Abstract
Incompatibility (Inc) HI2 plasmids are large (typically > 200 kb), transmissible plasmids that encode antimicrobial resistance (AMR), heavy metal resistance (HMR) and disinfectants/biocide resistance (DBR). To better understand the distribution and diversity of resistance-encoding genes among IncHI2 plasmids, computational approaches were used to evaluate resistance and transfer-associated genes among the plasmids. Complete IncHI2 plasmid (N = 667) sequences were extracted from GenBank and analyzed using AMRFinderPlus, IntegronFinder and Plasmid Transfer Factor database. The most common IncHI2-carrying genera included Enterobacter (N = 209), Escherichia (N = 208), and Salmonella (N = 204). Resistance genes distribution was diverse, with plasmids from Escherichia and Salmonella showing general similarity in comparison to Enterobacter and other taxa, which grouped together. Plasmids from Enterobacter and other taxa had a higher prevalence of multiple mercury resistance genes and arsenic resistance gene, arsC, compared to Escherichia and Salmonella. For sulfonamide resistance, sul1 was more common among Enterobacter and other taxa, compared to sul2 and sul3 for Escherichia and Salmonella. Similar gene diversity trends were also observed for tetracyclines, quinolones, β-lactams, and colistin. Over 99% of plasmids carried at least 25 IncHI2-associated conjugal transfer genes. These findings highlight the diversity and dissemination potential for resistance across different enteric bacteria and value of computational-based approaches for the resistance-gene assessment.
Similar content being viewed by others
Introduction
Bacterial plasmids contain genes that impart selective advantage to the host species. Plasmids are key vehicles for the distribution of antimicrobial resistance (AMR), disinfectant and biocide resistance (DBR), and heavy metal resistance (HMR) elements among bacteria. A number of different resistance plasmid types have been identified in the Enterobacteriaceae that can be classified into various incompatibility (Inc) groups based on their predicted compatibility to coexist in a single bacterium1, with some of the key groups being IncA/C, FIB, HI1, HI2, I1, P and W, which are associated with resistance to multiple different antibiotics2,3,4. Among these, IncHI2 plasmids are an interesting group due to their large in size, typically greater than 200 kb, ability to encode for AMR, HMR and DBR, and transmission potential to facilitate resistance dissemination among enteric bacteria5,6,7,8,9.
The IncHI2 plasmids are typically associated with members of the family Enterobacteriaceae; including Enterobacter, Escherichia, Salmonella, Klebsiella and Citrobacter species5,6,10,11,12. Strains carrying these plasmids originate from a wide range of locations within Asia, Europe, North and South America, Africa and Australia5,6,10,11,13,14,15,16 and variety of sources, including human patients, foods, food animals, and the environment5,17,18,19,20.
These plasmids have gained significant public health interest due to their ability to carry AMR genes to a number of clinically important antimicrobials including colistin, carbapenems and other β-lactams, and quinolones19,21,22,23,24,25. Resistance to these antimicrobials has been on the rise globally, which has led to increased morbidity and mortality26. In the U.S., a study of hospitalized patients in 2017, found that 12.9% of Enterobacteriaceae were resistant to extended-spectrum β-lactamases (ESBL), 1.2% were resistant to carbapenems and 6.9% were multidrug resistant27. Plasmids were key contributors to the emergence of this resistance26.
IncHI2 plasmids often also carry HMR genes including for arsenic, copper, mercury, nickel, silver and tellurite7,8,11,28. The characterization of DBR and HMR is important since disinfectants, biocides and metal ions have been widely used for infection control and limiting microbial contamination in the environment29,30,31. Many IncHI2 plasmids are also known to be conjugative and can subsequently transmit the AMR, DBR and HMR genes from the resistant donor strains to previously susceptible recipients leading to the spread of a wide range of resistance elements in a single transfer event9.
With the wide availability of whole genome sequencing (WGS), the ability to assess the genetics of bacterial pathogens has been greatly enhanced though a wide variety of tools and bioinformatics approaches32. Computational programs such as PlasmidFinder, PubMLST, ResFinder, AMRFinderPlus, and IntegronFinder can identify and subtype plasmids and identify different genes associated with AMR phenotypes33,34,35,36. In addition, with the increasing access to long-read sequencing technologies, there is an enhanced ability to close plasmid sequences and determine the locations of resistance determinants and mobile genetic elements (MGEs), such as transposons, insertion sequences and integrons that make up the AMR mobilome that can facilitate the spread of genes among different plasmids and bacterial populations37. Understanding the genetic composition of plasmids is important to aid in epidemiological investigations and identifying measures to limit the spread of AMR.
The current project takes advantage of different bioinformatics tools available to assess the resistance genetics and molecular epidemiology of IncHI2 plasmid sequences available through the GenBank database to aid in the understanding the distribution of the plasmids among foods, the environment and humans. The IncHI2 plasmids identified were classified using plasmid multilocus sequencing typing (pMLST) and assessed to predict AMR, DBR, HMR and conjugal transfer gene content and the presence of integrons within the plasmids. The data allowed for evaluation of the distribution of resistance genes among the assembled plasmids from different bacteria taxa, and exploration of geographical and temporal sources. Understanding the epidemiology of the resistance mobilome can help with the development of strategies to limit the spread of multidrug and HMR carrying plasmids among bacterial pathogens in the environment, food animals and humans37,38.
Methods and materials
Selection of enteric plasmids for sequence analyses and data acquisition
The GenBank database was screened for IncHI2 plasmids based on their plasmid replicon sequence39. To select plasmid sequences for the project, the IncHI2-associated plasmid replicon target sequence described by Carattoli et al. (2005) was used for BLAST searching by narrowing the search to “Microbe” genomes and “Complete plasmids” in the Microbial Nucleotide BLAST database to return all sequences that were identified as highly similar sequences using default megablast settings39,40. These queries were conducted in December 2022 and returned 667 complete plasmid DNA sequences. Complete sequences for each plasmid were downloaded from GenBank in the FASTA and GenBank formats. These sequences were uploaded into the pMLST database program to categorize the plasmids into IncHI2 plasmid sequence types33. The identity, accession numbers, sequence type, bacterial host, and additional available metadata for each of the plasmids are provided in Table S1.
Identification of resistance genes and integrons using GalaxyTrakr tools
The FASTA files for plasmids noted above were analyzed using AMRFinderPlus database V3.2.136 with the AMRFinderPlus plugin within the GalaxyTrakr (https://account.galaxytrakr.org) operating environment as described previously37. AMRFinderPlus is a comprehensive database with 983 unique AMR genes, 82 unique HMR genes and 30 DBR genes. For several of these genes, there are multiple different variants of the genes in the database36. The output file describing the AMR, DBR and HMR genes was imported into Microsoft Excel and sorted by on sequence ID and gene type. For integron characterization, the analyses were performed using the IntegronFinder within GalaxyTrakr and the associated summary and integron annotation files were downloaded and imported into Excel for further analyses. The IntegronFinder output provides the number and locations of complete integrons and partial integrons containing either the insertion sites without the integrase nearby (CALIN) or containing the integrase without the insertion locations nearby (In0)41.
Plasmid transfer gene analysis
To predict which putative plasmid transfer genes are present in the IncHI2 plasmids, the FASTA-formatted plasmid sequence files were submitted to the FDA Virulence and Plasmid Transfer Factor (VPTF) Database and searched with the Plasmid Transfer Factor Profile Comparison tool (https://virulence.fda.gov/tools/virulenceassess)42. The VPTF database contains sequences for type 4 secretion system genes for multiple incompatibility groups of resistance plasmids associated with enteric bacteria. The resulting output was exported to Microsoft Excel and included a detailed table containing the plasmid transfer genes, their percent identity to the reference sequence, number of mismatched base pairs, location of the gene in both the query and reference sequences, e-value, and bit score of the gene. Using the PivotTable function, the data was transformed to a presence/absence matrix with the e-value cutoff for inclusion in the table as 10–3.
Statistical and phylogenetic analyses
To characterize the overall plasmid sequences, the GenBank formatted files for all 667 plasmids were uploaded into GalaxyTrakr and converted to GFF3 format required for analysis using Roary to determine the IncHI2 “plasmid pangenome”43. Default parameters were used for the analysis, with the exception of using a 90% presence cutoff for being considered part of the core set of genes. The Roary output was then used as an input into Scoary to facilitate comparison of phenotypic features to the genes present in the pangenome44. Features assessed included bacterial hosts, geographical location and sample type (human infection, animal, food, or environmental). Descriptive statistics including means and standard deviations were also calculated to facilitate comparison of the proportion of genetic elements among the plasmids collected from different hosts, locations, sample types and year of collection. For comparison of the distribution of AMR, DBR, and HMR genes across the different metadata variables, student t-tests and chi-square (χ2) test of independence was calculated in Microsoft Excel were used with statistical significance observed at p < 0.05. For these χ2 tests, data from Klebsiella plasmids were combined with the “other” taxa due to their relatively low numbers and the differences compared by drug class. For phylogenetic analyses, the presence/absence binary data was imported into the BioNumerics (Applied Maths, Austin, TX) program to assess the comparisons of the different profiles and sample characteristics. In BioNumerics, separate datasets were created for AMR, HMR and transfer-associated genes, while the DBR and integron-associated genes were combined due to genetic linkages and the limited data size (eight variables). For each group, the sample diversities were calculated using the Anderberg similarity coefficient45 and a dendrogram generated using the unweighted pair group means with averages (UPGMA) clustering techniques. In addition, the data were analyzed with BioNumerics to conduct principal component analyses (PCA) and generate minimal spanning trees based on the AMR, HMR and/or DBR/integron binary data. For the spanning tree analyses, the samples were identified by either bacterial genus, country of origin, year of isolation or sample type. A randomly selected plasmid representing each of the pMLST sequence types was selected for alignment using the BLAST Ring Image Generator (BRIG) program46. The nucleotide sequence for the ST1 representative (accession # NZ_LC590026.1) served as the reference sequence for the BRIG comparison and annotations.
Results and discussion
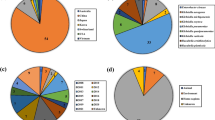
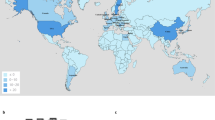
There were 667 IncHI2 plasmids that were detected using the study search parameters as of December of 2022. The details of the strains are provided in Table S1. Briefly, the dominant genera of the bacteria carrying the plasmids included Enterobacter (N = 209), Escherichia (N = 208), Salmonella (N = 204), and Klebsiella (N = 18), with the remaining taxa including Citrobacter, Kluyvera, Leclercia, Raoultella, Serratia and Yersinia species. On average, the sizes of the plasmids from Enterobacter were largest in size, averaging 319.5 kb (S.D. 45.6 kb), followed by the “other” taxa (289.5 kb; S.D. 56.3 kb), Escherichia (252.4 kb; S.D. 43.9 kb), Klebsiella (244.9 kb; S.D. 57.8 kb) and Salmonella (238.9 kb; S.D. 41.0 kb) (Fig. 1). The predominant geographical source locations included China (N = 346), Japan (N = 76), United States (N = 39), Taiwan (N = 27) and Australia (N = 23). In several cases (N = 105), the year that the bacteria were collected was not provided; however, in those provided, the top years were 2018 (N = 88), 2016 (N = 78), 2017 (N = 68), 2019 (N = 63) and 2015 (N = 54). Isolates were collected from humans (N = 360), animals (N = 167), foods (N = 39), environment (N = 34), and unknown sources (N = 68). Strains that carried the IncHI2 plasmids were members of the Enterobacteriaceae; although Serratia and Yersinia have recently been placed in the new family Yersiniaceae47.
Most of the isolates originated from China (346/667, 51.9%) and the majority were from human patients (360/667, 54.0%). When reported, there was a fairly even distribution of strains isolated each year from 2015 to 2019 (range of 54–88/year). Additionally, there were circumstances where isolates from a certain country were predominated by a particular bacterial species. For example, of the 76 isolates from Japan, 56 (73.7%) were identified as Enterobacter hormaechei. Conversely, the E. hormaechei were isolated from a wide range of countries and across several years, so it may serve as a key taxon to look at IncHI2 resistance genetics in detail (discussed below).
The results of the AMRFinderPlus analysis identified 170 unique AMR genes in one or more of the 667 IncHI2 plasmid sequences analyzed, with the most common genes detect being sul1 and sul2 (sulfonamide resistance), aadA2, aph(6)-Id, and aadA1 (aminoglycoside/streptomycin), floR (chloramphenicol), and tet(A) (tetracycline); however, the distribution of genes varied across the different host bacterial taxa (Table 1 and Tables S2 and S3). For example, among the sulfonamide resistance genes there was a significant difference in distribution (χ2, p < 0.001), sul1 was detected in plasmids isolated from the Enterobacter (77.0% positive), Klebsiella (77.8%) and “other” genera (75.0%) compared to Escherichia (55.7%) and Salmonella (64.2%). Conversely, sul2 and sul3 were more commonly detected among those from Escherichia (40.9% and 50.5%, respectively) and Salmonella (60.7% and 49.0%) isolates compared to Enterobacter (20.6% and 1.4%), Klebsiella (22.2% and 33.3%) and “other” genera (28.6% and 7.1%) (Table 1 and Figure S1A). Similar differences were seen with carbapenem and colistin resistance. For carbapenem resistance genes, blaIMP-1 was identified in 29.2% of plasmids from Enterobacter, but not found in the other plasmids. Other carbapenem resistance genes, including blaNDM-5 and blaNDM-9 were more common among plasmids from E. coli, Salmonella and Klebsiella, but not detected in those from Enterobacter (Fig. 2A). For colistin resistance there was also a significant difference in the distribution of resistance genes (χ2, p < 0.001), mcr-1.1 was more commonly detected in Escherichia (42.3%), Klebsiella (27.8%) and Salmonella (32.4%) in comparison to not being detected among the Enterobacter and less commonly among the “other” genera (10.7%). In contrast mcr-9.1 was detected in Enterobacter (43.5%), Klebsiella (33.3%) and “other” genera (46.4%), while only in 3.8% and 2.4% of plasmids from Escherichia and Salmonella, respectively (Fig. 2B). Likewise, the gene selection for several of the other drug classes showed significant taxa level distribution differences as well (χ2, all noted differences p < 0.001), including tetracycline genes (e.g. tet(A) vs. tet(B)), quinolones (e.g. oqxA and oqxB genes vs. qnrA and qnrB genes in Escherichia and Salmonella vs. Enterobacter), trimethoprim (e.g. dfrA12 vs. dfrA19), macrolides (e.g. ere(A) vs. mph(A) in Enterobacter vs. Escherichia and Salmonella), phenicols (e.g. catA vs. floR), and β-lactams (e.g. blaOXA-1 vs. blaSHV-12) (Figures S1B-G).
Percentage of isolates from a particular genus that has different AMR genes encoding resistance to different antimicrobial families: carbapenems (panel A) and colistin (panel B). The error bars indicate the standard deviation among the different taxa for the individual AMR gene displayed. Results for the other antimicrobial classes are shown in Figure S1.
The characterization and reporting of the resistance genes have been the focus of many manuscripts describing IncHI2 plasmids. A search of PubMed on “IncHI2 plasmids” identifies hundreds of papers, many of which have focused on the carriage of colistin, carbapenems and other β-lactam (primarily ESBL) resistance genes due to their importance for clinical medicine19,21,22,23,24,25,48,49. For example, Aoki et al. (2018) characterized blaIMP-1-carrying Enterobacter species isolated from patients in Japan and found that this carbapenem resistance gene was often carried on pST1 IncHI2 plasmids. These findings correspond to the results of the current study that found that the majority (59/61, 96.7%) of the blaIMP-1-carrying plasmids were isolated from Enterobacter in Japan50. Similarly, Timmermans et al. (2021) examined colistin resistance in E. coli isolated from different food animals in Belgium during the years of 2012–2016. Of the colistin resistant isolates in their study and the current study, mcr-1.1 was detected on IncHI2 plasmids in E. coli across multiple years51. These papers represent a small fraction of the IncHI2 literature focused on the characterization of these important resistance genes from the plasmids in bacteria from varied geographical locations, host sources and time periods, highlighting the global spread of IncHI2 plasmids23,52,53,54.
A potentially concerning reality is that in many cases IncHI2 plasmids are co-located with other resistance plasmids that carry overlapping genes encoding the resistance elements detected on the IncHI2 plasmids in the study. Khine et al. (2023) showed that mcr-1.1 in E. coli can be carried on IncI2 plasmids flanked by different insertion sequences. These plasmids could potentially facilitate transfer from the IncI2 plasmid to the IncHI2 plasmid if they are co-located in the same bacterium. The IncHI2 plasmids have several regions with insertion sequences that can lead to variability in the gene content of the different plasmids12,55. The most conserved regions in the IncHI2 plasmids appears to be the transfer regions of the plasmid. The presence of the transfer regions provides that many of these plasmids are conjugative and can lead to the spread between different bacteria in the same niche56.
In addition to the AMR genes, AMRFinderPlus was used to identify the HMR and DBR genes in the IncHI2 plasmids. There were 36 HMR genes detected, including multigene operons for arsenite efflux (ars), copper resistance (pco), copper/silver resistance (sil), mercury resistance (mer), nickel resistance (ncr), and tellurite resistance (ter) (Fig. 3). The most commonly detected resistance genes were the tellurite resistance genes terD, terW and terZ, which were detected in at least 94% of the strains from each genus. The arsenic resistance gene arsC was present in 87.1% of the IncHI2 plasmids isolated from Enterobacter, from 57.1% of the “other” taxa and 27.8% of the Klebsiella. arsC was less commonly detected in the plasmids from Escherichia (6.3%) and Salmonella (3.4%). There was a significant difference in distribution of many of the mercury resistance genes across the taxa, with many of the genes more common in the plasmids from Enterobacter, the “other” taxa and Klebsiella in comparison to those from Escherichia and Salmonella (Fig. 3A). Similarly, the distribution differences among the copper resistance genes (pco operon) were significant (χ2, p < 0.001), as each gene was less commonly detected in Enterobacter plasmids, with the exception of pcoS, where the gene was more frequently detected. Minimum spanning tree analyses indicated distinct lineages of Escherichia and Salmonella clustered together (Fig. 3B). Among the DBR genes there was also significant difference in the distribution of genes (χ2, p < 0.001), qacEΔ1 was more commonly detected in Enterobacter derived plasmids (75.6%) than those from Escherichia (39.9%) and Salmonella (44.1%; Figure S2). This higher proportion of qacEΔ1 in Enterobacter may correspond with the higher proportion of sul1 genes in the taxa, as both are common components of many of the class 1 integrons57. In contrast, qacL was present at much lower levels in Enterobacter plasmids (0.5%) compared to those from Escherichia (44.2%; Figure S2).
The percentage of isolates from a particular genus that have different HMR genes displayed (panel A). The error bars indicate the standard deviation among the different taxa for the individual AMR gene displayed. The lower panel (panel B) provides the results of minimum spanning tree analyses based on all the HMR genes identified among the plasmids in the study. The size and proportions in the circles are representative of the number of isolates within the group with identical gene profiles. The grey shading around the circles represents partitioning groups based on relative similarity of the groups.
Further analyses of the AMR, DBR and HMR resistance genes in relation to plasmid sequence type (pST), host bacterial genera/species, geographical and temporal origin were assessed using different comparative approaches (Fig. 4). When assessing the pST results the predominant STs were ST3 (N = 217), ST1 (N = 210), ST2 (N = 59) and ST4 (N = 33) (Table 2 and Table S1). The pMLST typing is based off two different loci (smr0018 and smr0199) and there were 135 plasmids that lacked one of the two loci, with the largest group having the allele 3 for smr0018 and no allele for smr0199 (N = 84). ST3 was made up of the smr0018 allele 3 and smr0199 allele 2; thus these 84 incomplete ST (3_NT) shared the smr0018 allele sequence with ST358. ST3 and 3_NT were most commonly identified in plasmid from Salmonella, Escherichia and Klebsiella (Table 2); whereas ST1 and other STs with smr0018 allele 1 (1_NT, ST14, and ST16) where most commonly detected from Enterobacter, other taxa, and Klebsiella plasmids. These plasmids were the largest sized IncHI2 plasmids, each group averaging over 280 kb in size, while all other STs (with the exception of NT_1) averaging less than 276 kb in size (Figure S3). ST2 was spread across each of the different genera, except for the other taxa plasmids. Of the Enterobacter strains that were ST2, the majority (8/11, 73%) were species other than E. hormaechei, which was the most common species among the Enterobacter found in the study.
Principal components analysis (PCA) plots of AMR gene presence/absence data for the 667 different plasmid sequences. The plasmids were characterized by: bacterial host taxa (panel A); plasmid sequence type (panel B); and sample type (panel C). Each of the circles represents a distinct genotype and may contain multiple members from the identified groups. The PCA analyses are displayed in 3D plots rotated to maximumly visualize the diversity of the groups.
The BRIG alignment of representatives of each the STs indicated highest levels of conservation appeared to be among the transfer region genes and other genes associated with the plasmid backbone (Fig. 5). When looking at the plasmid pangenome prediction based on Roary analysis, there were 70 genes that were present in 90% of the plasmids, which could be viewed as the “core” sequences of the IncHI2 plasmids. This 90% threshold, which is somewhat more lenient than for bacterial genomes, was chosen due to the high level of sequence plasticity among plasmids. The genes that made up this core genome included many of the transfer-associated genes, plasmid toxin/antitoxin genes and the tellurite resistance operon which correspond to the regions spanning for about 335 kbp to 85 kbp and 132 kpb to 145 kpb in the BRIG diagram in Fig. 5. The accessory genome, where a gene was detected in at least 2 plasmids, but outside the core genes, included 2,644 genes. Many of these accessory genes appear to related to plasmid recombination (e.g. transfer genes from other plasmid types) or are variants of the same gene, which is possibly due to the default 95% amino acid similarity cutoff used to define separate genes in Roary43. The accessory genome includes most of the resistance genes observed among the plasmids, which is consistent with the diverse results across the different taxa. The Scoary analyses also showed demonstrated differences in the gene content of the plasmids from the different bacterial taxa (Supplemental Data File). This phenomenon was most evident with the plasmids from the Enterobacter in which there were multiple genes associated with membrane transport and gene regulation that were only found in the IncHI2 plasmids from Enterobacter. As further data on geographic and source locations were included in the puzzle, it was shown that these more than 30 genes were associated (Scoary, p < 0.001) with a group of plasmids isolated from E. hormaechei isolated from human patients Japan across multiple years (Supplemental Data File).
Alignment of randomly selected plasmids representing each of the pMLST sequence types identified using the BRIG program. The inner ring is the GC content of the reference plasmid, which was the ST1 representative (accession # NZ_LC590026.1). The inner purple ring is the representative reference sequence for the BRIG comparison and annotations. The light yellow regions of shading in the center of the plasmid circles represents the core plasmid genetic regions, with these genes being present in at least 90% of the IncHI2 plasmids in the study as determined by Roary analysis.
Among the HMR genes, several of the mer and ter genes were conserved across many of the analyzed STs, which is not surprising since these are among the most commonly detected HMR genes among all the plasmids (e.g., terD, terW and terZ were detected in about 95% of the plasmids in the study; Fig. 3). Interestingly, when the sample types were examined, the plasmids from strains collected from environmental sources had on average a higher number (N = 10.3) of HMR and DBR genes than those from human patients (N = 9.3), foods (N = 9.1) and animals (N = 8.3). While these differences are not statistically significant, they do provide evidence that environmental exposures to heavy metals and disinfectants may provide selective pressure for resistance development.
When examining the distribution of AMR genes across the different taxa using PCA analyses, the aforementioned grouping of taxa is evident (Fig. 4A), with the Escherichia and Salmonella largely separating from the three other taxa. These grouping differences also pertain to the different plasmid STs (Fig. 4B), which is not too surprising since specific STs were typically associated with the different taxa (Table 2). There appears to be overlap of the AMR profiles is with the different sample types, with plasmids originating from human patients extensively overlapping with those originating from food, animals and the environment (Fig. 4C). When looking at the specific taxa, among the Salmonella strains, there was separation of the plasmid AMR gene profiles based on the pST. The majority of the plasmids fell into ST3 or the related 3_NT (148/204, 72.5%). Of the plasmids from other STs, each tended to cluster together with the other plasmids of the same ST. All six ST13 plasmids were isolated in the U.S. and had identical AMR gene profiles, yet they were isolated in different years (2002–2012) and different sources (animal and human; Figure S4). A majority of the ST2 plasmids (24/31, 77.4%) formed a separate cluster from most of the ST3 plasmids, while four of the remaining ST2 plasmids clustered with the ST4 and ST13 plasmids and two other ST2 plasmids shared a distinct gene profile with an NT_2.
Among the major genera carrying the IncHI2 plasmids, plasmids from Escherichia appear to be the most diverse, both in terms of AMR gene profiles (Fig. 4B) and pSTs. Plasmids from both Salmonella and Enterobacter had multiple groupings of more than five plasmids sharing common AMR gene profiles (Figures S4 and S5), whereas for Escherichia the largest group sharing a common AMR profile contained only five plasmids (Figure S6). Like was observed with the plasmids from Salmonella, there was some clustering of ST2 and ST4 for Escherichia plasmids, although the clusters were not as tight, which may be due to the Escherichia plasmids carrying larger numbers of different resistance genes than the Salmonella plasmids (118 vs. 91; Figure S6).
When we look at the host bacteria at the species level, all of Salmonella are S. enterica and a majority of the Escherichia are E. coli (197/208, 94.7%); whereas for the 209 Enterobacter strains, there is more diversity with 148 (70.8%) being E. hormaechei, 24 (16.3%) E. cloacae, 10 (4.8%) E. asburiae and remaining 17 (8.1%) spread across at least 6 other species. The E. hormaechei-associated plasmids are an interesting subset to explore due to their wide geographic and temporal distribution across a variety of host sources (Figure S5). When looking at the countries of origin of the strains carrying the plasmids, there are some clonal AMR clusters that were associated with Japan, The Netherlands, Canada, Mexico, and The Czech Republic. In several of the cases, plasmids with clonal AMR gene profiles were isolated across multiple years; for example, strains carrying plasmids from the Japanese cluster isolated from human patients from 2007 to 2020 (Figure S5).
The conjugal transfer of IncHI plasmids is interesting in a number of facets, including that the optimal conjugation temperatures, at least in vitro, appears to be in the range of 25 to 30 °C, although the plasmids can transfer at lower frequency at 37 °C57,59. These lower optimal conjugal transfer temperatures could lead to environmental locations playing an important role in spread of these plasmids. Additionally, the co-transfer of IncHI2 plasmids with other plasmid types can impact the efficiency of the plasmid transfer, potentially indicating a potential co-regulation of transfer and creating opportunities of cointegration of multiple plasmid-associated genes57. This cointegration of genes across plasmid types may lead to increase plasmid diversity and acquisition of resistance elements. When assessing the IncHI2-associated transfer genes specifically, the predominant genotype included all the genes except for traF and traG (N = 530, 79.5%; Fig. 6). This genotype was identified in plasmids from across the globe representing all genera, pST, and sources. The second most common genotype included all the genes except traF-traJ, trhF-trhH, trhR, and trhY (N = 91, 13.6%). Each of these plasmids also lacked the smr0199 allele as part of the pMLST scheme, including 1_NT, 2_NT, 3_NT, and 4_NT, which is likely due to this allele being located to close to these transfer genes that are missing in this group of plasmids60. The host isolates for these plasmids came from multiple continents, across a range of years and from all source categories, but were predominantly from Salmonella and Escherichia isolates.
The impacts of the differences in the transfer genes were not assessed in the current project; however, it is curious what the impact on plasmid transfer would be for those plasmids lacking multiple genes. Zhang et al. (2022) found that the deletions of the transfer gene region, and some of the AMR genes, may be a compensatory mechanism that reduces the cost of maintaining the plasmids in the host strain. Their experiments showed that strains carrying the IncHI2 plasmids that had the regions deleted during serial passaging of the bacteria had reduced fitness costs compared to the strains that did not undergo the deletions within the plasmids61. Part of the factors that appeared to contribute to the evolution of the plasmids with the compensatory gene deletions was the exposure to different antimicrobial agents. The tradeoff of the reduction of genes for the increased fitness was the likely reduction in the ability for the horizontal gene transfer of the plasmids like those lacking the smr0199 locus mentioned above (Fig. 6). The increase fitness and decreased ability for horizontal plasmid transfer may contribute to some clonal plasmids persisting in a bacterial species in a common geographical region over several years.
Class 1 integrons were detected in 75% of the plasmids (N = 498) (Figure S2). Based on the IntegronFinder and AMRFinder analyses, the integrons carried diverse sets of resistance genes, with some of the integron elements appeared complex with multiple resistance cassettes and integration sequences (Table S4). Zhao et al. (2018) noted that the integrons among the IncHI2 plasmids can be quite diverse, even among plasmids within the same pST and host taxa. The presence of these integrons allows for the accumulation of AMR genes within these plasmids facilitating the multidrug resistance phenotype and potential spread of resistance62.
While the study shows the importance of IncHI2 plasmids to AMR, DBR and HMR development and spread, the study has some caveats to consider in the overall interpretation. The first is that the results are based on the complete plasmid sequences that were available in GenBank as of December of 2022 which are reliant on the types of sequences completed and uploaded to the database. The host bacteria tend to skew to those associated with human illness and those encoding resistance, as these plasmids may have a higher public health significance. Secondly, the geographic origin of the sequences tended to be most heavily dominated by a few countries that have more heavily invested in long-read DNA sequencing technologies that are important for closing of the plasmids or possibly sets of bacteria/plasmids associated with individual focused studies. These potential issues will likely diminish in the near future with the increasing availability of new sequencing technologies and subsequent availability of high-quality closed plasmids. Even with these potential limitations, the study provides a valuable resource for the assessment of the IncHI2 plasmids in enteric bacteria across a wide range of species, geographical locations and time points that is important to understand the epidemiology of IncHI2 plasmids. The IncHI2 plasmids warrant continued surveillance to determine whether shifts in resistance profiles and bacterial host ranges can lead to increased public health risks due to the potential spread of large numbers of different resistance genes that can be carried on these plasmids.
Data availability
All data utilized in this study are publicly available through NCBI GenBank. GenBank Accession Numbers are provided in Table S1.
References
Novick, R. P. Plasmid incompatibility. Microbiol Rev 51, 381–395 (1987).
Carattoli, A. Resistance plasmid families in enterobacteriaceae. Antimicrob. Agents Chemother. 53, 2227–2238. https://doi.org/10.1128/AAC.01707-08 (2009).
Rozwandowicz, M. et al. Plasmids carrying antimicrobial resistance genes in Enterobacteriaceae. J. Antimicrob. Chemother. 73(5), 1121–1137. https://doi.org/10.1093/jac/dkx488 (2018).
Algarni, S., Ricke, S. C., Foley, S. L. & Han, J. The dynamics of the antimicrobial resistance mobilome of salmonella enterica and related enteric bacteria. Front. Microbiol. 13, 859854. https://doi.org/10.3389/fmicb.2022.859854 (2022).
Roy Chowdhury, P. et al. Identification of a novel lineage of plasmids within phylogenetically diverse subclades of IncHI2-ST1 plasmids. Plasmid 102, 56–61. https://doi.org/10.1016/j.plasmid.2019.03.002 (2019).
Lai, J. et al. Unique class 1 integron and multiple resistance genes co-located on IncHI2 plasmid is associated with the emerging multidrug resistance of Salmonella Indiana isolated from chicken in China. Foodborne Path Dis. 10, 581–588. https://doi.org/10.1089/fpd.2012.1455 (2013).
Ryan, D. & Colleran, E. Arsenical resistance in the IncHI2 plasmids. Plasmid 47, 234–240. https://doi.org/10.1016/s0147-619x(02)00012-4 (2002).
Whelan, K. F., Colleran, E. & Taylor, D. E. Phage inhibition, colicin resistance, and tellurite resistance are encoded by a single cluster of genes on the IncHI2 plasmid R478. J. Bacteriol. 177, 5016–5027. https://doi.org/10.1128/jb.177.17.5016-5027.1995 (1995).
Fang, L. et al. Co-spread of metal and antibiotic resistance within ST3-IncHI2 plasmids from E. coli isolates of food-producing animals. Sci. Rep. 6, 25312. https://doi.org/10.1038/srep25312 (2016).
Chen, W. et al. IncHI2 Plasmids are predominant in antibiotic-resistant salmonella isolates. Front. Microbiol. 7, 1566. https://doi.org/10.3389/fmicb.2016.01566 (2016).
Cain, A. K. & Hall, R. M. Evolution of IncHI2 plasmids via acquisition of transposons carrying antibiotic resistance determinants. J. Antimicrob. Chemother. 67, 1121–1127. https://doi.org/10.1093/jac/dks004 (2012).
Liu, R. et al. Genomic characterization of two Escherichia fergusonii isolates harboring mcr-1 gene from farm environment. Front. Cell Infect. Microbiol. 12, 774494. https://doi.org/10.3389/fcimb.2022.774494 (2022).
Alzahrani, K. O. et al. Complete genome sequence of a colistin-susceptible salmonella Enterica Serovar Minnesota strain harboring mcr-9 on an IncHI2/IncHI2A plasmid, isolated from chicken meat. Microbiol. Resour. Announc. 10, e0082621. https://doi.org/10.1128/mra.00826-21 (2021).
Zakaria, A. S., Edward, E. A. & Mohamed, N. M. Genomic insights into a Colistin-resistant Uropathogenic Escherichia coli strain of O23:H4-ST641 lineage harboring mcr-11 on a conjugative IncHI2 plasmid from Egypt. Microorganisms https://doi.org/10.3390/microorganisms9040799 (2021).
Pot, M. et al. Dissemination of extended-spectrum-β-Lactamase-Producing Enterobacter cloacae complex from a hospital to the nearby environment in guadeloupe (French west indies): ST114 lineage coding for a successful IncHI2/ST1 plasmid. Antimicrob. Agents Chemother. https://doi.org/10.1128/aac.02146-20 (2021).
Pontes, L. D. S. et al. Letter to the editor: Escherichia fergusonii harboring IncHI2 plasmid containing mcr-1 Gene-A novel reservoir for colistin resistance in Brazil. Microb Drug Resist 27, 721–725. https://doi.org/10.1089/mdr.2020.0041 (2021).
Diaconu, E. L. et al. Emergence of IncHI2 plasmids with mobilized colistin resistance (mcr)-9 Gene in ESBL-producing, multidrug-resistant salmonella typhimurium and its monophasic variant st34 from food-producing animals in Italy. Front Microbiol 12, 705230. https://doi.org/10.3389/fmicb.2021.705230 (2021).
Zhang, S. et al. High prevalence of multidrug-resistant Escherichia coli and first detection of IncHI2/IncX4-plasmid carrying mcr-1 E. coli in retail ready-to-eat foods in China. Int. J. Food Microbiol. 355, 109349. https://doi.org/10.1016/j.ijfoodmicro.2021.109349 (2021).
Trujillo-Soto, T., Machuca, J., Arca-Suárez, J., Rodríguez-Iglesias, M. & Galán-Sánchez, F. Co-Occurrence of mcr-1 and qnrS1 on an IncHI2 Plasmid in Clinical Isolates of Salmonella Typhimurium in Spain. Vector Borne Zoonotic Dis. 19, 662–665. https://doi.org/10.1089/vbz.2018.2398 (2019).
Tkadlec, J. et al. The intestinal carriage of plasmid-mediated colistin-resistant Enterobacteriaceae in tertiary care settings. Antibiotics (Basel) https://doi.org/10.3390/antibiotics10030258 (2021).
Nakayama, T. et al. IncHI2 plasmid encoding bla(CTX-M-55) and mcr-1.1 in Salmonella enterica SE20-C72–2 and Escherichia coli EC20-C72–1 isolates from the edible river fish anabas testudineus. Microbiol. Resour. Announc. 12, e0014923. https://doi.org/10.1128/mra.00149-23 (2023).
Fu, S. et al. First detection of bla(NDM-1)-haboring incHI2 plasmid in Escherichia coli strain isolated from goose in China. Foodborne Path Dis. 20, 244–250. https://doi.org/10.1089/fpd.2022.0071 (2023).
Marathe, N. P., Radisic, V., Salvà-Serra, F., Moore, E. R. B. & Grevskott, D. H. Emergence of new IncHI2 multidrug-resistance plasmids carrying VIM-1 metallo-β-lactamase in Escherichia coli in Norway. Lancet Microbe https://doi.org/10.1016/s2666-5247(22)00389-5 (2023).
Galetti, R., Andrade, L. N., Sazinas, P., Jelsbak, L. & Darini, A. L. C. pST15-IncHI2 plasmids co-harboring mcr-9 and several other antibiotic resistance genes in heavy metal tolerant Enterobacter cloacae complex isolates from hospital infections. J. Glob. Antimicrob.Resist. 31, 187–188. https://doi.org/10.1016/j.jgar.2022.09.002 (2022).
Lian, X. et al. oqxAB-positive IncHI2 plasmid pHXY0908 increase Salmonella enterica serotype typhimurium strains tolerance to ciprofloxacin. Front. Cell Infect. Microbiol. 9, 242. https://doi.org/10.3389/fcimb.2019.00242 (2019).
Lynch, J. P. 3rd., Clark, N. M. & Zhanel, G. G. Escalating antimicrobial resistance among Enterobacteriaceae: focus on carbapenemases. Expert Opin. Pharmacother. 22, 1455–1473. https://doi.org/10.1080/14656566.2021.1904891 (2021).
Gupta, V. et al. National prevalence estimates for resistant Enterobacteriaceae and Acinetobacter species in hospitalized patients in the United States. Int. J. Infect. Dis. 85, 203–211. https://doi.org/10.1016/j.ijid.2019.06.017 (2019).
Zhai, Y. et al. Complete nucleotide sequence of pH11, an IncHI2 plasmid conferring multi-antibiotic resistance and multi-heavy metal resistance genes in a clinical Klebsiella pneumoniae isolate. Plasmid 86, 26–31. https://doi.org/10.1016/j.plasmid.2016.04.001 (2016).
Deng, W. et al. Antibiotic resistance in Salmonella from retail foods of animal origin and its association with disinfectant and heavy metal resistance. Microb. Drug Resist. 24, 782–791. https://doi.org/10.1089/mdr.2017.0127 (2018).
Johnson, T. J. et al. Plasmid replicon typing of commensal and pathogenic Escherichia coli isolates. Appl. Environ. Microbiol. 73, 1976–1983 (2007).
Han, J. et al. DNA sequence analysis of plasmids from multidrug resistant Salmonella enterica serotype Heidelberg isolates. PLoS ONE 7, e51160. https://doi.org/10.1371/journal.pone.0051160 (2012).
Mao, C. et al. Curation, integration and visualization of bacterial virulence factors in PATRIC. Bioinformatics 31, 252–258. https://doi.org/10.1093/bioinformatics/btu631 (2015).
Carattoli, A. et al. In silico detection and typing of plasmids using PlasmidFinder and plasmid multilocus sequence typing. Antimicrob. Agents Chemother. 58, 3895–3903. https://doi.org/10.1128/AAC.02412-14 (2014).
Zankari, E. et al. Identification of acquired antimicrobial resistance genes. J Antimicrob. Chemother. 67, 2640–2644. https://doi.org/10.1093/jac/dks261 (2012).
Tyson, G. H. et al. WGS accurately predicts antimicrobial resistance in Escherichia coli. J. Antimicrob. Chemother. https://doi.org/10.1093/jac/dkv186 (2015).
Feldgarden, M. et al. Validating the AMRFinder tool and resistance gene database by using antimicrobial resistance genotype-phenotype correlations in a collection of isolates. Antimicrob. Agents Chemother. https://doi.org/10.1128/AAC.00483-19 (2019).
Algarni, S. et al. In silico analyses of diversity and dissemination of antimicrobial resistance genes and mobile genetics elements, for plasmids of enteric pathogens. Front. Microbiol. 13, 1095128. https://doi.org/10.3389/fmicb.2022.1095128 (2022).
Johansson, M. H. K. et al. Detection of mobile genetic elements associated with antibiotic resistance in Salmonella enterica using a newly developed web tool: MobileElementFinder. J. Antimicrob. Chemother. 76, 101–109. https://doi.org/10.1093/jac/dkaa390 (2021).
Carattoli, A. et al. Identification of plasmids by PCR-based replicon typing. J.Microbiol. Methods 63(3), 219–228. https://doi.org/10.1016/j.mimet.2005.03.018 (2005).
Altschul, S. F., Gish, W., Miller, W., Myers, E. W. & Lipman, D. J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410. https://doi.org/10.1016/S0022-2836(05)80360-2 (1990).
Neron, B. et al. IntegronFinder 2.0: identification and analysis of integrons across bacteria, with a focus on antibiotic resistance in Klebsiella. Microorganisms https://doi.org/10.3390/microorganisms10040700 (2022).
Algarni, S. et al. Development of an antimicrobial resistance plasmid transfer gene database for enteric bacteria. Front. Bioinform. 3, 1279359. https://doi.org/10.3389/fbinf.2023.1279359 (2023).
Page, A. J. et al. Roary: rapid large-scale prokaryote pan genome analysis. Bioinformatics 31, 3691–3693. https://doi.org/10.1093/bioinformatics/btv421 (2015).
Tang, A. D. et al. Full-length transcript characterization of SF3B1 mutation in chronic lymphocytic leukemia reveals downregulation of retained introns. Nat. Commun. 11, 1438. https://doi.org/10.1038/s41467-020-15171-6 (2020).
Boriah, S., Chandola, V. & Kuma, V. in Proceedings of the 2008 SIAM International Conference on Data Mining (SDM). 243–254 (Society for Industrial and Applied Mathematics).
Alikhan, N. F., Petty, N. K., Ben Zakour, N. L. & Beatson, S. A. BLAST ring image generator (BRIG): simple prokaryote genome comparisons. BMC Genom. 12, 402. https://doi.org/10.1186/1471-2164-12-402 (2011).
Janda, J. M. & Abbott, S. L. The changing face of the family Enterobacteriaceae (Order: “Enterobacterales”): new members, taxonomic issues, geographic expansion, and new diseases and disease syndromes. Clin. Microbiol. Rev. https://doi.org/10.1128/CMR.00174-20 (2021).
Bortolaia, V. et al. Co-localization of carbapenem (blaOXA-162) and colistin (mcr-1) resistance genes on a transferable IncHI2 plasmid in Escherichia coli of chicken origin. J. Antimicrob. Chemother. 76, 3063–3065. https://doi.org/10.1093/jac/dkab285 (2021).
Pauly, N. et al. Co-occurrence of the blaVIM-1 and blaSHV-12 genes on an IncHI2 plasmid of an Escherichia coli isolate recovered from German livestock. J. Antimicrob. Chemother. 76, 531–533. https://doi.org/10.1093/jac/dkaa436 (2021).
Aoki, K. et al. Molecular characterization of IMP-1-Producing enterobacter cloacae complex isolates in Tokyo. Antimicrob. Agents Chemother. https://doi.org/10.1128/AAC.02091-17 (2018).
Timmermans, M., Wattiau, P., Denis, O. & Boland, C. Colistin resistance genes mcr-1 to mcr-5, including a case of triple occurrence (mcr-1, -3 and -5), in Escherichia coli isolates from faeces of healthy pigs, cattle and poultry in Belgium, 2012–2016. Int. J. Antimicrob. Agents 57, 106350. https://doi.org/10.1016/j.ijantimicag.2021.106350 (2021).
Zając, M. et al. The first description of the complete genome sequence of multidrug-resistant Salmonella enterica serovar monophasic Typhimurium (1,4,[5],12:i:-) isolate with the mcr-1.1 gene on IncHI2 found in pig in Poland. J. Glob. Antimicrob. Resist. 33, 218–220. https://doi.org/10.1016/j.jgar.2023.04.008 (2023).
Manageiro, V. et al. Genomic analysis of a mcr-9.1-harbouring IncHI2-ST1 plasmid from enterobacter ludwigii isolated in fish farming. Antibiotics (Basel) https://doi.org/10.3390/antibiotics11091232 (2022).
Allen, J. L., Doidge, N. P., Bushell, R. N., Browning, G. F. & Marenda, M. S. Healthcare-associated infections caused by chlorhexidine-tolerant Serratia marcescens carrying a promiscuous IncHI2 multi-drug resistance plasmid in a veterinary hospital. PLoS ONE 17, e0264848. https://doi.org/10.1371/journal.pone.0264848 (2022).
Khine, N. O., Wongsurawat, T., Jenjaroenpun, P., Hampson, D. J. & Prapasarakul, N. Comparative genomic analysis of Colistin resistant Escherichia coli isolated from pigs, a human and wastewater on colistin withdrawn pig farm. Sci. Rep. 13, 5124. https://doi.org/10.1038/s41598-023-32406-w (2023).
Tan, W. et al. Cotransfer of resistance to cephalosporins, colistin, and fosfomycin mediated by an IncHI2/pSH16G4928-like plasmid in ESBL-producing monophasic Salmonella Typhimurium strains of pig origin. J. Aappl. Microbiol. https://doi.org/10.1093/jambio/lxac060 (2023).
Zhao, H., Chen, W., Xu, X., Zhou, X. & Shi, C. Transmissible ST3-IncHI2 plasmids are predominant carriers of diverse complex IS26-Class 1 integron arrangements in multidrug-resistant Salmonella. Front. Microbiol. 9, 2492. https://doi.org/10.3389/fmicb.2018.02492 (2018).
García-Fernández, A. & Carattoli, A. Plasmid double locus sequence typing for IncHI2 plasmids, a subtyping scheme for the characterization of IncHI2 plasmids carrying extended-spectrum beta-lactamase and quinolone resistance genes. J. Antimicrob. Chemother. 65, 1155–1161. https://doi.org/10.1093/jac/dkq101 (2010).
Forns, N., Banos, R. C., Balsalobre, C., Juarez, A. & Madrid, C. Temperature-dependent conjugative transfer of R27: role of chromosome- and plasmid-encoded Hha and H-NS proteins. J. Bacteriol. 187, 3950–3959. https://doi.org/10.1128/JB.187.12.3950-3959.2005 (2005).
Gilmour, M. W., Thomson, N. R., Sanders, M., Parkhill, J. & Taylor, D. E. The complete nucleotide sequence of the resistance plasmid R478: defining the backbone components of incompatibility group H conjugative plasmids through comparative genomics. Plasmid 52, 182–202. https://doi.org/10.1016/j.plasmid.2004.06.006 (2004).
Zhang, J. F. et al. A trade-off for maintenance of multidrug-resistant IncHI2 plasmids in Salmonella enterica serovar typhimurium through adaptive evolution. mSystems 7, e0024822. https://doi.org/10.1128/msystems.00248-22 (2022).
Gillings, M. et al. The evolution of class 1 integrons and the rise of antibiotic resistance. J. Bacteriol. 190, 5095–5100. https://doi.org/10.1128/JB.00152-08 (2008).
Acknowledgements
This research was supported by the National Center for Toxicological Research (NCTR) and U.S. Food and Drug Administration (FDA). Dr. Dereje Gudeta was supported in part by an appointment to the Research Participation Program at the NCTR administered by the Oak Ridge Institute for Science and Education (ORISE) through an interagency agreement between the U.S. Department of Energy and the U.S. FDA. This manuscript reflects the views of the authors and does not necessarily reflect those of the Food and Drug Administration.
Author information
Authors and Affiliations
Contributions
SA, JH, and SLF conceived of the study. SA and SLF extracted the data from GenBank and SA, DDG, JH, RN, and SLF analyzed and interpreted the data. All authors contributed to the drafting and final revision of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Algarni, S., Gudeta, D.D., Han, J. et al. Genotypic analyses of IncHI2 plasmids from enteric bacteria. Sci Rep 14, 9802 (2024). https://doi.org/10.1038/s41598-024-59870-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-59870-2
- Springer Nature Limited