Abstract
The early genome-wide association studies (GWAS) found a significant association between lung cancer and rs1051730 (15q25) polymorphism. However, the subsequent studies reported consistent and inconsistent results in different populations. Three meta-analysis studies were thus performed to reevaluate the association. But their results remain inconsistent. After that, some new GWAS studies reported conflicting results again. We think that the divergence of these results may be due to small-scale samples or heterogeneity among different populations. Therefore, we reevaluated the association by collecting more samples (N = 33,617 cases and 116,639 controls) from 31 studies, which incorporate 8 new studies and 23 previous studies used by one or more of the three meta-analysis studies. We observed a significant association between lung cancer and rs1051730 in pooled population by using allele (OR = 1.30, 95% CI = 1.27–1.34, P < 0.0001), dominant (OR = 1.41, 95% CI = 1.29–1.55, P < 0.0001), recessive (OR = 1.53, 95% CI = 1.42–1.65, P < 0.0001) and additive (OR = 1.75, 95% CI = 1.61–1.90, P < 0.0001) models. Through the subgroup analysis, we observed a significant heterogeneity only in East Asian population (P = 0.006, I2 = 66.9%) and the association is significant in all subgroups (OR = 1.2976, 95% CI = 1.2622–1.3339 (European ancestry), OR = 1.5025, 95% CI = 1.2465–1.8110 (African), OR = 1.7818, 95% CI = 1.3915–2.2815 (East Asian), P < 0.0001). We believe that these results will contribute to understanding the genetic mechanism of lung cancer.
Similar content being viewed by others
Introduction
Lung cancer is one of the most dangerous diseases and the morbidity and mortality are increasing constantly around the world today1. According to the statistics from American Cancer Society (ACS), the new cases of lung cancer were 0.221 million and dead 0.158 million in the United States in 2015, which was one of the most frequently diagnosed cancers and the leading causes of cancer death in men and women2. Even though, the majority of studies proved that over 80 percent lung cancer occurrence is associated with the habit of smoking, while only 20 percent of these smokers will develop into lung cancer, which suggests that lung cancer occurrence possesses genetic susceptibility1,3.
Many single nucleotide polymorphism (SNP) sites in association with lung cancer were obtained by the genome–wide association studies (GWAS)4. For example, Amos et al.5, Thorgeirsson, et al.6 and Shiraishi et al.7 found that the rs1051730 SNP was associated with lung cancer in European ancestry and Japanese population, respectively. Amos et al. analyzed 956 cases and 1,830 controls from the UK and discovered the odds ratio (OR) = 1.32, 95% confidence interval (CI) is from 1.23 to 1.39 and P < 10E–08. Thorgeirsson, et al. analyzed 665 cases and 28,752 controls from the Iceland, 269 cases and 1,474 controls from the Spain, 90 cases and 2,018 controls from the Netherlands and the combined result showed that OR = 1.31, 95% CI is from 1.19 to 1.44 and P = 1.5E–08. Shiraishi et al. analyzed 1,250 cases and 936 controls from the Japan and found the OR = 2.3, 95% CI is from 1.5 to 3.7 and P = 0.0028.
However, the subsequent studies reported consistent8,9,10,11,12,13,14,15,16,17,18 and inconsistent19,20 results. For example, Wu et al.19 used 1,151 cases and 1,150 controls from China and Girard et al.20 used 94 cases and 95 controls from America to investigate the association between rs1051730 and lung cancer and obtained negligible or weak association.
Facing the inconsistent results above, Gu et al.21, Hu et al.22 and Zhan et al.23 collected different samples to re-assess the association between rs1051730 and lung cancer by meta-analysis, respectively. However, the results of the three meta-analysis studies are also inconsistent. Gu et al.21 and Hu et al.22 found that the association between rs1051730 and lung cancer is not significant, while it is significant according to the study of Zhan et al.23.
After these studies, five articles24,25,26,27,28, which include eight studies, investigated the association between rs1051730 and lung cancer. But the results of eight studies remain inconsistent. For example, He et al.27 used 301 cases and 318 controls from China, Hansen et al.28 used 448 cases and 611 controls from America and found a negligible association between rs1051730 and lung cancer, while other researches24,25,26 reported opposite consequences.
We considered that the divergence of these results may be due to small-scale samples or heterogeneity among different populations. Here, we collected all the samples of the three meta-analysis studies and the eight studies from the five new articles and thus obtained a larger sample sizes (33,617 cases and 116,639 controls) from 31 studies to reevaluate the association between rs1051730 polymorphism and lung cancer based on the method that was frequently used by Liu et al. to study Alzheimer’s Disease29,30,31,32 and colorectal cancer33.
Results
Literature and Study acquisition as well as Data extraction
By searching PubMed with keywords (details shown in the method section), we obtained 20 articles, which include 16 articles used in the three previous meta-analysis researches and 4 new articles. Moreover, we obtained another article through the reference search in Google Scholar. Finally, we got 31 studies from the 21 articles according to the six inclusion criteria (details shown in the method section). The workflow was showed in Fig. 1. After that, we collected the relative data according to 11 terms for each study and 7 of the 11 terms were listed in Table 1.
The flow chart of collecting articles for analyzing the association between rs1051730 polymorphism and lung cancer.
The criteria of selecting articles are: (1) the study was designed according to the method of case-control. (2) the study evaluated the association of rs1051730 polymorphism and lung cancer. (3) the number of cases and controls was provided in the study. (4) the study provided the population of each individual. (5) the study provided the number of rs1051730 genotypes both in cases and controls or provided enough data to calculate the genotypes. (6) the study provided the OR value with 95% CI and the P value or provided enough data to calculate them.
Heterogeneity Test
According to the kind of genotype shown in Table 1, we used 30 studies in the allele model, 17 studies in the dominant model, 14 studies in the recessive model and 13 studies in the additive model, respectively. We didn’t get a significant heterogeneity in the allele model (P = 0.0161 and I2 = 39.1%), recessive model (P = 0.4208 and I2 = 2.7%) and additive model (P = 0.1791 and I2 = 26.3%). However, it is significant in the dominant model (P = 0.0023 and I2 = 56.4%).
Meta-analysis
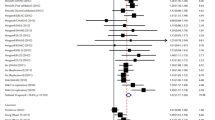
Because the heterogeneity is significant in the dominant model, we used the random effect model to analyze it. And because the heterogeneity isn’t significant in the allele, recessive and additive models, we used the fix effect model to analyze them and get a relatively strong association between the rs1051730 and lung cancer in the four models (OR = 1.30 and 95% CI = 1.27–1.34 in the allele model, OR = 1.41 and 95% CI = 1.29–1.55 in the dominant model, OR = 1.53 and 95% CI = 1.42–1.65 in the recessive model, OR = 1.75 and 95% CI = 1.61–1.90 in additive model, respectively). The P values of Z test are less than 0.0001 in all models, which proved that these OR values are believable. These conclusions were described in Figs 2, 3, 4, 5.
Forest plot for the meta-analysis of rs1051730 polymorphism using allele model.
According to Table 1, 30 studies that provided the data of genotype A/G and AA/GA/GG and GA/GG were used in the allele model (A vs G).
Forest plot for the meta-analysis of rs1051730 polymorphism using dominant model.
According to Table 1, 17 studies that provided the data of genotype AA/GA/GG and GA/GG were used in the dominant model (AA+AG vs GG).
Forest plot for the meta-analysis of rs1051730 polymorphism using recessive model.
According to Table 1, 14 studies that provided the data of genotype AA/GA/GG as well as AA/GA and GG were used in the recessive model (AA vs AG+GG).
Forest plot for the meta-analysis of rs1051730 polymorphism using additive model.
According to Table 1, 13 studies that provided the data of genotype AA/GA/GG were used in the additive model (AA vs GG).
Subgroup Analysis
Because the allele model included the maximum number of studies, we further used it to perform the subgroup analysis. We found that there is no significant heterogeneity in European ancestry (P = 0.2296 and I2 = 18.4%) and African (P = 0.1586 and I2 = 42.2%) populations, while in East Asian population the heterogeneity is significant (P = 0.006 and I2 = 66.9%). So we further split East Asian population into Japanese and Chinese subgroups. The heterogeneity was not found in the Japanese population (P = 0.8387 and I2 = 0), but it remains significant in the Chinese population (P = 0.0753 and I2 = 56.5%). Then, we removed each study from Chinese population orderly and found that there was no significant heterogeneity after the study of Wu et al. had been removed. After meta-analysis and Z test, we found the association between the rs1051730 and lung cancer is relatively strong in all populations. The results were described in Table 2. Forest plots of each subgroup meta-analysis were shown in Figure S1–S7.
Publication Bias Analysis and Sensitivity Analysis
We used Begg’s test and Egger’s test to measure the publication bias for the allele, dominant, recessive and additive models and didn’t find a significant publication bias in all these models. The P values of Begg’s test and Egger’s test for the allele, dominant, recessive and additive models are 0.4077 and 0.3897, 0.1061 and 0.0869, 0.1838 and 0.2044, 0.1314 and 0.1619, respectively. The funnel plot (Fig. 6) reflected the result directly. And then, the result of sensitivity analysis showed that the association between rs1051730 and lung cancer doesn’t extremely change when we removed each of the studies in the four models orderly. The detail information was in supplement materials (Table S5–S8).
Funnel plot of publication bias analysis in the allele, dominant, recessive and additive model.
This funnel plot shows the result of the publication bias analysis for rs1051730 polymorphism with lung cancer using allele (a), dominant (b), recessive (c) and additive (d) model. The X-axis represents OR value and the Y-axis represents standard error of each study, respectively.
Discussion
Nicotine receptor protein abnormal expression is one of the reasons for lung cancer occurrence3. CHRNA3, a gene coding a part of nicotinic acetylcholine receptor protein subunits, includes a SNP rs1051730. The elder GWAS studies showed that a significant association between the rs1051730 polymorphism and lung cancer in the European ancestry and Japanese population5,6,7. However, the subsequent studies that researched the association in various populations or in different scales reported consistent and inconsistent results. We think the divergence of these results might be due to the small-scale samples or heterogeneity among different populations. So we summarized these studies in a larger scale and in a more comprehensive population and found that the association between rs1051730 polymorphism and lung cancer is significant by using the allele model (OR = 1.30, 95% CI = 1.27–1.34, P < 0.0001), dominant model (OR = 1.41, 95% CI = 1.29–1.55, P < 0.0001), recessive model (OR = 1.53, 95% CI = 1.42–1.65, P < 0.0001) and additive model (OR = 1.75, 95% CI = 1.61–1.90, P < 0.0001). The farther subgroup analysis showed a significant association in European ancestry (OR = 1.2976, 95% CI = 1.2622–1.3339, P < 0.0001) and African (OR = 1.5025, 95% CI = 1.2465–1.8110, P < 0.0001) populations. But in East Asian population, we found a weak association with a non–significant result of Z test (OR = 1.4759, CI = 0.9957–2.1878, P = 0.0526) and the heterogeneity is significant (P = 0.006 and I2 = 66.9%). So we further split East Asian population into Japanese and Chinese subgroups. The result indicated that there is a significant association in the Japanese population (OR = 2.2654, 95% CI = 1.5675–3.2741, P < 0.0001), while the result of Z test is still non-significant in the Chinese population (P = 0.4389). We removed each study from Chinese population orderly and found a significant association (OR = 1.4235, 95% CI = 1.0146–1.9972, P = 0.041) after the study of Wu et al. had been removed. We think this phenomenon may be caused by a relatively small sample size in the study of Wu et al. Moreover, we removed the study of Wu et al from East Asian population and result of meta-analysis showed that the association is significant in East Asian population (OR = 1.7818, 95% CI = 1.3915–2.2815, P < 0.0001). Consequently, the association between rs1051730 polymorphism and lung cancer is significant in all of these populations. In addition, the result of sensitivity analysis reflects the conclusion is robust and the publication bias isn’t significant.
Before submitting this paper, we used keyword “rs1051730”, “lung cancer” and “meta” to search in PubMed and obtained six articles21,22,23,34,35,36, which include the researches of Gu et al.21, Hu et al.22 and Zhan et al.23. Among these three articles, Gu et al.21 integrated 16 studies to assess the risk of rs1051730 in East Asian, European and African populations by using allelic and dominant models. Hu et al.22 also collected 16 studies in the same populations, but merely the allelic model was used. They obtained a similar result that the risk is high in European and African populations, but it is weak in East Asian. Zhan et al.23 only assessed the East Asian, they combined 4 studies and the result shows a significant association between the rs1051730 and lung cancer. The other three articles didn’t research the association between the rs1051730 polymorphism and lung cancer. They evaluated the association between rs1051730 and habit of smoking34,35,36, or the association between rs1051730 and cotinine levels36 by meta-analysis.
Our work is different from the others. We collected all 31 studies, which include 23 studies used by the three previous meta-analysis researches and 8 new studies. We analyzed the association between rs1051730 and lung cancer in European ancestry, African and East Asian populations. The association is significant in European ancestry and African populations, which is consistent with the result of Gu et al.21 and Hu et al.22. However, we found that the association remains significant in East Asian population, which is not consistent with the result of Gu et al.21 and Hu et al.22.
The rs1051730 polymorphism is in CHRNA3 on 15q25. CHRNA3 is one of members in a multigene family of nicotinic acetylcholine receptor (nAChR gene cluster) which can code various nicotinic acetylcholine receptor protein subunits include: α3, α4, α7, α9, α10, β2 and β4 nAChR subunits37. These subunits are expressed on the bronchial epithelial cells of human being and primates38. Through combining with the nicotinic acetylcholine receptor protein, nicotine promotes tumor cell proliferation, invasion, migration and induces blood vessel formation. At the same time, it provides a protection for the tumor cell to avoid the programmed cell death39. In addition, Arredondo et al.40 found that α3, α4, α7, α9, α10, β2 and β4 nAChR subunits can form the high-affinity sites to bind 4-(methylnitro-samino)-1-(3-pyridyl)-1-butanone (NNK), a cancerogen produced through nicotine nitrosylation, thus to increase the risk of lung cancer. Moreover, CHRNA3 also contains other two SNP: rs578776 and rs938682 polymorphism. The rs17486278, rs11637635 as well as rs7178270 polymorphism belong to CHRNA5 and CHRNA4, respectively. They may be also associated with the susceptibility of non-small cell lung cancer was reported in a study41. In addition, another gene AGPHD1 also in 15q25 contains the rs8034191 polymorphism and many studies reported its association with lung cancer5,8,9,20,24. We expect that more research on them could be performed in the future.
Methods
Literature and Study acquisition
We collected all the articles which were used to perform meta-analysis by Gu et al.21, Hu et al.22 and Zhan et al.23. And then, we searched all the possible articles in PubMed (http://www.ncbi.nlm.nih.gov/pubmed) with keywords: “rs1051730” and “lung cancer”, or “rs1051730” and “Carcinoma”, or “rs1051730” and “tumor”, or “CHRNA3” and “lung cancer”, or “CHRNA3” and “Carcinoma”, or “CHRNA3” and “tumor”. All of these literatures had been collected before the PubMed’s last update on April 7 2015. In addition, we selected the related references in these articles both from PubMed and the three meta-analysis researches by using Google Scholar (http://scholar.google.com/). All the selected articles were written in English.
After that, we selected the studies in all obtained articles according to the following criteria: (1) The study was designed according to the method of case-control. (2) The study evaluated the association of rs1051730 polymorphism and lung cancer. (3) The number of cases and controls was provided in the study. (4) The study provided the population of each individual. (5) The study provided the number of rs1051730 genotypes both in cases and controls or provided enough data to calculate the genotypes. (6) The study provided the OR value with 95% CI and the P value or provided enough data to calculate these.
Data extraction
We extracted the following information from each study we have selected: (1) The first author of each article. (2) The publication year of each article. (3) The population and ethnicity of individual in each study. (4) The number of cases and controls in each study. (5) The number of rs1051730 genotypes both in cases and controls. (6) The OR value with 95% CI and the P value in each study. (7) The genotyping platform. If the information didn’t be provided directly, we used program R (http://www.r-project.org/) to work them out.
Choice of genetic model
The rs1051730 polymorphism has two alleles A and G. Among them, A is the minor allele. We analyzed the association between rs1051730 polymorphism and lung cancer by using the allele model (A allele versus G allele), the dominant model (AA+GA versus GG), the recessive model (AA versus GA+GG) and the additive model (AA versus GG), respectively. According to Table 1, among these analyses, the studies that provided the kind of genotype A/G and AA/GA/GG and GA/GG were used in the allele model, the studies that provided the kind of genotype AA/GA/GG and GA/GG were used in the dominant model, the studies that provided the kind of genotype AA/GA/GG as well as AA/GA and GG were used in the recessive model, the studies that provided the kind of genotype AA/GA/GG were used in the additive model. These classification data according to genotype were shown in supplement materials (Table S1–S4).
Heterogeneity Test
We used two quantities, Cochran’s Q and I2, to measure the heterogeneity among the different ethnic groups. Q approximately follows a χ2 distribution with k-1 degrees of freedom (where k is the number of studies) and the P value can be used to measure the significance level of heterogeneity. I2 = (Q-(K-1))/Q*100%, which ranges from 0 to 100%. Usually, we would tentatively assign adjectives of low, moderate and high to I2 values of 25%, 50% and 75%42. In our study, we deemed that the different ethnic groups have significant heterogeneity when P < 0.01 and I2 > 50%29,30,31,32,33.
Meta-analysis and Subgroup Analysis
Two models can be used in meta-analysis: the random effect model and the fix effect model43. After heterogeneity test, we used the software package meta (http://cran.r-project.org/web/packages/meta/index.html) to perform the meta-analysis with the random effect model, if the heterogeneity is significant and used the fix effect model, if the heterogeneity is not. And then, we used Z test to examine the significance of meta-analysis. We further conducted a subgroup analysis after meta-analysis. We split original studies into smaller groups according to population and conducted the meta-analysis to each subgroup respectively.
Publication Bias Analysis and Sensitivity Analysis
Begg’s test44 and Egger’s test45 are two methods to evaluate the publication bias. When the P values are less than 0.05, publication bias is deemed significant. We measured the publication bias of each group, which include the allele model, the dominant model, the recessive model and the additive model, by the Begg’s and Egger’s test. Meanwhile, we drew the funnel plot to show the bias of each group directly. And then, we removed each study in these groups orderly to measure the influence of each study. We used the software package named meta of program R for all these calculations.
Additional Information
How to cite this article: Han, Z. et al. Analyzing large-scale samples confirms the association between the rs1051730 polymorphism and lung cancer susceptibility. Sci. Rep. 5, 15642; doi: 10.1038/srep15642 (2015).
References
Jemal, A. et al. Global cancer statistics. CA Cancer J Clin 61, 69–90 (2011).
Siegel, R. L. et al. Cancer statistics, 2015. CA Cancer J Clin 65, 5–29 (2015).
Alberg, A. J. & Samet, J. M. Epidemiology of lung cancer. Chest 123, 21S–49S (2003).
Yang, I. A., Holloway, J. W. & Fong, K. M. Genetic susceptibility to lung cancer and co-morbidities. J Thorac Dis 5 Suppl 5, S454–462 (2013).
Amos, C. I. et al. Genome-wide association scan of tag SNPs identifies a susceptibility locus for lung cancer at 15q25.1. Nat Genet 40, 616–622 (2008).
Thorgeirsson, T. E. et al. A variant associated with nicotine dependence, lung cancer and peripheral arterial disease. Nature 452, 638–642 (2008).
Shiraishi, K. et al. Contribution of nicotine acetylcholine receptor polymorphisms to lung cancer risk in a smoking-independent manner in the Japanese. Carcinogenesis 30, 65–70 (2009).
Zienolddiny, S. et al. The TERT-CLPTM1L lung cancer susceptibility variant associates with higher DNA adduct formation in the lung. Carcinogenesis 30, 1368–1371 (2009).
Schwartz, A. G., Cote, M. L., Wenzlaff, A. S., Land, S. & Amos, C. I. Racial differences in the association between SNPs on 15q25.1, smoking behavior and risk of non-small cell lung cancer. J Thorac Oncol 4, 1195–1201 (2009).
Kohno, T. et al. Contribution of the TP53, OGG1, CHRNA3 and HLA-DQA1 genes to the risk for lung squamous cell carcinoma. J Thorac Oncol 6, 813–817 (2011).
VanderWeele, T. J. et al. Genetic variants on 15q25.1, smoking and lung cancer: an assessment of mediation and interaction. Am J Epidemiol 175, 1013–1020 (2012).
Yang, L. et al. Functional polymorphisms of CHRNA3 predict risks of chronic obstructive pulmonary disease and lung cancer in Chinese. PLoS One 7, e46071 (2012).
Ren, J. H. et al. Association between CHRNA3 rs1051730 genotype and lung cancer risk in Chinese Han population: a case-control study. J Huazhong Univ Sci Technolog Med Sci 33, 897–901 (2013).
Sakoda, L. C. et al. Chromosome 15q24-25.1 variants, diet and lung cancer susceptibility in cigarette smokers. Cancer Causes Control 22, 449–461 (2011).
Amos, C. I. et al. Nicotinic acetylcholine receptor region on chromosome 15q25 and lung cancer risk among African Americans: a case-control study. J Natl Cancer Inst 102, 1199–1205 (2010).
Wassenaar, C. A., Dong, Q., Amos, C. I., Spitz, M. R. & Tyndale, R. F. Pilot Study of CYP2B6 Genetic Variation to Explore the Contribution of Nitrosamine Activation to Lung Carcinogenesis. Int J Mol Sci 14, 8381–8392 (2013).
Liu, P. et al. Familial aggregation of common sequence variants on 15q24-25.1 in lung cancer. J Natl Cancer Inst 100, 1326–1330 (2008).
Landi, M. T. et al. A genome-wide association study of lung cancer identifies a region of chromosome 5p15 associated with risk for adenocarcinoma. Am J Hum Genet 85, 679–691 (2009).
Wu, C. et al. Genetic variants on chromosome 15q25 associated with lung cancer risk in Chinese populations. Cancer Res 69, 5065–5072 (2009).
Girard, N. et al. Analysis of genetic variants in never-smokers with lung cancer facilitated by an Internet-based blood collection protocol: a preliminary report. Clin Cancer Res 16, 755–763 (2010).
Gu, M. et al. Strong association between two polymorphisms on 15q25.1 and lung cancer risk: a meta-analysis. PLoS One 7, e37970 (2012).
Hu, B. et al. Quantitative assessment of the influence of common variations (rs8034191 and rs1051730) at 15q25 and lung cancer risk. Tumour Biol 35, 2777–2785 (2014).
Zhan, P. & Song, Y. CHRNA3 rs1051730 polymorphism and lung cancer susceptibility in Asian population: a meta-analysis. Transl Lung Cancer Res 4, 104–108 (2015).
Kaur-Knudsen, D., Nordestgaard, B. G. & Bojesen, S. E. CHRNA3 genotype, nicotine dependence, lung function and disease in the general population. Eur Respir J 40, 1538–1544 (2012).
Liu, P. et al. A second genetic variant on chromosome 15q24-25.1 associates with lung cancer. Cancer Res 70, 3128–3135 (2010).
Kaur-Knudsen, D., Bojesen, S. E., Tybjaerg-Hansen, A. & Nordestgaard, B. G. Nicotinic acetylcholine receptor polymorphism, smoking behavior and tobacco-related cancer and lung and cardiovascular diseases: a cohort study. J Clin Oncol 29, 2875–2882 (2011).
He, P. et al. CHRNA3 polymorphism modifies lung adenocarcinoma risk in the Chinese Han population. Int J Mol Sci 15, 5446–5457 (2014).
Hansen, H. M. et al. Fine mapping of chromosome 15q25.1 lung cancer susceptibility in African-Americans. Hum Mol Genet 19, 3652–3661 (2010).
Liu, G. et al. Analyzing large-scale samples confirms the association between the ABCA7 rs3764650 polymorphism and Alzheimer’s disease susceptibility. Mol Neurobiol 50, 757–764 (2014).
Shen, N. et al. An Updated Analysis with 85,939 Samples Confirms the Association Between CR1 rs6656401 Polymorphism and Alzheimer’s Disease. Mol Neurobiol 51, 1017–1023 (2015).
Chen, H. et al. Analyzing 54,936 Samples Supports the Association Between CD2AP rs9349407 Polymorphism and Alzheimer’s Disease Susceptibility. Mol Neurobiol 52, 1–7 (2015).
Li, X. et al. CD33 rs3865444 Polymorphism Contributes to Alzheimer’s Disease Susceptibility in Chinese, European and North American Populations. Mol Neurobiol 52, 414–421 (2015).
Liao, M. et al. Analyzing large-scale samples confirms the association between rs16892766 polymorphism and colorectal cancer susceptibility. Sci Rep 5, 7957 (2015).
Thorgeirsson, T. E. et al. Sequence variants at CHRNB3-CHRNA6 and CYP2A6 affect smoking behavior. Nat Genet 42, 448–453 (2010).
Ware, J. J., van den Bree, M. B. & Munafo, M. R. Association of the CHRNA5-A3-B4 gene cluster with heaviness of smoking: a meta-analysis. Nicotine Tob Res 13, 1167–1175 (2011).
Munafo, M. R. et al. Association between genetic variants on chromosome 15q25 locus and objective measures of tobacco exposure. J Natl Cancer Inst 104, 740–748 (2012).
Fu, X. W., Lindstrom, J. & Spindel, E. R. Nicotine activates and up-regulates nicotinic acetylcholine receptors in bronchial epithelial cells. Am J Respir Cell Mol Biol 41, 93–99 (2009).
Millar, N. S. & Gotti, C. Diversity of vertebrate nicotinic acetylcholine receptors. Neuropharmacology 56, 237–246 (2009).
Warren, G. W. & Singh, A. K. Nicotine and lung cancer. J Carcinog 12, 1 (2013).
Arredondo, J., Chernyavsky, A. I. & Grando, S. A. The nicotinic receptor antagonists abolish pathobiologic effects of tobacco-derived nitrosamines on BEP2D cells. J Cancer Res Clin Oncol 132, 653–663 (2006).
Li, Z., Bao, S., Xu, X., Bao, Y. & Zhang, Y. Polymorphisms of CHRNA5-CHRNA3-CHRNB4 Gene Cluster and NSCLC Risk in Chinese Population. Transl Oncol 5, 448–452 (2012).
Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ 327, 557–560 (2003).
Demets, D. L. Methods for combining randomized clinical trials: strengths and limitations. Stat Med 6, 341–350 (1987).
Begg, C. B. & Mazumdar, M. Operating characteristics of a rank correlation test for publication bias. Biometrics 50, 1088–1101 (1994).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634 (1997).
Acknowledgements
This work was supported by supported by the National Natural Science Foundation of China (No: 61571152), the National High-tech R&D Program of China (863 Program) [Nos: 2012AA02A601, 2012AA020409, 2012AA02A604, 2014AA021505, 2015AA020101, 2015AA020108] and the National Science and Technology Major Project [No: 2013ZX03005012].
Author information
Authors and Affiliations
Contributions
Q.H.J., Y.D.W. and Z.J.H. conceived and initiated the project. Q.H.J., Z.J.H., T.J.Z. and X.L.W. searched the database and extracted the information from each study. Q.H.J., Z.J.H., R.M. and J.X.W. analyzed the data. Y.B., R.J.W. and R.J.T. prepared the Figures 1–6. Z.J.H. wrote the manuscript. Q.H.J. revised the manuscript. All authors discussed the results and contributed to the final manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Han, Z., Jiang, Q., Zhang, T. et al. Analyzing large-scale samples confirms the association between the rs1051730 polymorphism and lung cancer susceptibility. Sci Rep 5, 15642 (2015). https://doi.org/10.1038/srep15642
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep15642
- Springer Nature Limited