Abstract
Fermented traditional Chinese medicines (TCMs) have been identified as a low-cost and promising feed additive to to alleviate weaning stress in young livestock and poultry effectively. This study investigated the impact of probiotic fermentation on the metabolite content of BanQi (Radix Isatidis and Astragalus membranaceus) extract while also examined the effects of both fermented-BanQi (FBQ) and unfermented-BanQi (UBQ) on growth performance, serum biochemistry, intestinal villi, and gut microbiota in weaned lambs. This study demonstrated that compared with UBQ, FBQ contained significantly higher levels of free amino acids (e.g., phenylalanine and isoleucine), short peptides (e.g., Val–Leu–Pro–Val–Pro–Gln and Gly–Leu), and the active ingredients (e.g., vindesine and reserpine) (P < 0.05). The addition of FBQ to the diet significantly increased the final body weight and average daily gain of weaned lambs (P < 0.05). In addition, FBQ significantly increased the total protein level in the serum and the villus length of the jejunum and ileum in lambs, while significantly reduced the levels of aspartate aminotransferase (AST) and urea (P < 0.05). Sequencing of the intestinal flora showed that FBQ improved the diversity of intestinal flora and promoted the enrichment of beneficial bacteria in the lamb intestine, such as Mogibacterium and Butyrivibrio, compared to NC or UBQ groups (P < 0.05). Fermentation with Bacillus subtilis can enhance the content of free amino acids, peptides, and active ingredients in BanQi extract, making it an effective method to improve the efficacy of traditional Chinese medicine. Adding FBQ to the diet can improve the growth performance of weaned lambs, and its mechanism may be related to increasing the height of intestinal villi and increasing the diversity of intestinal flora.
Similar content being viewed by others
Introduction
In intensive livestock production, early weaning (8-week-old) is commonly used to shorten the reproductive cycle of ewes and improve productivity and profitability1. However, early weaning can impose stress on lambs, leading to a slowdown in growth performance and a decline in overall health status, ultimately resulting in feed wastage. The causes of weaning stress are multifaceted, with the primary factors being the alterations in the feeding environment and diet, as well as the decrease in maternal antibody levels, which collectively lead to significant changes in the gut microbiome of young ruminants, affecting the colonization of beneficial bacteria2. Research indicates that the gut microbiota of young ruminants is highly susceptible to external environmental influences, and weaning stress significantly impacts the diversity of the gut microbiome, triggering inflammatory responses and disrupting gut homeostasis3. Consequently, the incorporation of exogenous feed additives to modulate the gut microbiome has emerged as an effective strategy to alleviate weaning stress and enhance lamb growth performance.
Traditional Chinese medicine (TCM), as feed additives, can improve the growth performance and immunity of weaned livestock, alleviating the stress of weaning4. Sun et al5. found that the supplementation of Traditional Chinese Medicine residue contains can improve the growth performance of weaned piglets and reduce the relative abundance of Escherichia coli and Treponema hyodysenteriae in the intestine. Additionally, the addition of polysaccharides from traditional Chinese medicine to the diet can increase the average daily gain (ADG) and the levels of IgA, IgG, and IgM in the serum of lambs, while enhancing the relative abundance of Fibrobacteria in the intestine6. Radix Isatidis, called “Banlangen” has been traditionally used to treat influenza, epidemic hepatitis, and epidemic encephalitis7. Astragalus membranaceus, also known as Huangqi, contains astragalus polysaccharides, saponins, flavonoids, and other active components, Astragalus membranaceus root powder is used as a natural feed additive in lamb diets to improve average daily weight gain, antioxidant status, and immune function with an optimal level of 10 g/kg of diet4,8. Our previous studies have found that BanQi (Radix Isatidis and Astragalus membranaceus) polysaccharides can enhance animal immune function and have antiviral effects (Patent certificate: ZL 200,610,160,048.4). However, the high feeding cost of traditional Chinese medicines and their polysaccharides has severely limited their long-term large-scale application in livestock and poultry production.
In the processing of traditional Chinese medicine, fermentation is a commonly used processing technology. Through the action of microorganisms, medicinal materials are fermented under appropriate temperature, humidity, and moisture conditions to enhance their original characteristics and produce new efficacies9. Probiotics can produce a variety of digestive enzymes such as proteases, amylases, and cellulases, which facilitate the breakdown of large molecular nutrients in traditional Chinese medicine (TCM) into smaller components like peptides, amino acids, and monosaccharides10,11. In livestock production, fermentation can enhance the effectiveness of traditional Chinese medicine in improving the growth performance and immune function of livestock and poultry, while also reducing the application costs12,13. Feeding fermented TCM to weaned livestock has been shown to increase intestinal villus length, improve intestinal flora compositions, and strengthen the intestinal barrier, thereby improving growth14,15.
Currently, many studies are leveraging fermented traditional Chinese medicine (TCM) to improve animal production performance or enhance disease resistance15,16. Nevertheless, research on the overall metabolic changes before and after the fermentation of TCM remains scarce. Even the existing studies on metabolites tend to focus on a specific category or a few select substances, significantly hindering the large-scale application of fermented TCM17,18. For the above reasons, we made a non-targeted metabonomics analysis of Ban Qi extract and FBQ to explore the influence of probiotic fermentation on metabolites of traditional Chinese medicine, and fed them to weaned lambs to explore the influence of fermented-TCM on growth performance, intestinal villi morphology and intestinal flora of lambs, to provide theoretical support for the application of fermented-TCM in lamb feeding.
Materials and methods
Herbal extraction and fermentation strains
Radix Isatidis and Astragalus membranaceus were obtained from the Yi Guo Ren Pharmacy in Zhengzhou City, Henan Province, China. A total of 100 g of Radix Isatidis slices and 200 g of Astragalus membranaceus slices are accurately weighed and then soaked in 3000 ml of water, which is then brought to a boil and allowed to simmer for 120 min. The medicinal extract was concentrated to 300 ml, which is the unfermented Banqi extract (UBQ). The strain of Bacillus subtilis used for fermentation was isolated and preserved in our laboratory (CGMCC No. 24193; deposited at the China General Microbiological Culture Collection Center). To initiate fermentation, the strain was cultured in Lysogeny Broth (LB) medium at 37 °C for 24 h and then adjusted to a concentration of 3 × 109 CFU/mL.
Optimization of fermentation conditions
To maximize the number of Viable B. subtilis in the fermented extract, we conducted an orthogonal experiment to explore the effects of different ratios of LB medium to extract (75:25, 50:50, 25:75, and 0:100), fermentation time (28, 32, 36, 40, and 48 h), fermentation temperature (30, 33, 35, 37, and 40 °C), and inoculum size of B. subtilis (3%, 5%, 7%, and 10%). The results showed that the maximum number of Viable B. subtilis was achieved when the ratio of LB medium to extract was 75:25, fermentation time was 36 h, fermentation temperature was 35 °C, and inoculum size was 5%, with a count of 1.21 × 1010 CFU/mL (Supplementary data 1). This is the fermented BanQi extract (FBQ).
Extraction of metabolites from UBQ or FBQ and non-targeted metabolomics analysis
An appropriate amount of FBQ or UBQ was added into pre-cooled methanol: acetonitrile: water (2:2:1) solution and mixed well, sonicated at 4 °C for 30 min, allowed to stand at − 20 °C for 10 min, and centrifuged at 14,000×g for 20 min. The resulting supernatant was dried under a vacuum. The samples were separated using an Agilent 1290 Infinity LC C-18 ultra-performance liquid chromatography (UPLC) column. The temperature of the column was 40 °C, the flow rate was 0.4 mL/min, and the injection volume was 2 μL. The mobile phase A was water, 25 mM ammonium acetate, and 0.5% formic acid, whereas mobile phase B was methanol. During the operation, the samples were placed in four autosamplers and the quality control (QC) samples were lined up separately. Each sample was subjected to electrospray ionization tests in the positive and negative ion modes; the UPLC chromatograms were analyzed using a TOF 6600 triple mass spectrometer (AB SCIEX, USA). The acquisition time was 0.2 s/segment for TOF-MS scans and 0.05 s/segment for product ion scans. Secondary mass spectrometry results were derived from the correlation data. In the high-sensitivity mode, the collision energy was 30 eV and the cluster voltage was 60 V (positive and negative ion modes). The correlation data collection parameters were set to measure approximately 10 candidate ions per cycle, excluding 4 Da isotope.
Animal diets and experimental design
Eighteen weaned Hu sheep (50 days old, 15.79 ± 0.79 kg) were randomly divided into three groups, with six sheep in each group. The NC group was fed a basal diet without any additives. The design of the basal diet referred to the NY/T 816-2004 Standard of Meat Sheep Feeding, and its ingredients and nutritional levels are shown in Table 1. Meanwhile, the UBQ and FBQ groups were supplemented with 6 ml/lamb/day of UBQ or FBQ, respectively, in addition to the basal diet.
Each lamb was housed individually in cages (1.5 m × 1.2 m) and had ad libitum access to feed and water. During the 28-day experimental period, the ambient temperature was maintained at (25 ± 2)°C with a relative humidity of (50 ± 5)%. All experimental procedures involving sheep adhered to the rules and regulations outlined in the Regulations on the Administration of Laboratory Animal Protection for Experimental Use (2013) and received approval from the Institutional Animal Care and Use Committee (IACUC) of Henan Agricultural University in Henan, China (Approval No.: 11–0085). Furthermore, all experiments were conducted in accordance with the ARRIVE guidelines.
Growth performance analysis
At the start of the experiment (Day 1) and at the end of the experiment (Day 28), record the body weights of the sheep before morning feeding. Calculate the Average Daily Feed Intake (ADFI) and Average Daily Gain (ADG), then divide ADFI by ADG to calculate the Feed-to-weight ratio.
Serum biochemical parameters analysis
On the 14th and 28th days of the experiment, blood samples were collected from all lambs before feeding using disposable blood collection tubes. The serum was then separated and stored at − 20 °C. An automated biochemical analyzer (chemray 800, China) was used to measure the levels of total protein (TP), total cholesterol (TC), urea, glucose (GLU), aspartate aminotransferase (AST), and alanine aminotransferase (ALT) in the serum.
Hematoxylin and eosin staining analysis
At the end of the experiment, three lambs were randomly selected from each treatment group and euthanized by intravenous injection of 90 mg/kg pentobarbital sodium. Approximately 3 cm of tissue samples from the duodenum, jejunum, and ileum were collected and fixed in 2.5% glutaraldehyde-polyoxymethylene. The intestinal tissues were dehydrated in a series of ethanol solutions with increasing concentrations (70%, 80%, 95%, 100%), cleaned with xylene, and embedded in liquid paraffin. The paraffin blocks were then sectioned into 5 μm slices and placed on glass slides. These sections were stained with hematoxylin and eosin for histological examination. The slices were observed and captured using an optical microscope (Olympus BX5, Tokyo, Japan) and measurements of villus height, villus width, and crypt depth were performed using ImageJ (NIH, Bethesda, Maryland, USA).
Gut flora composition analysis
The contents of the duodenum, jejunum, and ileum of the slaughtered lambs were collected and immediately stored at − 80 °C until analysis of the flora composition using Illumina high-throughput sequencing technology. The total DNA of microbial communities was extracted from the samples using the OMEGA Soil DNA Kit (M5635-02) (Omega Bio-Tek, Norcross, GA, USA) as per the manufacturer's instructions. The quality of the DNA extraction was evaluated by performing 1% agarose gel electrophoresis and UV spectrophotometry. The V3-V4 region of the bacterial 16S rRNA gene was amplified by PCR using the forward primer 338F (5′-ACTCCTACGGGAGGCAGCA-3′) and the reverse primer 806R (5′-GGACTACHVGGGTWTCTAAT-3′). The PCR products were recovered by 2% agarose gel electrophoresis and libraries were constructed using the NEXTflexTM Rapid DNA-Seq Kit according to the manufacturer's instructions. Sequencing was performed on the MiSeq PE300 platform (Illumina, San Diego, CA, USA). The raw sequencing data were quality controlled using Fastp (version 0.20.0), and assembled using FLASH (version 1.2.7)19,20. The sequences were clustered using the UPARSE software (version 7.1) with a similarity threshold of 97% and chimeras were removed. The species classification annotation of each sequence was performed using the RDP classifier software (version 2.2) and the Silva 16S rRNA database (version 138) as a reference, with a comparison threshold of 70%.
Data processing and analysis
In non-targeted metabolomics research, the raw mass spectrometry data is converted to mzXML format and processed using MSDAIL software for peak alignment, retention time correction, and peak area extraction. The extracted data from MSDAIL is then used for metabolite identification, data preprocessing, experimental data quality assessment, and correlation analysis. The experimental data of the lambs was initially processed using SPSS 22.0 (SPSS Inc., Chicago, Illinois, USA) and Excel 2016. Differences between treatments were examined using Tukey’s test, with P < 0.05 considered significant. The results are presented as mean values with standard error of the mean (SEM).
Results
Quality assessment of non-targeted metabolomics data
The total ion chromatograms (TIC) of the samples are shown in Fig. 1, with overlapping TIC for each treatment, indicating the experiment's reproducibility. The peak response intensity of FBQ substance is usually higher than that of UBQ.
Non-target metabolomic analysis of UBQ and FBQ
The metabolites of the samples were structurally identified by comparing their molecular weights, secondary fragmentation spectra, and retention times in the plant metabolome database, and the identification results were verified. The overall metabolite profile of the BanQi extracts is depicted in Fig. 2A, where the five samples from each treatment group are clustered together, indicating that fermentation altered the composition of metabolites present in the BanQi extracts. In total, 998 metabolites were identified; the proportion of each type of metabolite is shown in Fig. 2B. Among them, lipids and lipid-like molecules (20.641%), phenylpropanoids and polyketides (17.936%), benzenoids (11.323%), organic acids and derivatives (11.122%), and organic oxygen compounds (10.922%) were the main metabolites. Based on P < 0.05 and |log2FC|> 1152 and 222 metabolites were significantly increased and decreased in FBQ, respectively, compared to UBQ (Fig. 2C).
Heat map visualization of metabolites identified by UBQ and FBQ, metabolite ratio map, and volcano map. (A) Heat map visualization. Metabolite levels in each sample are normalized, with red representing metabolites of higher relative abundance and blue representing metabolites of lower mass. (B) Types and proportions of metabolites identified from UBQ and FBQ. (C) Metabolite volcano map. UBQ (Unfermented BanQi extract), FBQ (Fermented Banqi extract).
Multivariate statistical analysis of identified metabolites in UBQ and FBQ
Principal component analysis (PCA) was performed on 13 BanQi extracts (five UBQ, five FBQ, and three QC samples) using an unsupervised model to assess the overall differences between samples. The QC sample dataset was highly concentrated, indicating good stability of the testing method. Furthermore, PCA showed a high degree of aggregation within the sample treatment, and a clear separation was observed between FBQ and UBQ samples, indicating that fermentation can result in stable alterations of metabolites in BanQi extracts (Fig. 3A–B). After sevenfold cross-validation, an OPLS-DA model was established to compare the fermentation properties of the BanQi extracts. The closer the prediction parameters R2X, R2Y, and Q2 of the model are to 1, the more stable and reliable the model. The parameters of the OPLS-DA model used in this study are presented in Supplementary Table S1. The R2Y and Q2 scores of the positive ion model were both higher than 0.9, and the R2Y and Q2 scores of the negative ion model were both higher than 0.85, indicating the feasibility of the model, the stability of the positive ion model is superior to that of the negative ion model. Differential metabolites were screened using OPLS-DA VIP > 1, P < 0.05, and |log2FC|> 1, and 67 differentially expressed metabolites were identified. To further investigate the changes in these metabolites within the BanQi extracts, we categorized them into 42 up-regulated and 25 down-regulated metabolites and visualized them using a heatmap (Fig. 3C–D; Supplementary Tables S2-3).
Multivariate statistical analysis of identified metabolites. (A) PCA analysis of metabolite in positive ion mode. (B) PCA analysis of metabolite in negative ion mode. (C) Visualization of up-regulated differential metabolite heat map. (D) Visualization of down-regulated differential metabolite heat map. UBQ (Unfermented BanQi extract), FBQ (Fermented Banqi extract).
Effect of fermentation on the metabolite content of BanQi extract
In this study, FBQ had significantly increased phenylalanine (tenfold change; NEG_760), leucine (sevenfold change; NEG_330), and isoleucine (sixfold change; NEG_329) levels compared to UBQ (P < 0.05) . In addition, FBQ contained significantly increased small peptides such as Val–Leu–Pro–Val–Pro–Gln (57-fold change; POS_15211), Gly–Leu (32-fold change; NEG_1150), isoleucyl-isoleucine (30-fold change; NEG_2521), cyclo (Pro–Leu) (12-fold change; POS_2340), and leucyl-leucine (11-fold change; POS_3382) compared to UBQ (P < 0.05) (Fig. 3C; Supplementary Table S3). These results suggest that fermentation of the BanQi extract decomposed macromolecular proteins into small-molecule polypeptides and free amino acids. Besides, Compared with UBQ, FBQ had significantly increased vindesine (107-fold change; NEG_15093), echujin (52-fold change; POS_18158), reserpine (50-fold change; POS_14370), yunaconitine (44-fold change; POS_15359), and prednisone (36-fold change; POS_7962) (P < 0.05) (Fig. 3C; Table S3).
Growth performance
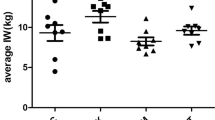
All lambs performed well and remained healthy during the experiment without any disease occurrence. The initial average body weights of the NC, UBQ, and FBQ groups were approximately 15.78, 15.82, and 15.77 kg, respectively, with no significant difference. After 28 days of feeding, the final body weight of lambs in the FBQ group (21.75 kg) was significantly higher than that in the NC group (P < 0.05). Furthermore, compared with the NC group, the average daily gain of lambs in the FBQ group increased significantly, and the feed-to-weight ratio decreased significantly (P < 0.05). The final body weight of lambs in the UBQ group increased slightly, but the difference was not significant. These results indicate that FBQ enhanced the growth performance of weaned lambs fed with Banqi extract (Table 2).
Serum biochemical parameters
As shown in Table 3, at 14d of the experiment, compared to the NC, the FBQ group exhibited significantly reduced in serum total protein (TP), urea levels, and aspartate aminotransferase (AST) (P < 0.05). The FBQ treatment group also showed lower levels of total cholesterol (TC), glucose (GLU), and alanine aminotransferase (ALT), but the differences were not statistically significant. Similarly, the UBQ treatment group displayed slight decreases in TP, TC, UREA, AST, and ALT levels, yet these differences were also not significant. On the 28th day of the experiment, no significant differences were observed in serum biochemical parameters among the treatment groups.
Morphology of small intestine
The morphology of the villus in duodenum, jejunum, and ileum is shown in Fig S1. Compared to the NC group, the villus in the duodenum, jejunum, and ileum of weaned lambs fed UBQ or FBQ appeared more orderly and compact. Table 4 showns the data on villus height, villus width, and crypt depth in each intestinal segment. Compared to the NC group, UBQ significantly reduced the villus height and villus width of lamb ileum (P < 0.05), while FBQ elevated the villus height in all intestinal segments, with a significant increase in the jejunum and ileum (P < 0.05). Additionally, the villus width in the duodenum and ileum of lambs in the FBQ group was significantly decreased (P < 0.05). The treatments in each group did not significantly affect the crypt depth of the small intestine.
Intestinal microbiota diversity and composition
Duodenum flora diversity and composition
The duodenum flora of lambs in the FBQ treatment had the highest quantity of OTUs compared to the UBQ or NC group (Fig. 4A). The α-diversity index revealed that the richness (Chao 1 index) and diversity (Simpson index) were significantly increased in the FBQ treatment compared to the NC (P < 0.05; Fig. 4B). β-diversity based on Bray–Curtis distances showed no crossover between treatment, indicating that FBQ significantly changed the composition of duodenal intestinal flora. (Fig. 4C). At the phylum level, compared with other treatments, the abundance of Firmicutes and Actinobacteria increased, while the abundances of Proteobacteria and Cyanobacteria decreased in the gut of lambs fed FBQ (Fig. 4D). LDA Effect Size (Lefse) analysis was used to Screening the dominant strains in each treatment (LDA threshold = 3). Overall, 17 dominant colonies were found in the duodenum across treatment. The FBQ treatment was significantly enriched in L7A_E11 and Mogibacterium, the NC was significantly enriched in Acinetobacter, and the UBQ treatment showed no colonies enriched at any genus level (P < 0.05) (Fig. 4E). In addition, we used the PICRUSt2 tool to map the 16S sequences to the genes and pathways that these bacterial species may harbor. After obtaining the relative abundance of signaling pathways, we filtered for metabolic pathways that showed significant differences between different treatment using a criterion of |log2FC|> 1. The results showed that compared to the NC, the enrichment of Proteasome, Secondary bile acid biosynthesis, Bacterial invasion of epithelial cells, Xylene degradation, N-Glycan biosynthesis, Staphylococcus aureus infection, Galactose metabolism, and Sphingolipid metabolism in FBQ treatment group increased significantly, while the enrichment of Isoflavonoid biosynthesis and Steroid hormone biosynthesis pathways decreased significantly (P < 0.05) (Fig. 4 F1). In addition, compared with the NC group, the enrichment of Proteasome, N-Glycan biosynthesis, and Malaria pathways in the UBQ treatment group increased significantly (P < 0.05). No significantly downregulated signaling pathways were predicted in the UBQ treatment (Fig. 4 F2).
Microbial diversity of duodenum of lambs (A) OTU Clustering Map. (B) Alpha Diversity (C) Beta Diversity. (D) Flora composition at the phylum level. (E) Differential flora analysis. (F) Functional Prediction Analysis of Gut flora. Dietary treatments: NC (Basic Diet), UBQ (Basic Diet + 6 ml/lamb/day Unfermented BanQi extract), FBQ (Basic Diet + 6 ml/lamb/day Fermented BanQi extract). FBQ-A, UBQ-A, and NC-A represent the duodenum of lambs in the FBQ, UBQ, and NCs, respectively.
Jejunum flora diversity and composition
OTU clustering of jejunum flora showed that FBQ increased colony diversity (Fig. 5A). α-diversity showed that the Simpson index of the UBQ group was significantly lower than that of the FBQ group (P < 0.05). β-diversity showed that there were significant differences in the composition and structure of intestinal flora among the three groups (Fig. 5C). The phylum-level results showed that FBQ lambs had increased abundances of Firmicutes and Tenericutes and a decreased abundance of Cyanobacteria compared to the NC; Elusimicrobia were found only in the UBQ treatment (Fig. 5D). LEFSE analysis revealed that 15 biomarkers, including YRC22 and Aeriscardovia, were significantly enriched in the jejunum of FBQ lambs, Herbaspirillum and Paenibacillus were significantly enriched in the jejunum of UBQ lambs, and Zoogloea and Clostridium was significantly enriched in the jejunum of NC lambs (P < 0.05) (Fig. 5E). The functional prediction of the jejunal flora showed that in the FBQ treatment, the enrichment of Polyketide sugar unit biosynthesis, Photosynthesis—antenna proteins, and Bacterial invasion of epithelial cells was significantly higher compared to the NC (P < 0.05), while the enrichment of Isoflavonoid biosynthesis was significantly lower than the NC (P < 0.05) (Fig. 5 F1). In comparison to the NC, the UBQ treatment exhibited a significant increase in the enrichment of Photosynthesis-antenna proteins and African trypanosomiasis (P < 0.05), while the enrichment of the mRNA surveillance pathway significantly decreased (P < 0.05) (Fig. 5 F2).
Microbial diversity of jejunum of lambs. (A) OTU Clustering Map. (B) Alpha Diversity. (C) Beta Diversity. (D) Flora composition at the phylum level. (E) Differential flora analysis. (F) Functional Prediction Analysis of Gut flora. Dietary treatments: Dietary treatments: NC (Basic Diet), UBQ (Basic Diet + 6 ml/lamb/day Unfermented BanQi extract), FBQ (Basic Diet + 6 ml/lamb/day Fermented BanQi extract). FBQ-B, UBQ-B, and NC-B represent the jejunum of lambs in the FBQ, UBQ, and NCs, respectively.
Ileum flora diversity and composition
Similar to the duodenum, the FBQ increased diversity in the Ileum flora compared to the other treatments. The UBQ treatment showed decreased abundance and diversity of flora compared to the NC group (Fig. 6A–C). At the phylum level, the highest abundances of Proteobacteria and Elusimicrobia were found in the ileum of the UBQ treatment (Fig. 6D). A total of 31 biomarkers were obtained using the LEFSE analysis. Butyrivibrio, Selenomonas, Eggerthella, Pseudoramibacter, Eubacterium, Lawsonia, and Streptococcus were all significantly enriched in the FBQ treatment, Clostridium, Chlamydia, and SMB53 were significantly enriched in the NC (P < 0.05), whereas the UBQ treatment showed no colony enrichment at the genus level (Fig. 6E). In addition, we also performed functional prediction of gut flora. The results showed that compared to the NC, FBQ significantly enriched the signal pathways of Bacterial invasion of epithelial cells, Penicillin and cephalosporin biosynthesis, and Biosynthesis of type II polyketide backbone, while the enrichment of Isoflavonoid biosynthesis, Staphylococcus aureus infection, mRNA surveillance pathway, Pathogenic Escherichia coli infection, and Lysosome decreased significantly (P < 0.05) (Fig. 6 F1). Compared with NC, UBQ significantly enriched the signal pathways of African trypanosomiasis and Lipopolysaccharide biosynthesis, while Lysosome, Staphylococcus aureus infection, and mRNA surveillance pathway were significantly reduced (Fig. 6 F2).
Microbial diversity of ileum of lambs (A) OTU Clustering Map. (B) Alpha Diversity (C) Beta Diversity. (D) Flora composition at the phylum level. (E) Differential flora analysis. (F) Functional Prediction Analysis of Gut flora. Dietary treatments: NC (Basic Diet), UBQ (Basic Diet + 6 ml/lamb/day Unfermented BanQi extract), FBQ (Basic Diet + 6 ml/lamb/day Fermented BanQi extract). FBQ-C, UBQ-C, and NC-C represent the ileum of lambs in the FBQ, UBQ, and NCs, respectively.
Discussion
To explore the influence of fermentation on the metabolites of traditional Chinese medicine, we analyzed the compositional changes of BanQi extract before and after fermentation using non-targeted metabolomics. FBQ had significantly increased Val–Leu–Pro–Val–Pro–Gln, Gly–Leu, isoleucyl-isoleucine, cyclo (Pro–Leu), and leucyl-leucine in small molecular quantities. These results show that in vitro fermentation can break down large-molecule proteins into small-molecule peptides, which is extremely important for young animals with immature absorption capabilities. Phenylalanine, leucine, and isoleucine are important building blocks for protein synthesis in animals and are catalyzed by hydroxylases to produce tyrosine, which reduces the effects of stressors21. Leucine and its metabolite β-hydroxy-β-methylbutyrate have been shown to activate the mTORC1 signaling pathway, induce skeletal muscle protein synthesis, attenuate insulin-independent protein degradation, and improve growth performance22,23. Isoleucine has been shown to restore growth performance in low-protein-fed piglets by improving intestinal development and the plasma metabolic profile and activating the hepatic Insulin-like Growth Factor 1 (IGF-1) pathway24. Vindesine, an analog of vinca alkaloid, and its analogs are classical drugs for NSCLC treatment. The pharmacological effects of aconitine include inotropic, analgesic, anti-inflammatory, anti-rheumatic, and neurotransmitting effects25,26. Reserpine is used together with prednisone as an antipsychotic and antihypertensive27,28, these free amino acids and medicinal components are enhanced after fermentation. Our study shows that BanQi extract fermented with B. subtilis can effectively enhance medicinal components of traditional herb medicine and convert biological macromolecules into small molecules, which are better suited for animal absorption.
Astragalus membranaceus is a widely used fermented traditional Chinese medicine in animal feeding. Studies have shown that the addition of fermented Astragalus membranaceus to the feed can enhance the growth performance, immune function, antioxidant capacity, and intestinal flora regulation of broiler chickens, with a 2% concentration achieving even better results. The results of this study showed that compared to UBQ, FBQ treatment was more effective in improving the growth performance of lambs, with a supplement dose of approximately 0.6% by weight. Serum total protein, albumin, creatinine, urea, and glucose concentrations have also been described as valuable parameters of hepatic injury and function29. In the middle of the experiment (14 d), we observed that TP, AST, and urea in the serum of FBQ lambs were all significantly lower than those in the NC. Increasing the villus height indicates enhancing the efficiency of digestion and absorption, thus long villi are correlated with improved gut health. The FBQ treatment significantly increased the villus height of the jejunum and ileum, whereas the UBQ treatment did not show such a significant change. These results suggest that supplementing with FBQ can improve the digestive and absorptive capacity of lambs while also protecting liver health.
The intestinal microbiota is an important component of intestinal health, and intestinal flora with high diversity may be more stable and healthier than that with low diversity30. It is noteworthy that FBQ could increase the abundance and diversity of microbial communities in the duodenum, jejunum, and ileum of lambs, whereas UBQ could decrease the diversity of microbial communities in the ileum and jejunum. This may be part of the reason why FBQ is more effective in enhancing the growth performance and intestinal villus height of lambs. Mogibacterium can produce phenylacetic acid, which is necessary for cellulose breakdown in ruminant strains31, whereas Butyrivibrio is the primary butyric acid-producing strain that plays a crucial role in the degradation of fibers and other complex polysaccharide32,33. These two flora were significantly enriched in the intestines of FBQ lambs. Acinetobacter is commonly found in soil and water. A. baumannii, A. nosocomial, A. pittii, A. ursingii, A. haemolyticus, and A. calcoaceticus are known to pose severe threats to human health34. Clostridium is a medically significant genus that includes pathogens that cause several diseases in adults and children, such as C. difficile and C. perfringens, which cause antibiotic-associated diarrhea and colitis35,36. Two species, Chlamydia trachomatis and C. pneumoniae, belonging to the genus Chlamydia, have been reported to cause various diseases37. The above-mentioned flora were all significantly enriched in the intestinal segments of the NC. The results of gut flora functional prediction showed significant differences in the abundance of signal pathways, such as Proteasome, Secondary bile acid biosynthesis, Penicillin and cephalosporin biosynthesis, Staphylococcus aureus infection, and Pathogenic Escherichia coli infection, between the NC and FBQ treatment. This suggests that feeding FBQ can regulate the energy metabolism of gut flora and modulate the sensitivity of gut flora to pathogenic microorganisms such as Escherichia coli and Staphylococcus aureus. Therefore, feeding FBQ to weaned lambs could be beneficial for reducing the abundance of harmful bacteria and increasing the enrichment of cellulose-degrading bacteria, thus improving the cellulose digestion capacity of lambs for improved feed utilization.
Conclusions
This study shows that probiotic fermentation is an effective method to enhance the efficacy of TCM. Feeding fermented BanQi extract can increase the height of intestinal villus and the abundance of cellulose-digesting bacteria, and reduce the abundance of pathogenic bacteria, which may be an effective dietary supplement for weaned lambs. This study provides experimental support for using fermented TCM to improve the growth performance of weaned lambs.
Data availability
All datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Li, C. et al. Effect of early weaning on the intestinal microbiota and expression of genes related to barrier function in lambs. Front. Microbiol. 9, 1431. https://doi.org/10.3389/fmicb.2018.01431 (2018).
Pascual-Alonso, M. et al. Effect of postweaning handling strategies on welfare and productive traits in lambs. J. Appl. Anim. Welf. Sci. 18, 42–56. https://doi.org/10.1080/10888705.2014.941107 (2015).
Li, Y. et al. Yeast peptides improve the intestinal barrier function and alleviate weaning stress by changing the intestinal microflora structure of weaned lambs. Microorganisms https://doi.org/10.3390/microorganisms11102472 (2023).
Hao, X. et al. Effects of Astragalus membranaceus roots supplementation on growth performance, serum antioxidant and immune response in finishing lambs. Asian-Australas. J. Anim. Sci. 33, 965–972. https://doi.org/10.5713/ajas.19.0295 (2020).
Sun, W. et al. Effects of dietary traditional Chinese medicine residues on growth performance, intestinal health and gut microbiota compositions in weaned piglets. Front Cell Infect. Microbiol. 13, 1283789. https://doi.org/10.3389/fcimb.2023.1283789 (2023).
Chen, H. et al. Response of growth performance, blood biochemistry indices, and rumen bacterial diversity in lambs to diets containing supplemental probiotics and Chinese medicine polysaccharides. Front Vet. Sci. 8, 681389. https://doi.org/10.3389/fvets.2021.681389 (2021).
Wang, Y., Qiao, C. Z., Liu, S. & Hang, H. M. Evaluation on antiendotoxic action and antiviral action in vitro of tetraploid Isatis indigotica. Zhongguo Zhong yao za zhi = Zhongguo zhongyao zazhi = China J. Chin. Mater. Medica 25, 327–329 (2000).
Liu, Y., Weng, W., Gao, R. & Liu, Y. New insights for cellular and molecular mechanisms of aging and aging-related diseases: Herbal medicine as potential therapeutic approach. Oxid. Med. Cell. Longev. 2019, 4598167. https://doi.org/10.1155/2019/4598167 (2019).
Li, L. et al. The application of fermentation technology in traditional Chinese medicine: A review. Am. J. Chin. Med. 48, 899–921. https://doi.org/10.1142/s0192415x20500433 (2020).
Xu, J., Crowley, M. F. & Smith, J. C. Building a foundation for structure-based cellulosome design for cellulosic ethanol: Insight into cohesin-dockerin complexation from computer simulation. Prot. Sci. Publ. Prot. Soc. 18, 949–959. https://doi.org/10.1002/pro.105 (2009).
Wang, L., Zhang, G., Xu, H., Xin, H. & Zhang, Y. Metagenomic analyses of microbial and carbohydrate-active enzymes in the rumen of holstein cows fed different forage-to-concentrate ratios. Front. Microbiol. 10, 649. https://doi.org/10.3389/fmicb.2019.00649 (2019).
Xu, Y. et al. Monitoring of the bacterial and fungal biodiversity and dynamics during Massa Medicata Fermentata fermentation. Appl. Microbiol. Biotechnol. 97, 9647–9655. https://doi.org/10.1007/s00253-013-5187-0 (2013).
Spatuzza, C. et al. Effects of berberine and red yeast on proinflammatory cytokines IL-6 and TNF-α in peripheral blood mononuclear cells (PBMCs) of human subjects. Front. Pharmacol. 5, 230. https://doi.org/10.3389/fphar.2014.00230 (2014).
Chen, G. et al. Fermented Chinese herbal medicine promoted growth performance, intestinal health, and regulated bacterial microbiota of weaned piglets. Anim. Open Access J. MDPI https://doi.org/10.3390/ani13030476 (2023).
Zhou, H., Wang, C., Ye, J., Chen, H. & Tao, R. Effects of dietary supplementation of fermented Ginkgo biloba L. residues on growth performance, nutrient digestibility, serum biochemical parameters and immune function in weaned piglets. Anim. Sci. J. Nihon Chikusan Gakkaiho 86, 790–799. https://doi.org/10.1111/asj.12361 (2015).
Wang, J., Cao, F., Su, E., Zhao, L. & Qin, W. Improvement of animal feed additives of Ginkgo leaves through solid-state fermentation using Aspergillus niger. Int. J. Biol. Sci. 14, 736–747. https://doi.org/10.7150/ijbs.24523 (2018).
Niu, K., Wang, H., Kim, S. K., Wassie, T. & Wu, X. Stepwise co-fermented traditional Chinese medicine byproducts improve antioxidant and anti-inflammatory effects in a piglet model. J. Sci. Food Agric. 104, 1166–1177. https://doi.org/10.1002/jsfa.13002 (2024).
Zhu, X. et al. Enhancement of anti-acne effect of Scutellaria baicalensis extract by fermentation with symbiotic fungus Penicillium decumbens. J. Biosci. Bioeng. 130, 457–463. https://doi.org/10.1016/j.jbiosc.2020.06.008 (2020).
Chen, S., Zhou, Y., Chen, Y. & Gu, J. fastp: An ultra-fast all-in-one FASTQ preprocessor. Bioinform. (Oxf. Engl.) 34, i884–i890. https://doi.org/10.1093/bioinformatics/bty560 (2018).
Magoč, T. & Salzberg, S. L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinform. (Oxf. Engl.) 27, 2957–2963. https://doi.org/10.1093/bioinformatics/btr507 (2011).
Banderet, L. E. & Lieberman, H. R. Treatment with tyrosine, a neurotransmitter precursor, reduces environmental stress in humans. Brain Res. Bull. 22, 759–762. https://doi.org/10.1016/0361-9230(89)90096-8 (1989).
Suryawan, A. et al. Enteral leucine supplementation increases protein synthesis in skeletal and cardiac muscles and visceral tissues of neonatal pigs through mTORC1-dependent pathways. Pediatric Res. 71, 324–331. https://doi.org/10.1038/pr.2011.79 (2012).
Wilkinson, D. J. et al. Effects of leucine and its metabolite β-hydroxy-β-methylbutyrate on human skeletal muscle protein metabolism. J. Physiol. 591, 2911–2923. https://doi.org/10.1113/jphysiol.2013.253203 (2013).
Habibi, M. et al. A mixture of valine and isoleucine restores the growth of protein-restricted pigs likely through improved gut development, hepatic IGF-1 pathway, and plasma metabolomic profile. Int. J. Mol. Sci. https://doi.org/10.3390/ijms23063300 (2022).
Honerjäger, P. & Meissner, A. The positive inotropic effect of aconitine. Naunyn-Schmiedeberg’s Arch. Pharmacol. 322, 49–58. https://doi.org/10.1007/bf00649352 (1983).
Hikino, H. et al. Antiinflammatory principles of Aconitum roots. J. Pharmacobio-Dyn. 3, 514–525. https://doi.org/10.1248/bpb1978.3.514 (1980).
Hu, G. et al. New fluorescent substrate enables quantitative and high-throughput examination of vesicular monoamine transporter 2 (VMAT2). ACS Chem. Biol. 8, 1947–1954. https://doi.org/10.1021/cb400259n (2013).
Liu, D. et al. Preparation of colon-targeted acetylharpagide tablets and its release properties in vivo and in vitro. Front. Pharmacol. 9, 832. https://doi.org/10.3389/fphar.2018.00832 (2018).
Mathur, S. et al. Fumonisin B(1) is hepatotoxic and nephrotoxic in milk-fed calves. Toxicol Sci. 60, 385–396. https://doi.org/10.1093/toxsci/60.2.385 (2001).
Li, C. L. et al. Intestinal morphologic and microbiota responses to dietary Bacillus spp. in a broiler chicken model. Front Physiol. 9, 1968. https://doi.org/10.3389/fphys.2018.01968 (2018).
Morrison, M., Mackie, R. I. & Kistner, A. 3-Phenylpropanoic acid improves the affinity of Ruminococcus albus for cellulose in continuous culture. Appl. Environ. Microbiol. 56, 3220–3222. https://doi.org/10.1128/aem.56.10.3220-3222.1990 (1990).
Wallace, R. J., McKain, N., Shingfield, K. J. & Devillard, E. Isomers of conjugated linoleic acids are synthesized via different mechanisms in ruminal digesta and bacteria. J. Lipid Res. 48, 2247–2254. https://doi.org/10.1194/jlr.M700271-JLR200 (2007).
Shomorony, I. et al. An unsupervised learning approach to identify novel signatures of health and disease from multimodal data. Genome Med. 12, 7. https://doi.org/10.1186/s13073-019-0705-z (2020).
Weber, B. S., Harding, C. M. & Feldman, M. F. Pathogenic acinetobacter: From the cell surface to infinity and beyond. J. Bacteriol. 198, 880–887. https://doi.org/10.1128/jb.00906-15 (2015).
Rupnik, M., Wilcox, M. H. & Gerding, D. N. Clostridium difficile infection: New developments in epidemiology and pathogenesis. Nat. Rev. Microbiol. 7, 526–536. https://doi.org/10.1038/nrmicro2164 (2009).
Preidis, G. A. & Versalovic, J. Targeting the human microbiome with antibiotics, probiotics, and prebiotics: Gastroenterology enters the metagenomics era. Gastroenterology 136, 2015–2031. https://doi.org/10.1053/j.gastro.2009.01.072 (2009).
Peng, Y. et al. The glycolipid exoantigen derived from Chlamydia muridarum activates invariant natural killer T cells. Cell. Mol. Immunol. 9, 361–366. https://doi.org/10.1038/cmi.2012.19 (2012).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (31972731) and Key Research and Development Program of Henan Province (221111111300).
Author information
Authors and Affiliations
Contributions
J.F. and H.C. completed the whole experiment and wrote the main manuscript text, Z.M. and C.Y. participated in the data processing, M.Y., Y.J. and C.N. assisted in the experimental design, and H.Z. was the main sponsor and sponsor of the study. All the authors have reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Fan, J., Cui, H., Mu, Z. et al. Non-targeted metabolomics analysis of fermented traditional Chinese medicine and its impact on growth performance, serum biochemistry, and intestinal microbiome of weaned lambs. Sci Rep 14, 20385 (2024). https://doi.org/10.1038/s41598-024-71516-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-024-71516-x
- Springer Nature Limited