship between exercise and diffuse myocardial fibrosis in healthy athletes.
Similar content being viewed by others
Introduction
The beneficial effect of physical activity on reducing mortality and risk of cardiovascular disease in the general population is well established1,2,3. There is a non-linear dose–response relationship between physical activity and cardiovascular health. The most sedentary individuals benefit the most from increasing exercise doses3. However, the long-term cardiac consequences of elite athleticism and exercise doses far beyond what is recommended for the improvement and sustainment of cardiovascular health is still a matter of controversy.
It has long been hypothesized that excessive physical activity can induce permanent cardiac damage even in normal hearts4. Under this hypothesis, elite athleticism may in itself cause cardiac disease and not merely trigger arrhythmia in susceptible individuals. Consequent myocardial fibrosis can be either focal or diffuse, which can be visualised by cardiovascular magnetic resonance (CMR) imaging as areas of late gadolinium enhancement (LGE) and by T1 mapping as increased native T1 time and extracellular volume (ECV), respectively5,6. Both of these conditions have been found to be associated with increased mortality in clinical populations7,8. However, in athletes the prognostic significance of LGE is largely unknown9,10,11,12, whereas for T1 mapping it is uncharted.
Athletes have been found to have increased prevalence of LGE on CMR12,13, but not in general to be at increased risk of diffuse myocardial fibrosis13,14,15,16,17,18,19,20. However, studies utilizing CMR T1 mapping in athletes with LGE have been ambiguous, but may indicate a global myocardial involvement14,15,19,21. Further, there may be a dose–response relationship between exercise exposure and LGE in athletes12,19,22,23,24. However, no previous study has comprehensively investigated the association between exercise dose and diffuse myocardial fibrosis in athletes, and the limited available data on exercise history and its association with diffuse myocardial fibrosis in athletes are scarce and conflicting18,25.
In light of these observations, we hypothesize that there might be an association between exercise dose and diffuse myocardial fibrosis in athletes. The aim of this study was therefore to investigate the potential association between exercise and markers of diffuse myocardial fibrosis by CMR in healthy male competitive endurance athletes.
Methods
Study population
From April 2016 to February 2017, we enrolled 27 adult male competitive endurance athletes from Olympic to elite master level. Athletes were defined as subjects engaged in regular exercise training and participating in official sports competition7, and were required to have exercised for > six consecutive years at doses equivalent to > 24 metabolic equivalents of task (MET)-hours per week; three times the minimum recommended exercise dose for healthy adults7,26. The athletes were healthy and reported no use of prescription drugs or performance enhancing substances. We recruited 16 healthy male controls from adult blood donors aged 16 to 65 years. Disregarding the requirements of athletic status and history, controls were subject to the same inclusion and exclusion criteria as their athletic peers. We excluded individuals with signs or symptoms of cardiovascular disease, significant valvular or congenital heart disease, current comorbidity potentially influencing cardiovascular performance, antihypertensive or other vasoactive drug therapy or a body mass index (BMI) ≥ 30 kg/m2. The study complied with the Declaration of Helsinki and was approved by the Regional Committee for Medical and Health Research Ethics in South-Eastern Norway. All study participants gave written informed consent.
Exercise history
All the athletes underwent a structured interview based on a questionnaire (Supplementary Fig. S1 and S2), retrospectively assessing their exercise history from 12 years of age. We defined exercise as any planned or structured action with the objective of improving or maintaining physical fitness or health3. Exercise duration was defined as the actual time in motion and accumulated exercise duration calculated as the sum of weekly durations from reported time intervals. Exercise intensity was estimated using the 2011 Compendium of Physical Activities and reported as MET defined as the ratio of the work metabolic rate to a standard resting metabolic rate of 1 kcal/kg/hour26. Exercise dose expressed as MET-hours was calculated as the product of exercise duration and exercise intensity3.
CMR protocol
All subjects underwent CMR on the same 3 Tesla MRI scanner (Philips Ingenia; Philips Healthcare, Best, The Netherlands). ECG-gated balanced steady state free precession (bSSFP) two-dimensional cine sequences during breath hold were performed in standardized long axis and multiple short axis projections covering both ventricles from base to apex for anatomical and functional evaluation.
T1 mapping was performed in accordance with current recommendations6, using 5(3)3 modified Look-Locker Inversion recovery (MOLLI) sequences for native and 4(1)3(1)2 MOLLI sequences for post-contrast images.
A gadolinium-based contrast agent (0.2 mmol/kg of Dotarem/gadoterate meglumine; Guerbet, Aulnay-sous-Bois, France) was injected to perform post-contrast T1 mapping and evaluate the presence of LGE in the steady state ≥ 10 min after contrast administration.
CMR analysis
Analyses of ventricular volumes and mass were performed semi-automatically and according to current recommendations27 in the bSSFP cine sequences using Segment version 3.2 R853128. The endocardial and epicardial borders were delineated in end-diastole and end-systole for volumetric assessment paying careful attention to systolic basal descent of the atrioventricular plane. A constant of 1.05 g/ml for myocardial tissue was applied for calculation of left ventricular mass (LVM)27. Papillary muscles were excluded from the analysis of LVM to allow for comparison with reference values of left ventricular volumes and mass specific for endurance athletes29. Left ventricular cellular mass (CM) and extracellular mass (ECM) were calculated by Eqs. (1) and (2)20 as
T1 mapping was performed in Sectra Picture Archive and Communication System (PACS) (Sectra AB, Linköping, Sweden), excluding any areas of LGE. ECV was calculated by Eq. (3)30 as
EVF was derived from blood sampling in all but one athlete where synthetic ECV31 was reported with EVF derived from blood T1 due to missing data. Blood sampling was standardized and performed prior to the CMR examination after a minimum of 15 min of rest in a seated position, predominantly from the right antecubital vein.
Visual assessment of LGE was performed in Sectra PACS and semi-automatically quantified using Segment. The distribution of LGE was classified as insertion point, non-ischemic and ischemic.
Statistics
Descriptive statistics are reported as mean ± standard deviation for parametric data, median (interquartile range, IQR) for non-parametric data and numbers (%) for categorical data. Between-group differences for continuous variables were evaluated using independent samples T-test for parametric data and independent samples Mann–Whitney U Test for non-parametric data. Pearson Chi-Square test was applied to compare distributions of categorical outcomes, whereas Fisher’s Exact test was applied in the case of low expected frequencies (< 5) in the analyses. The upper limit of normality (ULN) of the T1 mapping results were defined as the mean plus two standard deviations of the normal data6. Bivariate associations between exercise history data and CMR results were analysed calculating Spearman’s correlation coefficients due to non-parametric distribution of data. We used linear regression to assess associations between exercise history and CMR data, and adjusted for age, EVF and the presence of LGE in multivariable models. Accumulated exercise dose was chosen from the exercise metrics for the regression model as it expressed the strongest correlations to myocardial structure in univariable analysis and was the most comprehensive of the exercise metrics encompassing both duration and intensity over time. Assumptions of the linear regression models were investigated by histograms and normality plots of standardized residuals and plots of residuals versus fitted values. Associations between exercise history and the presence of LGE were assessed by logistic regression. Non-parametric distribution of the data were handled by logarithmic transformation or non-parametric tests. A two-tailed p-value of < 0.05 was considered statistically significant. Intra and inter observer variability was assessed by analysing intraclass correlation coefficients for native T1 time and ECV of ten study participants. Statistical analyses were performed using STATA version 17.
Results
Baseline characteristics and exercise history
Twenty-seven male athletes and 16 male controls were included in the study. The athletes had performed a median of 367 h of exercise per year since 12 years of age (Table 1). Five of the athletes were unwilling to detail their exercise history further than reporting average exercise intensity. The athletes had lower resting heart rate than the controls. There were no differences in body size or blood pressure between the groups. Importantly, there was no between-group difference in EVF, which is used for ECV calculation in the T1 mapping. The athletes had higher values of Troponin T than controls, where three of the athletes compared to none of the controls had values exceeding the upper reference limit of ≤ 14 ng/L for Troponin T (15, 22 and 80 ng/L).
Cardiovascular magnetic resonance imaging
All athletes and controls underwent CMR. One athlete was excluded from the LGE and ECV calculations due to lack of contrast administration and two athletes were excluded from the T1 mapping analysis due to inadequate image quality in the T1 sequences and biologically implausible native T1 time (889 ms).
Cardiac dimensions and function
The left and right ventricles were larger in the athletes than the controls (Table 2). Seven (26%) of the athletes and none of the controls exceeded the ULN for endurance athletes of indexed left and right ventricular end-diastolic volumes (ULN 121 ml/m2 and 126 ml/m2 respectively, p = 0.04 for difference). Total left ventricular mass was higher in athletes than controls. Twenty-five (93%) of the athletes and one (6%) of the controls had LVM exceeding the ULN for endurance athletes (ULN 76 g/m2, p < 0.01 for difference). Both cellular and extracellular mass were higher in the athletes than in the controls, and the ratio of cellular to extracellular mass was also higher in the athletes.
Late gadolinium enhancement
LGE was observed in five (19%) of the athletes and in none of the controls. The LGE was localized in the right ventricular posterior septal insertion point in two (8%) athletes and as non-ischemic LGE in three (12%) athletes. None of the athletes or controls had LGE suggestive of ischemia. The median extent of LGE in those five athletes was 1.0 (0.7–4.0) % of left ventricular mass. There were no clear differences between athletes with or without LGE in regards to exercise history, and no association between exercise history and presence of LGE (Table 3).
T1 mapping
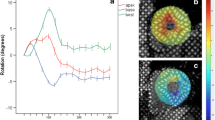
Athletes had shorter native T1 time (1214 ms vs. 1268 ms, p < 0.01) and lower ECV (21.5% vs. 23.8%, p = 0.03) in the interventricular septum than controls (Fig. 1). There were no differences in native T1 time (1220 ms vs. 1212 ms, p = 0.56) or ECV (21.5% vs. 21.5%, p = 0.89) between athletes with or without LGE. Native T1 mapping in athletes with and without LGE and a healthy control is illustrated in Fig. 2.
LGE and native T1 mapping by CMR. Illustrations of athletes with (A) non-ischemic LGE and (B) LGE in the right ventricular inferior insertion point. (C) Native T1 mapping with regions of interest. Colour-coded native T1 maps in (D) LGE-negative athlete: native T1 time 1204 ms, ECV 20.4%, (E) LGE-positive athlete: native T1-time 1222 ms, ECV 21.0% and (F) healthy control: native T1 time 1259 ms, ECV 24.3%. ECV extracellular volume, LGE late gadolinium enhancement.
None of the athletes had abnormally long native T1 times, whereas two of the athletes had ECV exceeding the ULN (29.6% and 31.4% vs. 27.8%). Besides older age (49 and 47 years vs. mean 41 years), there were no clear differences between these two and the other athletes regarding baseline characteristics, exercise history or cardiac dimensions and function. None of the two athletes displayed LGE.
Exercise history, T1 mapping and left ventricular mass
Accumulated, but not average, exercise duration and dose were negatively associated with the native T1 time (Table 4). None of the parameters of exercise exposure were associated with ECV. All parameters of exercise exposure were associated with left ventricular total mass and left ventricular cellular mass, but there was no association between exercise exposure and left ventricular extracellular mass. The associations between accumulated exercise dose and native T1 time, ECV and left ventricular cellular and extracellular mass are demonstrated in Fig. 3.
The accumulated exercise dose was negatively associated with the native T1 time and positively associated with all three parameters of left ventricular mass, but not ECV (Table 5). In multivariable analysis adjusting for age, EVF and presence of LGE, accumulated exercise dose remained negatively associated with the native T1 time and positively associated with total left ventricular mass and left ventricular cellular mass. There was no independent association between accumulated exercise dose and left ventricular extracellular mass.
Reproducibility
Intraclass correlation coefficients (ICC) for native T1 time and ECV were excellent for both intra and inter observer reanalysis at 0.95 and 0.99 for native T1 time and 0.99 and 0.98 for ECV, respectively (all p < 0.01).
Discussion
Our study is the first to examine the association between exercise dose and diffuse myocardial fibrosis in endurance athletes using CMR. We did not find an association between athleticism and diffuse myocardial fibrosis. Our main results were: (1) athletes had shorter native T1 time and lower ECV than controls; (2) there were moderate inverse associations between exercise exposure and native T1 time, and there was no association between exercise exposure and ECV. Our results seem therefore to oppose the hypothesis of exercise-induced diffuse myocardial fibrosis.
Native T1 time and ECV
Our observation of shorter native T1 time and lower ECV in athletes than controls is in contrast to what one would expect in exercise-induced diffuse myocardial fibrosis. These results indicate that diffuse myocardial fibrosis is not prevalent in healthy male endurance athletes despite a high cumulative exercise load and a marked expression of the athlete’s heart phenotype. Our results are in line with most previous CMR studies on endurance athletes, showing either neutral results or shorter native T1 time and lower ECV in athletes14,15,16,17,19,20. ECV is a validated surrogate marker of diffuse myocardial fibrosis in the absence of infiltration or acute myocardial injury, and ECV is strongly related with histology6,32. Higher ECV is associated with adverse outcomes in clinical populations8, but the prognostic value of T1 mapping in athletes is unknown.
Left ventricular mass
The athletes had higher left ventricular mass than controls. As expected, cellular as well as extracellular mass was enlarged. However, the cellular enlargement was more pronounced than the extracellular enlargement which is reflected in the lower ECV observed in the athletes in our study. This suggests that the physiologic remodelling of the left ventricular myocardium observed in the athlete’s heart is predominantly caused by myocyte hypertrophy with a relatively smaller increase of the interstitium. Our results are in apparent contrast to what one would expect in diffuse myocardial fibrosis and in line with previous CMR studies on athletic remodelling observing greater relative expansion of the cellular than the extracellular compartment of the myocardium in athletes16,20.
Our results also show that athletes with the largest exercise exposure have the greatest degree of athletic remodelling. This is in line with the current perception of the athlete’s heart33. However, only the left ventricular cellular mass was independently associated with exercise exposure, whereas the extracellular mass was not. These results might indicate that there is a dose–response relationship between exercise exposure and left ventricular cellular expansion in healthy male endurance athletes, but probably not for extracellular mass.
In summary, our observations regarding exercise exposure and myocardial remodelling indicate that the enlargement of left ventricular mass in the athlete’s heart phenotype is accompanied by a relative expansion of the cellular compartment of the myocardium with a dose–response relationship between exercise exposure and myocyte hypertrophy.
Exercise exposure and diffuse myocardial fibrosis
Exercise exposure was independently associated with shorter native T1 time and expressed no association with ECV in our athletes. This contradicts the supposition that exercise may cause diffuse myocardial fibrosis, where native T1 time and ECV would be expected to increase6. Existing data on the association between exercise exposure and diffuse myocardial fibrosis in athletes are scarce and limited to one study on veteran runners which found no association between total training volume and ECV18 and another study on a mixed group of athletes which found a possible association between increasing number of exercise years and longer native T1 time25. Whereas the former is in line with results from our study, our results contradict the latter. However, no previous study has comprehensively evaluated the association between exercise dose and diffuse myocardial fibrosis in endurance athletes using CMR. Our results indicate that exercise is not adversely associated with diffuse myocardial fibrosis.
Late gadolinium enhancement
We observed LGE in five of the athletes in our study and in none of the controls. Several studies14,15,16,17,18,19,21,22,23,24,34,35 and reviews9,12,13 have addressed the issue of LGE in athletes during the recent years reporting prevalences ranging from 0 to 50% in often small-scale studies. Hence, the prevalence of LGE in the athletes in our study is comparable to previous literature. Multiple pathophysiological mechanisms for this have been proposed, including genetic predisposition, pulmonary artery pressure and/or volume overload, myocarditis, myocardial ischemia and repetitive microdamage from repeated bouts of intense exercise12,13,14,15,17,18,19,21,22,23,34. The LGE has typically been distributed in three different patterns: right ventricular septal insertion point, non-ischemic and ischemic13. Some studies have suggested a dose–response relationship between exercise exposure and LGE in athletes12,19,22,23,24, but the evidence is conflicting14,18,21,35. We found no difference in exercise history between athletes with or without LGE, nor any association between exercise history and presence of LGE. However, as the study sample was quite small and the number of athletes with LGE limited, these neutral results should be interpreted with caution as further evaluation of the potential association between exercise metrics and LGE was beyond the scope of this study to address.
Interestingly, there were no differences in native T1 time or ECV between athletes with or without LGE in our study sample, suggesting that LGE in these athletes was not associated with diffuse fibrosis. There have been conflicting results in the literature regarding this issue where some studies have reported higher ECV in the remote myocardium of athletes with LGE19,21, whereas other studies have found no such results14,15.
The presence of LGE is a risk factor for adverse outcomes in clinical populations, but the prognostic implication of non-specific LGE in asymptomatic athletes is uncertain9,10,11,12. However, it may not be a benign phenomenon as higher prevalence of LGE have been found in athletes evaluated for ventricular arrhythmia compared to asymptomatic athletic controls36,37 with reduced survival free from major arrhythmic events in LGE positive athletes37. In pathology studies, myocardial fibrosis has been attributed to up to 6% of cases of sudden cardiac death (SCD) in athletes38.
Athlete’s heart
The athletes in our study displayed the characteristic phenotype of the athlete’s heart39 including symmetrical chamber enlargement, enlarged left ventricular mass and lower resting heart rate compared to controls. The extent of remodelling may be exemplified by 26% of athletes exceeding the upper reference values for ventricular volumes and 93% for left ventricular mass when comparing with proposed reference values for dimensional parameters in the male athlete’s heart29. As a profound degree of remodelling would be expected in these athletes considering their participation in competitive endurance sports at Olympic to elite master level, we perceive this extent of remodelling as an indicator of the sample being representative of the study population.
Troponin T
Baseline characteristics in athletes and controls excluding athletic traits were well matched. We did, however, observe a difference in Troponin T values between athletes and controls where three of the athletes and none of the controls exceeded the upper reference limit for Troponin T for our laboratory. Athletes were not detrained prior to investigation, hence this could be explained by post exercise rise in cardiac troponins which is a well-known and usually physiologic phenomenon40. Although being asymptomatic, this could alternatively be a sign of sub-clinical cardiac disease in these athletes. The cross-sectional study design with no follow-up data did not permit further investigation of this observation.
Strengths and limitations
This was a single-centre, observational study with inherent limitations in regards to external validity and causal inference. We recruited healthy athletes only, which may have introduced a selection bias. Our study sample is small, leaving the results prone to type-2 error. Also, we evaluated male athletes only. Our results therefore do not necessarily translate to women.
Exercise exposure was evaluated retrospectively as this was considered to be the most feasible option. All the athletes were actively competitive, well-performing and asymptomatic, and we do not consider the exercise history data of the athletes to be subject to bias beside the inherent potential of inaccuracy of retrospective data. We did not evaluate the exercise history of the control group as this would have introduced a potential for recall bias in the data due to expected differences in interest regarding exercise and sports between the groups. The lack of exercise history for the control group could have introduced a risk of type-2 error if the controls also had been exercising extensively. However, the groups were distinctively different based on the profound expression of the athlete’s heart phenotype in the athlete group; hence there is no evidence suggestive of this in our data.
We performed cardiac magnetic resonance imaging using a 3 Tesla MRI scanner. Whereas T1 and ECV mapping are typically performed either at 1.5 Tesla or 3 Tesla scanners6, parametric mapping results obtained by different magnetic field strengths cannot be used interchangeably41. Higher magnetic field strength in 3 Tesla CMR results in a higher signal-to-noise ratio, contrast-to-noise ratio and may improve the precision of T1 mapping, but has disadvantages due to increased energy deposits and potential loss of homogeneity in the magnetic and radiofrequency fields41. There is no clear preference of either field strength in CMR T1 mapping41, but the choice of field strength will directly affect the absolute values of the parametric mapping results as the T1 relaxation time increases with increasing strength of the magnetic field41. However, this is only one amongst several sources of variability in CMR parametric mapping as relaxation times also vary between vendors, scanners and pulse sequences6. Hence, the methodology of CMR T1 mapping is highly dependent on the application of local reference values in the interpretation of results and participants being examined under similar conditions6,41, which we have taken great care to optimize in this study.
Correct ECV calculation is dependent on standardization of blood sampling for EVF determination to reduce variability. We ensured that blood sampling of all study participants were performed under similar conditions to avoid bias. Potential random variability might have been further reduced by performing blood sampling in the supine position as postural changes in EVF may influence ECV calculation42.
Finally, we have only performed T1 mapping of the left ventricular myocardium, whereas it is the right ventricle which experiences the greatest acute impact of exercise and displays the greatest degree of chronic remodelling in response to exercise22,43. Parametric mapping of the right ventricular myocardium is inherently challenging due to the thin wall of the right ventricle and risk of partial volume effects5.
We would encourage future studies on athletic remodelling and myocardial fibrosis to report exercise dose to further increase the external validity of our results across different CMR conditions and study samples, as well taking potential gender differences into account. There is also a need for prospective data on the long-term consequences of exercise-induced cardiac remodelling which was beyond the scope of this study to address, but which might be remedied by the ongoing multicentre Pro@Heart cohort study44.
Conclusions
Healthy male endurance athletes had lower ECV and shorter native T1 time than controls despite high cumulative exercise exposure and a profound expression of the athlete’s heart phenotype. Our results indicate that diffuse myocardial fibrosis is not prevalent in healthy male endurance athletes. Moreover, we did not find any adverse dose–response relationship between exercise exposure and diffuse myocardial fibrosis. These results offer reassurance and might help mitigate concerns regarding the potential for adverse long-term cardiac remodelling of elite athleticism in healthy athletes.
Data availability
The data underlying this article cannot be shared publicly due to the privacy of individuals that participated in the study. The data will be shared on reasonable request to the corresponding author.
References
Eijsvogels, T. M., Molossi, S., Lee, D. C., Emery, M. S. & Thompson, P. D. Exercise at the extremes: The amount of exercise to reduce cardiovascular events. J. Am. Coll. Cardiol. 67, 316–329. https://doi.org/10.1016/j.jacc.2015.11.034 (2016).
Shiroma, E. J. & Lee, I. M. Physical activity and cardiovascular health: Lessons learned from epidemiological studies across age, gender, and race/ethnicity. Circulation 122, 743–752. https://doi.org/10.1161/circulationaha.109.914721 (2010).
Wasfy, M. M. & Baggish, A. L. Exercise dose in clinical practice. Circulation 133, 2297–2313. https://doi.org/10.1161/circulationaha.116.018093 (2016).
Rowe, W. J. Extraordinary unremitting endurance exercise and permanent injury to normal heart. Lancet 340, 712–714. https://doi.org/10.1016/0140-6736(92)92243-9 (1992).
Kellman, P. & Hansen, M. S. T1-mapping in the heart: accuracy and precision. J. Cardiovasc. Magn. Reson. 16, 2. https://doi.org/10.1186/1532-429x-16-2 (2014).
Messroghli, D. R. et al. Clinical recommendations for cardiovascular magnetic resonance mapping of T1, T2, T2* and extracellular volume: A consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) endorsed by the European Association for Cardiovascular Imaging (EACVI). J. Cardiovasc. Magn. Reson. 19, 75. https://doi.org/10.1186/s12968-017-0389-8 (2017).
Pelliccia, A. et al. 2020 ESC Guidelines on sports cardiology and exercise in patients with cardiovascular disease: The Task Force on sports cardiology and exercise in patients with cardiovascular disease of the European Society of Cardiology (ESC). Eur. Heart J. 42, 17–96. https://doi.org/10.1093/eurheartj/ehaa605%JEuropeanHeartJournal (2020).
Schelbert, E. B. et al. Myocardial fibrosis quantified by extracellular volume is associated with subsequent hospitalization for heart failure, death, or both across the spectrum of ejection fraction and heart failure stage. J. Am. Heart Assoc. 4, e002613. https://doi.org/10.1161/jaha.115.002613 (2015).
Androulakis, E. & Swoboda, P. P. The role of cardiovascular magnetic resonance in sports cardiology; current utility and future perspectives. Curr. Treat. Options Cardiovasc. Med. 20, 86. https://doi.org/10.1007/s11936-018-0679-y (2018).
La Gerche, A. et al. Cardiac imaging and stress testing asymptomatic athletes to identify those at risk of sudden cardiac death. JACC Cardiovasc. Imaging 6, 993–1007. https://doi.org/10.1016/j.jcmg.2013.06.003 (2013).
Maestrini, V., Torlasco, C., Hughes, R. & Moon, J. C. Cardiovascular magnetic resonance and sport cardiology: A growing role in clinical dilemmas. J. Cardiovasc. Transl. Res. 13, 296–305. https://doi.org/10.1007/s12265-020-10022-7 (2020).
van de Schoor, F. R. et al. Myocardial fibrosis in athletes. Mayo Clin. Proc. 91, 1617–1631. https://doi.org/10.1016/j.mayocp.2016.07.012 (2016).
Małek, ŁA. & Bucciarelli-Ducci, C. Myocardial fibrosis in athletes-current perspective. Clin. Cardiol. 43, 882–888. https://doi.org/10.1002/clc.23360 (2020).
Banks, L. et al. Left ventricular fibrosis in middle-age athletes and physically active adults. Med. Sci. Sports Exerc. 52, 2500–2507. https://doi.org/10.1249/mss.0000000000002411 (2020).
Małek, ŁA. et al. Cardiovascular magnetic resonance with parametric mapping in long-term ultra-marathon runners. Eur. J. Radiol. 117, 89–94. https://doi.org/10.1016/j.ejrad.2019.06.001 (2019).
McDiarmid, A. K. et al. Athletic cardiac adaptation in males is a consequence of elevated myocyte mass. Circ. Cardiovasc. Imaging 9, e003579. https://doi.org/10.1161/circimaging.115.003579 (2016).
Mordi, I., Carrick, D., Bezerra, H. & Tzemos, N. T1 and T2 mapping for early diagnosis of dilated non-ischaemic cardiomyopathy in middle-aged patients and differentiation from normal physiological adaptation. Eur. Heart J. Cardiovasc. Imaging 17, 797–803. https://doi.org/10.1093/ehjci/jev216 (2016).
Pujadas, S. et al. Myocardial remodelling and tissue characterisation by cardiovascular magnetic resonance (CMR) in endurance athletes. BMJ Open Sport Exerc. Med. 4, e000422. https://doi.org/10.1136/bmjsem-2018-000422 (2018).
Tahir, E. et al. Myocardial fibrosis in competitive triathletes detected by contrast-enhanced CMR correlates with exercise-induced hypertension and competition history. JACC Cardiovasc. Imaging 11, 1260–1270. https://doi.org/10.1016/j.jcmg.2017.09.016 (2018).
Treibel, T. A. et al. Left ventricular hypertrophy revisited: Cell and matrix expansion have disease-specific relationships. Circulation 136, 2519–2521. https://doi.org/10.1161/circulationaha.117.029895 (2017).
Domenech-Ximenos, B. et al. Prevalence and pattern of cardiovascular magnetic resonance late gadolinium enhancement in highly trained endurance athletes. J. Cardiovasc. Magn. Reson. 22, 62. https://doi.org/10.1186/s12968-020-00660-w (2020).
La Gerche, A. et al. Exercise-induced right ventricular dysfunction and structural remodelling in endurance athletes. Eur. Heart J. 33, 998–1006. https://doi.org/10.1093/eurheartj/ehr397 (2012).
Möhlenkamp, S. et al. Running: The risk of coronary events : Prevalence and prognostic relevance of coronary atherosclerosis in marathon runners. Eur. Heart J. 29, 1903–1910. https://doi.org/10.1093/eurheartj/ehn163 (2008).
Wilson, M. et al. Diverse patterns of myocardial fibrosis in lifelong, veteran endurance athletes. J. Appl. Physiol. 1985(110), 1622–1626. https://doi.org/10.1152/japplphysiol.01280.2010 (2011).
Görmeli, C. A. et al. Assessment of myocardial changes in athletes with native T1 mapping and cardiac functional evaluation using 3 T MRI. Int. J. Cardiovasc. Imaging 32, 975–981. https://doi.org/10.1007/s10554-016-0866-4 (2016).
Ainsworth, B. E. et al. 2011 compendium of physical activities: a second update of codes and MET values. Med. Sci. Sports Exerc. 43, 1575–1581. https://doi.org/10.1249/MSS.0b013e31821ece12 (2011).
Schulz-Menger, J. et al. Standardized image interpretation and post-processing in cardiovascular magnetic resonance—2020 update : Society for cardiovascular magnetic resonance (SCMR): Board of trustees task force on standardized post-processing. J. Cardiovasc. Magn. Reson. 22, 19. https://doi.org/10.1186/s12968-020-00610-6 (2020).
Heiberg, E. et al. Design and validation of Segment–freely available software for cardiovascular image analysis. BMC Med. Imaging 10, 1. https://doi.org/10.1186/1471-2342-10-1 (2010).
D’Ascenzi, F. et al. Cardiac magnetic resonance normal reference values of biventricular size and function in male athlete’s heart. JACC Cardiovasc. Imaging 12, 1755–1765. https://doi.org/10.1016/j.jcmg.2018.09.021 (2019).
Reiter, G., Reiter, C., Kräuter, C., Fuchsjäger, M. & Reiter, U. Cardiac magnetic resonance T1 mapping. Part 1: Aspects of acquisition and evaluation. Eur. J. Radiol. 109, 223–234. https://doi.org/10.1016/j.ejrad.2018.10.011 (2018).
Treibel, T. A. et al. Automatic measurement of the myocardial interstitium synthetic extracellular volume quantification without hematocrit sampling. JACC Cardiovasc. Imaging 9, 54–63. https://doi.org/10.1016/j.jcmg.2015.11.008 (2016).
Reiter, U., Reiter, C., Kräuter, C., Fuchsjäger, M. & Reiter, G. Cardiac magnetic resonance T1 mapping. Part 2: Diagnostic potential and applications. Eur. J. Radiol. 109, 235–247. https://doi.org/10.1016/j.ejrad.2018.10.013 (2018).
Pelliccia, A. et al. European Association of Preventive Cardiology (EAPC) and European Association of Cardiovascular Imaging (EACVI) joint position statement: Recommendations for the indication and interpretation of cardiovascular imaging in the evaluation of the athlete’s heart. Eur. Heart J. 39, 1949–1969. https://doi.org/10.1093/eurheartj/ehx532 (2018).
Breuckmann, F. et al. Myocardial late gadolinium enhancement: Prevalence, pattern, and prognostic relevance in marathon runners. Radiology 251, 50–57. https://doi.org/10.1148/radiol.2511081118 (2009).
Merghani, A. et al. Prevalence of subclinical coronary artery disease in masters endurance athletes with a low atherosclerotic risk profile. Circulation 136, 126–137. https://doi.org/10.1161/circulationaha.116.026964 (2017).
Lie, Ø. H. et al. Cardiac phenotypes and markers of adverse outcome in elite athletes with ventricular arrhythmias. JACC Cardiovasc. Imaging 14, 148–158. https://doi.org/10.1016/j.jcmg.2020.07.039 (2021).
Zorzi, A. et al. Nonischemic left ventricular scar as a substrate of life-threatening ventricular arrhythmias and sudden cardiac death in competitive athletes. Circ. Arrhythm Electrophysiol. 9, e004229. https://doi.org/10.1161/circep.116.004229 (2016).
de Noronha, S. V. et al. Aetiology of sudden cardiac death in athletes in the United Kingdom: A pathological study. Heart 95, 1409–1414. https://doi.org/10.1136/hrt.2009.168369 (2009).
Pelliccia, A. et al. European Association of Preventive Cardiology (EAPC) and European Association of Cardiovascular Imaging (EACVI) joint position statement: Recommendations for the indication and interpretation of cardiovascular imaging in the evaluation of the athlete’s heart. Eur. Heart J. 39, 1949–1969. https://doi.org/10.1093/eurheartj/ehx532%JEuropeanHeartJournal (2017).
Cantinotti, M. et al. Cardiac troponin-T release after sport and differences by age, sex, training type, volume, and intensity: A critical review. Clin. J. Sport Med. 32, e230–e242. https://doi.org/10.1097/jsm.0000000000000940 (2022).
Holtackers, R. J., Wildberger, J. E., Wintersperger, B. J. & Chiribiri, A. Impact of field strength in clinical cardiac magnetic resonance imaging. Invest. Radiol. 56, 764–772. https://doi.org/10.1097/rli.0000000000000809 (2021).
Engblom, H. et al. Importance of standardizing timing of hematocrit measurement when using cardiovascular magnetic resonance to calculate myocardial extracellular volume (ECV) based on pre- and post-contrast T1 mapping. J. Cardiovasc. Magn. Reson. 20, 46. https://doi.org/10.1186/s12968-018-0464-9 (2018).
La Gerche, A. & Heidbuchel, H. Can intensive exercise harm the heart? You can get too much of a good thing. Circulation 130, 992–1002. https://doi.org/10.1161/circulationaha.114.008141 (2014).
De Bosscher, R. et al. Rationale and design of the PROspective ATHletic Heart (Pro@Heart) study: Long-term assessment of the determinants of cardiac remodelling and its clinical consequences in endurance athletes. BMJ Open Sport Exerc. Med. 8, e001309. https://doi.org/10.1136/bmjsem-2022-001309 (2022).
Acknowledgements
The authors would like to acknowledge all study participants for their contribution to the study.
Funding
ProCardio Center for Innovation is funded by the South-Eastern Norway Regional Health Authority (project number 2017207) and the Research Council of Norway (project number 309762).
Author information
Authors and Affiliations
Contributions
Conceptualization and design: TE, KHH, LGK, ØHL. Data acquisition and image analysis: KA, LGK, ØHL, KB, ABK, GB, EH, CL, TE. Statistical analysis: KA, TE. Drafting, revising, rewriting and approval of the final manuscript: KA, LGK, ØHL, KB, ABK, GB, EH, CL, KHH, TE. All authors met the ICMJE criteria for authorship and no contributors fulfilling these criteria have been excluded from authorship. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Andresen, K., Klæboe, L.G., Lie, Ø.H. et al. No adverse association between exercise exposure and diffuse myocardial fibrosis in male endurance athletes. Sci Rep 14, 6581 (2024). https://doi.org/10.1038/s41598-024-57233-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-57233-5
- Springer Nature Limited