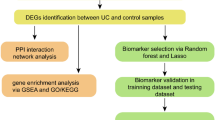
Abstract
Ulcerative colitis (UC) is characterized by an abnormal immune response, and the pathogenesis lacks clear understanding. The cGAS-STING pathway is an innate immune signaling pathway that plays a significant role in various pathophysiological processes. However, the role of the cGAS-STING pathway in UC remains largely unclear. In this study, we obtained transcriptome sequencing data from multiple publicly available databases. cGAS-STING related genes were obtained through literature search, and differentially expressed genes (DEGs) were analyzed using R package limma. Hub genes were identified through protein–protein interaction (PPI) network analysis and module construction. The ConsensuClusterPlus package was utilized to identify molecular subtypes based on hub genes. The therapeutic response, immune microenvironment, and biological pathways of subtypes were further investigated. A total of 18 DEGs were found in UC patients. We further identified IFI16, MB21D1 (CGAS), TMEM173 (STING) and TBK1 as the hub genes. These genes are highly expressed in UC. IFI16 exhibited the highest diagnostic value and predictive value for response to anti-TNF therapy. The expression level of IFI16 was higher in non-responders to anti-TNF therapy. Furthermore, a cluster analysis based on genes related to the cGAS-STING pathway revealed that patients with higher gene expression exhibited elevated immune burden and inflammation levels. This study is a pioneering analysis of cGAS-STING pathway-related genes in UC. These findings provide new insights for the diagnosis of UC and the prediction of therapeutic response.
Similar content being viewed by others
Introduction
Ulcerative colitis (UC) is a chronic inflammatory disease that can affect the rectum and extend to pancolitis, significantly impacting the quality of life. The global prevalence of UC is expected to reach 5 million cases by 2023, with an increasing incidence rate each year1. While the development of biologics targeting tumour necrosis factor (TNF) has provided hope for UC patients, the rates of primary non-response and secondary loss of response remain high2. Other emerging biological therapies and small molecule drugs, such as vedolizumab and Janus kinase inhibitors, have shown limited therapeutic efficacy, with clinical remission rates ranging from only 10–30%3,4. Long-term treatment with these drugs may have side effects such as severe infections, neurological disorders, malignancies and thrombosis. Therefore, revealing the pathogenesis and exploring new therapeutic targets are crucial for patients with UC.
The pathogenesis of UC is not fully understood and may be influenced by a combination of genetic susceptibility, environmental factors, gut microbiota, and immune response5. As the first defense of host immunity, innate immunity acts first when pathogens invade the body6, and plays an important role in the development of UC. Recent studies found that Cyclic GMP-AMP (cGAMP) synthase (cGAS)-stimulator of interferon genes (STING) signaling pathway is one of the important pattern recognition and effector pathways in the innate immune system. Cyclic GMP-AMP (cGAMP) synthase (cGAS) is activated when it recognizes and binds to double-stranded DNA (dsDNA), resulting in the synthesis of 2´3´-cGAMP. 2ʹ3ʹ-cGAMP as a second messenger binds and activates stimulator of interferon genes (STING), which subsequently triggers a signalling cascade to activate the innate immune system through production of immune and inflammatory factors, such as type I interferon (IFN-I)7,8. Abnormal cGAS-STING pathway activation may lead to the occurrence of autoimmune diseases. Increasing evidence suggests that this pathway plays a significant role in inflammation-related diseases9,10. Consequently, the cGAS-STING signaling pathway is expected to be a new target for the treatment of autoimmune diseases.
Inhibition of cGAS-STING pathway to reduce abnormal inflammatory response has become a new research direction for the treatment of various autoimmune diseases such as systemic lupus erythematosus11. The latest study shows that the clinical development of inhibitor has begun, and clinical trials are planned to begin12. Interestingly, previous study has reported that the cGAS-STING pathway is involved in the pathogenesis, progression, and therapeutic response of UC13. The level of STING is increased in the colon of patients with UC14. However, our understanding of the role of the cGAS-STING pathway in UC process is remains limited. This lack of knowledge may be a key factor affecting future clinical diagnosis and treatment.
In this study, we aimed to systematically summarized the cGAS-STING pathway-related genes and identified hub genes in UC. In addition, patients with UC were classified based on the cGAS-STING pathway-related genes. The correlations between hub genes and anti-TNF therapy efficacy were further investigated and validated. Finally, we analyzed the expression of hub genes in cancer.
Materials and methods
Patients
Human colon tissue samples were obtained from patients with UC prior to the treatment of infliximab. The patients with UC were divided into two groups based on their response to infliximab. The participants were recruited from the Second Hospital of Hebei Medical University. All procedures were conducted in accordance with the principles of the Declaration of Helsinki and approved by the Ethics Committee of the Second Hospital of Hebei Medical University.
Data collection and processing
The microarray data and clinical information of the patients were obtained from multiple public databases. All databases are listed in Table 1. We used ulcerative colitis as keyword to retrieve gene expression profiles and corresponding clinical data from the Gene Expression Omnibus (GEO) database (http://www.ncbi.nlm.nih.gov/geo). The GSE38713 dataset contains RNA sequencing results of colonic mucosal tissues from 15 UC patients with active disease and 13 healthy controls15. The GSE47908 dataset contains RNA sequencing results of colonic mucosal tissues from 39 patients with UC and 15 controls16. After normalizing the data, the batch effect was removed using ComBat function from the "sva" package between the two datasets17. The "limma" package (Version 3.42.2) was used to identify differentially expressed genes (DEGs) between inflammatory and healthy tissues18. The adjust P value < 0.05 were statistically signifcant.
Identification of cGAS-STING pathway-related differentially expressed genes
A literature search identified 30 genes that have been implicated in the cGAS-STING pathway (Supplementary Table S1). To demonstrate the reliability of the data, we used the original gene ID from the GEO data. For instance, CGAS and STING were previously known as MB21D1, TMEM173. Next, we used a Venn diagram tool to determine the overlap between the DEGs and the 30 genes related to the cGAS-STING pathway.
Protein–protein interaction network construction and evaluation of hub genes
Using a protein–protein interaction network (PPI network, Version 11.5, https://string-db.org) to study interactions between DEGs, we defined statistical significance as an interaction score > 0.7 and obscured individual target protein nodes. Subsequently, the identification of hub genes was performed using the molecular complex detection (MCODE) plug-in in Cytoscape software (Version 3.9.1). To assess the diagnostic value of hub genes, we employed receiver operating characteristic curves (ROC) and the area under the curve (AUC) by "pROC" package.
The relationship between hub genes and anti-TNF therapy
GSE16879 and GSE12251 were merged to investigate the correlation between anti -TNF therapy and hub genes expression19,20. GSE73661 was used as the validation database21. Cluster analysis was performed using the "ConsensusClusterPlus" package, and DEGs between the various clusters were found using the "limma" package (adjusted P < 0.05).
Functional enrichment analysis
The "clusterProfiler" package was used to perform Gene Ontology (GO), Kyoto Encyclopedia of Genes and Genomes (KEGG), and Gene Set Enrichment Analysis (GSEA) based on the DEGs between various clusters. The "Hallmarks" pathway was chosen for GSEA.
Immune landscape analysis
We used single sample gene set enrichment analysis (ssGSEA) through "GSVA" package22 to evaluate the difference of immune cell infiltration and alternations of immune-related pathways between different clusters.
Kaplan–Meier (KM) plotter database
The Kaplan Meier plotter (http://kmplot.com/analysis) online database was used to analysis the relationship between IFI16 and immunotherapy outcomes. The hazard ratio (HR) with 95% confidence intervals and log rank P value were calculated.
Immunohistochemistry staining
Colon tissue samples were fixed in 4% paraformaldehyde and subsequently embedded in paraffin. The tissue was sliced into 4 μm thickness for immunostaining. Sections were used for analyzing the expression of IFI16 with IFI16 Rabbit mAb (1:400 dilution; 14970, Cell Signaling Technology, MA, USA). Images were captured by CX40 optical microscope (Olympus, Tokyo, Japan) and and three fields were randomly selected for analysis. The expression of IFI16 in each of the randomly selected fields was quantified using ImageJ.
Statistical analysis
The statistical analyzes were performed in R software (version 3.6.0) and GraphPad Prism (version 9.0.0). Use the “ggplot2” package (Version 3.3.3) to visualize the data, and choose the appropriate statistical methods based on the features of the data format for statistical analysis (“stats” package and “car” package). Wilcoxon rank-sum test or T-test was used to analyze the continuous variables according to the normality. Fisher's exact test was used to examine the differences of categorical variables. P < 0.05 was considered statistically significant.
Ethics statement
The studies involving human participants were reviewed and approved by Ethics Committee of the Second Hospital of Hebei Medical University. The patients provided their written informed consent to participate in this study.
Results
Identification of cGAS-STING pathway expression landscape in patients with UC
To compare mRNA expression differences between UC patients and controls, datasets GSE38713 and GSE47908 were combined. Batch effect elimination was performed on the merged dataset before data analysis (Supplementary Fig. S1). In a comparative analysis of mucosal gene expression between UC patients and controls, the volcano map and heat map respectively display the cGAS-STING pathway expression landscape (Fig. 1A–D). A total of 18 genes were selected for further investigation based on the overlapping genes found in all three circumstances (Fig. 1E). In summary, patients with UC showed decreased mRNA expression of DTX4, ENPP1, TRAF6, and increased mRNA expression of TMEM173 (STING), IFI16, CXCL11, CXCL9, PARP1, RELA, CXCL10, TBK1, TRIM21, MB21D1 (CGAS), MAP3K14, NLRC3, MRE11A, ATM.
Identification of hub genes and diagnositc value
We established interactive associations between 18 DEGs using the PPI network (Fig. 2A). By using Cytoscape's MCODE plug-in, we found a module made up of four hub genes. IFI16, MB21D1 (CGAS), TMEM173 (STING), and TBK1 were identified as hub genes that are up-regulated in inflammation (Fig. 2B). The diagnostic value of four hub genes for UC was next assessed, and we discovered that IFI16 has the highest value (AUCs of 0.893 and 0.922, respectively) (Fig. 2C,D).
Predictive values and expression changes of hub genes in anti-TNF treatment
Three datasets (GSE16879, GSE12251, and GSE73661) provided the data for UC patients who had received the anti-TNF therapy. To look into the relationship between hub genes expression and anti-TNF therapy, GSE16879 and GSE12251 were combined. We examined the expression of hub genes in responders and non-responders at the start of anti-TNF treatment. It was discovered that non-responders expressed IFI16, MB21D1 (CGAS), and TBK1 at higher levels than responders (Fig. 3A,B). Anti-TNF therapy was found to decrease the expressions of IFI16, and TMEM173 (STING) in the majority of responders (Fig. 3C,D). Meanwhile, IFI16 has the highest predictive value, the AUC of IFI16 reached 0.804 and 0.850 (Fig. 3E). The results suggest that poor anti-TNF therapy outcomes are associated with a high level of the cGAS-STING pathway, and IFI16 is a promising predictive biomarker for this clinical therapeutic decision.
Expression differences of hub genes between responders and non-responders with anti-TNF treatment. (A, B) Comparison of hub gene expression between responders and non-responders with anti-TNF therapy based on GSE16879 + GSE12251 datasets and GSE73661. (C, D) Comparison of hub gene expression in responders before and after treatment. (E) ROC curve show the predictive values of hub genes in the anti-TNF treatment.
Expression of IFI16 in UC patients receiving infliximab therapy
We collected colon tissue samples from 21 patients with UC to investigate the changes in IFI16 expression between infliximab responders and non-responders. The baseline characteristics of responders and non-responders are shown in Table 2. The results of the immunohistochemistry staining showed that non-responders had higher levels of IFI16 expression compared to responders (Fig. 4A,B).
Identification of different subtypes based on hub genes
A cluster analysis of UC patients undergoing anti-TNF therapy was conducted based on four hub genes. The best clusters were observed when K = 2 (Fig. 5A,B). While TMEM173 (STING) was downregulated in cluster 1, IFI16, MB21D1 (CGAS), and TBK1 were generally upregulated in cluster 1 (Fig. 5C,D). Compared to patients in cluster 1, patients in cluster 2 exhibit a greater response rate to anti-TNF treatment (Fig. 5E).
Classification of UC patients according to hub genes. (A) Using unsupervised consensus clustering, patients with UC of GSE16879 + GSE12251 are divided into two groups based on hub genes. (B) The item-consensus plot reveals that K = 2 is the best grouping. The expression of hub genes in each cluster is depicted by a heatmap (C) and box plots (D). (E) The stacked column chart shows the proportion of respondents and non-responders in two groups.
Functional enrichment and immunological infiltration analysis
Additional functional enrichment analysis was performed in order to clarify the variations in function between the two clusters (Supplementary Table S2). Multiple immune-related pathways were shown to be enhanced in cluster 1 by GO analysis, including leukocyte migration, leukocyte activation involved in immune response, immune receptor activity, macrophage activation, and inflammsome complex (Fig. 6A). Based on KEGG pathway analysis, TNF signaling pathway, Toll-like receptor signaling pathway, and NOD-like receptor signaling pathway were all active in cluster 1 (Fig. 6B). According to the GSEA results, cluster 1 had increased levels of INFLAMMATORY_RESPONSE, ALLOGRAFT REJECTION, INTERFERON_GAMMA_RESPONSE, EPITHELIAL MESENCHYMAL TRANSITION and TNFA_SIGNALING_VIA_NFKB, while cluster 2 had increased levels of SPERMATOGENESIS, FATTY_ACID_METABOLISM, PANCREAS BETA CELLS, E2F TARGETS and OXIDATIVE_PHOSPHORYLATION (Fig. 6C,D).
Immune infiltration analysis showed similar results. Using ssGSEA, the abundance of 28 immune cells was calculated. Cluster 1 was concentrated in both innate and adaptive immune cells (Fig. 7A). Similarly, cluster 1 was enriched in several immune-related pathways, including those that promote inflammation and the IFN response (Fig. 7B). After examining the connection between immune infiltration and hub gene expression, we discovered that IFI16 is linked to the greatest level of immune cell infiltration (Fig. 7C). According to these investigations, cluster 1 exhibited a higher immunological burden and degree of inflammation than cluster 2.
cGAS-STING may inhibit cancer development by enhancing immune infiltration
The development of colitis-associated colorectal cancer (CAC) is one of the most serious outcomes of UC. We investigated the expression of hub genes associated with the inflammation-carcinogenesis process. Interestingly, we found that the expression of the cGAS-STING pathway actually decreased during the process of colitis-carcinoma transformation. Particularly, the gene expression of IFI16 (murine homolog IFI204) was significantly decreased (Fig. 8A). This phenomenon also occurs in sporadic rectal cancer (Fig. 8B). The Kaplan–Meier survival curve demonstrates that patients with high expression of IFI16 have better overall survival (Fig. 8C). Additionally, immune infiltration analysis reveals that IFI16 is associated with more abundant immune infiltration (Fig. 8D). It is widely recognized that hot tumors with abundant immune infiltration are crucial for the effectiveness of immunotherapy. Therefore, we hypothesize that IFI16 could serve as a biomarker for the application of immunotherapy. To explore this further, we examined the relationship between IFI16 and immunotherapy outcomes using the KM plotter database. The results indicate that patients with high IFI16 expression who received PD-1 and CTLA4 monoclonal antibodies had improved overall survival and progression-free survival (Fig. 8E). These findings suggest that IFI16 may play a role in regulating tumor progression through the immune microenvironment.
The role of cGAS-STING pathway-related hub genes in cancer. (A) Expression changes of hub genes during colitis-carcinoma transformation in mice and humans. (B) Expression of IFI16 in tumor tissues from the rectal cancer database. (C) Survival curves for overall survival. (D) Immune cell infiltration analysis. (E) The K–M survival curve shows that patients received immunotherapy with high expression of IFI16 are associated with better prognosis.
Discussion
The cGAS-STING pathway is crucial for mammalian cells to detect exogenous DNA and activate innate immune responses. This pathway is widely associated with various pathophysiology processes such as infections, stress, and inflammation23. UC is a result of genetic susceptibility, environmental factors, and gut microbiota interactions, leading to a weakening of the intestinal barrier function and activation of the intestinal immune response24. It has been found that cGAS-STING pathway is correlated with the development of UC14,25,26. Therefore, this study explored the role of cGAS-STING pathway-related genes in the process of UC. Furthermore, we also discovered that these genes play a significant role in predicting drug efficacy and the progression of colitis-carcinoma transformation.
Firstly, we searched for 30 genes related to the cGAS-STING pathway by retrieving relevant papers. Meanwhile, we screened the RNA sequencing data of UC from the GEO database. From the sequencing data, we identified 18 DEGs that overlapped with the cGAS-STING pathway-related genes. Based on these genes, we comprehensively mapped the cGAS-STING pathway in UC, suggesting that these genes may play important roles in the development of UC. Mechanistically, cGAS is a primary sensor of cytosolic DNA. Upon binding dsDNA, cGAS dimers assemble on dsDNA, resulting in enzymatic activation of cGAS and synthesis of cGAMP. cGAMP binds to STING and activates tank-binding kinase 1 (TBK1), promoting the autophosphorylation of TBK1, phosphorylation of STING at Ser366, and recruitment of interferon regulatory factor 3 (IRF3), then induce the release of IFN-I, NF-κB mediated proinflammatory cytokines and chemokines8,9. DTX4 is a negative regulator of TBK1. Previous studies have shown that NLRP4 recruited the E3 ubiquitin ligase DTX4 to TBK1 for Lys48 (K48)-linked Lys670 polyubiquitination, which led to the degradation of TBK127. Additionally, ENPP1 regulates extracellular cGAMP to reduce STING signaling28. Consistent with our study, these genes may be involved in cGAS-STING signaling pathways as negative regulators in UC. However, the expression of STAT6 varied in different databases. It has been reported that activation of the cGAS-STING pathway recruits STAT6 to the endoplasmic reticulum, resulting in STAT6 phosphorylation by TBK1 and Tyr(641) at Ser(407) and subsequent transfer to the nucleus to mediate immune signaling29. STAT6 activation has been detected in the inflamed colonic epithelium of patients with active inflammatory bowel disease (IBD). By weakening the function of intestinal epithelial barrier, non-hematopoietic STAT6 increases intestinal permeability, aggravates experimental colitis induced by dextran sulphate sodium (DSS) and further leads to CAC30. In conclusion, our study provides a comprehensive summary and identification of genes associated with the cGAS-STING pathway in UC. However, the role of these genes in UC has not been fully revealed, and further research in the future holds significant clinical value.
Next we identified four hub genes associated with the cGAS-STING pathway in UC, including IFI16, MB21D1 (CGAS), TMEM173 (STING), and TBK1. MB21D1 (CGAS), TMEM173 (STING), and TBK1 are considered to be the key signaling molecules in cGAS-STING pathway31. Recent studies have shown that the cGAS-STING pathway is activated in patients with UC32. Animal experiments found that dysbiosis can promote the accumulation of STING in intestinal myeloid cells, resulting in intestinal inflammation14. These results suggested that the cGAS-STING pathway may play an important role in UC. In addition, IFI16 (interferon-γ inducible protein 16) belongs to the PYHIN-200 family, which encodes evolutionarily related human proteins33. The murine homolog of IFI16 is IFI204 (p204)34. While there have been numerous studies on the function of cGAS in recognizing exogenous DNA, fewer studies have focused on the function of IFI16 in the cGAS-STING-TBK1 pathway. It has been found that IFI16 and cGAS work synergistically to promote STING activation. Moreover, IFI16 is essential for cGAMP-stimulated downstream signaling, which promotes TBK1 recruitment and activation in the STING complex35,36. Given the unclear pathogenesis and complexity of UC, diagnosing the disease remains challenging. Therefore, convenient and economical diagnostic methods are urgently needed. In our analysis, we found that IFI16 had the highest diagnostic efficacy among the four hub genes for UC. Therefore, IFI16 is expected to be a biomarker for the diagnosis of UC. Additionally, IFI16 has the potential to serve as a diagnostic and prognostic biomarker for various diseases, including tumors and rheumatoid arthritis37,38.
Early identification of non-responders in UC treatment remains a challenge that needs to be addressed. Our study focused on examining the expression of hub genes in patients who responded or did not respond to anti-TNF therapy. We found that IFI16 exhibited the highest predictive value in determining whether a patient would respond to the treatment or not. Furthermore, we observed a decrease in the expression of genes in responders during treatment, which could be attributed to the inhibition of the cGAS-STING pathway by anti-TNF drugs. Notably, a clinical study found that Anti-IFI16 IgG titers may be of great clinical relevance. Patients with low Anti-IFI16 IgG titers prior to infliximab treatment demonstrated a significantly higher probability of clinical response or remission39. Subsequently, we validated this finding using human samples and discovered that non-responders had higher levels of IFI16 protein compared to responders. These findings suggest that IFI16 has the potential to serve as a promising biomarker in the future.
Different sets of functional genes can be used to group samples, with each cluster having distinct pathogenic mechanisms and clinical prognostic features40,41. Through unsupervised consensus clustering, we have identified two subtypes based on cGAS-STING pathway-associated genes. Cluster1, which has a high proportion of non-responders to anti-TNF therapy, exhibits abundant immune cell infiltration, activated immune responses, and inflammatory responses. These findings indicate that cGAS-STING pathway-related genes are involved in immune regulation in UC.
CAC is the most serious complications of UC. The process of CAC typically involves inflammation, dysplasia, and carcinoma. Inflammation is the initiator of CAC, while abnormal immune response is the trigger of inflammation-carcinogenesis transformation42,43. Encouragingly, the incidence of CAC appears to be decreasing over time, which could potentially indicate advancements in medical treatment and colonoscopy44,45,46. However, it is important to note that CAC still remains the primary cause of death and colectomy in patients with UC42. Interestingly, the cGAS-STING signaling pathway may have different roles in inflammation and cancer. On the one hand, activation of cGAS-STING signaling is associated with the severity of intestinal inflammation. On the other hand, it plays a key role in preventing tumorigenesis and infection13. Colorectal cancer (CRC) patients with high STING expression had increased intratumoral CD8+ T cells infiltration and decreased frequency of lymphovascular infiltration. Additionally, patients with higher STING expression had longer overall survival and recurrence-free survival compared to those with lower STING expression47. Another study found that STING−/− mice alleviated colitis but exacerbated the development of CAC. STING inhibited the development of CAC by regulating tumor cell proliferation, adhesion, invasion and inflammatory responses48. This is consistent with our finding that IFI16 expression decreases in inflammation-carcinogenesis transformation process. Furthermore, tumor patients with high IFI16 expression had better overall survival and immunotherapy outcomes. In conclusion, we found that cGAS-STING pathway-related genes may play different roles in colitis and tumors. IFI16 is expected to be a biomarker for predicting the efficacy of immunotherapy in CRC patients.
The mechanism of cGAS-STING pathway in UC has not been fully elucidated. In this study, we investigated the role and function of cGAS-STING pathway in UC at the gene level. We also demonstrated its potential therapeutic predictive value and provided a new perspective for further research. However, our study has some limitations. Although we observed that cGAS-STING pathway-related genes might have different roles in UC and CAC, we did not further explore the underlying reasons in depth. Our study is mainly based on online database, and we will probably further clarify the roles of cGAS-STING pathway genes in UC and CAC by cell and animal experiments in the future.
Conclusion
In conclusion, our study systematically summarized the cGAS-STING pathway-related genes in UC and identified IFI16, MB21D1 (CGAS), TMEM173 (STING), and TBK1 as hub genes. These hub genes are associated with the immune landscape, subtypes of UC patients and the effects of drug treatment. Notably, we identified IFI16 as a promising biomarker for predicting the efficacy of anti-TNF therapy and might playing an important role in CAC. Our findings provide new insights for exploring the molecular mechanisms and therapy of UC and cancer. Further exploration and validation of mechanisms will be necessary.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
Abbreviations
- UC:
-
Ulcerative colitis
- TNF:
-
Tumour necrosis factor
- IFN-I:
-
Type I interferon
- cGAS:
-
Cyclic GMP-AMP synthase
- STING:
-
Stimulator of interferon genes
- GEO:
-
Gene expression omnibus
- DEGs:
-
Differentially expressed genes
- MCODE:
-
Molecular complex detection
- PPI:
-
Protein–protein interaction
- ROC:
-
Receiver operating characteristic curves
- AUC:
-
Area under the curve
- GO:
-
Gene ontology
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- GSEA:
-
Gene set enrichment analysis
- ssGSEA:
-
Single sample gene set enrichment analysis
- HR:
-
Hazard ratio
- CAC:
-
Colitis-associated colorectal cancer
- TBK1:
-
Tank-binding kinase 1
- IRF3:
-
Interferon regulatory factor 3
- IBD:
-
Inflammatory bowel disease
- DSS:
-
Dextran sulphate sodium
- IFI16:
-
Interferon-γ inducible protein 16
- CRC:
-
Colorectal cancer
References
Le Berre, C., Honap, S. & Peyrin-Biroulet, L. Ulcerative colitis. Lancet 402(10401), 571–584 (2023).
Qiu, Y. et al. Systematic review with meta-analysis: Loss of response and requirement of anti-TNFα dose intensification in Crohn’s disease. J. Gastroenterol. 52(5), 535–554 (2017).
Singh, S. et al. First- and second-line pharmacotherapies for patients with moderate to severely active ulcerative colitis: An updated network meta-analysis. Clin. Gastroenterol. Hepatol. 18(10), 2179–2191 (2020).
Bressler, B. Is there an optimal sequence of biologic therapies for inflammatory bowel disease?. Ther. Adv. Gastroenterol. 16, 17562848231159452 (2023).
Zhang, L. et al. Early-life exposures and the microbiome: implications for IBD prevention. Gut (2023).
Danne, C. et al. Neutrophils: From IBD to the gut microbiota. Nat. Rev. Gastroenterol. Hepatol. 21(3), 184–197 (2024).
Ablasser, A. & Chen, Z. J. cGAS in action: Expanding roles in immunity and inflammation. Science 363, 6431 (2019).
Chen, C. & Xu, P. Cellular functions of cGAS-STING signaling. Trends Cell Biol. 33(8), 630–648 (2023).
Decout, A. et al. The cGAS-STING pathway as a therapeutic target in inflammatory diseases. Nat. Rev. Immunol. 21(9), 548–569 (2021).
Zhou, J. et al. Significance of the cGAS-STING pathway in health and disease. Int. J. Mol. Sci. 24(17), 13316 (2023).
Skopelja-Gardner, S., An, J. & Elkon, K. B. Role of the cGAS-STING pathway in systemic and organ-specific diseases. Nat. Rev. Nephrol. 18(9), 558–572 (2022).
Mullard, A. Biotechs step on cGAS for autoimmune diseases. Nat. Rev. Drug Discov. 22(12), 939–941 (2023).
Yang, Y. et al. cGAS-STING signaling pathway in intestinal homeostasis and diseases. Front. Immunol. 14, 1239142 (2023).
Shmuel-Galia, L. et al. Dysbiosis exacerbates colitis by promoting ubiquitination and accumulation of the innate immune adaptor STING in myeloid cells. Immunity 54(6), 1137-1153.e8 (2021).
Planell, N. et al. Transcriptional analysis of the intestinal mucosa of patients with ulcerative colitis in remission reveals lasting epithelial cell alterations. Gut 62(7), 967–976 (2013).
Bjerrum, J. T. et al. Transcriptional analysis of left-sided colitis, pancolitis, and ulcerative colitis-associated dysplasia. Inflamm. Bowel Dis. 20(12), 2340–2352 (2014).
Zhang, Y. et al. MAPK8 and CAPN1 as potential biomarkers of intervertebral disc degeneration overlapping immune infiltration, autophagy, and ceRNA. Front. Immunol. 14, 1188774 (2023).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43(7), e47 (2015).
Arijs, I. et al. Mucosal gene expression of antimicrobial peptides in inflammatory bowel disease before and after first infliximab treatment. PLoS ONE 4(11), e7984 (2009).
Arijs, I. et al. Mucosal gene signatures to predict response to infliximab in patients with ulcerative colitis. Gut 58(12), 1612–1619 (2009).
Arijs, I. et al. Effect of vedolizumab (anti-α4β7-integrin) therapy on histological healing and mucosal gene expression in patients with UC. Gut 67(1), 43–52 (2018).
Hänzelmann, S., Castelo, R. & Guinney, J. GSVA: Gene set variation analysis for microarray and RNA-seq data. BMC Bioinform. 14, 7 (2013).
Wottawa, F. et al. The role of cGAS/STING in intestinal immunity. Eur. J. Immunol. 51(4), 785–797 (2021).
Ramos, G. P. & Papadakis, K. A. Mechanisms of disease: Inflammatory bowel diseases. Mayo Clin. Proc. 94(1), 155–165 (2019).
Aden, K. et al. ATG16L1 orchestrates interleukin-22 signaling in the intestinal epithelium via cGAS-STING. J. Exp. Med. 215(11), 2868–2886 (2018).
Flood, P. et al. DNA sensor-associated type I interferon signaling is increased in ulcerative colitis and induces JAK-dependent inflammatory cell death in colonic organoids. Am. J. Physiol. Gastrointest. Liver Physiol. 323(5), G439–G460 (2022).
Cui, J. et al. NLRP4 negatively regulates type I interferon signaling by targeting the kinase TBK1 for degradation via the ubiquitin ligase DTX4. Nat. Immunol. 13(4), 387–395 (2012).
Carozza, J. A. et al. ENPP1’s regulation of extracellular cGAMP is a ubiquitous mechanism of attenuating STING signaling. Proc. Natl. Acad. Sci. USA 119(21), e2119189119 (2022).
Chen, H. et al. Activation of STAT6 by STING is critical for antiviral innate immunity. Cell 147(2), 436–446 (2011).
Lin, Y. et al. Non-hematopoietic STAT6 induces epithelial tight junction dysfunction and promotes intestinal inflammation and tumorigenesis. Mucosal. Immunol. 12(6), 1304–1315 (2019).
Hopfner, K. P. & Hornung, V. Molecular mechanisms and cellular functions of cGAS-STING signalling. Nat. Rev. Mol. Cell Biol. 21(9), 501–521 (2020).
Chen, C. et al. Atrial natriuretic peptide attenuates colitis via inhibition of the cGAS-STING pathway in colonic epithelial cells. Int. J. Biol. Sci. 18(4), 1737–1754 (2022).
Gariglio, M. et al. The multifaceted interferon-inducible p200 family proteins: From cell biology to human pathology. J. Interferon Cytokine Res. 31(1), 159–172 (2011).
Kim, H. et al. PRMT5 control of cGAS/STING and NLRC5 pathways defines melanoma response to antitumor immunity. Sci. Transl. Med. 12, 551 (2020).
Jønsson, K. L. et al. IFI16 is required for DNA sensing in human macrophages by promoting production and function of cGAMP. Nat. Commun. 8, 14391 (2017).
Almine, J. F. et al. IFI16 and cGAS cooperate in the activation of STING during DNA sensing in human keratinocytes. Nat. Commun. 8, 14392 (2017).
Alunno, A. et al. Circulating interferon-inducible protein IFI16 correlates with clinical and serological features in rheumatoid arthritis. Arthritis Care Res. 68(4), 440–445 (2016).
Yu, B. et al. IFI16 can be used as a biomarker for diagnosis of renal cell carcinoma and prediction of patient survival. Front. Genet. 12, 599952 (2021).
Caneparo, V. et al. Distinct anti-IFI16 and anti-GP2 antibodies in inflammatory bowel disease and their variation with infliximab therapy. Inflamm. Bowel Dis. 22(12), 2977–2987 (2016).
Karim, M. R. et al. Deep learning-based clustering approaches for bioinformatics. Brief. Bioinform. 22(1), 393–415 (2021).
Gao, X. et al. Pyroptosis burden is associated with anti-TNF treatment outcome in inflammatory bowel disease: New insights from bioinformatics analysis. Sci. Rep. 13(1), 15821 (2023).
Shah, S. C. & Itzkowitz, S. H. Colorectal cancer in inflammatory bowel disease: Mechanisms and management. Gastroenterology 162(3), 715–730 (2022).
Rajamäki, K. et al. Genetic and epigenetic characteristics of inflammatory bowel disease-associated colorectal cancer. Gastroenterology 161(2), 592–607 (2021).
Jess, T. et al. Cancer risk in inflammatory bowel disease according to patient phenotype and treatment: A Danish population-based cohort study. Am. J. Gastroenterol. 108(12), 1869–1876 (2013).
Choi, C. H. et al. Forty-year analysis of colonoscopic surveillance program for neoplasia in ulcerative colitis: An updated overview. Am. J. Gastroenterol. 110(7), 1022–1034 (2015).
Mozooni, Z. et al. The role of interferon-gamma and its receptors in gastrointestinal cancers. Pathol. Res. Pract. 248, 154636 (2023).
Chon, H. J. et al. STING signaling is a potential immunotherapeutic target in colorectal cancer. J. Cancer 10(20), 4932–4938 (2019).
Gong, W. et al. STING-mediated Syk signaling attenuates tumorigenesis of colitis-associated colorectal cancer through enhancing intestinal epithelium pyroptosis. Inflamm. Bowel Dis. 28(4), 572–585 (2022).
Acknowledgements
We thank the database for its open source, and also express our gratitude to the patients who provided the samples for research.
Funding
This work was supported by the National Natural Science Foundation of China (No. 82270545 and No. 82070563) and Hebei Province Graduate Innovation Funding Project (No. CXZZBS2023114).
Author information
Authors and Affiliations
Contributions
XL. Z. were involved in project conceiving and funding acquisition, C. W. and X. G were involved in data collection, data curation, investigation, formal analyses, software performance, visualization, experimental analysis and original manuscript writing. YC. L., CY. L., ZM. M., DL. S. and XN. L. were involved in study supervision, investigation, and manuscript editing. All of authors read and approved the final manuscript. All authors have read and approved the content and agree to submit for consideration for publication in the journal.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, C., Gao, X., Li, Y. et al. A molecular subtyping associated with the cGAS-STING pathway provides novel perspectives on the treatment of ulcerative colitis. Sci Rep 14, 12683 (2024). https://doi.org/10.1038/s41598-024-63695-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-63695-4
- Springer Nature Limited