Abstract
Guillain–Barré syndrome (GBS) is an acute autoimmune polyneuropathy with substantial geographic variations in demography, antecedent events, clinical manifestations, electrophysiological sub-types, diagnostic findings, treatment modalities, and prognostic indicators. However, there is limited contemporary data on GBS patient profiles and prognostic factors from low-resource settings like Ethiopia. The objective of this study is to investigate the clinical profile, factors associated with mortality, and hospital outcomes among GBS patients admitted to Tikur Anbessa Specialized Hospital (TASH) in Addis Ababa, Ethiopia. A retrospective cross-sectional study was conducted among 60 GBS patients admitted to TASH from January 2018 to December 2022. Data on demographics, clinical features, treatments, complications, and outcomes were extracted from medical records. Bivariate and multivariate logistic regression analyses identified factors associated with mortality and poor hospital outcomes. The cohort had a mean age of 28.5 years, with 76.7% aged 14–34 years. Males comprised 61.7% of cases. Ascending paralysis (76.7%) was the predominant presentation. Absent or reduced reflexes were seen in 91.7% of patients. The most common antecedent event was gastroenteritis (26.7%), followed by upper respiratory tract infection (URTI) (15%) and vaccination (11.7%). The mean interval from symptom onset to hospital presentation was 8.77 days, and the peak symptom severity was 4.47 days. The axonal variant (75.5%) was the most common subtype, followed by the demyelinating variant (24.5%). Intravenous immunoglobulin was administered to 41.7% of patients. Respiratory failure requiring invasive mechanical ventilator (MV) support occurred in 26.7% of cases. The mortality rate was 10%, with mechanical ventilation being the only factor significantly associated with mortality (95% CI 2.067–184.858; P < 0.010). At discharge, 55% had a good outcome, and 45% had a poor outcome, according to the Hughes Functional Disability Scale (HFDS). Mechanical ventilation (AOR 0.024, 95% CI 0.001–0.607) and a GBS disability score > 3 (AOR 0.106, 95% CI 0.024–0.467) were factors significantly associated with poor hospital outcomes. GBS in this cohort primarily affected individuals of young age, commonly preceded by gastroenteritis and characterized by a high frequency of the axonal variant. Mechanical ventilation was found to be significantly linked to mortality. Alongside mechanical ventilation requirements, severe disability upon presentation emerged as a crucial determinant of poor outcomes upon discharge, underscoring the importance of early identification of high-risk patients and prompt interventions.
Similar content being viewed by others
Introduction
Guillain–Barré syndrome (GBS) is an acute polyradiculoneuropathy characterized by immune-mediated damage to the peripheral nervous system, leading to varying degrees of motor dysfunction, sensory impairment, and autonomic instability 1. It represents the most common cause of acute flaccid paralysis globally, exerting a substantial burden on healthcare systems due to the intensity of care required during the acute phase and the long-term rehabilitation requirements 2,3. Epidemiological data from North America and Europe indicate an annual incidence of GBS ranging from 0.8 to 1.9 cases per 100,000 person-years 4.
GBS exhibits notable variations in incidence, demographic distribution, preceding events, clinical manifestations, electrophysiological subtypes, diagnostic approaches, therapeutic interventions, and prognostic outcomes across different geographical regions 5,6,7,8. These variations can be attributed to multifaceted factors. Firstly, regional differences in the prevalence and strains of infectious agents such as cytomegalovirus (CMV), Epstein-Barr virus (EBV), and Campylobacter contribute to regional discrepancies in GBS incidence rates 9. Moreover, variations in hygiene practices across regions affect exposure to these pathogens, potentially influencing GBS development 10. Dietary habits and nutrient deficiencies also affect disease progression 11. Environmental factors unique to specific regions also serve as potential triggers for the onset of GBS 12. Furthermore, genetic variations among populations influence susceptibility to GBS and disease severity 12,13. In regions with limited access to advanced diagnostic tools, underdiagnosis or misdiagnosis of GBS subtypes may occur, impacting reported incidence rates 10. Moreover, slight differences in diagnostic criteria and disease reporting practices across regions further complicate the accurate assessment of GBS burden 5,10. The absence of affordable and effective treatments significantly worsens outcomes in low- and middle-income countries. Furthermore, socioeconomic factors such as poverty, inadequate infrastructure, and healthcare disparities further compound the difficulties in accessing timely and appropriate care 10.
Most comprehensive studies investigating GBS patient profiles and outcomes originate from high-income regions, particularly North America and Europe. Consequently, there exists a need for more contemporary data on GBS from low- and middle-income countries, including Africa, with limited representation from Ethiopia, thereby impeding a comprehensive understanding of geographical variations in the disease. Moreover, existing studies from the region need to be updated, to accurately depict the current GBS landscape in Ethiopia.
This study aims to address this gap in the literature by thoroughly investigating the clinical profile and factors associated with mortality and hospital outcomes among patients diagnosed with GBS admitted to Tikur Anbessa Specialized Hospital (TASH), Ethiopia. By elucidating the contemporary epidemiological, clinical, and prognostic features of GBS in the Ethiopian context, this research endeavors to provide invaluable insights into managing and treating the condition within the local healthcare setting.
Methodology
Study design and setting
A retrospective cross-sectional chart review study was conducted at TASH, focusing on patients admitted to the medical intensive care unit (MICU) and medical ward who were diagnosed with GBS during the period from January 1, 2018, to December 30, 2022. The inclusion criteria encompassed patients aged 14 years and older whose clinical records provided comprehensive information. Excluded from the study were individuals with missing and incomplete medical documentation. Data encompassing clinical and paraclinical variables, inclusive of sociodemographic factors, primary presenting symptoms, symptom and in-hospital stay duration, antecedent events, complications, utilized treatment modalities, mechanical ventilation requirement, and investigation outcomes such as lumbar puncture cytochemistry and nerve conduction studies, were obtained.
Patients were stratified based on GBS diagnostic certainty as per Brighton’s criteria 14, alongside their functional status at hospital admission, assessed utilizing the Hughes Functional Disability Scale (HFDS), also known as the GBS disability score 15,16 (see Supplementary Table S1). The classification of patients' nerve conduction studies into electrophysiological variants of GBS relied on Rajabally's electrophysiological criteria following a single nerve conduction study 17.
Operational definitions
In our study, dysautonomia is defined by the presence of blood pressure fluctuations (hypertension or hypotension), occurrences of postural hypotension (a drop of 20 mmHg in systolic blood pressure or 10 mmHg in diastolic blood pressure within 5 min of rising from a supine or seated position), and manifestations of cardiac dysrhythmias (tachycardia or bradycardia) attributable solely to autonomic nervous system dysfunction 18,19. Assessment of the need for mechanical ventilator support encompassed evaluations of respiratory rate, single breath count, incapacity to lift the head, and oxygen saturation levels. A poor outcome was identified by the inability to ambulate independently, denoted by a GBS disability score of 3 or higher upon hospital discharge 20.
Ethical approval
The present research received ethical clearance from the Institution of Health Research Ethics Review Committee of Tikur Anbessa Specialized Hospital, Internal Medicine Department. The study was conducted in strict accordance with the relevant guidelines and regulations set forth by the committee. Informed consent was waived by the Institutional Health Research Ethics Review Committee of TASH due to the retrospective nature of the study, following established protocols.
Statistical analysis
We utilized SPSS version 26 for data analysis. Before analysis, data completeness was ensured. Socio-demographic characteristics were presented in tabular format, detailing both numbers and percentages. A bivariate analysis was conducted to identify independent variables at a significance level of 5%, which were subsequently incorporated into the multivariate binary logistic regression analysis. In the multivariate logistic regression, a 95% confidence interval was calculated for the adjusted odds ratio (AOR), with variables exhibiting a p-value ≤ 0.05 considered statistically associated with poor hospital outcomes among GBS patients.
Ethics approval and consent to participate
Ethical clearance for the study was obtained from the Institution of Health Research Ethics Review Committee of Tikur Anbessa Specialized Hospital, Internal Medicine Department. Officials at various levels within the study area were duly informed through official letters issued by the Internal Medicine Department. Throughout the study, strict measures were implemented to uphold the confidentiality of collected information, and the privacy of participants was meticulously maintained, ensuring compliance with ethical standards and safeguarding the rights of all involved individuals. Informed consent was waived due to the retrospective nature of the study by the Institutional Health Research Ethics Review Committee of TASH.
Results
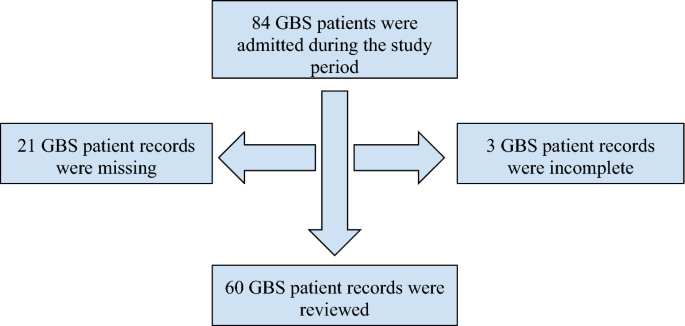
During the study period spanning from January 2018 to December 2022, a total of 60 GBS patient charts were thoroughly reviewed and included in the analysis for the study (see Fig. 1).
Sociodemographic and clinical profile of patients
The study exhibited a mean age of 28.5 ± 12.5 years, ranging from 14 to 70 years. The male-to-female ratio was calculated as 1.61, with males comprising 37 individuals (61.7%). Analysis of the age distribution revealed that most cases, comprising 46 (76.7%), fell within the age bracket of 14–34 years (see Table 1).
Ascending weakness emerged as the predominant presenting symptom among GBS patients, accounting for 46 (76.7%) cases. Bulbar nerve involvement (cranial nerves IX and X) resulting in dysphagia was observed in 11 patients (18.3%), while cranial nerve VII involvement causing facial palsy was noted in 6 patients (10%). Details are provided in Table 1.
The primary antecedent event identified in this study was gastroenteritis, observed in 16 (26.7%) cases. Post-vaccination GBS was seen in 7 (11.7%) cases. Of the 7 vaccination instances, 6 pertained to anti-rabies vaccines and 1 to the COVID-19 vaccine. Notably, COVID-19 infection preceded the onset of GBS in 1 patient. Conversely, 27 (45%) patients exhibited no antecedent infection. 16 (26.7%) patients required mechanical ventilation (see Table 1). Additionally, six patients presented with comorbid illnesses, including 4 cases of hypertension (HTN), 1 case of dilated cardiomyopathy (DCMP), and 1 case of chronic myeloid leukemia (CML).
The mean interval from the onset of symptoms to presentation at the hospital was 8.77 (± 7.25) days, ranging from 1 to 40 days. Additionally, the mean duration from the initial symptom to peak symptomatology was 4.47 (± 4.78) days, ranging from 1 to 21 days. Hospitalization durations varied widely, ranging from 2 to 180 days, with a mean stay of 26.08 (± 31.08) days. Among the 16 patients who required mechanical ventilation (MV) support, the mean duration of MV support was 25.50 (± 18.79) days, ranging from 8 to 82 days.
Diagnosis, laboratory, and nerve conduction profile of patients
Regarding the laboratory tests, lumbar puncture was conducted on 47 patients, revealing albuminocytological dissociation in 39 cases (82.9%). Nerve conduction studies were performed on 45 individuals. The predominant GBS variant observed in this study was the axonal variant, present in 34 out of 45 cases (75.5%), followed by the demyelinating variant in 11 out of 45 cases (24.5%). Among the axonal variant cases, 28 cases (82.3%) were classified as acute motor axonal neuropathy (AMAN), while 6 cases (17.7%) were classified as acute motor and sensory axonal neuropathy (AMSAN). None of the 33 patients who underwent serological testing for HIV yielded reactive results (see Table 2).
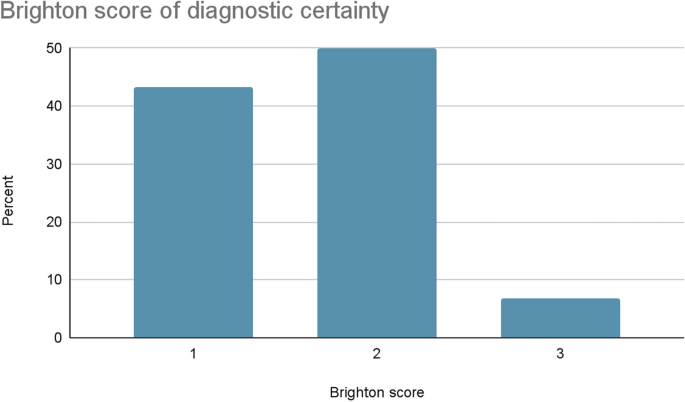
The diagnostic certainty of patients in this study is depicted in Fig. 2. A Brighton score of 2 was the most common score, observed in half of the patients, totaling 30 cases (50%). Similarly, nearly half of the patients had a Brighton score of 1, comprising 26 cases (43.3%).
Treatment and outcomes of patients
Intravenous immunoglobulin (IVIg) treatment was administered to 25 patients, accounting for 41.7% of the cohort. Additionally, one patient received steroids for a severe hospital-acquired infection, while none of the patients underwent plasmapheresis. Notably, specific treatment was not provided to 34 patients (56.7%), with only supportive care being administered.
Upon bivariate logistic regression analysis, IVIg treatment did not demonstrate an association with either death (p = 0.22) or hospital outcome (p = 0.90). Furthermore, a Mann–Whitney U test revealed that the length of hospital stays for patients receiving IVIg (mean rank = 34.9 days) was not significantly different from those not receiving IVIg (mean rank = 27.3 days), with a p-value of 0.096).
Despite a shorter duration of mechanical ventilation support observed in patients who received IVIg (mean = 20.6 days, SD = 11.7 days) compared to those who did not (mean = 30.3 days, SD = 23.7 days), this difference was not statistically significant according to t-test analysis (t(16) = − 1.041, p = 0.316).
The hospital mortality rate among patients diagnosed with GBS in this study was determined to be 10%, with 6 out of 60 patients succumbing to their condition. The causes of death were attributed to sudden cardiac arrest in 3 patients, respiratory arrest in 2 patients, and uncontrolled urosepsis in 1 patient. Notably, the requirement for mechanical ventilation support was significantly associated with death on bivariate analysis (5 out of 6 cases; 95% CI 2.067–184.858; p < 0.010).
Common complications observed in this study included infections in 19 cases (31.7%), comprising catheter-associated urinary tract infections (CA-UTI) in 12 cases, hospital-acquired pneumonia (HAP) in 10 cases, COVID-19 infection in 1 case, and thrombophlebitis in 1 case. Autonomic dysfunction was noted in 17 cases (28.3%), while bed sores were observed in 4 cases (6.7%). Additionally, tracheoesophageal fistula (TEF) occurred in 3 cases (5%), and pneumothorax was documented in 2 cases (3.3%).
Among the total of 60 patients admitted in this study, 33 patients (55%) had a good outcome at discharge, while 27 patients (45%) experienced a poor outcome, as indicated by a high Hughes score.
Factors associated with poor hospital outcome (high Hughes score) at discharge
In bivariate binary logistic regression analysis conducted at a 95% level of significance (p < 0.05), several factors were identified as significantly associated with poor hospital outcomes. These factors included respiratory failure at presentation, the requirement for MV support, autonomic dysfunction, infection, and a GBS functional disability score > 3 at admission, as delineated in Table 3.
However, upon conducting multivariable binary logistic regression analysis, only the need for MV support and a GBS functional disability score > 3 at admission were found to be significantly associated with a poor hospital outcome at discharge (p < 0.05).
Limitations
The limitation of our study is its retrospective nature, relying on chart reviews, which are contingent upon the accuracy and completeness of documentation. Additionally, the relatively small sample size represents another limitation, diminishing the statistical power of the findings and impeding their generalizability to broader patient populations.
Discussion
Sociodemographic and clinical profiles of patients
GBS affects all age groups, with prevalence generally increasing with age 21,22. While common in children, it is less frequent than in adults 23. Notably, studies show a bimodal distribution of the disease 22,24. The first peak occurs between ages 15 and 34, a trend corroborated by our study. The second peak occurs after age 50. Some studies reported mean ages of 30 and 29.3 years 25,26. Conversely, others have documented comparatively older mean ages, ranging from 40.69 to 52.6 years 22,27,28,29,30. The age-related variations in GBS may stem from immune system changes 31, declining nerve repair mechanisms 32, and varied exposure to infectious agents 9.
GBS is more prevalent in males than females, with ratios ranging from 1.1:1 to 1.7:1 23,33. Interestingly, while girls and adolescent females are more likely to develop GBS, this trend reverses in older age groups 34. The higher prevalence in males may be due to sex differences in immune response, but factors like sex hormones, genetics, and environmental influences also play significant roles, warranting further investigation 33.
In our study, the predominant GBS presentation was ascending paralysis, consistent with other studies 35,36,37. The mean interval from symptom onset to hospital presentation in Ethiopia improved from 11.2 days two decades ago to 8.77 days in our study, likely due to better awareness and healthcare access 38,39. IVIg use increased to 41.7% from 6.2%, indicating improved treatment 38. However, the mean hospital stay remains longer than in Thailand (14.2 days) and the Netherlands (17 days), reflecting ongoing healthcare challenges in Ethiopia 29,40.
Albuminocytological dissociation (ACD), a hallmark diagnostic feature of GBS with reported incidences ranging from 44 to 81%, was observed in 82.9% of participants in our study 14,41,42. This high prevalence may be due to delayed healthcare presentation, lumbar puncture procedures conducted later in disease progression, and the absence of localized variants in our cohort 43,44.
In our study, the predominant variant of GBS was axonal, accounting for 75.5%. This aligns with findings from studies in northern China, India, and Mexico 26,45,46. However, it contrasts with studies in southern China, the Balkans, Wuhan-China, Thailand, and Canada, where acute inflammatory demyelinating polyneuropathy (AIDP) is more common. 14,29,47,48. The difference may be attributed to a higher prevalence of preceding gastroenteritis and a younger age distribution in our cohort, factors often associated with axonal variants.
Factors associated with mortality and poor hospital outcomes
In our study, the observed mortality rate of 10% in GBS patients aligns with the reported range (1–18%) and is higher among those requiring mechanical ventilation (12–20%) 49. Mortality was primarily associated with the need for MV, reflecting the severity of nerve involvement and risks such as ventilator-associated pneumonia (VAP) and ventilator-induced lung injury (VILI) 50,51. These complications underscore the challenges and increased mortality risks associated with MV in GBS. Additionally, a significant subset (45%) experienced poor outcomes at discharge, characterized by a GBS disability score > 3 at discharge. Factors significantly associated with a poor hospital outcome (p < 0.05) include the requirement for MV support and a GBS disability score > 3 at admission. A GBS disability score > 3 at admission can exacerbate complications like pneumonia and deep vein thrombosis (DVT) 52,53. Early mobilization and proactive management strategies are crucial to mitigate these risks and improve patient recovery and outcomes.
Conclusion
In conclusion, this retrospective cross-sectional study provides valuable insights into the contemporary clinical profile and factors influencing the outcomes and mortality of GBS patients in Ethiopia. The study addresses a notable gap in the literature by examining this neurological condition within the context of a low-resource setting. Key findings revealed a predominance of the axonal variant of GBS, with the majority of patients presenting with ascending paralysis. Mechanical ventilation requirements and a GBS disability score > 3 at admission emerged as significant risk factors associated with poor hospital outcomes. Moreover, the need for mechanical ventilation was identified as a predictor of mortality risk. While the observed overall mortality rate aligned with global estimates, a substantial proportion of discharged patients exhibited residual functional disability. These findings underscore the complexities of managing GBS and highlight the need for early identification of high-risk patients, prompt initiation of appropriate treatments, and the implementation of comprehensive rehabilitation strategies tailored to the local healthcare environment. By elucidating the challenges and prognostic factors in the Ethiopian context, this study provides a foundation for developing targeted interventions and optimizing resource allocation to improve care delivery and mitigate the burden of GBS in similar resource-constrained settings.
Data availability
The data supporting the findings of this study will be available from the corresponding author upon reasonable request.
References
van den Berg, B. et al. Guillain–Barré syndrome: Pathogenesis, diagnosis, treatment and prognosis. Nat. Rev. Neurol. 10(8), 469–482. https://doi.org/10.1038/nrneurol.2014.121 (2014).
Frenzen, P. D. Economic cost of Guillain–Barré syndrome in the United States. Neurology 71(1), 21–27. https://doi.org/10.1212/01.wnl.0000316393.54258.d1 (2008).
Kim, A.-Y., Lee, H., Lee, Y.-M. & Kang, H.-Y. Epidemiological features and economic burden of Guillain–Barré syndrome in South Korea: A nationwide population-based study. J. Clin. Neurol. Seoul Korea 17(2), 257–264. https://doi.org/10.3988/jcn.2021.17.2.257 (2021).
Abbassi, N. & Ambegaonkar, G. Guillain–Barre syndrome: A review. Paediatr. Child Health 29(11), 459–462. https://doi.org/10.1016/j.paed.2019.07.008 (2019).
Doets, A. Y. et al. Regional variation of Guillain–Barré syndrome. Brain J. Neurol. 141(10), 2866–2877. https://doi.org/10.1093/brain/awy232 (2018).
Arends, S. et al. Electrodiagnosis of Guillain–Barre syndrome in the International GBS Outcome Study: Differences in methods and reference values. Clin. Neurophysiol. Off. J. Int. Fed. Clin. Neurophysiol. 138, 231–240. https://doi.org/10.1016/j.clinph.2021.12.014 (2022).
Webb, A. J. S., Brain, S. A. E., Wood, R., Rinaldi, S. & Turner, M. R. Seasonal variation in Guillain–Barré syndrome: A systematic review, meta-analysis and Oxfordshire cohort study. J. Neurol. Neurosurg. Psychiatry 86(11), 1196–1201. https://doi.org/10.1136/jnnp-2014-309056 (2015).
Yao, J., Liu, Y., Liu, S. & Lu, Z. Regional differences of Guillain–Barré syndrome in China: From south to north. Front. Aging Neurosci. https://doi.org/10.3389/fnagi.2022.831890 (2022).
Hao, Y. et al. Antecedent infections in Guillain–Barré syndrome: A single-center, prospective study. Ann. Clin. Transl. Neurol. 6(12), 2510. https://doi.org/10.1002/acn3.50946 (2019).
Papri, N. et al. Guillain–Barré syndrome in low-income and middle-income countries: Challenges and prospects. Nat. Rev. Neurol. 17(5), 285–296. https://doi.org/10.1038/s41582-021-00467-y (2021).
Gao, Y. et al. Serum folate correlates with severity of Guillain–Barré syndrome and predicts disease progression. BioMed Res. Int. 2018, 5703279. https://doi.org/10.1155/2018/5703279 (2018).
Acosta-Ampudia, Y., Monsalve, D. M. & Ramírez-Santana, C. Identifying the culprits in neurological autoimmune diseases. J. Transl. Autoimmun. 2, 100015. https://doi.org/10.1016/j.jtauto.2019.100015 (2019).
Safa, A., Azimi, T., Sayad, A., Taheri, M. & Ghafouri-Fard, S. A review of the role of genetic factors in Guillain–Barré syndrome. J. Mol. Neurosci. MN 71(5), 902–920. https://doi.org/10.1007/s12031-020-01720-7 (2021).
Fokke, C. et al. Diagnosis of Guillain–Barré syndrome and validation of Brighton criteria. Brain J. Neurol. 137(Pt 1), 33–43. https://doi.org/10.1093/brain/awt285 (2014).
Hughes, R. A. C., Newsom-Davis, J. M., Perkin, G. D. & Pierce, J. M. Controlled trial of prednisolone in acute polyneuropathy. Lancet 312(8093), 750–753. https://doi.org/10.1016/S0140-6736(78)92644-2 (1978).
van Koningsveld, R. et al. A clinical prognostic scoring system for Guillain–Barré syndrome. Lancet Neurol. 6(7), 589–594. https://doi.org/10.1016/S1474-4422(07)70130-8 (2007).
Rajabally, Y. A., Hiew, F. L. & Winer, J. B. Influence of timing on electrodiagnosis of Guillain–Barré syndrome in the first six weeks: A retrospective study. J. Neurol. Sci. 357(1), 143–145. https://doi.org/10.1016/j.jns.2015.07.018 (2015).
Chakraborty, T., Kramer, C. L., Wijdicks, E. F. M. & Rabinstein, A. A. Dysautonomia in Guillain–Barré syndrome: Prevalence, clinical spectrum, and outcomes. Neurocrit. Care 32(1), 113–120. https://doi.org/10.1007/s12028-019-00781-w (2020).
Zaeem, Z., Siddiqi, Z. A. & Zochodne, D. W. Autonomic involvement in Guillain–Barré syndrome: An update. Clin. Auton. Res. Off. J. Clin. Auton. Res. Soc. 29(3), 289–299. https://doi.org/10.1007/s10286-018-0542-y (2019).
“Frontiers | Disability evaluation in patients with Guillain–Barre syndrome and SARS-CoV-2 infection”. https://doi.org/10.3389/fneur.2023.1191520/full (accessed 02 Apr 2024).
Sejvar, J. J., Baughman, A. L., Wise, M. & Morgan, O. W. Population incidence of Guillain–Barré syndrome: A systematic review and meta-analysis. Neuroepidemiology 36(2), 123–133. https://doi.org/10.1159/000324710 (2011).
Shrivastava, M., Nehal, S. & Seema, N. Guillain–Barre syndrome: Demographics, clinical profile and seasonal variation in a tertiary care centre of central India. Indian J. Med. Res. 145(2), 203–208. https://doi.org/10.4103/ijmr.IJMR_995_14 (2017).
Al-Hakem, H. et al. Guillain–Barré syndrome in Denmark: A population-based study on epidemiology, diagnosis and clinical severity. J. Neurol. 266(2), 440–449. https://doi.org/10.1007/s00415-018-9151-x (2019).
McGrogan, A., Madle, G. C., Seaman, H. E. & de Vries, C. S. The epidemiology of Guillain–Barré syndrome worldwide. A systematic literature review. Neuroepidemiology 32(2), 150–163. https://doi.org/10.1159/000184748 (2009).
Ralot, T. K., Parmar, S., Gujar, R., Sarkar, S. & Meghwal, H. Study of clinical profile and prognosis in various subtypes of Guillain–Barre syndrome patients. IP Indian J. Neurosci. 4(4), 204–208 (2020).
Verma, R., Chaudhari, T. S., Raut, T. P. & Garg, R. K. Clinico-electrophysiological profile and predictors of functional outcome in Guillain–Barre syndrome (GBS). J. Neurol. Sci. 335(1–2), 105–111. https://doi.org/10.1016/j.jns.2013.09.002 (2013).
AlKahtani, N. A. et al. Guillain–Barré syndrome in adults in a decade: The largest, single-center, cross-sectional study from the Kingdom of Saudi Arabia. Cureus 15(6), e40995. https://doi.org/10.7759/cureus.40995 (2023).
González-Suárez, I., Sanz-Gallego, I., Rodríguez de Rivera, F. J. & Arpa, J. Guillain–Barré syndrome: natural history and prognostic factors: A retrospective review of 106 cases. BMC Neurol. 13, 95. https://doi.org/10.1186/1471-2377-13-95 (2013).
Kulkantrakorn, K. & Sukphulloprat, P. Outcome of Guillain–Barré syndrome in tertiary care centers in Thailand. J. Clin. Neuromuscul. Dis. 19(2), 51–56. https://doi.org/10.1097/CND.0000000000000176 (2017).
Peric, S. et al. Clinical and epidemiological features of Guillain–Barré syndrome in the Western Balkans. J. Peripher. Nerv. Syst. JPNS 19(4), 317–321. https://doi.org/10.1111/jns.12096 (2014).
Hughes, R. A. & Rees, J. H. Clinical and epidemiologic features of Guillain–Barré syndrome. J. Infect. Dis. 176(Suppl 2), S92-98. https://doi.org/10.1086/513793 (1997).
Hagen, K. M. & Ousman, S. S. The neuroimmunology of Guillain–Barré syndrome and the potential role of an aging immune system. Front. Aging Neurosci. 12, 613628. https://doi.org/10.3389/fnagi.2020.613628 (2021).
McCombe, P. A., Hardy, T. A., Nona, R. J. & Greer, J. M. Sex differences in Guillain–Barré syndrome, chronic inflammatory demyelinating polyradiculoneuropathy and experimental autoimmune neuritis. Front. Immunol. 13, 1038411. https://doi.org/10.3389/fimmu.2022.1038411 (2022).
Sipilä, J. O. T., Soilu-Hänninen, M., Ruuskanen, J. O., Rautava, P. & Kytö, V. Epidemiology of Guillain–Barré syndrome in Finland 2004–2014. J. Peripher. Nerv. Syst. 22(4), 440–445. https://doi.org/10.1111/jns.12239 (2017).
Amante, J. M. G. & Preysler, M. G. Clinical profile and outcomes of Guillain–Barre syndrome: A four-year retrospective study at a tertiary hospital in the Philippines. Clin. Profile Outcomes Guillain–Barre. Syndr. Four-Year Retrosp. Study Tert. Hosp. Philipp. 73(1), 1 (2021).
Head, V. A. & Wakerley, B. R. Guillain–Barré syndrome in general practice: Clinical features suggestive of early diagnosis. Br. J. Gen. Pract. 66(645), 218–219. https://doi.org/10.3399/bjgp16X684733 (2016).
Maawali, S. M. A., Shibani, A. Y. A., Nadeem, A. S. & Al-Salti, A. M. Guillain–Barre syndrome: Demographics, clinical features, and outcome in a single tertiary care hospital, Oman. Neurosci. J. 25(5), 369–374. https://doi.org/10.17712/nsj.2020.5.20200057 (2020).
Melaku, Z., Zenebe, G. & Bekele, A. Guillain–Barré syndrome in Ethiopian patients. Ethiop. Med. J. 43(1), 21–26 (2005).
Assefa, Y. et al. Primary health care contributions to universal health coverage, Ethiopia. Bull. World Health Organ. 98(12), 894-905A. https://doi.org/10.2471/BLT.19.248328 (2020).
van Leeuwen, N. et al. Hospital admissions, transfers and costs of Guillain–Barré syndrome. PLoS One 11(2), e0143837. https://doi.org/10.1371/journal.pone.0143837 (2016).
Bourque, P. R., Brooks, J., McCudden, C. R., Warman-Chardon, J. & Breiner, A. Age matters. Neurol. Neuroimmunol. Neuroinflamm. 6(4), e576. https://doi.org/10.1212/NXI.0000000000000576 (2019).
Bourque, P. R., Brooks, J., Warman-Chardon, J. & Breiner, A. Cerebrospinal fluid total protein in Guillain–Barré syndrome variants: Correlations with clinical category, severity, and electrophysiology. J. Neurol. 267(3), 746–751. https://doi.org/10.1007/s00415-019-09634-0 (2020).
Nishimoto, Y., Odaka, M., Hirata, K. & Yuki, N. Usefulness of anti-GQ1b IgG antibody testing in Fisher syndrome compared with cerebrospinal fluid examination. J. Neuroimmunol. 148(1), 200–205. https://doi.org/10.1016/j.jneuroim.2003.11.017 (2004).
Guillain–Barré syndrome in Taiwan: A clinical study of 167 patients. J. Neurol. Neurosurg. Psychiatry. https://jnnp.bmj.com/content/63/4/494 (accessed 28 Mar 2024).
Ho, T. W. et al. Guillain–Barré syndrome in northern China. Relationship to Campylobacter jejuni infection and anti-glycolipid antibodies. Brain J. Neurol. 118(Pt 3), 597–605. https://doi.org/10.1093/brain/118.3.597 (1995).
Ruiz-Sandoval, J. L. et al. Clinical characteristics and predictors of short-term outcome in Mexican adult patients with Guillain–Barré syndrome. Neurol. India 69(1), 107–114. https://doi.org/10.4103/0028-3886.310063 (2021).
Liu, S. et al. Guillain–Barré syndrome in southern China: Retrospective analysis of hospitalised patients from 14 provinces in the area south of the Huaihe River. J. Neurol. Neurosurg. Psychiatry 89(6), 618–626. https://doi.org/10.1136/jnnp-2017-316930 (2018).
Zeng, Y. et al. Clinical features and the validation of the brighton criteria in Guillain–Barré syndrome: Retrospective analysis of 72 hospitalized patients in three years. Eur. Neurol. 81(5–6), 231–238. https://doi.org/10.1159/000503101 (2019).
Netto, A. B., Taly, A. B., Kulkarni, G. B., Rao, U. G. S. & Rao, S. Mortality in mechanically ventilated patients of Guillain–Barré syndrome. Ann. Indian Acad. Neurol. 14(4), 262–266. https://doi.org/10.4103/0972-2327.91942 (2011).
Melone, M.-A. et al. Early mechanical ventilation in patients with Guillain–Barré syndrome at high risk of respiratory failure: A randomized trial. Ann. Intensive Care 10, 128. https://doi.org/10.1186/s13613-020-00742-z (2020).
Orlikowski, D. et al. Prognosis and risk factors of early onset pneumonia in ventilated patients with Guillain–Barré syndrome. Intensive Care Med. 32(12), 1962–1969. https://doi.org/10.1007/s00134-006-0332-1 (2006).
Parry, S. M. & Puthucheary, Z. A. The impact of extended bed rest on the musculoskeletal system in the critical care environment. Extreme Physiol. Med. 4, 16. https://doi.org/10.1186/s13728-015-0036-7 (2015).
Teasell, R. & Dittmer, D. K. Complications of immobilization and bed rest. Part 2: Other complications. Can. Fam. Physician 39, 1440 (1993).
Author information
Authors and Affiliations
Contributions
Z.D.T. contributed to the study’s conception, design, initial drafting, and data analysis. Y.T.K. and B.D.M. contributed to manuscript revision, data analysis, and final intellectual content assembly. D.K.H. and L.H.J. guided the design, initial drafting, and data analysis phases. A.T.A. and M.S.H. contributed to data acquisition and proofreading.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tewedaj, Z.D., Huluka, D.K., Kebede, Y.T. et al. A retrospective analysis of the clinical profile and factors associated with mortality and poor hospital outcomes in adult Guillain–Barre syndrome patients. Sci Rep 14, 15520 (2024). https://doi.org/10.1038/s41598-024-65265-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-65265-0
- Springer Nature Limited