Abstract
In order to determine the effect of nystagmus on objective visual acuity (VA) estimates, we compared subjective (VApsych) and objective (VEP, VAVEP) VA estimates in participants with nystagmus. For this purpose, 20 participants with nystagmus (NY) caused by idiopathic infantile nystagmus, albinism, achiasma or acquired nystagmus were recruited in this study. Estimates of BCVA (best corrected visual acuity) were determined psychophysically (VApsych; FrACT, Freiburg visual acuity test) and electrophysiologically (VAVEP; EP2000) according to ISCEV (International Society of Clinical Electrophysiology of Vision) guidelines. For each participant the eye with the stronger fixation instability [Nidek microperimeter (MP-1), Nidek Instruments] was included for further analysis. VApsych vs VAVEP were compared via paired t-tests and the correlation of the difference between VApsych and VAVEP (∆VA) vs the degree of fixation instability was tested with Pearson correlation (r). We found VAVEP to be better than VApsych [by 0.12 Logarithm of the Minimum Angle of Resolution (logMAR); mean ± standard error (SE) of VAVEP vs VApsych: 0.176 ± 0.06 vs. 0.299 ± 0.06, P = 0.017] and ∆VA to be correlated linearly with the degree of fixation instability (r2 = 0.21,p = 0.048). In conclusion, on average we report a small VA overestimation, around 1 line, for VAVEP compared to VApsych in NY. This overestimation depended on the magnitude of the fixation instability. As a rule of thumb, a reduction of the fixation probability in the central 4° from 100 to 50% leads on average to a VAVEP overestimation of around 0.25 logMAR, i.e. 2.5 lines.
Similar content being viewed by others
Introduction
Subjective visual acuity testing is a ubiquitous routine tool for everyday clinical practice in ophthalmology and often the first step to check the integrity of retinal function and subsequent structures of the visual system. Critically, this measure of visual acuity (VApsych) is challenged by the subjective nature of the responses, depends on the compliance of the participants, and is hence vulnerable e.g. to malingering1. This creates a need for an objective measure of visual acuity, which might be met by visual evoked potential (VEP) based visual acuity estimates (VAVEP). In fact, previous research in this field has demonstrated the value of this approach2. However, limits to the applicability of VAVEP naturally are expected for certain conditions, which might be particularly prone to systematic VA misestimations. The identification of such conditions is instrumental to avoid the misinterpretation of a patient's VAVEP results. Within this context, it is uncertain whether VEP-acuity estimations maintain their validity in the presence of nystagmus.
As a matter of course, VEP-recordings depend on the capacity of a visual stimulus to drive responses in the visual cortex. One critical parameter is the spatial frequency of the stimulus, as only stimuli that can be resolved will generate responses. It is this dependence of the VEP on spatial frequency of the visual stimulus that is exploited for the estimation of the resolution limit in acuity-VEP paradigms. Specifically, these paradigms target the detection of the spatial frequency limit beyond which stimuli fail to elicit a response2. Another critical parameter for VEP recordings is the stimulation mode. This includes the distinction of pattern-reversal vs pattern-pulse stimulation, which is of particular importance when targeting populations with nystagmus. Notably, nystagmus reduces pattern-reversal VEPs, while VEPs to pattern-pulse stimulation are relatively spared3,4. Acuity-VEP paradigms are often based on pattern-pulse stimulation2. As a consequence, responses from participants with nystagmus are expected to be only little affected, preserving the validity of VEP-acuity estimates in nystagmus, or might even be overestimated. In fact, some reports do suggest the potential of an overestimation of VEP-acuity for nystagmus5,6,7. A systematic assessment of this issue with a contemporary approach to determine VEP-acuity is at present missing. At the same time, however, as nystagmus is a common feature in patients with low vision, it is of critical importance for clinical applications, whether the validity of VAVEP measures is confounded by nystagmus.
We aimed to investigate the validity of VAVEP in the presence of nystagmus by employing a VEP paradigm that is based on patten-pulse stimulation8. Specifically, we compared VApsych and VAVEP estimations in a cohort of patients (NY) with nystagmus due to different etiologies. To accurately determine the effect of eye motion on VAVEP misestimation, fundus imaging was employed to quantify fixation instabilities. We hypothesized that VAVEP might be overestimated compared to VApsych in NY particularly, if fixation instabilities are pronounced.
Methods
This prospective observational study was conducted at the ophthalmic department of the Otto-von-Guericke University (OVGU), Magdeburg, Germany.
Participants
Participants were included after an ophthalmological examination: 20 participants with nystagmus (NY; 9 females; age (mean, range across all participants): \(38, 20-64\text{y}\)) due to the idiopathic infantile nystagmus syndrome (n = 10), albinism (n = 6), achiasma (n = 1), or acquired nystagmus (n = 3), as detailed in Table 1. Optic nerve misrouting in albinism9,10 and achiasma11,12 was confirmed via the misrouting VEP13,14, a VEP method detailed further in previous publications9,15,16. To study the effect of nystagmus on measures of VA, only the individual's eye with stronger fixation instability determined by microperimetry test (see below) was included in the analysis. Exclusion criteria were epilepsy, dizziness, neurological diseases unrelated to nystagmus and any eye diseases affecting visual function, e.g., diabetic retinopathy.
Microperimetry—Fixation stability
Fixation stability of participants within 2° and 4° was quantified by tracking fundus-motion at 25 Hz with a fundus-controlled microperimeter (MP-1 microperimeter, Nidek, Padua, Italy), for an epoch of 30 s, where participants were asked to fixate a central target. Eyes with stronger fixation instability, using fixation proportion within the central ± 2°, were selected for the analysis. See Table 1 for characteristics, including fixation instability and BCVA, of each participant's selected eye.
Subjective VA estimation
Estimates of best corrected visual acuity (BCVA) for each eye were determined psychophysically (VApsych) using the “Freiburg Visual Acuity and Contrast Test” [FrACT; VApsych,17] applying Landolt Cs at a viewing distance of 114 cm (as for the VAVEP estimation). Every measurement was performed twice to reassure the validity of the measurements.
Objective VA estimation
Objective VEP-acuity testing (VAVEP) was estimated according to International Society for Clinical Electrophysiology of Vision (ISCEV) standards18 and followed the procedure described previously8. Briefly, steady state (ss)-VEPs were recorded using pattern-pulse stimulation at 7.5 Hz as detailed in Table 2. The ssVEPs were Fourier analyzed. For each spatial frequency (SF [cpd] = 1/√2 × check size) the response magnitude at the stimulation frequency (7.5 Hz) and a noise estimate, the average of the response of the two neighboring frequencies (6.5 and 8.5 Hz) were obtained to determine the ‘true’, i.e. noise-corrected, amplitude A*(SF)19,20,21 and the significance-level of the response p(SF)21. A stepwise heuristic algorithm8 was applied to calculate the upper SP where the log amplitude response extrapolated to zero, i.e., SF0. SF0 was converted to VEP acuity [decimal VAVEP = SF0/17.6 cpd, which corresponds to logMAR VAVEP = log(SF0/17.6 cpd)]. For all participants tested, the heuristic algorithm produced an estimated VAVEP (100% testability).
Analysis and statistics
Only one eye of each participant was included, namely the eye with the stronger fixation instability. As the data passed the Shapiro Wilk test for normality, parametric statistical testing was applied. The VApsych vs VAVEP estimates were compared using a paired t-test. Pearson correlation (r) was applied to test whether the difference between VApsych and VAVEP (∆VA) correlated with the degree of fixation loss within 4° determined by Pearson correlation (r).
Ethical approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by Ethics Committee of Faculty of medicine, Otto-von-Guericke University, Magdeburg (153/18).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Results
For a qualitative overview of the VEP-recordings obtained, VEP traces for a representative participant with nystagmus (NY) and a healthy control (HC) are juxtaposed in Fig. 1A together with the acuity estimation (Fig. 1B) according to the heuristic algorithm published previously8. The group's data (20 participants with nystagmus, for each individual's eye with stronger fixation instability) are depicted in Fig. 2 (cyan symbols). For comparison, results previously reported for participants without nystagmus by Bach et al. 2008 and Hoffmann et al. 2017 are added (gray symbols). Overall, there was a significant overestimation of VAVEP vs VApsych for nystagmus by on average − 0.12 logMAR (mean ± SE of VAVEP vs VApsych: \(0.18\pm 0.06\text{ vs} \, 0.30\pm 0.06,\text{ p}= 0.017\)). Still, 16 (80%) of the 20 eyes were within the 95% confidence interval determined in Bach et al. 2008 for participants without nystagmus.
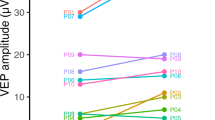
VEP data from two examples. Visual evoked potential visual acuity (VAVEP) and subjective VA (VEPpsych) are given for two individuals, i.e., a healthy control (HC) and a participant with nystagmus (NY). A) VEP-results underlying the estimation of VAVEP. For each participant, NY (JTE807) and a HC (OEX914), two panels are given. The left panel depicts the raw VEP traces for the different check sizes ranging from 0.046° to 0.370° . The right panel depicts the power spectrum and the respective significance of the response’s amplitude at 7.5 Hz, i.e., the stimulation frequency, which enter the spatial frequency tuning curve. B) Spatial frequency tuning curve and VAVEP estimate for NY and HC. VAVEP is derived from the extrapolated spatial frequency (SF) limit determined from the regression line (strong black line) according to Bach et al.8, as detailed in Methods. Significant responses are denoted with an asterix, non-significant with open symbols. For NY VApsych and VAVEP (0.73 vs 0.36) match less closely than for HC (− 0.09 vs − 0.03).
Objective vs subjective visual acuity. Participants with nystagmus [large symbol: mean ± SEM for VAVEP] are given in comparison to previously published data from participants without nystagmus8,24). For our nystagmus cohort, 4 out of 20 eyes fell above the 95% CI established in Bach et al. 2008, indicating an acuity overestimation.
To test whether the VAVEP overestimation in nystagmus was associated with nystagmus severity, we employed a measure of fixation instability (fixation proportion within the central ± 2°) as a surrogate measure of nystagmus severity. We tested the correlation of the acuity differences (∆VA = VAVEP–VApsych) with fixation instability. In fact, there was a weak, albeit significant correlation between fixation instability and the \(\Delta \) VA (r2 = 0.21, p = 0.048; Fig. 3). Consequently, 21% of the variance in the data can be attributed to the strength of the participants' fixation instability.
Discussion
We tested whether nystagmus affects the relationship of VAVEP and VApsych. In our cohort of 20 eyes, we report an overestimation of visual acuity, i.e., better VA, for nystagmus by around 1 line, i.e., 0.12 logMAR. The difference between VAVEP and VApsych correlated significantly with the degree of fixation loss, indicating that the VAVEP estimates were particularly affected by higher degree of nystagmus.
VAVEP offer a complementary or alternative option to assess VA, in cases where VApsych appears questionable. However, as recently reported2, consistency between VAVEP and VApsych is dependent on the etiology of visual dysfunction and acuity loss. Comparable VAVEP and VApsych reductions were reported in media opacities or retinal pathologies, while a VAVEP and VApsych mismatch is more likely in optic nerve, neurological diseases or amblyopia. For patients, where the stimulation is subject to retinal image slip, due to nystagmus, the matter was still unresolved. For our cohort we demonstrate the effect to be minor on average, i.e. an overestimation of around 1 line, but that stronger effects are more likely for higher degree of nystagmus-related fixation loss. Previous research on NY as a separate disease entity was up to now limited as reflected by a few studies from the 80-ies and 90-ies: In a small number of NY patients (n = 5) VAVEP was reported to be poorer than VApsych by 0.15 logMAR5. In a cohort of 14 NY children, Gottlob et al.6 found slightly and non-significantly better estimates of VAVEP (Sinusoidal sweep VEP, 0.48 log min arc) than VApsych (recognition VA using Allen picture cards, 0.51 log min arc) and VAVEP vs VApsych not to be correlated. Westall et al.7 also reported a non-significant trend of a VAVEP overestimation by 0.09 logMAR compared to acuity-card VA in a group of NY children. Different stimulation paradigms, sample size, and study design of these earlier studies, might account for the discrepant findings between studies.
Practical considerations, limitations and outlook
In this study, we assessed the VEP-based VA estimation in comparison to subjective VA measures in an important disease cohort, nystagmus. We highlighted the importance of taking fixation stability in consideration when evaluating VAVEP in nystagmus, as 21% of the variance in the data can be attributed to fixation instability. As a rule of thumb, a reduction of the fixation probability in the central 4° from 100 to 50% leads on average to a VAVEP overestimation of around 0.25 logMAR, i.e. 2.5 lines.
At present, the mechanisms that might mediate this overestimation of VAVEP compared to VApsych are unclear. They might be associated with different stimulation schemes used for the VA estimation, as pulsed patterns are used for VAVEP as opposed to continuous stimulation for VApsych. As an alternative to a methodological cause, there might be a physiological cause. E.g., an association of nystagmus with other pathophysiologies that might lead to VA overestimation, e.g. amblyopia25. Further research is needed to address this issue. Additional insights into the underlying mechanisms might be uncovered in studies that employ fixation-monitoring via eye-tracking during the VEP recordings for a quantitative account of the fixation instabilities and ultimately correct for eye-movements that during VEP recording. Moreover, investigations that specifically address the dependence of VAVEP -overestimation in nystagmus on etiology, ocular co-morbidity and type and strength of nystagmus might be of promise to specify the clinical implications of the influence of nystagmus on VEP-acuity.
Conclusion
This study reported a slight but significant overestimation of VAVEP compared to VApsych in the presence of nystagmus. The differences between objective and subjective VA estimates depended on the magnitude of fixation loss, i.e., higher magnitude of instability leads to higher differences between objective and subjective VA measures. This dependence needs to be taken into account when evaluating VAVEP estimates in nystagmus, specifically when fixation instabilities are pronounced.
Data availability
Data are available upon request. Please contact the corresponding author, MBH, for data requests.
References
Gräf, M. & Jomaa, M. Probleme bei der augenärztlichen Bescheinigung von Blindheit. Ophthalmologe 101(11), 1121–1125. https://doi.org/10.1007/s00347-004-1045-x (2004).
Hamilton, R. et al. VEP estimation of visual acuity: A systematic review. Doc. Ophthalmol. 142(1), 25–74. https://doi.org/10.1007/s10633-020-09770-3 (2021).
Saunders, K. J., Brown, G. & McCulloch, D. L. Pattern-onset visual evoked potentials: More useful than reversal for patients with nystagmus. Doc. Ophthalmol. Adv. Ophthalmol. 94(3), 265–274. https://doi.org/10.1007/BF02582984 (1997).
Hoffmann, M. B., Seufert, P. S. & Bach, M. Simulated nystagmus suppresses pattern-reversal but not pattern-onset visual evoked potentials. Clin. Neurophysiol. 115(11), 2659–2665. https://doi.org/10.1016/j.clinph.2004.06.003 (2004).
Sokol, S., Hansen, V. C., Moskowitz, A., Greenfield, P. & Towle, V. L. Evoked potential and preferential looking estimates of visual acuity in pediatric patients. Ophthalmology 90(5), 552–562. https://doi.org/10.1016/s0161-6420(83)34524-3 (1983).
Gottlob, I., Wizov, S., Odom, J. & Reinecke, R. Predicting optotype visual acuity by swept spatial visual-evoked potentials. Clin. Vis. Sci. 8, 417–423 (1993).
Westall, C. A., Ainsworth, J. R. & Buncic, J. R. Which ocular and neurologic conditions cause disparate results in visual acuity scores recorded with visually evoked potential and Teller Acuity Cards?. J. Am. Assoc. Pediatr. Ophthalmol. Strabismus 4(5), 295–301. https://doi.org/10.1067/mpa.2000.107898 (2000).
Bach, M., Maurer, J. P. & Wolf, M. E. Visual evoked potential-based acuity assessment in normal vision, artificially degraded vision, and in patients. Br. J. Ophthalmol. 92(3), 396–403. https://doi.org/10.1136/bjo.2007.130245 (2008).
Al-Nosairy, K. O., Quanz, E. V., Biermann, J. & Hoffmann, M. B. Optical coherence tomography as a biomarker for differential diagnostics in nystagmus: Ganglion cell layer thickness ratio. J. Clin. Med. 11(17), 4941. https://doi.org/10.3390/jcm11174941 (2022).
Kruijt, C. C. et al. The phenotypic spectrum of albinism. Ophthalmology 125(12), 1953–1960. https://doi.org/10.1016/j.ophtha.2018.08.003 (2018).
Hoffmann, M. B. et al. Plasticity and stability of the visual system in human achiasma. Neuron 75(3), 393–401. https://doi.org/10.1016/j.neuron.2012.05.026 (2012).
Pilat, A., McLean, R. J., Vanina, A., Dineen, R. A. & Gottlob, I. Clinical features and imaging characteristics in achiasmia. Brain Commun. 5(4), fcad219. https://doi.org/10.1093/braincomms/fcad219 (2023).
Hoffmann, M. B. & Dumoulin, S. O. Congenital visual pathway abnormalities: A window onto cortical stability and plasticity. Trends Neurosci. 38(1), 55–65. https://doi.org/10.1016/j.tins.2014.09.005 (2015).
Apkarian, P., Reits, D., Spekreijse, H. & Van Dorp, D. A decisive electrophysiological test for human albinism. Electroencephalogr. Clin. Neurophysiol. 55(5), 513–531. https://doi.org/10.1016/0013-4694(83)90162-1 (1983).
Hoffmann, M. B., Lorenz, B., Morland, A. B. & Schmidtborn, L. C. Misrouting of the optic nerves in albinism: Estimation of the extent with visual evoked potentials. Invest. Ophthalmol. Vis. Sci. 46(10), 3892–3898. https://doi.org/10.1167/iovs.05-0491 (2005).
Hoffmann, M. B. et al. Optic nerve projections in patients with primary ciliary dyskinesia. Invest. Ophthalmol. Vis. Sci. 52(7), 4617–4625. https://doi.org/10.1167/iovs.11-7194 (2011).
Bach, M. The freiburg visual acuity test–automatic measurement of visual acuity. Optom. Vis. Sci. Off. Publ. Am. Acad. Optom. 73(1), 49–53. https://doi.org/10.1097/00006324-199601000-00008 (1996).
Hamilton, R. et al. ISCEV extended protocol for VEP methods of estimation of visual acuity. Doc. Ophthalmol. 142(1), 17–24. https://doi.org/10.1007/s10633-020-09780-1 (2021).
Norcia, A. M., Clarke, M. & Tyler, C. W. Digital filtering and robust regression techniques for estimating sensory thresholds from the evoked potential. IEEE Eng. Med. Biol. Mag. 4, 26–32 (1985).
Norcia, A. M., Tyler, C. W., Hamer, R. D. & Wesemann, W. Measurement of spatial contrast sensitivity with the swept contrast VEP. Vis. Res. 29, 627–637 (1989).
Meigen, T. & Bach, M. On the statistical significance of electrophysiological steady-state responses. Doc. Ophthalmol. 98(3), 207–232 (1999).
Bach M. Visual Evoked Potentials “EP2000” – Computer system by Michael Bach. Published 2007. https://michaelbach.de/ep2000/
Acharya, J. N., Hani, A., Cheek, J., Thirumala, P. & Tsuchida, T. N. American clinical neurophysiology society guideline 2: Guidelines for standard electrode position nomenclature. J. Clin. Neurophysiol. 33(4), 308. https://doi.org/10.1097/WNP.0000000000000316 (2016).
Hoffmann, M. B., Brands, J., Behrens-Baumann, W. & Bach, M. VEP-based acuity assessment in low vision. Doc. Ophthalmol. 135(3), 209–218. https://doi.org/10.1007/s10633-017-9613-y (2017).
Wenner, Y., Heinrich, S. P., Beisse, C., Fuchs, A. & Bach, M. Visual evoked potential-based acuity assessment: Overestimation in amblyopia. Doc. Ophthalmol. 128(3), 191–200. https://doi.org/10.1007/s10633-014-9432-3 (2014).
Acknowledgements
We thank the study participants and the German nystagmus network (Nystagmus Netzwerk e.V.) for their support.
Funding
Open Access funding enabled and organized by Projekt DEAL. Supported by the German Research Foundation (DFG; 435838478 HO-2002/22–1 & HE-3504/9–1).
Author information
Authors and Affiliations
Contributions
EVQ, MBH, KA-N: Study concept and design. EVQ, JK, FK: Investigation. EVQ, MBH, KA-N: Data analysis. EVQ, MBH, KA-N: Original manuscript draft. All authors: comments on results, revision and approval of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Quanz, E.V., Kuske, J., Stolle, F.H. et al. Effect of nystagmus on VEP-based objective visual acuity estimates. Sci Rep 14, 16797 (2024). https://doi.org/10.1038/s41598-024-66819-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-66819-y
- Springer Nature Limited