Abstract
Postoperative delirium (POD) is a common complication in older patients with hepatocellular carcinoma (HCC) that adversely impacts clinical outcomes. We aimed to evaluate the risk factors for POD and to construct a predictive nomogram. Data for a total of 1481 older patients (training set: n=1109; validation set: n=372) who received liver resection for HCC were retrospectively retrieved from two prospective databases. The receiver operating characteristic (ROC) curve, calibration plot, and decision curve analysis (DCA) were used to evaluate the performance. The rate of POD was 13.3% (148/1109) in the training set and 16.4% (61/372) in the validation set. Multivariate analysis of the training set revealed that factors including age, history of cerebrovascular disease, American Society of Anesthesiologists (ASA) classification, albumin level, and surgical approach had significant effects on POD. The area under the ROC curves (AUC) for the nomogram, incorporating the aforementioned predictors, was 0.798 (95% CI 0.752–0.843) and 0.808 (95% CI 0.754–0.861) for the training and validation sets, respectively. The calibration curves of both sets showed a degree of agreement between the nomogram and the actual probability. DCA demonstrated that the newly established nomogram was highly effective for clinical decision-making. We developed and validated a nomogram with high sensitivity to assist clinicians in estimating the individual risk of POD in older patients with HCC.
Similar content being viewed by others
Hepatocellular carcinoma (HCC) ranks as the sixth most commonly diagnosed cancer and the third deadliest globally1. Globally, the incidence rates of HCC exhibit a positive correlation with age, reaching their peak around the age of 75 years2,3. Owing to rapid aging of the global population and a record-high average life expectancy, there is a growing incidence of older patients diagnosed with HCC4,5.
Postoperative delirium (POD) is a prevalent and serious complication marked by acute and varying alterations in mental condition, attentional capabilities, and consciousness levels following liver resection6,7,8. Studies have shown a correlation between POD and unfavorable consequences, including heightened mortality rates, extended hospital stays, and elevated medical costs. Additionally, POD may contribute to lasting and more substantial declines in cognitive functions and daily life activities9,10,11,12,13.
Roughly one-third of POD cases are considered preventable, making it a suitable focus for surgical quality improvement endeavors14,15. In practice, uniformly implementing all effective delirium prevention strategies for every older surgical patient throughout their perioperative course is often impractical, despite being theoretically possible. Given the resource constraints and infrequent implementation of these interventions in most centers, recommendations have been made to focus on identifying patients with the highest risk16,17,18.
Previous research has established nomograms for POD in malignant tumors, including gynecologic cancers19, gastric cancer 20, colorectal cancer21, and head and neck cancer22. However, the accuracy of these nomograms varies widely and may not necessarily be applicable to HCC. In this study, we aimed to identify the risk factors for POD in older patients with HCC and to develop a corresponding nomogram.
Materials and methods
Patients
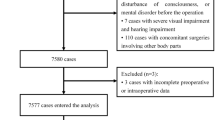
A nomogram was developed through a retrospective analysis of a prospectively registered database including 1109 patients with HCC at Mengchao Hepatobiliary Hospital of Fujian Medical University between March 2015 and June 2020. Concurrently, for external validation, we included data from 372 patients treated at Fujian Cancer Hospital between March 2018 and August 2020 (Fig. 1). The inclusion criteria were: (1) individuals aged 65 years and older; (2) patients who underwent elective hepatectomy; and (3) availability of sufficient data. The exclusion criteria were: (1) preoperative cognitive impairment; (2) history of severe nervous system disorders and dementia; (3) language barriers; hearing or vision impairments resulting in an inability to communicate; and (4) brain metastases.
This study was performed in accordance with the Declaration of Helsinki. Written informed consent was obtained from all participants. This research was approved by the ethics committee of Mengchao Hepatobiliary Hospital of Fujian Medical University.
Definitions
The diagnosis of POD was conducted through the application of the Confusion Assessment Method (CAM), a widely used diagnostic algorithm known for its demonstrated high sensitivity and specificity in identifying delirium23,24. POD frequently initiates in the recovery room and can persist for up to 5 days following the surgical procedure18. Assessment of POD can occur daily for a consecutive 5-day period post surgery, whenever patients manifest an abrupt alteration in mental status.
Cerebrovascular disease, as defined in this study, included conditions such as cerebral infarction, cerebral hemorrhage, stenosis (such as stenosis of the carotid, vertebral stenosis, or intracranial stenosis), and aneurysms25.
Ischemic heart disease (IHD) refers to inadequate blood supply to the heart, resulting in myocardial ischemia. This encompasses acute myocardial infarction, chronic stable angina, chronic IHD, and its associated heart failure25,26.
Chronic pulmonary disease is characterized by the presence of at least one of the following conditions: asthma, chronic obstructive pulmonary disease, and restrictive lung disease27.
Intraoperative hypotension is characterized by instances in which the patient experiences a systolic blood pressure below 80 mmHg or encounters at least one episode of systolic blood pressure that falls more than 20% below the baseline28.
Statistical analysis
The data were analyzed using both IBM SPSS 24.0 (IBM Corp) and R software (version 4.1.1). Parameters with a normal distribution are expressed as mean ± standard deviation and analyzed using the Student t-test. Parameters not following a normal distribution are expressed as median and interquartile range and analyzed using the Mann–Whitney test. Categorical variables are presented as frequency and percentage, and their comparisons were conducted using the chi-squared test. The least absolute shrinkage and selection operator (LASSO) regression model was used to select the optimal predictive variables. Subsequently, the identified key features were integrated into multivariable logistic regression analysis. Forest plots, constructed using GraphPad Prism, were used to visualize the results. Predictors found to be statistically significant were used to establish a nomogram system for diagnosing POD. The discriminative performance of the nomogram was assessed using the area under the receiver operating characteristic curve (AUC). Calibration curves were generated to evaluate the concordance between predicted and observed probabilities. Additionally, a decision curve analysis (DCA) was conducted to assess the clinical utility of the nomogram. Statistical significance was defined as a two-tailed P-value < 0.05.
Results
Patient characteristics
In the training set, the overall rate of POD was 13.3% (148 of 1109 patients). In the validation set, this rate was slightly higher at 16.4% (61 of 372 patients). Notable differences between the two sets included factors such as sex, body mass index, presence of hypertension, history of previous abdominal surgery, platelet levels, blood urea nitrogen, duration of the surgical procedure, and a requirement for intraoperative blood transfusion (Table 1).
Selection of independent predictors for POD and development of the nomogram in the training set
From a pool of 40 related dependent variables, eight potential predictors with non-zero coefficients in the LASSO regression model were selected based on data from the training set. These predictors include age, diabetes mellitus, history of cerebrovascular disease, smoking, ASA classification, albumin level, tumor number, and surgical approach (Fig. 2). To facilitate analysis and clinical implementation, continuous variables were dichotomized according to the ROC curve analysis. The optimal cutoff values for age and albumin level were determined to be 71 (AUC = 0.711) and 37.2 g/L (AUC = 0.688), respectively (Fig. 3). The multivariable logistic regression analysis incorporated these predictors, with age, history of cerebrovascular disease, ASA classification, albumin level, and surgical approach recognized as independent risk factors for POD (Fig. 4).
Construction and validation of the nomogram
The nomogram was created using these independent factors as its foundation (Fig. 5). The AUCs of the nomogram model were 0.798 (95% CI 0.752–0.843) in the training set and 0.808 (95% CI 0.754–0.861) in the validation set (Fig. 6). The calibration curves generated by the nomogram exhibited strong concordance between observed outcome frequencies and predicted probabilities in both sets (Fig. 7). The results of DCA are shown in Fig. 8, which demonstrated that the nomogram used in our study had superior effectiveness compared with treating all patients or providing no treatment. This advantage was observed when the threshold probability ranged from 9 to 91% in the training set and from 5 to 87% in the validation set.
Calibration curves of the nomogram in the training set (A) and validation set (B). The horizontal axis depicts the anticipated likelihood of POD, and the vertical axis illustrates the actual occurrence of diagnosed POD relative to the total cases. The diagonal dashed line represents the perfect prediction of the ideal model. The solid line represents the prediction of the nomogram; a closer fit to the diagonal dashed line represents the result after bias correction by bootstrapping (1000 repetitions).
Decision curve analysis of the nomogram in the training set (A) and validation set (B). The net benefit is quantified along the y-axis. The red line denotes predictions from the nomogram, the green line signifies the assumption of POD occurrence in all patients, and the blue line signifies the assumption of no POD occurrence in any patient.
Discussion
The incidence of POD was 13.3% in the training set and 16.1% in the validation set. Consistent with prior research, Nomi et al. reported a POD rate of 14.2%29. Yoshimura et al. found a POD rate of 17.0% in patients undergoing hepatectomy30. However, Ishihara et al. reported a lower POD incidence of 7.5%6, and Chen et al. found this to be 8.4%31. Variability in POD rates may be attributed to several factors: a lack of consistent definitions and assessment methods for POD by researchers, the diverse clinical characteristics of patients, and inaccuracies in estimation owing to the use of retrospective research methods.
This research involved developing and validating a new nomogram for predicting POD in older HCC patients. The model demonstrated strong capabilities in both discrimination and calibration, which showed its clinical value. The model's robustness was further enhanced by external validation, affirming its applicability across different patient groups and clinical settings. To our knowledge, this represents the inaugural predictive model for POD in individuals diagnosed with HCC.
Previous studies have indicated a correlation between the risk of POD and the emergence of postoperative complications32,33. However, such data are not accessible before or during surgery and thus cannot be integrated into predictive models.
In this study, a significant correlation between advanced age and an increased risk of POD was observed. Older patients experience a decline in physical capabilities, brain tissue integrity, and stress response regulation, along with diminished levels of key central neurotransmitters like acetylcholine and epinephrine34. Age-related constriction of blood vessels reduces cerebral oxygenation, which can potentially lead to postoperative cerebral impairment35,36. Moreover, alterations in drug metabolism and response owing to aging may increase the adverse effects of medications, thereby increasing the likelihood of POD37.
The present study identified that a history of cerebrovascular disease is an independent risk factor for POD. Cerebrovascular disease can lead to cognitive impairment, dementia, and neurocognitive deficits, which is postulated to increase delirium possibly through altered brain networks and a reduced ability to integrate sensory inputs38. Long-term susceptibility to delirium should be regarded as an integral aspect of the overall cerebrovascular disease burden39,40. Several studies have indicated that cognitive dysfunction and reduced functional capacity are associated with a heightened risk of POD41,42,43.
The ASA physical status classification system is commonly applied to evaluate a patient's ability to withstand anesthesia, primarily based on their overall compromised health and the presence of multiple comorbidities44. Research has indicated that an ASA classification ≥ 3 is associated with an increased risk of complications and decreased overall survival after hepatectomy45,46,47. Our study indicated that an ASA classification ≥ 3 is a risk factor for POD, as evidenced in numerous studies on this topic48,49,50,51. Whereas we found no statistically significant differences in common comorbidities such as diabetes and hypertension between the groups, it is considered that the cumulative impact of various comorbidities might heighten baseline vulnerability in older patients. This susceptibility, combined with the stress of surgery, could be a contributing factor to the development of POD52,53.
Numerous research findings indicate that a lower patient albumin level increases their likelihood of experiencing POD, a conclusion that aligns with the findings of our study6,54,55,56,57. Hypoalbuminemia affects drug metabolism, antioxidant defense, and toxin processing because albumin is the primary transport protein in blood plasma. Reduced albumin levels may result in cognitive dysfunction owing to toxic effects and oxidative injuries58,59. Appropriate medical intervention can yield lower albumin levels, potentially aiding in the reduction of POD risk.
This study showed that an open approach independently increases the risk of POD. A laparoscopic approach may reduce operative stress and postoperative systemic inflammation, which are known to be linked to the occurrence of POD29,60,61,62.
There are a number of limitations in this study. First, this research was a retrospective evaluation conducted using a prospectively registered database. Recognizing the intrinsic biases inherent in this type of study design is crucial. Prior studies have highlighted several risk factors linked to POD, including preoperative depression and anxiety63,64,65,66,67,68. Nevertheless, these factors were not incorporated into our analysis owing to certain constraints. Second, the experiment was conducted in only two centers, both of which are located in the same city. To further validate the model, it is necessary to use a more extensive sample size and conduct studies across various centers in different regions.
Conclusion
In older patients with HCC, factors such as age, cerebrovascular disease history, ASA classification, albumin levels, and the type of surgical procedure are identified as independent predictors of POD. In this study, we developed and externally validated a new, precise nomogram for personalized assessment and clinical decision-making.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249 (2021).
Xu, L., Xu, Y., Li, G. & Yang, B. Perioperative factors related to the prognosis of elderly patients with hepatocellular carcinoma. Eur. J. Med. Res. 27, 280 (2022).
Petrick, J. L. et al. International trends in hepatocellular carcinoma incidence, 1978–2012. Int. J. Cancer 147, 317–330 (2020).
Liu, C., Wu, J. & Chang, Z. Trends and age-period-cohort effects on the prevalence, incidence and mortality of hepatocellular carcinoma from 2008 to 2017 in Tianjin, China. Int. J. Environ. Res. Public Health 18, 6034 (2021).
Zhang, C. H., Cheng, Y., Zhang, S., Fan, J. & Gao, Q. Changing epidemiology of hepatocellular carcinoma in Asia. Liver Int. 42, 2029–2041 (2022).
Ishihara, A. et al. Preoperative risk assessment for delirium after hepatic resection in the elderly: A prospective multicenter study. J. Gastrointest. Surg. 25, 134–144 (2021).
Lin, X. et al. The hotspots and publication trends in postoperative delirium: A bibliometric analysis from 2000 to 2020. Front. Aging Neurosci. 14, 982154 (2022).
Jin, Z., Hu, J. & Ma, D. Postoperative delirium: Perioperative assessment, risk reduction, and management. Br. J. Anaesth. 125, 492–504 (2020).
Park, E. A. & Kim, M. Y. Postoperative delirium is associated with negative outcomes and long-term mortality in elderly Koreans: A retrospective observational study. Medicina (Kaunas) 55, 618 (2019).
Tsukakoshi, D. et al. Association between postoperative delirium and heart rate variability in the intensive care unit and readmissions and mortality in elderly patients with cardiovascular surgery. Heart Vessels 38, 438–447 (2023).
Shi, Z. et al. Postoperative delirium is associated with long-term decline in activities of daily living. Anesthesiology 131, 492–500 (2019).
Markar, S. R., Smith, I. A., Karthikesalingam, A. & Low, D. E. The clinical and economic costs of delirium after surgical resection for esophageal malignancy. Ann. Surg. 258, 77–81 (2013).
Janssen, T. L. et al. Long-term outcomes of major abdominal surgery and postoperative delirium after multimodal prehabilitation of older patients. Surg. Today 50, 1461–1470 (2020).
Burton, J. K. et al. Non-pharmacological interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst. Rev. 7, CD013307 (2021).
Berian, J. R. et al. Postoperative delirium as a target for surgical quality improvement. Ann. Surg. 268, 93–99 (2018).
Hughes, C. G. et al. American society for enhanced recovery and perioperative quality initiative joint consensus statement on postoperative delirium prevention. Anesth. Analg. 130, 1572–1590 (2020).
American Geriatrics Society abstracted clinical practice guideline for postoperative delirium in older adults. J. Am. Geriatr. Soc. 63, 142-150 (2015).
Aldecoa, C. et al. European Society of Anaesthesiology evidence-based and consensus-based guideline on postoperative delirium. Eur. J. Anaesthesiol. 34, 192–214 (2017).
Xiang, D., Xing, H. & Zhu, Y. A predictive nomogram model for postoperative delirium in elderly patients following laparoscopic surgery for gynecologic cancers. Support Care Cancer 31, 24 (2022).
Chen, J., Ji, X. & Xing, H. Risk factors and a nomogram model for postoperative delirium in elderly gastric cancer patients after laparoscopic gastrectomy. World J. Surg. Oncol. 20, 319 (2022).
Mosk, C. A. et al. Low skeletal muscle mass as a risk factor for postoperative delirium in elderly patients undergoing colorectal cancer surgery. Clin. Interv. Aging 13, 2097–2106 (2018).
Choi, N. Y. et al. Development of a nomogram for predicting the probability of postoperative delirium in patients undergoing free flap reconstruction for head and neck cancer. Eur. J. Surg. Oncol. 43, 683–688 (2017).
Inouye, S. K. et al. Clarifying confusion: The confusion assessment method. A new method for detection of delirium. Ann. Intern. Med. 113, 941–948 (1990).
Smulter, N., Lingehall, H. C., Gustafson, Y., Olofsson, B. & Engström, K. G. Validation of the confusion assessment method in detecting postoperative delirium in cardiac surgery patients. Am. J. Crit. Care 24, 480–487 (2015).
Kumar, V., Bishayee, K., Park, S., Lee, U. & Kim, J. Oxidative stress in cerebrovascular disease and associated diseases. Front. Endocrinol. (Lausanne) 14, 1124419 (2023).
Kang, H. S. et al. An elevated likelihood of stroke, ischemic heart disease, or heart failure in individuals with gout: a longitudinal follow-up study utilizing the National Health Information database in Korea. Front. Endocrinol. (Lausanne) 14, 1195888 (2023).
Hong, C. M. & Galvagno, S. M. Jr. Patients with chronic pulmonary disease. Med. Clin. North Am. 97, 1095–1107 (2013).
Wang, J. et al. Bibliometric and visual analysis of intraoperative hypotension from 2004 to 2022. Front. Cardiovasc. Med. 10, 1270694 (2023).
Nomi, T. et al. Effect of laparoscopic liver resection on postoperative delirium in elderly patients with hepatocellular carcinoma. J. Hepatobiliary Pancreat. Sci. 30, 1111–1118 (2023).
Yoshimura, Y. et al. Risk factors for postoperative delirium after liver resection for hepatocellular carcinoma. World J. Surg. 28, 982–986 (2004).
Chen, Y. L. et al. Low hemoglobin level is associated with the development of delirium after hepatectomy for hepatocellular carcinoma patients. PLoS One 10, e0119199 (2015).
Takeuchi, M. et al. Incidence and risk factors of postoperative delirium in patients with esophageal cancer. Ann. Surg. Oncol. 19, 3963–3970 (2012).
Kunz, J. V., Spies, C. D., Bichmann, A., Sieg, M. & Mueller, A. Postoperative anaemia might be a risk factor for postoperative delirium and prolonged hospital stay: A secondary analysis of a prospective cohort study. PLoS One 15, e0229325 (2020).
Kolk, A., Schwarzer, C., Wolff, K. D., Grill, F. & Weingart, J. Factors associated with postoperative delirium in patients undergoing complex head and neck flap surgery. J. Oral Maxillofac. Surg. 80, 372-379.e5 (2022).
Kang, T. et al. Incidence & risk factors of postoperative delirium after spinal surgery in older patients. Sci. Rep. 10, 9232 (2020).
Liu, Y., Shen, W. & Tian, Z. Using machine learning algorithms to predict high-risk factors for postoperative delirium in elderly patients. Clin. Interv. Aging 18, 157–168 (2023).
Xu, Y. et al. Prediction model for delirium in patients with cardiovascular surgery: Development and validation. J. Cardiothorac. Surg. 17, 247 (2022).
Gold, B. T. et al. Clinically silent Alzheimer’s and vascular pathologies influence brain networks supporting executive function in healthy older adults. Neurobiol. Aging 58, 102–111 (2017).
Pendlebury, S. T., Thomson, R. J., Welch, S. & Rothwell, P. M. Cognitive predictors of delirium on long-term follow-up after TIA and stroke: Population-based cohort study. Cerebrovasc. Dis. 51, 288–295 (2022).
Liu, F., Huang, J., Hei, G., Wu, R. & Liu, Z. Effects of sulforaphane on cognitive function in patients with frontal brain damage: Study protocol for a randomised controlled trial. BMJ Open 10, e037543 (2020).
Zhou, Q. et al. Predictors of postoperative delirium in elderly patients following total hip and knee arthroplasty: A systematic review and meta-analysis. BMC Musculoskelet. Disord. 22, 945 (2021).
Honda, S. et al. Risk factors for postoperative delirium after gastrectomy in gastric cancer patients. World J. Surg. 42, 3669–3675 (2018).
Hiraki, M. et al. A clinical risk analysis of early post-operative delirium after laparoscopic colorectal cancer surgery in elderly patients: A retrospective study. Int. J. Colorectal Dis. 36, 1461–1468 (2021).
Mayhew, D., Mendonca, V. & Murthy, B. A review of ASA physical status—Historical perspectives and modern developments. Anaesthesia 74, 373–379 (2019).
Ueno, M. et al. Recent trends in hepatectomy for elderly patients with hepatocellular carcinoma. Surg. Today 44, 1651–1659 (2014).
Takagi, K. et al. Sarcopenia and American Society of anesthesiologists physical status in the assessment of outcomes of hepatocellular carcinoma patients undergoing hepatectomy. Acta Med. Okayama 70, 363–370 (2016).
Ng, K. et al. Development and validation of a novel nomogram predicting 10-year actual survival after curative hepatectomy for hepatocellular carcinoma. Surgeon 19, 329–337 (2021).
Zhang, X. et al. Predictive nomogram for postoperative delirium in elderly patients with a hip fracture. Injury 50, 392–397 (2019).
Malik, A. T., Quatman, C. E., Phieffer, L. S., Ly, T. V. & Khan, S. N. Incidence, risk factors and clinical impact of postoperative delirium following open reduction and internal fixation (ORIF) for hip fractures: An analysis of 7859 patients from the ACS-NSQIP hip fracture procedure targeted database. Eur. J. Orthop. Surg. Traumatol. 29, 435–446 (2019).
Vacas, S., Grogan, T., Cheng, D. & Hofer, I. Risk factor stratification for postoperative delirium: A retrospective database study. Medicine (Baltimore) 101, e31176 (2022).
Sánchez Acedo, P., Eguaras Córdoba, I., Zazpe Ripa, C., Herrera Cabezón, J. & Tarifa Castilla, A. Prospective study of factors associated with postoperative delirium after urgent abdominal surgery. Cir. Esp. (Engl. Ed.) 98, 450–455 (2020).
Liu, J. et al. High ASA physical status and low serum uric acid to creatinine ratio are independent risk factors for postoperative delirium among older adults undergoing urinary calculi surgery. Clin. Interv. Aging 18, 81–92 (2023).
Zhu, Y. et al. Risk factors for postoperative delirium in patients undergoing major head and neck cancer surgery: A meta-analysis. Jpn. J. Clin. Oncol. 47, 505–511 (2017).
Venkatakrishnaiah, N. K. et al. Identification of factors contributing to the development of postoperative delirium in geriatric patients with hip fractures- A prospective study. J. Family Med. Prim. Care 11, 4785–4790 (2022).
Okawa, Y. et al. The assessment of risk factors for postoperative delirium using cubic spline curves in gastroenterological surgery. Surg. Today 51, 1969–1977 (2021).
Li, G. H. et al. Development and validation of a risk score for predicting postoperative delirium after major abdominal surgery by incorporating preoperative risk factors and surgical Apgar score. J. Clin. Anesth. 75, 110408 (2021).
Kim, H., Chung, S., Joo, Y. H. & Lee, J. S. The major risk factors for delirium in a clinical setting. Neuropsychiatr. Dis. Treat. 12, 1787–1793 (2016).
Llewellyn, D. J., Langa, K. M., Friedland, R. P. & Lang, I. A. Serum albumin concentration and cognitive impairment. Curr. Alzheimer Res. 7, 91–96 (2010).
Oh, E. S. et al. Preoperative risk factors for postoperative delirium following hip fracture repair: A systematic review. Int. J. Geriatr. Psychiatry 30, 900–910 (2015).
Beloosesky, Y. et al. Cytokines and C-reactive protein production in hip-fracture-operated elderly patients. J. Gerontol. A Biol. Sci. Med. Sci. 62, 420–426 (2007).
Lin, S. et al. Surgical outcomes of hand-assisted laparoscopic liver resection vs. open liver resection: A retrospective propensity score-matched cohort study. Chin. J. Cancer Res. 31, 818–824 (2019).
Hołówko, W. et al. Early adoption of laparoscopic liver surgery in Poland: A national retrospective cohort study. Int. J. Surg. (2023).
Yang, Q. et al. A retrospective analysis of the incidence of postoperative delirium and the importance of database selection for its definition. BMC Psychiatry 23, 88 (2023).
Liu, Q., Li, L., Wei, J. & Xie, Y. Correlation and influencing factors of preoperative anxiety, postoperative pain, and delirium in elderly patients undergoing gastrointestinal cancer surgery. BMC Anesthesiol. 23, 78 (2023).
Ishibashi, H. et al. Postoperative delirium in lung cancer anatomical resection-analysis of risk factors and prognosis. World J. Surg. 46, 1196–1206 (2022).
Aitken, S. J., Blyth, F. M. & Naganathan, V. Incidence, prognostic factors and impact of postoperative delirium after major vascular surgery: A meta-analysis and systematic review. Vasc. Med. 22, 387–397 (2017).
Mou, Q. et al. Preoperative anxiety as an independent predictor of postoperative delirium in older patients undergoing elective surgery for lumbar disc herniation. Aging Clin. Exp. Res. 35, 85–90 (2023).
Pakrad, F., Pakrad, E., Darvishi, N. & Poorolajal, J. Preoperative anxiety and depression increases the incidence of delirium after coronary artery bypass graft surgery. J. Perianesth. Nurs. 35, 496–501 (2020).
Acknowledgements
This work was supported by Fujian Cancer Hospital and Mengchao Hepatobiliary Hospital of Fujian Medical University. We thank Analisa Avila, MPH, ELS, of Liwen Bianji (Edanz) (www.liwenbianji.cn) for editing the language of a draft of this manuscript.
Funding
This work was supported by the Startup Fund for scientific research, Fujian Medical University (grant number: 2022QH1158); Natural Science Foundation of Fujian Province (grant number: 2020J011151, 2023J011280); Fujian Cancer Hospital Project (Grant number: 2024YN01).
Author information
Authors and Affiliations
Contributions
Mingfang Yan collected the data and helped prepare the tables. Zhaoyan Lin contributed to the manuscript writing and revision. Huizhe Zheng contributed to the idea and design. Lai Jinglan designed the study and drew the figures. Yuming Liu collected the data and drafted the initial manuscript. Lin Zhenmeng collected the data and revised the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yan, M., Lin, Z., Zheng, H. et al. Development of an individualized model for predicting postoperative delirium in elderly patients with hepatocellular carcinoma. Sci Rep 14, 11716 (2024). https://doi.org/10.1038/s41598-024-62593-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-62593-z
- Springer Nature Limited