Abstract
Diabetic peripheral neuropathy is one of the diabetes most common microvascular complications. It is very prevalent in Sub-Saharan Africa due to a combination of causes, including rising diabetes prevalence, limited healthcare resources, and a lack of access to competent medical care. However, just a few studies have been undertaken in the study area. Institution-based cross-sectional study was conducted in the Amhara region referral hospitals, in 2022. By using a systematic random sampling technique, a total of 627 respondents were included. The data was entered into EPI Data version 4.6 and exported to SPSS version 25 for further analysis. A binary logistic regression was used to determine the relationship between the dependent and predictor variables. The association between predictor variables and the dependent variable was determined using multivariate logistic regression [p value < 0.05, 95% confidence interval]. The overall prevalence of diabetic peripheral neuropathy among the study participants was 48.2% (95% CI; 44.2, 52.1). Aged between 40 and 60 years (AOR = 4:27; 95% CI 2.62, 6.94), and 60 years and older (AOR = 4:47; 95% CI 2.40, 8.35), participants who have lived alone (AOR = 2:14; 95% CI 1.21, 3.79), patients with comorbidity (AOR = 1:83; 95% CI 1.22, 2.76), and being physically inactive (AOR = 1:69; 95% CI 1.14, 2.49) were significantly associated with Diabetic peripheral neuropathy. Diabetic peripheral neuropathy was high among diabetic patients. Healthcare providers should prioritize regular screening and early intervention for individuals at higher risk, particularly those aged 40 and above, those living alone, patients with comorbid conditions, and those who are physically inactive. Implementing community-based support programs, encouraging physical activity, and providing comprehensive management plans for diabetes and associated comorbidities can help mitigate the risk and improve the quality of life for these patients.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Introduction
The International Diabetes Federation (IDF) Diabetes Atlas 10th edition reports that 537 adults are living with diabetes, and three in four adults with diabetes live in low- and middle-income countries. It is expected to project 643 million people (11.3% of the global population) by 2030 and 783 million (12.2% of the global population) by 20451.
Diabetes peripheral neuropathy (DPN) is a significant global health issue, with a substantial burden, particularly in low- and middle-income countries. Globally, it is estimated that around 50% of individuals with diabetes will develop some form of neuropathy during their lifetime1,2.
Diabetes Peripheral neuropathy is one of the most common microvascular complications of diabetes mellitus3. It manifests as distal symmetrical polyneuropathy (DSPN), also known as DPN, which causes nerve damage in the extremities, particularly the feet3,4. DPN increases the risk of ulceration, noninvasive amputation, and foot infection, all of which can lead to long-term disability, as well as imposing significant economic and psychological burdens on Type 2 Diabetic Mellitus (T2DM) patients3. DPN is associated with high morbidity, mortality, and a lower quality of life4. It affects up to half of diabetics and is distinguished by pain, paresthesia, and sensory loss5. Approximately, 10–15% of diabetic patients have neuropathy symptoms at the time of diagnosis, and 50% of diabetics eventually develop DPN, which might affect 236 million people worldwide by 20303,6. Approximately 25% of persons with type 2 diabetes had diabetic complications at the time of their initial diagnosis7.
DPN is very prevalent in Sub-Saharan Africa due to a combination of causes, including rising diabetes prevalence, limited healthcare resources, and a lack of access to competent medical care. According to the International Diabetes Federation, the number of adults with diabetes in this region will increase by 143% by 2045, worsening the risk of DPN. Many Sub-Saharan African countries’ healthcare systems are unprepared to manage chronic illnesses such as diabetes, resulting in delayed diagnoses and inadequate management of consequences1,8.
DPN is a substantial burden in Ethiopia due to the fast-increasing prevalence of diabetes and the insufficient healthcare infrastructure available to handle its effects. The prevalence of diabetes in Ethiopia has been increasing, with an estimated 2.3 million adults living with the disease, a figure that is expected to rise further in the future1,9. DPN is one of the most common complications among Ethiopian diabetic patients, resulting in significant morbidity and healthcare expenses. A study conducted in Ethiopia discovered that roughly 29% of diabetes patients suffered from peripheral neuropathy, a condition that has a substantial impact on their quality of life due to persistent discomfort, an increased risk of foot ulcers, and the possibility of amputation. Ethiopia's problems are aggravated by a lack of qualified healthcare providers, poor medical supplies, and low public awareness of diabetes and its complications, all of which lead to delayed diagnosis and ineffective DPN management. Addressing this burden would necessitate collaborative efforts to improve diabetes treatment, such as improved screening and management measures, stronger training for healthcare workers, and expanded health education activities9,10,11.
There has been little study in Ethiopia to determine potential determinants of diabetic peripheral neuropathy. Furthermore, no previous investigations have been undertaken in the study area. Thus, the purpose of this study was to assess the prevalence and risk factors for diabetic peripheral neuropathy among type 2 adult diabetes patients in Ethiopia’s Amhara region referral hospitals.
Methods
Study area, and period
From May 1 to 30, 2022, an institution-based cross-sectional study design was conducted at referral hospitals in the Amhara region. The Amhara region is one of Ethiopia’s 12 Regional States. According to the 2020 Ethiopian fiscal year report, the region’s overall population projection is 22,191,890 (11,317,864 males and 10,874,026 females)12. According to the Amhara National Regional Health Bureau’s Annual Performance Report, the region has 81 hospitals. Among those, there are 08 Referral Hospitals, namely; the University of Gondar (UoG), Dessie, Felege Hiwot, Tibebe Gion, Debre-Markos, Woldia, Debre Tabor, and Debre Birhan12. All those Referral Hospitals serve the population found in the region and provide multidimensional aspects of care to clients who need healthcare services, including surgical, medical, pediatrics, maternal, and other services. Those hospitals also have special units and clinics; like Diabetic Mellitus (DM), Anti-retro Viral Therapy (ART) care and support services, cardiology, respiratory, renal, neurology, dermatology, and sexually transmitted diseases, an intensive care unit, and many other services.
Four hospitals were chosen at random from the entire number of Referral Hospitals: Debre Tabor Referral Hospital, Dessie Referral Hospital, Tibebe Gion Comprehensive Referral Hospital, and Debre Markos Referral Hospital. These hospitals serviced around 2064 Type 2 diabetes patients every month.
Population, sample size, and sampling procedure
The source and study populations included adult type 2 diabetic patients receiving follow-up care at referral hospitals in the Amhara region, as well as those selected at random between May 1 and 30, 2022.
All type-2 DM adult patients at selected hospitals attending follow-up care from May 1 to 30, 2022 were included. However, those type 2 DM with either leprosy or peripheral nerve injury were excluded.
The sample size was determined by using a single population proportion formula considering the following: 95% confidence interval (CI), 0.536 population proportion from the previous study13, and 5% marginal error. N = [(Zα/2)2*p(1−p)]/(d)2. Where n is the initial sample size, Z α/2 is the standardized normal distribution value for the 95% CI 1.96, P is the proportion of peripheral neuropathy, and d is the margin of error is 5%. By adding 10% for non-response rate and 1.5 for design effect our final sample of 627 type 2 diabetic patients on follow-up was included.
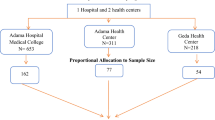
After a random sampling of four hospitals, proportional allocation was done for each referral hospital. A systematic random sampling technique (k = 3) was employed to select study participants (Fig. 1).
The first participant for each referral hospital was selected using the lottery method. For the selected participant, the charts were obtained from the chartroom, and a face-to-face interview in a convenient and private room was conducted.
Outcome and covariates
The outcome variable was DPN. It was assessed by using the Michigan Neuropathy Screening Instrument (MNSI)14. The MNSI consists of two steps: The first step of MNSI is the history questionnaire, which assesses the presence of neuropathic symptoms. This part consists of 15 items, 13 of which assess symptoms of DPN; item number 4 is a measure of impaired circulation, and item number 10 is a measure of general asthenia and was not included in the scoring. Responses of “yes” to items 1–3, 5–6, 8–9, 11–12, and 14–15 each counted as one point. A “no” response to items 7 and 13 counted as 1 point. The score ranges from 0 to 13, and a score that is ≥ 7 indicates the presence of neuropathic symptoms.
The second step of the MNSI is the physical examination part, which scores from 0 to 5 points. It was measured by examining the appearance of feet for deformities, dry skin, calluses, or infections; the presence or absence of ulceration; the examination of vibration sense by tuning-fork to 128 Hz. The detection of the ankle reflex using the hammer to detect the quality of stretch reflex pathway function; and the monofilament test. A 5.07/10g monofilament made in China was used to detect loss of pressure sensation on the feet. It is an objective instrument used in screening the diabetic foot for loss of protective sensation. Each monofilament was used to test 10 patients to avoid diagnosing errors. The participant, whose eyes were closed, was asked to respond yes if he or she felt the filament. Nine sites on the plantar surface of the foot and one on the dorsum were tested. 8 correct responses out of 10 applications were considered normal and scored 0, 1–7 correct responses indicated reduced sensation and scored 0.5, and no correct answers translated into absent sensation and scored 1.
Finally, the patient’s history version of the Michigan neuropathy screening instrument (MNSI) questionnaire score was ≥ 7 abnormal responses, and in the legs and/or lower extremity examination version of MNSI scores was ≥ 2.5 was considered as DPN.
19 variables were used as the independent (covariate) variables. It was grouped into three components: socio-demographic (age, sex, residence, marital status, family size, living status, educational status, occupational status, wealth index, and family history of DM), behavioral factors (alcohol intake, smoking, khat chewing, and physical exercise), and clinical factors (FBS (fasting blood sugar) (mg/dl), treatment regimen, BMI (body mass index) (kg/m2), abdominal circumference (cm), and comorbidity).
The wealth index is the asset-based index of a household’s material well-being or economic status that can be used for all low- and middle-income countries15. For this study, 14 and 26 questions were prepared to measure the wealth level of urban and rural residents, respectively. All categorical variables were labeled as one and zero. Similarly, continuous variables were categorized to be between zero and one.
For behavioral factors; alcohol, khat, and cigarette smoking, participants in this study were classified as current users if they had used psychostimulant substances like alcohol, khat, or cigarettes at least once within the previous 30 days16.
Patients performing moderate-to-intense physical activity for more than 150 min per week were considered physically active13,17.
For this research, comorbidity is defined as the existence of one or more chronic conditions in diabetic patients which can be classified as concordant and discordant comorbidities18. Waist circumference (WC) measurements were as follows: adults aged 20 or older were classified into three health risk categories according to cut-offs recommended by the WHO and the National Institute of Health (NIH)17.
Data collection procedure
Data was collected through a structured questionnaire, an MNSI, a standard stadiometer, and a weight scale. MNSI is a well-known instrument used to assess peripheral neuropathy among patients with DM with a sensitivity of 80% and a specificity of 95%14,19. Some questionnaires were adapted by reviewing different literature like wealth index and sociodemographic questionnaires15,20. The English version of the questionnaire was translated into the local language (Amharic) and again translated back to English by language experts.
Anthropometric measurements were conducted to measure the height and weight of the study participants. Body weight was measured while wearing light clothes on an adjusted scale. Height was measured by the meter, standing upright on a flat surface. BMI was calculated as kg/m2 to determine the nutritional status of the body’s composition. Data collection was carried out through face-to-face interviews. Patient record document review and physical examinations were also done.
Data quality assurance
Four data collectors and four MSc Nurses as supervisors were recruited for data collection who had experience in data collection. Data collectors, including the supervisor, were trained together for two days. During the training, the importance of obtaining the respondent's verbal consent and respecting their right to respond or not to respond to any part of the questions was given emphasis, in addition to ensuring privacy and confidentiality. After the supervisor and the data, collectors were assured of the clarity and completeness of the questionnaires, the data collectors received the data collection formats with a detailed clarification of each question to facilitate a uniform understanding of the meaning of each question.
A pre-test on 5% of the sample population was conducted at the University of Gondar Comprehensive Specialized Hospital Diabetic Clinic to assess the relevance of the questions. Consistency, accuracy, and internal consistency were checked, and Cronbach’s alpha value was 0.75. The collected data were checked daily for accuracy and completeness by the principal investigator and supervisors.
Data analysis
After coding and checking for completeness and consistency, the data was entered into Epi-Data version 4.6 and then exported to SPSS version 25 for analysis.
A binary logistic regression model was fitted to investigate the association between each factor and outcome variable, and factors that had a p value < 0.25 were included in a multivariable logistic regression model and adjusted for a confounding effect. Finally, an adjusted odds ratio with a 95% confidence interval and p value of less than 0.05 was used to report factors independently associated with DPN. The Hosmer and Lemeshow goodness-of-fit test was checked for model fitness with a significance value of 0.861.
Ethics approval and consent to participate
The study was approved by the Research Ethical Review Committee of the School of Nursing, College Medicine and Health Sciences on behalf of the University of Gondar review board under reference number S/N0042/22. The School Nursing Research and Ethical Review Committee accepted and approved that, this study was carried out by relevant guidelines/regulations, Helsinki declarations, and approval to take oral consent /informed consent. A formal letter indicating the approval was obtained and submitted to all four referral hospitals, and the Amhara region health bureau.
Oral consent/informed consent was obtained from each participant for an interview and to participate in the physical examination. In all steps, confidentiality was maintained. Finally, after the whole process of data collection, the questionnaire was kept safe throughout the whole process of the research work.
Results
Socio-demographic haracteristics of participants.
The study included 627 participants and had a 100% response rate. More than half (55.18%) of the participants were male. Participants’ mean and standard deviation (SD) ages were 46.61 ± 14.6 years and two-fifths (43%) of them were aged 50 or older. Two-thirds of the participants (69.22%) were urban dwellers. The majority of participants (58.53%) reported being married. The participant family size averaged 4.34 ± 2 (Table 1).
Clinical characteristics of participants.
Among the 627 study participants, 329 (52.5%) had a normal BMI, whereas two-fifths (28.7%) were overweight. Almost half (47.85%) of subjects took an insulin injection medication. The participants’ mean and SD fasting blood sugar (FBS) were 204.03 ± 58.7 and their BMI was 24.03 ± 3.6 (Table 2).
Behavioral characteristics of participants
Among the study participants, 270 (43.1%) drank alcohol, while only 47 (7.5%) smoked. Two-thirds (61.4%) of participants engaged in at least 150 min of physical exercise every week (Table 3).
Prevalence of diabetic peripheral neuropathy and associated factors
In this study, the overall prevalence of DPN was 48.2% (95% CI 44.2–52.1). Following bivariate analysis, thirteen variables with a p value less than 0.25 were selected as candidates for multivariable logistic regression analysis. In multivariable logistic regression analysis, participants’ age, living status, comorbidities, and physical activity all had a statistically significant relationship with DPN.
Participants aged 40 to 60 years were 4.27 times more likely to develop DPN than those under 40 years (95% CI: 2.62, 6.94). Furthermore, participants aged 60 and older were 4.47 times more likely to have DPN than individuals under the age of 40 (95% CI: 2.40, 8.35). The other factor identified was living status. Participants who lived alone were 2.14 times more likely to develop DPN than those who lived with their families (95% CI: 1.21, 3.79). Similarly, diabetic patients with comorbidity were 1.83 times more likely to develop DPN than those with no history of other medical conditions (95% CI: 1.22, 2.76). Finally, diabetic patients with physically inactive were 1.69 times more likely to develop DPN than those with physically active participants (95% CI: 1.14, 2.49) (Table 4).
Discussion
Diabetic peripheral neuropathy is a common and significant consequence of type 2 diabetes that causes damage to the peripheral nerves. This syndrome affects a large proportion of adults with type 2 diabetes, causing symptoms like pain, numbness, and paralysis in the extremities. The current study aimed to determine the prevalence of DPN and its associated factors in adult patients with type 2 diabetes.
As a result, the total prevalence of DPN was 48.2%, which is comparable to the prevalence reported in other countries such as the United States, Malaysia, and Ethiopia, which were 51%, 50.7%, and 52.2%, respectively21,22,23. Our findings outperformed those of studies conducted in Jordan (39.5%), Arabian Gulf Region (33.3%), China (33.1%), Libya (30.5%), Uganda (29.4%), and Ethiopia (25.4%)24,25,26,27,28,29. The higher DPN prevalence rate in this study may be due to differences in medical care access, sample size, study site, and diagnostic criteria. Furthermore, our data revealed that older participants were more sensitive to DPN. The gap could be attributed to the fact that more than 65% of study participants were above the age of 40. Furthermore, the study in Uganda was limited to newly diagnosed patients, whereas this study included all patients. The study from Ethiopia had a smaller sample size than this one.
However, the current study showed a lower prevalence of DPN when compared to other studies reported from Yemen and Iran, with a DPN prevalence of 56.2% and 75.1%, respectively30,31. The higher prevalence of those studies could be due to differences in study setting, tool used, and study design. For instance, the Iranian study used a large sample size and used nerve conduction velocity which is the gold standard test.
In the multivariable binary logistic regression analysis, the age of respondents, living status, comorbidity, and physical activity were statistically significant with DPN. This finding revealed that a patient’s age above 40 years was an independent predictor of DPN. This report was supported by previous studies13,23,25,32,33. The association between diabetic peripheral neuropathy and advanced age is multifaceted comprising the interaction of prolonged diabetes duration, chronic hyperglycemia, vascular alterations, comorbidities, and age-related physiological changes3,34,35,36.
Our findings revealed that persons who live alone are more likely to develop DPN. This could be because a living arrangement was found to be significantly associated with unmet healthcare needs among young adults (aged 18–30 years), with those living alone reporting greater unmet needs than those living with others, parents, or parents and spouse37. Another study reported that elderly individuals living with their spouses were significantly more likely to receive preventive care like routine physical check-ups38. Thus, living with another individual appears to increase healthcare encounters, which may in turn impact public perceptions of access to care.
Moreover, our data showed that physical inactivity was strongly associated with the prevalence of DPN13,23,39,40. The possible reason for this association might be that physical exercise can increase microvascular circulation, release neurotropic factors, attenuate oxidative stress, and improve the physiological well-being of the body41. The severity of concordant comorbidities, such as obesity, cardiovascular disease, and musculoskeletal illnesses, has an impact on physical inactivity in the research population. Obesity, for example, might hinder mobility, and cardiovascular disease can impair exercise tolerance. Musculoskeletal problems, like arthritis, can cause pain and discomfort while exercising41,42. It is recommended to follow health professional suggestions before engaging in any exercise.
Finally, a significant association was found between DNP and comorbidities. Our finding result was supported by previous studies18,39,43. It can be explained by the multifactorial nature of diabetes and its complications. Comorbid conditions such as hypertension, hyperlipidemia, and cardiovascular diseases often coexist with diabetes and contribute to the development and progression of DPN through various mechanisms. Hypertension, for instance, can exacerbate microvascular complications in diabetic patients by damaging blood vessels and reducing blood flow to peripheral nerves, leading to nerve damage and DPN21. Similarly, hyperlipidemia contributes to atherosclerosis, which can impair blood supply to peripheral nerves, resulting in neuropathic changes3,44. Moreover, Concordant comorbidities may share similar pathophysiological mechanisms with diabetes, such as hypertension. These conditions may lead to common underlying factors like obesity and insulin resistance. On the other hand, discordant comorbidities, like HIV/AIDS, do not share these mechanisms and require distinct management approaches, which can complicate the overall treatment plan for diabetic patients18,35.
As a limitation, the cross-sectional nature of the study inferring causal association correlations is challenging. Because participants are concerned about being identified if they admit to using alcohol, smoking, or khat chewing, they may submit inaccurate information, causing the results to be underestimated. Furthermore, exercise was solely by asking participants to walk and exercise every day, which may have resulted in subjective bias.
Conclusion
This study shows a notably high prevalence of Diabetic Peripheral Neuropathy (DPN) at 48.2% among the studied population, underscoring the critical need for focused healthcare interventions. The findings highlight a significant association between DPN and various predictors, specifically participants' age, living status, comorbidities, and levels of physical activity. These relationships suggest that targeted strategies addressing these variables could be important in managing and potentially mitigating the impact of DPN. Future research should explore tailored interventions that consider these factors to effectively reduce the prevalence and severity of DPN in vulnerable populations.
Data availability
Based on reasonable request all data is available at the corresponding author.
Abbreviations
- CI:
-
Confidence interval
- BMI:
-
Body mass index
- DM:
-
Diabetes mellitus
- DPN:
-
Diabetic peripheral neuropathy
- DSPN:
-
Distal symmetrical polyneuropathy
- FBS:
-
Fasting blood sugar
- KMO:
-
Keiser–Meyer–Olkin
- MNSI:
-
Michigan neuropathy screening instrument
- NIH:
-
National Institute of Health (NIH)
- T2DM:
-
Type 2 diabetic mellitus
- UoG:
-
University of Gondar
- WC:
-
Waist circumference
- WHO:
-
World Health Organization
References
Home, Resources, diabetes L with, Acknowledgement, FAQs, Contact, et al. IDF Diabetes Atlas [Internet]. [cited 2024 Jun 24]. Available from: https://diabetesatlas.org/.
Beran, D. & Yudkin, J. S. Diabetes care in sub-Saharan Africa. Lancet 368(9548), 1689–1695 (2006).
Tesfaye, S. & Selvarajah, D. Advances in the epidemiology, pathogenesis, and management of diabetic peripheral neuropathy. Diabetes Metab. Res. 28(S1), 8–14 (2012).
Callaghan, B. C., Cheng, H. T., Stables, C. L., Smith, A. L. & Feldman, E. L. Diabetic neuropathy: Clinical manifestations and current treatments. Lancet Neurol. 11(6), 521–534 (2012).
Tesfaye, S. Recent advances in the management of diabetic distal symmetrical polyneuropathy. J. Diabetes Invest. 2(1), 33–42 (2011).
Prospective Study of Neuropathic Symptoms Preceding Clinically Diagnosed Diabetic Polyneuropathy: ADDITION-Denmark | Diabetes Care | American Diabetes Association [Internet]. [cited 2024 Jun 24]. Available from: https://diabetesjournals.org/care/article/42/12/2282/36203/Prospective-Study-of-Neuropathic-Symptoms.
Ejigu, A. Brief communication: Patterns of chronic complications of diabetic patients in Menelik II Hospital Ethiopia. Ethiop. J. Health Dev. 14(1), 113–116 (2000).
Home, Resources, diabetes L with, Acknowledgement, FAQs, Contact, et al. Factsheets | IDF Diabetes Atlas [Internet]. [cited 2024 Jun 24]. Available from: https://diabetesatlas.org/regional-factsheets/.
Gedamu, M., Mulusew, C. & Senbeta, M. F. Diabetic complications and associated factors among diabetic patients in Tikur Anbessa specialized hospital, Addis Ababa Ethiopia. Int. J. Diabetes Clin. Res. 9, 160 (2022).
Feleke, Y. & Enquselassie, F. Cost of hospitalization of diabetic patients admitted at Tikur Anbessa Specialized Hospital, Addis Ababa Ethiopia. Ethiop. Med. J. 45(3), 275–282 (2007).
Diabetes Ethiopia 2016 country profile [Internet]. [cited 2024 Jun 24]. Available from: https://www.who.int/publications/m/item/diabetes-eth-country-profile-ethiopia-2016.
Ethiopia Health Sector Transformation Plan (2020/21–2024/25) | Global Financing Facility [Internet]. [cited 2024 Jun 24]. Available from: https://www.globalfinancingfacility.org/resource/ethiopia-health-sector-transformation-plan-202021-202425.
Abdissa, D. et al. Prevalence and determinants of peripheral neuropathy among type 2 adult diabetes patients attending Jimma University Medical Center, Southwest Ethiopia, 2019, an institutional-based cross-sectional study. J. Diabetes Res. 29, 9562920 (2020).
Herman, W. H. et al. Use of the Michigan neuropathy screening instrument as a measure of distal symmetrical peripheral neuropathy in type 1 diabetes: results from the diabetes control and complications trial/epidemiology of diabetes interventions and complications. Diabet. Med. 29(7), 937–944 (2012).
Smits, J. & Steendijk, R. The international wealth index (IWI). Soc. Indic. Res. 122(1), 65–85 (2015).
Jemere, T., Mossie, A., Berhanu, H. & Yeshaw, Y. Poor sleep quality and its predictors among type 2 diabetes mellitus patients attending Jimma University Medical Center, Jimma, Ethiopia. BMC Res. Notes. 12(1), 488 (2019).
WHO Consultation on Obesity (1999: Geneva S, Organization WH. Obesity : preventing and managing the global epidemic : report of a WHO consultation [Internet]. World Health Organization; 2000 [cited 2024 Jun 24]. Available from: https://iris.who.int/handle/10665/42330.
Chiang, J. I. et al. Multimorbidity, mortality, and HbA1c in type 2 diabetes: A cohort study with UK and Taiwanese cohorts. PLOS Med. 17(5), e1003094 (2020).
Moghtaderi, A., Bakhshipour, A. & Rashidi, H. Validation of Michigan neuropathy screening instrument for diabetic peripheral neuropathy. Clin. Neurol. Neurosurg. 1(108), 477–481 (2006).
Demographic E. Health Survey (EDHS) 2016: Key Indicators Report, Central Statistical Agency Addis Ababa, Ethiopia. The DHS Program ICF Rockville, Maryland, USA. 2016. - Google [Internet]. [cited 2024 Jun 24].
Pop-Busui, R., Lu, J., Lopes, N. & Jones, T. L. Z. Prevalence of diabetic peripheral neuropathy and relation to glycemic control therapies at baseline in the BARI 2D cohort. J. Peripher. Nerv. Syst. 14(1), 1–13 (2009).
Mimi, O., Teng, C. L. & Chia, Y. C. The prevalence of diabetic peripheral neuropathy in an outpatient setting. Med. J. Malays. 58(4), 533–538 (2003).
Jember, G. et al. Peripheral sensory neuropathy and associated factors among adult diabetes mellitus patients in Bahr Dar, Ethiopia. J. Diabetes Metab. Disord. 16(1), 16 (2017).
Khawaja, N. et al. The prevalence and risk factors of peripheral neuropathy among patients with type 2 diabetes mellitus; The case of Jordan. Diabetol. Metab. Syndr. 10(1), 8 (2018).
Li, L., Chen, J., Wang, J. & Cai, D. Prevalence and risk factors of diabetic peripheral neuropathy in Type 2 diabetes mellitus patients with overweight/obese in Guangdong province China. Primary Care Diabetes. 9(3), 191–195 (2015).
Elbarsha, A., Hamedh, M. & Elsaeiti, M. Prevalence and risk factors of diabetic peripheral neuropathy in patients with type 2 diabetes mellitus. Ibnosina J. Med. Biomed. Sci. 11(1), 25–28 (2019).
Kisozi, T. et al. Prevalence, severity, and factors associated with peripheral neuropathy among newly diagnosed diabetic patients attending Mulago hospital: A cross-sectional study. Afr. Health Sci. 17(2), 463–473 (2017).
Tilahun, A. N., Waqtola, C., Tewodros, G. M., Amare, D. W. & Yohannis, M. Major microvascular complications and associated risk factors among diabetic outpatients in Southwest Ethiopia. Endocrinol. Metab. Syndr. 6(4), 1–9 (2017).
Ponirakis, G. et al. Prevalence and risk factors for diabetic peripheral neuropathy, neuropathic pain, and foot ulceration in the Arabian Gulf region. J. Diabetes Invest. 13(9), 1551–1559 (2022).
Al Washali, A. Y., Azuhairi, A. A., Hejar, A. R. & Amani, Y. W. Prevalence and associated risk factors of diabetic peripheral neuropathy among diabetic patients in national center of diabetes in Yemen. Int. J. Public Health Clin. Sci. 1, 141–150 (2014).
Janghorbani, M. et al. Peripheral neuropathy in type 2 diabetes mellitus in Isfahan, Iran: Prevalence and risk factors. Int. J. Diabetes Metab. 14(3), 126–133 (2019).
DSouza, M. et al. Diabetic peripheral neuropathy and its determinants among patients attending a tertiary health care centre in Mangalore India. J. Public Health Res. 4(2), 450 (2015).
Kiani, J., Moghimbeigi, A., Azizkhani, H. & Kosarifard, S. The prevalence and associated risk factors of peripheral diabetic neuropathy in Hamedan Iran. Arch. Iran Med. 16(1), 17–19 (2013).
Boulton, A. J. M. et al. Diabetic neuropathies: a statement by the American diabetes association. Diabetes Care 28(4), 956–962 (2005).
Smith, A. G. & Singleton, J. R. Obesity and hyperlipidemia are risk factors for early diabetic neuropathy. J. Diabetes Complicat. 27(5), 436–442 (2013).
Ramji, N., Toth, C., Kennedy, J. & Zochodne, D. W. Does diabetes mellitus target motor neurons?. Neurobiol. Dis. 26(2), 301–311 (2007).
Marshall, E. G. Do young adults have unmet healthcare needs?. J. Adolesc. Health 49(5), 490–497 (2011).
Lau, D. T. & Kirby, J. B. The relationship between living arrangement and preventive care use among community-dwelling elderly persons. Am. J. Public Health 99(7), 1315–1321 (2009).
Al Maskari, F. prevalence and determinants of peripheral neuropathy in patients with type 2 diabetes attending a tertiary care center in the United Arab Emirates. J. Diabetes Metab. [Internet]. 5(03) (2014).
Gebabo, T. F., Zewdie, T. H., Shagaro, S. S. & Haile, F. Determinants of peripheral neuropathy among diabetic patients under follow-up in chronic care clinics of public hospitals at Gamo and Gofa zones, southern Ethiopia. PLOS ONE 16(2), e0246722 (2021).
Kluding, P. M. et al. The effect of exercise on neuropathic symptoms, nerve function, and cutaneous innervation in people with diabetic peripheral neuropathy. J. Diabetes Complicat. 26(5), 424–429 (2012).
Colberg, S. R. et al. Physical activity/exercise and diabetes: a position statement of the American diabetes association. Diabetes Care 39(11), 2065–2079 (2016).
Sendi, R. A., Mahrus, A. M., Saeed, R. M., Mohammed, M. A. & Al-Dubai, S. A. R. Diabetic peripheral neuropathy among Saudi diabetic patients: A multicenter cross-sectional study at primary health care setting. J. Family Med. Primary Care 9(1), 197 (2020).
Wiggin, T. D. et al. Elevated triglycerides correlate with progression of diabetic neuropathy. Diabetes 58(7), 1634–1640 (2009).
Acknowledgements
The authors would like to thank the study participants and data collectors for their unreserved contribution during data collection. Also, we would like to forward our gratitude to Amhara region referral hospital administrators, heads of the chronic outpatient department, and healthcare providers for their valuable support during data collection.
Funding
There was no funding for this research. However, the University of Gondar College of Medicine and Health Sciences has facilitated the process for this study.
Author information
Authors and Affiliations
Contributions
S.S.T., B.T.A., M.H.S., and A.J.F. were participated on conceptualization and data curation, methodology, software and validation, and writing for all manuscript. Consent for publication was secured during oral informed consent from the participant and approved by the Research Ethical Review Committee of the School of Nursing College Medicine and Health Sciences on behalf of the University of Gondar review board with reference number S/N0042/22.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Salih, M.H., Tsega, S.S., Alemayehu, B.T. et al. Peripheral neuropathy and associated factors among type 2 diabetic patients attending referral hospitals in the Amhara region, a multi-center cross-sectional study in Ethiopia. Sci Rep 14, 19662 (2024). https://doi.org/10.1038/s41598-024-70314-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-70314-9
- Springer Nature Limited