Abstract
Vector control is one of the principal strategies used for reducing malaria transmission. Long-lasting insecticidal bed nets (LLINs) are a key tool used to protect populations at risk of malaria, since they provide both physical and chemical barriers to prevent human-vector contact. This study aimed to assess the physical durability and insecticidal efficacy of LLINs distributed in Cruzeiro do Sul (CZS), Brazil, after 4 years of use. A total of 3000 LLINs (PermaNet 2.0) were distributed in high malaria risk areas of CZS in 2007. After 4 years of use, 27 ‘rectangular’ LLINs and 28 ‘conical’ LLINs were randomly selected for analysis. The evaluation of physical integrity was based on counting the number of holes and measuring their size and location on the nets. Insecticidal efficacy was evaluated by cone bioassays, and the amount of residual insecticide remaining on the surface of the LLINs was estimated using a colorimetric method. After 4 years of use, physical damage was highly prevalent on the rectangular LLINs, with a total of 473 holes detected across the 27 nets. The upper portion of the side panels sustained the greatest damage in rectangular LLINs. The overall mosquito mortality by cone bioassay was < 80% in 25/27 rectangular LLINs, with panel A (at the end of the rectangular bednet) presenting the highest mortality (54%). The overall mean insecticide concentration was 0.5 µg/sample, with the bednet roof containing the highest average concentration (0.61 µg/sample). On the conical LLINs, 547 holes were detected, with the bottom areas sustaining the greatest damage. The cone bioassay mortality was < 80% in 26/28 of the conical LLINs. The mean insecticide concentration was 0.3 µg/sample. After 4 years of use, the insecticidal efficacy of the LLINs was diminished to below acceptable thresholds.
Similar content being viewed by others
Introduction
The Brazilian Amazon continues to be the main malaria endemic area in Brazil, representing a challenge for public health authorities1. Since the beginning of the 2000’s, the Brazilian Ministry of Health (MOH) adopted prompt diagnosis and treatment as the principal strategies for reducing mortality and case severity2. Measures for vector control including the use of long-lasting insecticidal bed nets (LLINs) were implemented in highly endemic areas. The LLINs are distributed by the Brazilian MOH in two styles, conical for single or double beds and rectangular for hammocks3,4.
LLINs are an important malaria control tool for diminishing human/vector contact and are commonly provided to populations living in areas of malaria transmission risk3,5. Three municipalities in the Brazilian Amazon Region were the first to use LLINs as a strategy to control malaria: Cruzeiro do Sul, Mancio Lima and Rodrigues Alves. In 2007, both LLIN styles (conical and rectangular) were distributed in Cruzeiro do Sul (CZS) after a major malaria outbreak in 2006 with an API (Annual Parasite Index) of 571.5 malaria cases per 1000 inhabitants6,7. The main malaria vector in CZS, Anopheles darlingi, is present in both the intra- and peridomicile and its haematophagic activity occurs mainly between 6 and 9pm8.
In African countries, LLINs and indoor residual spraying (IRS) are the main malaria vector control measures used9. The information arising from experiences in Africa, including studies related to LLIN durability and effectiveness, has informed LLIN procurement decisions elsewhere, including determining the frequency with which they should be replaced10,11. In Brazil, data about malaria vectors, transmission dynamics and evaluations of vector control tools are comparatively scarce, and there are no previous studies evaluating long-term LLIN durability12. Thus, this study aimed to evaluate, for the first time, the physical durability and insecticidal efficacy of conical and rectangular LLINs after 4 years of use across CZS municipality.
Results
LLIN physical integrity
Only 1/27 of all rectangular LLINs analyzed had no detectable physical damage after 4 years of use. A total of 473 holes were found on the rectangular LLINs, resulting in a median of 8 holes (ranging from 0 to 63 holes; mean of 17.5 holes), with 51.8% in good condition, with pHI < 24. Damage was evenly distributed amongst the panels, with no statistically significant differences detected. Over half of the LLINs (16/27) had rips along the seams between the panels, resulting in a total of 40 rips, 70% of which were size 2. The area of greatest damage to the rectangular LLINs was along the top of the side panels of the nets, in about 45% of all rectangular nets.
Only 2/28 of the conical LLINs analyzed showed no evidence of physical damage after 4 years of use. A total of 547 holes were found on the conical LLINs, with a median of 13.5 holes/bednet (ranging from 0 to 70 holes; mean of 19.5), with only 28.6% in a good condition, with pHI < 24. The area of greatest damage was the bottom of the nets, in about 78% of all conical nets. Damage was evenly distributed amongst the panels, and on two of the conical nets, three rips were found along the seams between panels.
No significant differences were detected related to the number of holes between the LLIN types (p = 0.684). However, rectangular nets sustained more damage at the top and conical nets at the bottom (Fig. 1), showing significant statistical differences (Table 1).
WHO cone bioassays
In 25/27 of the rectangular nets tested, the mortality in cone bioassays was < 80%. The median mortality of the 27 LLINs was 60% (ranging from 4 to 92% - Supplementary Table 1). When mortality was analyzed using the same 5 panels tested by both WCT and CFT (A, B2, C, D2 and E2), we did not find statistically significant differences between the panels in terms of bioassay mortality (p = 0.706) (Fig. 2). A hundred per cent of the control mosquitoes survived in in all bioassays.
In 26/28 conical LLINs tested, the mortality was < 80%. The median mortality was 25.5% (ranging from 0 to 96% - Supplementary Table 2). When mortality was analyzed using the same 5 panels tested by both WCT and CFT (A, C, D, G and H), no statistically significant differences were detected among panels in terms of bioassay mortality (p = 0.334) (Fig. 2).
Colorimetric test (CFT)
The median concentration of surface insecticide on rectangular LLINs was 0.39 μg/sample (range: 0.003–2.458 µg/sample; mean of 0.5 µg/sample - Supplementary Table 1). Of the nets tested, 40.7% (n = 11) presented a mean concentration < 0.35 μg/sample, which was a threshold value previously proposed, below which the net could be considered as failed13. In the analysis of the sides, the roof (piece E2) showed a significantly higher concentration of insecticide, with 0.61 μg/sample (95% CI 0.446–0.779; p < 0.0001) followed by side C, with 0.54 μg/sample (95% CI 0.336–0.756) (Fig. 2).
The median concentration of surface insecticide on conical LLINs was 0.20 µg/sample (range: 0.00016 to 1.84 µg/sample, mean of 0.33 µg/sample - Supplementary Table 1). Of the nets tested, 85.7% (n = 24) had a mean concentration < 0.35 µg/sample. In the analysis of the panels, panel “A” contained significantly higher surface insecticide (0.41 µg/sample; 95% CI 0.29–0.53), in comparison to the other panels (p < 0.0001) (Fig. 2).
Comparison between WCT and CFT
In order to test the relationship between cone bioassay mortality and surface insecticide concentration from the CFT, we performed a logistic regression that showed a positive and statistically significant relationship (r2 = 0.2705; p < 0.0001), regardless of the type of mosquito net (rectangular or conical) (Fig. 3).
Discussion
The results of this study showed that after 4 years of use, nearly all (96.4%) of the LLINs (PermaNet 2.0—100 denier polyester-based) had sustained some degree of physical damage, but 40% of the all nets were still effective as a physical barrier (pHI < 24) The LLIN damages reported here are similar to those reported elsewhere. LLINs evaluations using PermaNet 2.0 after 3 years, showed in Ghana 52.1%10 and in Senagal 50% of the physical damages were size 1 holes (0.5–1.0 cm in diameter)14. In Laos, it was reported that 40% of LLINs presented physical damage after 2 years of use 150 denier polyethylene-based15 as well in Zanzibar, using the same brand and type of fabric of LLINs, high damage was observed in 68% of LLINs after 3 years of use16. In Cruzeiro do Sul, the LLINs Permanet 2.0 were in a good condition after 4 years of use, although Killian et al.17 had showed a slighter advantage of polyethylene-based LLINs on polyester-based, ones. In the same way, Allan et al.18 demonstrated that polyester-based had 4.22 times more chances of having a poor or very poor condition than polyethylene-based LLINs, and Skovmand and Bosselmann19 showed that multifibre polyester nets are weaker than commercialized mono-fibre polyethylene nets.
On the rectangular nets, the greatest damage was found in the upper region of the side panels. In the upper region, the damage may be explained by the local practice of placing a "rod" (wooden stick) along the upper portion of the side panels so that the nets are well-stretched, which can cause tears at the top. At the bottom, damage was likely due to increased manipulation due to being the place of entry and exit of people from the mosquito nets. On the conical LLINs, the greatest damage was recorded on the lower region, and this is likely because it is the site of contact with the bed platform (typically made of unfinished wood). In addition, people have the habit of knotting the nets to store them raised during the day, or fix them to walls with sharp materials, which can also damage them. Our findings highlight the importance of reminding the population about correct LLIN use to avoid compromising their durability (Supplementary file 1).
The cone bioassay tests found that the nets fell below the acceptable measure of bioefficacy after 4 years of use, since mortality was lower than 80% for nearly all the nets tested. However, it was not possible to determine when the LLINs began to lose their bioefficacy since only a single evaluation was performed. It is worth to note that LLINs were not designed to last 4 years but only 3. Ideally, annual evaluations to monitor the loss of insecticidal efficacy would more accurately identify the duration of the insecticidal effect of the LLINs and could be used to guide replacement strategies with greater precision. As previously observed in Laos, LLINs may need to be replaced more frequently than every 3 years15, and research from Ethiopia documented that the insecticidal efficacy of LLINs decreased over time20. In a study from Nicaragua on the durability of rectangular LLINs treated with deltamethrin, the median cone bioassay mortality after 36 months of use was only 2%21. Our results showed a median mortality of 33.4% (range: 0 to 96%) for both types of LLINs analyzed, after 4 years of use. When analyzing only rectangular nets, the median mortality was 60% (range: 4 to 92%). Despite the notable differences in bioefficacy compared to the study carried out in Nicaragua, our data were in alignment with the results of the correlation between colorimetric assays and bioassays conducted in Nicaragua, which also showed a positive correlation between the surface deltamethrin levels and mosquito mortality (Fig. 3).
Several factors may reduce the insecticidal efficacy of LLINs, such as frequent and inadequate washing, or drying in direct sunlight15, although, in this study area, most net users had informally reported following all washing and drying guidelines.
The colorimetric test detected significant differences between the surface insecticide concentrations found on the rectangular LLINs (0.5 µg/sample) compared to the conical LLINs (0.3 µg/sample). In addition, a statistically significant difference was observed between mortality in the cone bioassays between rectangular (49%) and conical (31%) LLINs. Green et al.13 demonstrated that LLINs used in Africa containing less than 0.35 µg/LLINs sample—10 mg/m2 that was 15% of the original insecticide concentration indicated that the LLIN had failed. However, in this study, of the 17 rectangular nets for which the value of the CFT was above 0.35 µg/sample, 14 presented mortality lower than 80% in the cone bioassay, and of the 4 conical nets with CFT above 0.35 µg/sample, 2 did not achieve 80% mortality in the biaossays. We cannot be sure if the apparent lack of sensitivity of the cutoff value of 0.35 µg/sample could be attributed to biological differences between the African and Brazilian anophelines, thus, further research is needed to evaluate the CFT cutoff value for Anopheles species in Brazil. However, the results of the cone bioassay and the colorimetric test were consistently well-associated, since when compared across the two types of nets used, no significant differences were detected. The colorimetric test was simple and fast, and has the additional benefit of not requiring live mosquitoes, which makes it a highly desirable method for this type of evaluation in Brazil where colonizing An. darlingi is particularly challenging. Furthermore, it is important to mention that the CFT measures the available insecticide on the surface, which is expected to be a more significant measure of bioefficacy than total insecticide content, as is often used for durability monitoring.
In the analysis of the panels of the rectangular LLINs, the position with the highest insecticide concentration was the roof, and on the conical LLINs, side A (upper side). Both are positions of little contact with people or objects. While one or two sides may retain disproportionately higher insecticide concentrations, this does not necessarily mean that the mosquito net as a whole provides an effective chemical barrier. It has been observed in studies in Africa that the levels of deltamethrin on the surface of an LLIN may diminish disproportionately over time, with a half-life of deltamethrin on the bottom portion of 2.4 months and on the upper portion of 5.6 months13. This suggests that the retention of the insecticide is closely related to the handling of the LLINs. As for the bottom side, independent of the user population, it likely presents a lower concentration of insecticide over time because it is the area of greatest manipulation (people entering/exiting, contact with bedding and other objects)13. However, Parker and cols using laboratory22 and field tests23 showed that it is possible for LLINs to still be effective if one of the sides is still impregnated with insecticides, especially on the roof, where these authors show a greater number of mosquitoes close to the nets. Our results showed that the roof of rectangular mosquito nets (Side E) and the top of conical mosquito nets (Side A) had a higher concentration of deltamethrin, which can determine the effectiveness of these mosquito nets even after 4 years of use.
In the municipality of Cruzeiro do Sul, the State Government implemented a set of actions to control the transmission of malaria, which involved investments in early diagnosis and treatment, recruitment of field personnel for entomological control, and implementing the use of LLINs and environmental control of breeding sites6. Those measures were implemented during a malaria outbreak, which resulted in a 67% reduction in the number of cases4. In the state of Amazonas, Brazil, it was observed that the effectiveness of LLINs was related to the socio-cultural profile of the population and the capacity of the local health teams to sensitize the community to appropriate LLIN use24. In contrast, in Rondônia State, no statistically significant differences in API were detected 1 year after LLIN installation when compared to municipalities without LLINs. However, the installation of LLINs in Rondônia was not accompanied by actions to evaluate their use, effectiveness, or durability, highlighting the need for further evaluations of this nature, with the aim of generating a body of evidence to support their widespread distribution in the Amazon Region25,26.
It should be noted that in CZS the number of malaria cases has been increasing since 2012, and that the increase in cases caused by P. falciparum is particularly worrying (35.4% in 2020)4. As a result, the health authorities continue to rely on LLINs as a key tool to prevent malaria. In addition, the potential development of deltamethrin resistance in the local mosquitoes is an important point to be considered and will need to be further evaluated and taken into account when selecting ongoing vector control tools. The data reported here fill an important gap regarding the effectiveness of a key malaria vector control tool in Brazil12, which can provide guidance for future malaria control program actions.
Methods
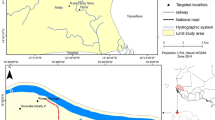
This study was performed in CZS (7° 37′ 51″ S, 72° 40′ 12″ W), in the northwest region of Acre State, in northern Brazil (Fig. 4). In December 2007, 3000 rectangular and conical PermaNet 2.0 (Vestergaard, Lausanne, Switzerland) deltamethrin-treated LLINs were distributed, and in December 2011, 27 rectangular and 28 conical LLINs were randomly selected by the local health authorities for the evaluation of their physical integrity and insecticidal efficacy after 4 years of use. The LLINs in the present work were the result of the first distribution of this vector control measure in Brazil. The collection of mosquito nets was carried out 4 years after distribution in an attempt to adapt to WHO standards and according to the possibility of the Brazilian Ministry of Health.
Annual parasite index (API) in Acre State in 2014. Cruzeiro do Sul municipality is indicated by the arrow (Map generated by the authors with the software https://www.mapchart.net/—Data source SIVEP—Malaria).
Physical integrity analysis
This analysis was based on a previously described protocol of Killian27 with modifications. LLINs were hung on a metal frame to facilitate the counting of holes. Hole sizes were categorized according to the following criteria: size 1 – hole with size smaller than the tip of index finger; size 2 – hole that permits the entrance of the tip of index finger but not a fist (≥ 1 cm and < 8 cm); size 3 – hole that permits the entrance of the closed fist (≥ 8 cm) and a proportionate holes index (pHI) based on the number of holes per category (1, 2 and 3) was calculated. The location of the holes was also recorded to assess which panels of the bednet were most affected as well as which locations on each panel incurred the most physical damage13.
Insecticidal efficacy
Based on the WHO cone bioassay test (WCT) protocol28, 10 pieces (20 × 20 cm) of each LLIN were evaluated, using 1 cone per piece (Figs. 5, 6). For the cone bioassays, wild-caught were used from Peixe-Boi municipality, Pará state, which are continuously monitored by our group using the CDC bottle bioassay and remain susceptible to the insecticides using the discriminating dose as recommended at Brogdon and Chan29. In each cone, five adult female, unfed, insecticide-susceptible An. darlingi were introduced and exposed for 3 min. At the end of this time, mosquitoes were transferred to a holding cup, where they were maintained and provided 10% sugar solution and mortality was recorded after 24 h. For each bioassay, one control was performed by exposing mosquitoes to a piece of non-impregnated bednet. The mortality index by bednet piece was determined by calculating the proportion of dead mosquitoes. For LLINs to be considered functional, they should result in mortality ≥ 80%28.
Surface insecticide quantification
The cyanopyrethroid field test (CFT) was used to measure surface levels of deltamethrin on 5 pieces of LLIN out of the same 10 used for the cone bioassays13. From the rectangular nets, the pieces were obtained from sides A, B2, C, D2 and E2, and on conical nets from sides A, C, D, G, and H (Figs. 5, 6). In brief, for each net piece, two 13-mm diameter Whatman 597 filter paper disks were wiped using magnets (magnetizing force = 35,000 Oersted) 30 times across 90 mm of net material on both the outer and inner surfaces. The amount of deltamethrin on both filter papers was measured by comparison with filter paper disks containing known quantities of deltamethrin (calibration standard). The sample disks and the calibration disks containing known amounts of deltamethrin were placed in 24-well polystyrene flat-bottomed tissue culture plates. A 0.2 ml aliquot of a solution containing 30 mg/ml of 1,2-dinitrobenzene and 4-nitrobenzaldehyde dissolved in methyl cellosolve (2-methoxyethanol) was added to each well. After allowing the disks to soak in the reagent for five minutes, the colorimetric reaction was activated with the addition of 0.05 ml 0.4 N sodium hydroxide. The reaction was allowed to proceed for five minutes whereupon the intensity of the purple color was recorded by digital photography using a standard digital camera. Deltamethrin concentrations were determined by comparing the color intensity of the sample disks to the calibration standards using image analysis techniques as described previously13.
Statistical analyses
Descriptive statistics were used to summarize the physical and chemical data from the analyzed LLINs. For the comparison between the damage found on the two LLIN types (rectangular and conical), t-tests (Mann–Whitney Rank test and Kruskal–Wallis test) and ANOVAs (nonparametric Friedman test), with Sidak's multiple comparisons tests were conducted. The chi-squared test was used to compare the observed mortality from the WCT and the amount of insecticide detected by CFT and linear regression was used to test the relationship between bioassay mortality and surface insecticide levels. The statistical tests were performed using Graphpad Prism version 7.00 for Windows (GraphPad Software, La Jolla California USA).
Conclusion
The LLINs after 4 years of use acted primarily as a physical barrier because many were no longer sufficiently effective as a chemical barrier. The value of the colorimetric test cut-off point of 0.35 µg/sample of insecticide and insecticide resistance among Anopheles species should be re-evaluated in countries like Brazil with mosquito mortality proportions < 80% in the majority of its bioassays. Additional evaluations of this nature will be useful to guide LLIN distributions and accompanying community engagement activities in Brazil.
Data availability
All data generated or analysed during this study are included in this published article and its supplementary information files.
Abbreviations
- LLINs:
-
Long-lasting insecticidal nets
- CZS:
-
Cruzeiro do Sul
- CFT:
-
Cyanopyrethroid field test
- CDC:
-
Centers for disease control and prevention
- SIVEP:
-
Sistema de Informações de Vigilância Epidemiológica
- API:
-
Annual parasite index
- WCT:
-
WHO cone test
References
World Health Organization. World Malaria Report: 20 years of global progress and challenges. World Health Organization (2020).
Brasil. Projeto para prevenção e controle da malária na Amazônia Brasileira. (2009).
Ministério da Saúde. Guia para Gestão Local do Controle da Malária—Controle Vetorial. Ministério da Saúde (2009).
Sivep-Malária, B. Ministério da Saúde & Secretaria de Vigilância em Saúde. SIVEP-MALÁRIA. http://www.saude.gov.br/sivep_malaria.
Santos, J. B. et al. Ação de mosquiteiros impregnados com deltametrina sobre a morbidade da malária em uma área da Amazônia Brasileira. Rev. Soc. Bras. Med. Trop. 31, 1–9 (1998).
Costa, K. M. de M. et al. Malária em Cruzeiro do Sul (Amazônia Ocidental brasileira): análise da série histórica de 1998 a 2008. Rev. Panam. Salud Pública 28, 353–360 (2010).
Santelli, A. C. et al. Effect of artesunate-mefloquine fixed-dose combination in malaria transmission in amazon basin communities. Malar. J. 11, 286 (2012).
Sucupira, I. M. C., dos Santos, M. M. M. & Póvoa, M. M. Mosquitos anofelinos envolvidos na transmissão da malária humana no município de Cruzeiro do Sul, estado do Acre, Amazônia brasileira. Rev. Pan-Amazônica Saúde 13, 45–54 (2022).
Nkya, T. E. et al. Six decades of malaria vector control in southern Africa: a review of the entomological evidence-base. Malar. J. 21, 279 (2022).
Smith, S. C. et al. Evaluation of bednets after 38 months of household use in northwest Ghana. Am. J. Trop. Med. Hyg. 77, 243–248 (2007).
Koenker, H. & Yukich, J. O. Effect of user preferences on ITN use: a review of literature and data. Malar. J. 16, 233 (2017).
Carlos, B. C., Rona, L. D. P., Christophides, G. K. & Souza-Neto, J. A. A comprehensive analysis of malaria transmission in Brazil. Pathog. Glob. Health 113, 1–13 (2019).
Green, M. D. et al. Evaluation of a rapid colorimetric field test to assess the effective life of long-lasting insecticide-treated mosquito nets in the Lao PDR. Malar. J. 12, 57 (2013).
Diouf, E. H. et al. Evaluation of the residual efficacy and physical durability of five long-lasting insecticidal nets (LLINs) in Senegal. Malar. J. 21, 1–15 (2022).
Shirayama, Y. et al. Maintenance behaviour and long-lasting insecticide-treated nets (LLITNs) previously introduced into Bourapar district, Khammouane province, Lao PDR. Public Health 121, 122–129 (2007).
Haji, K. A. et al. Challenges for malaria elimination in Zanzibar: pyrethroid resistance in malaria vectors and poor performance of long-lasting insecticide nets. Parasit. Vectors 6, 82 (2013).
Kilian, A. et al. Variation of physical durability between LLIN products and net use environments: summary of findings from four African countries. Malar. J. 20, 26 (2021).
Allan, R., O’Reilly, L., Gilbos, V. & Kilian, A. An observational study of material durability of three world health organization-recommended long-lasting insecticidal nets in Eastern Chad. Am. J. Trop. Med. Hyg. 87, 407–411 (2012).
Skovmand, O. & Bosselmann, R. Strength of bed nets as function of denier, knitting pattern, texturizing and polymer. Malar. J. 10, 87 (2011).
Wills, A. B. et al. Physical durability of PermaNet 2.0 long-lasting insecticidal nets over three to 32 months of use in Ethiopia. Malar. J. 12, 242 (2013).
Villalta, E. L. et al. Evaluation of the durability and use of long-lasting insecticidal nets in Nicaragua. Malar. J. 20, 106 (2021).
Parker, J. E. A. et al. Infrared video tracking of Anopheles gambiae at insecticide-treated bed nets reveals rapid decisive impact after brief localised net contact. Sci. Rep. 5, 13392 (2015).
Parker, J. E. A. et al. Host-seeking activity of a Tanzanian population of Anopheles arabiensis at an insecticide treated bed net. Malar. J. 16, 1–14 (2017).
Castro, D. B. de. A importância das equipes de atenção básica para a eficácia do uso de mosquiteiros impregnados como medida de controle da malária no Estado do Amazonas. in Memorias Convención Internacional de Salud Pública (2012).
Vieira, G. de D., Basano, S. de A., Katsuragawa, T. H. & Camargo, L. M. A. Insecticide-treated bed nets in Rondônia, Brazil: Evaluation of their impact on malaria control. Rev. Inst. Med. Trop. Sao Paulo 56, 493–497 (2014).
da Silva Ferreira Lima, A. C. et al. Evaluation of Long-lasting insecticidal nets (LLINs) for malaria control in an endemic area in Brazil. Parasit. Vectors 16, 162 (2023).
Kilian, A. et al. Evidence for a useful life of more than three years for a polyester-based long-lasting insecticidal mosquito net in Western Uganda. Malar. J. 10, 299 (2011).
World Health Organization. Guidelines for Laboratory and Field Testing of Long-Lasting Insecticidal Mosquito Nets. World Health Organization http://www.healthnettpo.org/files/807/who-cds-whopes-gcdpp-2005-11-testing.pdf (2005).
Brogdon, W. G. & Chan, A. Guidelines for evaluating insecticide resistance in vectors using the CDC bottle bioassay/methods in Anopheles research. (2010).
Acknowledgements
The authors gratefully acknowledge the assistance of site health authorities and laboratory teams who supported the study and research collaborations, and Michael Green for technical assistance with the CFT.
Disclaimer
The opinions expressed herein are those of the authors and do not necessarily reflect the views of the U.S. Centers for Disease Control and Prevention (CDC). OMML is a staff member of the Pan American Health Organization/World Health Organization (PAHO/WHO). The author alone is responsible for the views expressed in this publication and do not necessarily represent the decisions, policy or views of the PAHO/WHO.
Funding
Funding support for this evaluation was provided by the U.S. Agency for International Development (USAID) through the Latin America and Caribbean Regional Malaria Program, Instituto Evandro Chagas/Brazilian Ministry of Health and The Conselho Nacional de Desenvolvimento Científco e Tecnológico (CNPq—Brazil, Processos: 304827/2013-4 and 302292/2017-9). The funding source for this study had no role in study design, data collection, analysis, or interpretation.
Author information
Authors and Affiliations
Contributions
M.M.P., A.M.O. and A.L. conceived and supervised the project. M.M.P., A.M.O., S.D. and I.M.C.S. coordinated the study. A.C.F.S., C.C.R., I.M.C.S., E.R.R., M.M.M.S., E.L.C. and M.M.P. helped in the execution of the study and data collection. A.M.O., A.L., O.S.M.M.L., A.C.F.S., C.C.R., I.M.C.S. and M.M.P. contributed to data analysis. A.C.F.S. and C.C.R. drafted the manuscript. M.M.P., I.M.C.S., A.M.O., O.S.M.M.L. and A.L. reviewed and edited the manuscript. All authors read and approved the manuscript for publication.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Feio-dos-Santos, A.C., Reis, C.C., Sucupira, I.M.C. et al. Physical durability and insecticidal activity of long-lasting insecticidal nets in Cruzeiro do Sul, Brazil. Sci Rep 14, 9044 (2024). https://doi.org/10.1038/s41598-024-59172-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-59172-7
- Springer Nature Limited