Abstract
Airway mucus overproduction is one of the most common symptoms of asthma that causes severe clinical outcomes in patients. Despite the effectiveness of general asthma therapies, specific treatments that prevent mucus overproduction in asthma patients remain lacking. Recent studies have found that activation of GABAA receptors (GABAAR) is important for promoting mucus oversecretion in lung airway epithelia. Here, we report that luteolin, a natural flavonoid compound, suppresses mucus overproduction by functionally inhibiting the GABAergic system. This hypothesis was investigated by testing the effects of luteolin on goblet cell hyperplasia, excessive mucus secretion, and GABAergic transmission using histological and electrophysiological approaches. Our results showed that 10 mg/kg luteolin significantly decreased the number of goblet cells in the lung tissue and inhibited mucus overproduction in an in vivo asthma model induced by ovalbumin (OVA) in mice. Patch-clamp recordings showed that luteolin inhibited GABAAR-mediated currents in A549 cells. Furthermore, the inhibitory effects of luteolin on OVA-induced goblet cell hyperplasia and mucus overproduction were occluded by the GABAAR antagonist picrotoxin. In conclusion, our observations indicate that luteolin effectively attenuates mucus overproduction at least partially by inhibiting GABAARs, suggesting the potential for therapeutic administration of luteolin in the treatment of mucus overproduction in asthma patients.
Similar content being viewed by others
Introduction
Asthma is a major public health issue that affects as many as 300 million people worldwide (of all ages), with a global prevalence ranging from 1% to 21% in adults; up to 20% of children aged 6 to 7 years old experience severe wheezing episodes within a year1,2,3. Although current therapies have effectively decreased the rate of asthma hospitalizations and asthma-related deaths, a significant number of asthma patients do not respond well to existing therapies4,5,6.
Mucus overproduction is one of the most important pathological features of chronic inflammatory airway diseases that contribute to the morbidity and mortality associated with asthma, chronic obstructive pulmonary disease, and cystic fibrosis7,8,9. Mucus overproduction is characterized by enhanced mucin gene expression and mucin synthesis, excessive mucus secretion, goblet cell hyperplasia, and phlegm production8,10,11. Under normal conditions, mucus plays a protective role in the airway by preventing the entrance of invading pathogenic microorganisms, chemicals, and particles. However, excessive mucus secretion in the asthmatic airway reduces epithelial ciliary motility, damages local defensive function, promotes bacterial colonization, and increases recurrent infection12. More severely, airway obstruction because of mucus overproduction is one of the major causes of mortality in asthma patients. Mucin 5ac (Muc5ac) protein, encoded by the Muc5ac gene is the major type of mucin, which is the main biochemical component of mucus expressed in the airway epithelium in respiratory diseases11,13,14. Recent studies showed that chronic mucus hypersecretion increases the risk of lung cancers due to decreased carcinogen clearance15,16,17. Therefore, inhibition of mucus overproduction and mucus obstruction is a crucial therapeutic target for reducing serious airway complications.
The GABAergic system is the major inhibitory neurotransmitter system in the central nervous system (CNS), consisting of the neurotransmitter gamma-aminobutyric acid (GABA) and GABA receptors. A type GABA receptors (GABAARs), a class of pentameric ion channels highly permeable to chloride anions (Cl−), are the major form of ionotropic GABA receptors mediating inhibitory transmission in the brain and spinal cord18. Interestingly, recent studies showed that the GABAergic system, particularly GABAARs distributed in airway epithelial cells, is highly associated with mucus overproduction in respiratory diseases19,20. Allergic responses can cause the increased expression of both GABAARs and enzymes for GABA synthesis in airway epithelial cells. Activation of GABAARs causes depolarization of epithelial cells followed by Cl− efflux from the cell. The transfer of Cl− is proposed to create an osmotic gradient across the epithelium and lead to secretion of fluid into the airway lumen21. Furthermore, enhanced GABAergic signaling also results in epithelial proliferation and goblet cell hyperplasia20. Therefore, inhibition of GABAAR activation is considered a new strategy to relieve symptoms of mucus overproduction and attenuate the progression of respiratory diseases.
Flavonoids are a class of natural compounds containing two phenyl rings and a heterocyclic ring. Various flavonoids have been identified as positive, negative, or biphasic modulators of GABAARs22. However, it is difficult to infer from previous studies which compound directly targets airway GABAARs because GABAARs are assembled from a range of subunit isoforms and different flavonoids may have different or even opposite pharmacological behaviors depending on the subunit isoforms used. In the present study, we found that luteolin (3′,4′,5,7-tetrahydroxyflavone), a natural flavonoid found in Perilla frutescens and many other medical herbs23,24,25, directly inhibits GABAAR-mediated current responses in lung epithelial cells (A549). Furthermore, luteolin reduced goblet cell hyperplasia, mucin gene expression, and mucus accumulation in the airway in an in vivo model of mucus overproduction induced by ovalbumin (OVA), likely by inhibiting GABAAR activation. These data suggest the therapeutic potential of luteolin for treating mucus hypersecretion and related airway complications.
Results
Generation and characterization of the mucus overproduction model
Mucus overproduction was studied in a murine allergic asthma model induced by OVA sensitization and challenge26 (Fig. 1A). To demonstrate that asthma responses were successfully induced, we examined serum IgE levels as an indication of asthma severity in all experimental animals after OVA challenges27,28,29,30. Our data showed that mice receiving OVA (n = 112) treatments exhibited significantly higher IgE levels than the sham group (n = 40, P < 0.001; Fig. 1B). Histological assessments also showed that lung sections from OVA-treated mice displayed epithelial alteration, epithelial shedding, mucus hypersecretion, ciliated cell loss, and goblet cell hyperplasia in the airway (Fig. 1C). These changes are consistent with clinical observations of mucus hypersecretion in asthma patients8. Together, these results revealed hallmarks of mucus overproduction in the OVA-induced asthma model.
Experimental flow and verification of the OVA-induced mucus overproduction model.
(A) Schematic flow chart of the experimental design. (B) Validation test of serum IgE assay using blood samples collected on day 24 in the sham (n = 40) and OVA groups (n = 112). The bars represent means ± S.E.M. Significance differences were determined by two-tailed, unpaired Student’s t-test, ***indicates P < 0.01. (C) Morphological changes of the lung from sham and OVA-treated mice. The sections were stained with hematoxylin and eosin (H&E) and analyzed with a microscope at a magnification of ×63. ASM, airway smooth muscle cells hypertrophy and hyperplasia; AW, airway; GCH, goblet cell hyperplasia; M, excess mucus secretion.
Luteolin reduced OVA-induced airway goblet cell hyperplasia and mucus occlusion
We next tested whether luteolin can inhibit OVA-induced mucus overproduction. The periodic acid-Schiff (PAS) stain of airway sections showed that airway goblet cell hyperplasia and mucus overproduction were observed in OVA-treated mice but not in the sham group (Fig. 2A). In the OVA group, goblet cells have increased to 33.3 ± 5.3% of the total airway cells compared to the sham group (0.2 ± 0.13%, P < 0.001). Treatment with 1 mg/kg or 10 mg/kg luteolin significantly decreased goblet cell hyperplasia to 14.7 ± 2.7% and 10.3 ± 1.6%, respectively (P < 0.05 and P < 0.01 compared with the OVA group). The mean score of mucus occlusion of the airway diameter was 1.96 ± 0.15 in the OVA group, which was significantly higher than 0.07 ± 0.03 in the sham group (P < 0.001). With 1 mg/kg or 10 mg/kg luteolin treatment, the airway mucus occlusion scores significantly decreased to 1.16 ± 0.15 and 0.9 ± 0.14 (P < 0.05 and P < 0.01 compared to the OVA group), respectively.
Effects of luteolin on OVA-induced goblet cell hyperplasia, mucus occlusion and Muc5ac mucin expression.
(A) Representative lung tissue sections stained with PAS stain (×40 magnification) from the sham group or OVA-treated mice receiving vehicle or luteolin (0.1, 1, 10 mg/kg, i.p.). The number of goblet cells and mucus occlusion were significantly reduced in mice treated with 1 or 10 mg/kg (n = 9 and 8) but not with 0.1 mg/kg luteolin (n = 9), compared to mice receiving vehicles (n = 8). (B) Representative and quantitative data of Muc5ac gene expression assessed by RT-PCR (n = 7 for each group). (C) Representative and quantitative data of Muc5ac protein expression in each group by Western blot assay (n = 5 for each group). Full-length blots are presented in Supplementary Fig. 1A–D. The bars represent means ± S.E.M. Significant differences were determined by Kruskal-Wallis one-way ANOVA. ##P < 0.01 and ###P < 0.001 indicate significance differences compared to the sham group. *P < 0.05 and **P < 0.01 indicate significance differences compared to the OVA + vehicle group. Lut, luteolin.
The expression of Muc5ac mucin RNA and protein is a marker of goblet cell metaplasia in mouse airways14. Furthermore, Muc5ac is the main type of mucin in respiratory diseases with mucus hypersecretion, particularly in the asthmatic state13,31,32. The expression levels of Muc5ac mRNA increased significantly in the OVA group compared to the sham group (P < 0.05) in mouse lung tissue using reverse transcription polymerase chain reaction (RT-PCR) (Fig. 2B). When detected by Western blotting, Muc5ac protein produced bands with a high molecular mass of 400 to 600 kDa that mainly stayed near the gel entrance (Supplementary Fig. 1)33,34. To better separate bands of Muc5ac and the loading control alpha-tubulin, equal amount of samples were loaded onto 6% and 8% gels in parallel for the detection of Muc5ac and alpha-tubulin, respectively. The expression level of Muc5ac exhibited a significant increase, consistent with its mRNA levels in OVA-treated animals (Fig. 2C). We next examined the effect of luteolin on OVA-induced Muc5ac upregulation. Our results showed that treatment with luteolin (10 mg/kg) significantly attenuated the production of Muc5ac mRNA and protein (P < 0.05, Fig. 2B,C). Together, these results indicate a protective effect of luteolin on mucus overproduction in an in vivo asthma model.
Luteolin inhibited airway inflammation and airway hyper-reactivity (AHR) in OVA-treated mice
The OVA sensitization and airway challenges induced significant inflammatory responses, which were demonstrated as significant upregulation of interleukin-4 (IL-4), IL-5, and IL-13 levels in the bronchoalveolar lavage fluid (BALF). Treatment of mice with luteolin (1 or 10 mg/kg, i.p.) produced a decreased pattern of IL-4, IL-5, and IL-13 expression (Fig. 3A–C). AHR was tested in sham and OVA-treated mice receiving vehicle or 10 mg/kg luteolin by measuring airway resistance in response to methacholine on day 30 after the first immunization of OVA. The OVA-treated animals receiving vehicle only showed enhanced airway resistance compared to the sham group. Luteolin treatments significantly suppressed airway resistance in response to 12.5 and 25.0 mg/ml methacholine (Fig. 3D). These results indicate that luteolin has a protective effect against airway inflammation and hyperresponsiveness in OVA-treated animals.
Effects of luteolin on OVA-induced airway inflammation and hyper-reactivity.
(A–C) Expression levels of IL-4 (A), IL-5 (B), and IL-13 (C) in BALF were significantly higher in mice receiving OVA sensitization and challenge (n = 9) compared to mice receiving sham treatment (n = 6). In OVA-treated animals, BALF IL-4, IL-5 and IL-13 levels were dose-dependently inhibited by 0.1 mg/kg (n = 9), 1 mg/kg (n = 8), and 10 mg/kg luteolin (n = 8). (D) Pulmonary resistance in animals of the sham (n = 9), OVA+vehicle (n = 9), and OVA+10 mg/kg luteolin groups (n = 9) showed dose-dependent AHR in response to 3.125, 6.25, 12.5, and 25.0 mg/ml methacholine. The bars represent means ± S.E.M. Significance differences were determined by Kruskal-Wallis one-way ANOVA. #P < 0.05, ##P < 0.01, and ###P < 0.001 indicate significance differences compared to the sham group. *P < 0.05, **P < 0.01, and ***P < 0.001 indicate significance differences compared to the OVA+vehicle group. Lut, luteolin.
Expression of GABAA receptors in mouse lung tissue and A549 cells
To determine whether luteolin reduces mucus overproduction by inhibiting GABAARs, we used an RT-PCR assay to investigate the expression of different GABAAR subunit genes in both mouse lung tissue and A549 cells, a human alveolar basal epithelial cell line. Consistent with previous studies20,35,36, we found that various types of GABAAR subunits are differentially distributed in naive mouse lung tissue and A549 cells (Fig. 4). These data confirm that GABAARs are expressed in the lung and may be a possible molecular target of luteolin.
Expression of GABAAR subunits in mouse lung tissue and human alveolar epithelial A549 cells.
(A) The normalized mRNA expression levels of murine GABAAR α1-5, β1-3, σ, and π subunits in mouse lung tissue. (B) The normalized mRNA expression levels of human GABAAR α1-5, β1-3, σ, and π subunits in A549 cells.
Luteolin reduced GABAA receptor-mediated current responses in A549 cells
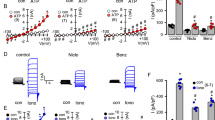
To determine whether luteolin reduces mucus overproduction by direct inhibition of GABAARs in the asthma model20, we tested the modulatory effect of luteolin on GABA-induced current responses in A549 cells using patch-clamp recordings. Our data showed that GABA (0.1–250 μM) evoked whole-cell currents in A549 cells in a dose-dependent manner with an EC50 of 0.69 μM (Fig. 5A, solid circles). At 10 μM, luteolin inhibited GABA-mediated currents, with greater inhibitory effects at higher GABA concentrations (with the highest reduction of 60.3% with 25 μM of GABA, Fig. 5A, open circles). Treatment with different concentrations of luteolin inhibited GABA (2.5 μM)-induced currents in a dose-dependent manner, with an IC50 of 8.2 μM (Fig. 5B). These results suggest that luteolin directly targets GABAARs and inhibits their functions in human alveolar basal epithelial cells.
Modulatory effects of luteolin on GABA-induced currents in A549 cells.
(A) Modulatory effects of luteolin (10 μM) on dose-response curves of GABA-induced currents in A549 cells (n = 12 to 18). (B) The dose-response curve of luteolin (0.1, 1, 5, 10 and 30 μM) blockade on GABA (2.5 μM)-mediated currents in A549 cells (n = 11). (C) Representative traces of GABA-induced currents in A549 cells and the inhibitory effects of luteolin or picrotoxin. (D) Summary of the effect of luteolin and picrotoxin on GABA-induced currents (n = 11). The bars represent means ± S.E.M. Significance differences were determined by Kruskal-Wallis one-way ANOVA. *P < 0.05, **P < 0.01, and NS indicates no significance. Lut, luteolin; Ptxn, picrotoxin.
We next compared the inhibitory effects between luteolin and picrotoxin, a well-established GABAAR antagonist18, on GABAAR activation in the airway. When current responses were induced with 2.5 μM of GABA in A549 cells, 50 μM of luteolin decreased GABAAR currents to 44.0 ± 4.5% of the original responses (Fig. 5C,D). Such an effect was comparable to the inhibitory effect of 5 μM picrotoxin, which reduced GABA currents to 50.1 ± 3.4% in A549 cells under the same conditions (P > 0.05 between luteolin and picrotoxin). These results demonstrate that luteolin has a strong negative modulatory effect on GABAAR activation and further indicate that the effect of luteolin on mucus overproduction may be due to blockade of GABAARs.
Effects of luteolin and picrotoxin on OVA-induced airway goblet cell hyperplasia and mucus occlusion in vivo
To further investigate the pharmacological mechanism of luteolin on mucus overproduction, we blocked GABAAR activation using the selective GABAAR antagonist picrotoxin and then tested whether luteolin continues to have an effect on mucus overproduction. To test this hypothesis, we treated OVA-sensitized and -challenged mice with the following: (1) picrotoxin (0.2 mg/kg), (2) luteolin (10 mg/kg), or (3) combined treatments of picrotoxin (0.2 mg/kg) and luteolin (10 mg/kg) (Fig. 6A). According to histological assessments, goblet cell hyperplasia and mucus occlusion induced by OVA were significantly reduced by luteolin (12.77 ± 3.10%, n = 8) and picrotoxin (10.33 ± 1.62%, n = 9) alone to a similar extent (P > 0.05, Fig. 6B). However, the combined treatment of luteolin and picrotoxin did not yield significantly better effects (P > 0.05 compared to luteolin alone or picrotoxin alone). Furthermore, the expression levels of Muc5ac mRNA and protein were also examined in animals receiving these treatments. Luteolin or picrotoxin alone decreased OVA-induced Muc5ac upregulation to a similar extent, and Muc5ac levels were not further decreased by combined treatment with both drugs (P > 0.05, Fig. 6C,D). Our results clearly show that luteolin did not produce any further effect after GABAARs were blocked by picrotoxin. Similarly, picrotoxin did not further extend the effect of luteolin, indicating that these two drugs target the same pathway to exert their anti-asthmatic effects by blocking GABAAR activation. Taken together with our electrophysiological data, these results strongly indicate that luteolin effectively attenuates mucus overproduction in the asthma model by inhibiting GABAergic signaling pathways.
Luteolin decreases airway mucus expression by blocking GABAAR activation.
(A) Schematic flow chart of the experimental design. (B) Representative PAS-staining (×40 magnification) of lung tissue sections from mice of the sham group (n = 8), OVA-sensitized and challenged mice treated with vehicle (n = 10), 0.2 mg/kg picrotoxin (n = 9), 10 mg/kg luteolin (n = 8), or with 10 mg/kg luteolin plus 0.2 mg/kg picrotoxin (n = 9). (C) Muc5ac gene expression was assayed by RT-PCR (n = 8 for each group). (D) Muc5ac protein expression assessed by Weston blot assay (n = 6 for each group). Full-length blots are presented in Supplementary Figure 1E–H. The bars represent means ± S.E.M. Significance differences were determined by Kruskal-Wallis one-way ANOVA. ###P < 0.001 indicates a significance difference compared to the sham group. *P < 0.05 and **P < 0.01 indicate significance differences compared to the OVA + vehicle group. NS indicates no significance. Lut, luteolin; Ptxn, picrotoxin.
Discussion
Luteolin has been predicted to exert its anti-asthmatic effects by acting as an anti-inflammatory drug25,37,38,39. However, we found that luteolin strongly inhibited GABAAR-mediated responses in lung epithelia cells. Moreover, the GABAAR antagonist picrotoxin occluded the effect of luteolin on goblet cell hyperplasia and mucus overproduction, suggesting a GABAAR-dependent mechanism. Our findings indicate that some flavonoid chemicals may exert their protective effect on mucus overproduction not only via an anti-inflammatory effect but also through inhibition of GABAergic signaling.
Distribution of GABA signaling in the airway
Many studies showed that GABA synthetic enzymes and GABAARs are expressed in the respiratory system and alveolar cell lines. The glutamic acid decarboxylase (GAD65/67) is expressed in mouse pulmonary neuroendocrine cells40, mouse alveolar epithelial type II cells41, monkey bronchial epithelial cells42, A549 cells20, and human airway epithilum43,44. Previous studies and our present findings both demonstrated the expression of mRNA or proteins of various GABAAR subunits, including α1-3, α5, β2, β3, γ1, γ3, ε, π, and σ, in the human lung epithelia cell line (A549), human lung epithelia, and mouse lung tissue20. We found that α3, α4, and β1 subunits are highly expressed in mouse lung tissue, whereas α3, α5, β2, β3, and π are the most abundant subunits in A549 cells20,36. Normally, the αβ composition is required to form a functional GABAAR, whereas αβγ2 is the major form of brain GABAAR responsible for inhibitory synaptic transmission. Interestingly, a major brain GABAAR subunit, γ2, is not likely to be present in the respiratory system35, which indicates that differences in the composition of functional GABAARs exist between those of the airway and the CNS. Furthermore, our electrophysiological results found that GABA induced large current responses in A549 cells with an EC50 of 0.69 μM, which is within the same range reported by other groups (EC50 of 2.45 μM)36. Because subunits, including α1-3, 5, β2, β3, γ1, 3, ε, and π are found in human airway epithelium42, luteolin also targets GABAARs in the human airway.
Mucus production and the GABAergic signaling
Recent studies indicated that the GABAergic signaling system is tightly associated with mucus accumulation in the airway in an animal asthma model. Nicotine treatment induced GAD and GABAAR expression and contributes to nicotine-induced overproduction of mucin in vitro and in vivo42. In the mouse second-hand cigarette smoke model, mucous cell metaplasia induced by cigarette smoke/nicotine requires GABAAR upregulation45. In the human airway epithelium of cigarette smokers, expression of the GABAergic system has been detected, and GAD67 and Muc5ac overproduction are associated with smoking44. The enhanced autocrine GABA signaling results in chloride efflux, depolarization, cell proliferation, and mucus overproduction of airway epithelia cells20,36. Together with these previous studies20, our study demonstrates that the GABAAR inhibitor picrotoxin prevents allergen-induced goblet cell transformation and mucus overproduction. These results indicate that the GABAergic system is a potential therapeutic target for reducing mucus overproduction.
Pharmacological effects of luteolin
As a natural flavonoid, luteolin has multiple biological activities, such as anti-inflammatory, antioxidant, anti-carcinogenic, and anti-allergic effects25,38. However, studies that used luteolin as a GABAAR modulator to treat asthmatic responses remain lacking. Some types of flavonoids have positive, negative, or neutralizing modulatory effects on GABAARs22. Here, we found that luteolin potentiated very low dose GABA (0.1 μM)-induced currents in A549 cells (Fig. 3). However, when current responses were induced by higher doses (2.5 μM GABA) in A549 cells, luteolin showed an inhibitory effect in a dose-dependent manner. The inhibitory effect of 50 μM luteolin on GABA currents was comparable to that of 5 μM picrotoxin, a well-established GABAAR antagonist. Furthermore, the effect of luteolin on mucus overproduction and goblet cell hyperplasia was not further extended by picrotoxin, suggesting that luteolin exhibits its effect through inhibition of GABAAR activation, thereby occluding the effect of picrotoxin.
Our results are consistent with previous reports on the protective effects of luteolin against mucus secretion in vitro and asthmatic responses in vivo. For example, luteolin reduces Muc5ac expression stimulated by epidermal growth factor or phorbol 12-myristate 13-acetate in NCI-H292 cells37. Luteolin inhibits bleomycin-induced lung fibrosis46 and alleviates OVA-induced bronchoconstriction and airway hyperreactivity39. Consistent with previous studies, we also found that luteolin suppressed OVA-induced IL upregulation and AHR, suggesting that luteolin may be used as an anti-asthmatic agent. Our results confirm that luteolin has strong inhibitory effects on mucus overproduction in the in vivo asthma model induced by OVA and advances current understanding of the effect of luteolin on the GABAAR signaling pathway.
Safety of luteolin as a GABAAR modulator
GABAAR antagonists are generally considered a group of convulsants. Systematic administration of GABAAR antagonists that cross the blood-brain barrier typically causes seizure activities and other side effects in the CNS. However, luteolin is generally considered safe47,48 because it is contained in many edible plants, such as broccoli, bird chili, and onion leaves49. We showed that luteolin acts as an allosteric modulator of GABAARs. After systemic administration of luteolin, we did not observe convulsive or other abnormal behaviors in animals during our study. This may be due to the limited permeability of luteolin through the blood brain barrier or its different modulatory effects on brain GABAARs. These possibilities must be tested.
Conclusion
Our observations indicate that luteolin effectively attenuates mucus overproduction and goblet cell hyperplasia in an animal asthma model at least partially by inhibiting GABAAR activities. These data suggest the potential for therapeutic administration of luteolin in the treatment of mucus overproduction in asthma patients.
Methods
Chemicals and Reagents
Luteolin, picrotoxin, ovalbumin and the PAS staining kit and other chemical reagents were purchased from Sigma (Sigma-Aldrich, St. Louis, MO, USA). Anti-Muc5ac and anti-alpha-tubulin antibodies were purchased from Santa Cruz Biotechnology (sc-21701, sc-12462-R, Dallas, TX, USA). Horseradish peroxidase (HRP)-conjugated secondary antibodies were purchased from Pierce Biotechnology (Thermo Fisher Scientific, Rockford, IL, USA). IL-4, IL-5 and IgE ELISA kits were purchased from BD Biosciences (San Diego, CA, USA). The IL-13 kit was purchased from R&D Systems (Minneapolis, MN, USA). OVA for intraperitoneal (i.p.) injection was adsorbed to aluminum hydroxide adjuvant (Santa Cruz, Dallas, TX, USA) at a ratio of 50 μg to 2 mg in 200 μl PBS. OVA for intratracheal (i.t.) injection was dissolved in saline at a final concentration of 2.5 mg/ml. Luteolin (Sigma-Aldrich, St. Louis, MO, USA) was prepared in DMSO and diluted with saline. Picrotoxin (Sigma-Aldrich, St. Louis, MO, USA) was prepared in DMSO and diluted with saline. Saline with 0.5% DMSO was used as a vehicle in control groups. The final concentration of DMSO in all reaction mixtures was less than 0.5%.
Animals
Adult male BALB/c mice, weighing 18–22 g, were obtained commercially from BioLasco co., Ltd. (Taipei, Taiwan). Animals were housed under controlled laboratory conditions with a 12-h dark-light cycle. The experiments that involved experimental animals were carried out in accordance to the Institutional Guidelines of the China Medical University for the Care and Use of Experimental Animals (IGCMU-CUEA) and were approved by the Institutional Animal Care and Use Committee (IACUC) of China Medical University (Taichung, Taiwan).
Murine model of asthma
The asthma model and validation were established according to a previously described protocol20,30,50. As shown in Fig. 1, mice were administered an i.p. injection of 50 μg of ovalbumin (OVA, adsorbed in 2 mg aluminum hydroxide in 200 μl PBS) on days 0, 7, and 14, followed by challenge of i.t. instillation of OVA (100 μg in 40 μl of saline) on days 21, 22, and 23. Mice of the OVA group were randomly allocated to six groups, receiving treatments of 0.1 mg/kg/day luteolin, 1 mg/kg/day luteolin, 10 mg/kg/day luteolin, 0.2 mg/kg/day picrotoxin, combined treatment of luteolin plus picrotoxin (10 and 0.2 mg/kg/day, respectively), or vehicle by i.p. injection once daily for 6 days. The mice were sacrificed on the next day after the last drug administration, and airway tissues were collected for further analysis.
Estimation of serum IgE using ELISA assay
Blood samples were drawn from the orbital sinus and collected on day 24 in the sham (n = 40) and the OVA group (n = 112). The serum IgE level was measured using an enzyme-linked immunosorbent assay antibody and ELISAs were performed according to the manufacturer’s instructions.
BALF Collection and Cytokine ELISA assay
Mice were anesthetized, and the trachea was cannulated while the thorax was gently massaged. Lungs were lavaged twice with 1 ml of saline. The covered lavage samples were cooled on ice and centrifuged at 1200 rpm for 10 min at 4 °C. The supernatant was collected and stored at −80 °C for ELISA assays. IL-4, IL-5 and IL-13 levels in BALF were measured using specific mouse IL-4, IL-5 and IL-13 ELISA kits and ELISAs were performed according to the manufacturer’s instructions.
Histopathological analysis
The left lung tissue was collected and fixed with 4% paraformaldehyde (wt/vol) in PBS at 4 °C for 24 h. The tissues were embedded in paraffin and cut into 2–5 μm sections. The slices were dewaxed by immersion in xylene, rehydrated in descending concentrations of an ethanol series (100% to 70%), and stained with hematoxylin and eosin (H&E). All slides were observed for pathological changes under light microscopy. Goblet cell hyperplasia and mucus occlusion were assayed with a PAS staining kit as previously described30,51. Slides were visualized using an upright light microscope (Zeiss, Oberkochen, Germany) with bright field illumination. Images with 40x or 63x magnification were acquired with Zeiss Plan-Apochromat 40x/0.95 and 63x/1.4 objectives, respectively. Images were captured with an AxioCam HRm camera and processed using the Axiovision LE Rel4.4 software (Zeiss, Oberkochen, Germany). The number of goblet cells was determined as the percentage of total airway epithelial cells in each airway examined. The cross-sectional area of the airway lumen and mucus plugging was outlined by the “freehand selections” function of ImageJ and was automatically calculated by the same software. The grade of mucus plugging of the airways was assessed as percent occlusion of airway lumen on the following scale: 0, no mucus; 1, <10% occlusion; 2, 30% occlusion; 3, <50% occlusion; and 4, greater than approximately 80% occlusion.
RNA isolation and reverse transcription polymerase chain reaction (RT-PCR)
Total RNA was isolated from murine lung tissue or cells using TRIzol reagent (Invitrogen Life Technologies, Carlsbad, CA, USA), according to the manufacturer’s instructions. Briefly, the RNA precipitate was washed twice by gentle vortexing with 70% ethanol, collected by centrifugation at 12000 rpm, dried under a vacuum for 5–10 min, dissolved in 200 μl RNase-free water (Promega, Madison, WI, USA), and incubated for 10–15 min at 55–60 °C. The RNA was quantified and assessed for purity by spectrophotometry at a wavelength of 260 nm. The integrity was assessed by 2% agarose gel electrophoresis and the RNA was visualized by ethidium bromide staining. The GABAAR subtype and Muc5ac mRNA transcripts were measured by RT-PCR as previously described30,52. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as the endogenous control gene. The PCR primers for mice and humans were designed according to the published cDNA sequences and synthesized by MDBio Inc (Taipei, Taiwan). The primers sequences are listed in Table 1. Mouse or human GAPDH mRNA was used as the internal control in all experiments. The mRNA expression levels of Muc5ac and GABAAR subunits were measured by band intensities using the Gel Analysis method in ImageJ 1.49 and were normalized to the level of GAPDH.
Protein extraction and Western blotting
The lung tissues were harvested and homogenized in RIPA lysis buffer containing proteinase inhibitors. After vortexing and a 10 min incubation on ice, the extract was centrifuged at 12000 rpm for 40 min at 4 °C. The supernatant protein extracts were collected and stored at −80 °C for future use. The protein concentration in the tissue extract was determined using a Bio-Rad protein assay kit.
For the detection of Muc5ac and alpha-tubulin, equal amounts of total protein sample (100 μg) were separated on 6% and 8% sodium dodecyl sulfate polyacrylamide gels, respectively, and transferred onto polyvinylidene difluoride (PVDF) membranes (Millipore, Bedford, MA, USA). To block nonspecific binding, the membranes were incubated for 1 h in blocking buffer (5% fat-free milk, 0.1 M Tris-HCl, 0.9% NaCl, 1% Tween-20, pH 7.4) at room temperature, and then incubated with anti-Muc5ac antibodies (1:1000) and alpha-tubulin antibodies (1:2000) overnight at 4 °C. Subsequently, membranes were incubated with an HRP-linked secondary antibody. The expression of proteins was detected using enhanced chemiluminescent (ECL) reagents (Millipore, Billerica, USA), and imaged on an ImageQuant LAS 4000 system (GE Healthcare, Buckinghamshire, UK). The protein levels of Muc5ac or alpha-tubulin were analyzed using Image J 1.49 software.
Invasive Measurements of Airway AHR
Airway resistance was assessed as an increase in pulmonary resistance in response to increasing concentrations of aerosolized methacholine in anesthetized mice. Briefly, mice were anesthetized with 70–90 mg/kg pentobarbital sodium. The trachea was exposed and cannulated with an 18-gauge tracheal tube. A suture around the trachea was then tied to prevent air leak. Mice were mechanically ventilated using a computer-controlled small animal ventilator (flexiVent; Scireq, Montreal, Canada) with the following parameters: respiratory rate of 150 breaths/min, tidal volume of 10 ml/kg, inhalation: exhaustion ratio of 2:3, and positive end-expiratory pressure of 2–3 cm H2O. Aerosolized PBS and increasing concentrations of methacholine (3.125, 6.25, 12.5, and 25.0 mg/ml) were delivered to the animal and readings were recorded every 4 min for 12 sec at each concentration. Pulmonary resistance was calculated using the flexiWare software.
Cell culture
The human alveolar basal epithelial cell line A549 was purchased from the American Type Culture Collection (A549, ATCC, Manassas, VA, USA). Cells were used at passage number 20–30 and maintained in Ham’s F12K medium with 10% fetal calf serum, 100 units/ml penicillin, and 100 mg/ml streptomycin at 37 °C in a 5% CO2 incubator with humidified atmosphere. All experiments were performed when cells were 80–90% confluent. For electrophysiological recording experiments, cells were seeded on poly-D-lysine-coated 12 mm glass coverslips in 24-well plates at a density of 5*104 cells per well.
Electrophysiology
The electrophysiology study was performed on A549 cells by examining GABAAR- mediated currents with whole-cell patch-clamp recordings53. Briefly, after removing the culture medium, cells were immersed in a solution containing (in mmol/l) 140 NaCl, 5.4 KCl, 10 HEPES, 1.0 MgCl2, 1.3 CaCl2, and 20 glucose (pH 7.4, 305–315 mOsm). Whole-cell recordings were performed under voltage-clamp mode using an Axopatch 200B (Molecular Devices, Sunnyvale, CA, USA). Whole-cell currents were recorded at a holding potential of −60 mV, and signals were low-pass filtered at 2 kHz and digitized at 10 kHz. GABA-mediated currents were evoked by a computer-controlled multibarrel fast perfusion system (Warner Instruments, Hamden, CT, USA). Maximum currents (Imax) were evoked by the highest concentration of GABA as determined by the dose-response curves. Modulatory effects of luteolin or picrotoxin on GABAARs were tested on 2.5 μM GABA-induced currents on the same cell with and without 50 μM luteolin or 5 μM picrotoxin in the extracellular solution.
Statistical analysis
All results are presented as the mean ± standard error of mean (S.E.M.). Statistical analysis was performed using the Prism statistical analysis program (GraphPad 6.0). Kruskal-Wallis one-way ANOVA test was used for statistical comparisons of 3 or more groups. Two-tailed unpaired Student’s t-test was used for statistical comparisons of two groups. P < 0.05 was considered significant for all tests. Statistical significance is presented as *P < 0.05; **P < 0.01; and ***P < 0.001.
Additional Information
How to cite this article: Shen, M.-L. et al. Luteolin Attenuates Airway Mucus Overproduction via Inhibition of the GABAergic System. Sci. Rep. 6, 32756; doi: 10.1038/srep32756 (2016).
References
Lai, C. K. et al. Global variation in the prevalence and severity of asthma symptoms: phase three of the International Study of Asthma and Allergies in Childhood (ISAAC). Thorax 64, 476–483, doi: 10.1136/thx.2008.106609 (2009).
Reddel, H. K. et al. A summary of the new GINA strategy: a roadmap to asthma control. The European respiratory journal 46, 622–639, doi: 10.1183/13993003.00853-2015 (2015).
To, T. et al. Global asthma prevalence in adults: findings from the cross-sectional world health survey. BMC public health 12, 204, doi: 10.1186/1471-2458-12-204 (2012).
Expert Panel Report 3 (EPR-3): Guidelines for the Diagnosis and Management of Asthma-Summary Report 2007. The Journal of allergy and clinical immunology 120, S94–138, doi: 10.1016/j.jaci.2007.09.043 (2007).
Lozano, R. et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380, 2095–2128, doi: 10.1016/S0140-6736(12)61728-0 (2012).
Vos, T. et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380, 2163–2196, doi: 10.1016/S0140-6736(12)61729-2 (2012).
Kaliner, M. et al. Human respiratory mucus. The American review of respiratory disease 134, 612–621, doi: 10.1164/arrd.1986.134.3.612 (1986).
Aikawa, T., Shimura, S., Sasaki, H., Ebina, M. & Takishima, T. Marked goblet cell hyperplasia with mucus accumulation in the airways of patients who died of severe acute asthma attack. Chest 101, 916–921 (1992).
Williams, O. W., Sharafkhaneh, A., Kim, V., Dickey, B. F. & Evans, C. M. Airway mucus: From production to secretion. American journal of respiratory cell and molecular biology 34, 527–536, doi: 10.1165/rcmb.2005-0436SF (2006).
Lai, H. & Rogers, D. F. New pharmacotherapy for airway mucus hypersecretion in asthma and COPD: targeting intracellular signaling pathways. Journal of aerosol medicine and pulmonary drug delivery 23, 219–231, doi: 10.1089/jamp.2009.0802 (2010).
Rose, M. C., Nickola, T. J. & Voynow, J. A. Airway mucus obstruction: mucin glycoproteins, MUC gene regulation and goblet cell hyperplasia. American journal of respiratory cell and molecular biology 25, 533–537, doi: 10.1165/ajrcmb.25.5.f218 (2001).
Fahy, J. V. & Dickey, B. F. Airway mucus function and dysfunction. The New England journal of medicine 363, 2233–2247, doi: 10.1056/NEJMra0910061 (2010).
Ordonez, C. L. et al. Mild and moderate asthma is associated with airway goblet cell hyperplasia and abnormalities in mucin gene expression. American journal of respiratory and critical care medicine 163, 517–523, doi: 10.1164/ajrccm.163.2.2004039 (2001).
Zuhdi Alimam, M. et al. Muc-5/5ac mucin messenger RNA and protein expression is a marker of goblet cell metaplasia in murine airways. American journal of respiratory cell and molecular biology 22, 253–260, doi: 10.1165/ajrcmb.22.3.3768 (2000).
Brenner, D. R., McLaughlin, J. R. & Hung, R. J. Previous lung diseases and lung cancer risk: a systematic review and meta-analysis. PloS one 6, e17479, doi: 10.1371/journal.pone.0017479 (2011).
Bruse, S. et al. Increased methylation of lung cancer-associated genes in sputum DNA of former smokers with chronic mucous hypersecretion. Respiratory research 15, 2, doi: 10.1186/1465-9921-15-2 (2014).
Wang, H. et al. Association between chronic obstructive pulmonary disease and lung cancer: a case-control study in Southern Chinese and a meta-analysis. PloS one 7, e46144, doi: 10.1371/journal.pone.0046144 (2012).
Macdonald, R. L. & Olsen, R. W. GABAA receptor channels. Annual review of neuroscience 17, 569–602, doi: 10.1146/annurev.ne.17.030194.003033 (1994).
Lu, W. Y. The potential use of GABAergic drugs in the treatment of asthma. Future medicinal chemistry 3, 145–147, doi: 10.4155/fmc.10.297 (2011).
Xiang, Y. Y. et al. A GABAergic system in airway epithelium is essential for mucus overproduction in asthma. Nature medicine 13, 862–867, doi: 10.1038/nm1604 (2007).
Widdicombe, J. H. Regulation of the depth and composition of airway surface liquid. Journal of anatomy 201, 313–318 (2002).
Hanrahan, J. R., Chebib, M. & Johnston, G. A. Interactions of flavonoids with ionotropic GABA receptors. Adv Pharmacol 72, 189–200, doi: 10.1016/bs.apha.2014.10.007 (2015).
Zand, R. S., Jenkins, D. J. & Diamandis, E. P. Steroid hormone activity of flavonoids and related compounds. Breast cancer research and treatment 62, 35–49 (2000).
Resende, F. A., de Oliveira, A. P., de Camargo, M. S., Vilegas, W. & Varanda, E. A. Evaluation of estrogenic potential of flavonoids using a recombinant yeast strain and MCF7/BUS cell proliferation assay. Plos one 8, e74881, doi: 10.1371/journal.pone.0074881 (2013).
Lopez-Lazaro, M. Distribution and biological activities of the flavonoid luteolin. Mini reviews in medicinal chemistry 9, 31–59 (2009).
Singer, M. et al. A MARCKS-related peptide blocks mucus hypersecretion in a mouse model of asthma. Nature medicine 10, 193–196, doi: 10.1038/nm983 (2004).
Baldacci, S., Omenaas, E. & Oryszczyn, M. P. Allergy markers in respiratory epidemiology. The European respiratory journal 17, 773–790 (2001).
Gould, H. J. & Sutton, B. J. IgE in allergy and asthma today. Nature reviews. Immunology 8, 205–217, doi: 10.1038/nri2273 (2008).
Satwani, H., Rehman, A., Ashraf, S. & Hassan, A. Is serum total IgE levels a good predictor of allergies in children? JPMA. The Journal of the Pakistan Medical Association 59, 698–702 (2009).
Lin, C. H. et al. Protective effects of the polyphenol sesamin on allergen-induced T(H)2 responses and airway inflammation in mice. PloS one 9, e96091, doi: 10.1371/journal.pone.0096091 (2014).
Hovenberg, H. W., Davies, J. R., Herrmann, A., Linden, C. J. & Carlstedt, I. MUC5AC, but not MUC2, is a prominent mucin in respiratory secretions. Glycoconjugate journal 13, 839–847 (1996).
Thornton, D. J. et al. Respiratory mucins: identification of core proteins and glycoforms. The Biochemical journal 316 (Pt 3), 967–975 (1996).
Fujisawa, T. et al. Regulation of airway MUC5AC expression by IL-1beta and IL-17A; the NF-kappaB paradigm. J Immunol 183, 6236–6243, doi: 10.4049/jimmunol.0900614 (2009).
Hao, Y., Kuang, Z., Xu, Y., Walling, B. E. & Lau, G. W. Pyocyanin-induced mucin production is associated with redox modification of FOXA2. Respiratory research 14, 82, doi: 10.1186/1465-9921-14-82 (2013).
Tyagi, N. et al. Differential expression of gamma-aminobutyric acid receptor A (GABA(A)) and effects of homocysteine. Clinical chemistry and laboratory medicine 45, 1777–1784, doi: 10.1515/CCLM.2007.342 (2007).
Xiang, Y. Y. et al. Isoflurane regulates atypical type-A gamma-aminobutyric acid receptors in alveolar type II epithelial cells. Anesthesiology 118, 1065–1075, doi: 10.1097/ALN.0b013e31828e180e (2013).
Lee, H. J. et al. Luteolin inhibited the gene expression, production and secretion of MUC5AC mucin via regulation of nuclear factor kappa B signaling pathway in human airway epithelial cells. Pulmonary pharmacology & therapeutics 31, 117–122, doi: 10.1016/j.pupt.2014.09.008 (2015).
Seelinger, G., Merfort, I. & Schempp, C. M. Anti-oxidant, anti-inflammatory and anti-allergic activities of luteolin. Planta medica 74, 1667–1677, doi: 10.1055/s-0028-1088314 (2008).
Das, M., Ram, A. & Ghosh, B. Luteolin alleviates bronchoconstriction and airway hyperreactivity in ovalbumin sensitized mice. Inflammation research 52, 101–106 (2003).
Yabumoto, Y. et al. Expression of GABAergic system in pulmonary neuroendocrine cells and airway epithelial cells in GAD67-GFP knock-in mice. Medical molecular morphology 41, 20–27, doi: 10.1007/s00795-007-0391-6 (2008).
Jin, N., Kolliputi, N., Gou, D., Weng, T. & Liu, L. A novel function of ionotropic gamma-aminobutyric acid receptors involving alveolar fluid homeostasis. The Journal of biological chemistry 281, 36012–36020, doi: 10.1074/jbc.M606895200 (2006).
Fu, X. W., Wood, K. & Spindel, E. R. Prenatal nicotine exposure increases GABA signaling and mucin expression in airway epithelium. American journal of respiratory cell and molecular biology 44, 222–229, doi: 10.1165/rcmb.2010-0109OC (2011).
Mizuta, K., Osawa, Y., Mizuta, F., Xu, D. & Emala, C. W. Functional expression of GABAB receptors in airway epithelium. American journal of respiratory cell and molecular biology 39, 296–304, doi: 10.1165/rcmb.2007-0414OC (2008).
Wang, G. et al. Smoking-mediated up-regulation of GAD67 expression in the human airway epithelium. Respiratory research 11, 150, doi: 10.1186/1465-9921-11-150 (2010).
Gundavarapu, S. et al. Role of nicotinic receptors and acetylcholine in mucous cell metaplasia, hyperplasia, and airway mucus formation in vitro and in vivo. The Journal of allergy and clinical immunology 130, 770–780 e711, doi: 10.1016/j.jaci.2012.04.002 (2012).
Chen, C. Y., Peng, W. H., Wu, L. C., Wu, C. C. & Hsu, S. L. Luteolin ameliorates experimental lung fibrosis both in vivo and in vitro: implications for therapy of lung fibrosis. Journal of agricultural and food chemistry 58, 11653–11661, doi: 10.1021/jf1031668 (2010).
Mittra, B. et al. Luteolin, an abundant dietary component is a potent anti-leishmanial agent that acts by inducing topoisomerase II-mediated kinetoplast DNA cleavage leading to apoptosis. Mol Med 6, 527–541 (2000).
Czeczot, H. et al. Isolation and studies of the mutagenic activity in the Ames test of flavonoids naturally occurring in medical herbs. Mutation research 240, 209–216 (1990).
Miean, K. H. & Mohamed, S. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. Journal of agricultural and food chemistry 49, 3106–3112 (2001).
Huang, F. et al. Calcium-activated chloride channel TMEM16A modulates mucin secretion and airway smooth muscle contraction. Proceedings of the National Academy of Sciences of the United States of America 109, 16354–16359, doi: 10.1073/pnas.1214596109 (2012).
Henderson, W. R. Jr., Banerjee, E. R. & Chi, E. Y. Differential effects of (S)- and (R)-enantiomers of albuterol in a mouse asthma model. The Journal of allergy and clinical immunology 116, 332–340, doi: 10.1016/j.jaci.2005.04.013 (2005).
Zhang, X. et al. Expression of gamma-aminobutyric acid receptors on neoplastic growth and prediction of prognosis in non-small cell lung cancer. Journal of translational medicine 11, 102, doi: 10.1186/1479-5876-11-102 (2013).
Zhou, N., Wang, C. H., Zhang, S. & Wu, D. C. The GLRA1 missense mutation W170S associates lack of Zn2+ potentiation with human hyperekplexia. The Journal of neuroscience 33, 17675–17681, doi: 10.1523/JNEUROSCI.3240-13.2013 (2013).
Acknowledgements
We thank Ms. Rita Y. Chen for editing the manuscript. This work was supported by grants from the Taiwan National Science Council (NSC 102-2320-B-039-038-MY3), the Ministry of Science and Technology (MOST 104-2320-B-039-045-MY3 and MOST 104-2320-B-039-048-MY3), China Medical University Hospital (DMR-104-080), and in part by the Taiwan Ministry of Health and Welfare Clinical Trial and Research Center of Excellence (MOHW105-TDU-B-212-133019). The funders had no role in the study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
M.L.S. designed and performed experiments, analyzed data, and wrote the manuscript. C.H.W. performed the electrophysiology experiments and analyzed data. C.H.L. assisted with experiments. N.Z., S.T.K. and D.C.W. conceived the study, supervised the experiments, and wrote the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Shen, ML., Wang, CH., Lin, CH. et al. Luteolin Attenuates Airway Mucus Overproduction via Inhibition of the GABAergic System. Sci Rep 6, 32756 (2016). https://doi.org/10.1038/srep32756
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep32756
- Springer Nature Limited
This article is cited by
-
Pulmonary inflammation caused by silica dioxide nanoparticles in mice via TXNIP/NLRP3 signaling pathway
Molecular & Cellular Toxicology (2020)