Abstract
Tono-Pen and TonoVet have been used in rabbits to measure intraocular pressure (IOP) and investigate the effect of IOP lowering therapies. Therefore, their reliability and accuracy are very important and deserve careful evaluation. Our results showed that the with-subject deviation (Sw) and intraclass correlation coefficient (ICC) of the TonoVet and Tono-Pen were 0.61 mmHg/0.83 mmHg and 0.97/0.94, respectively for intrasession repeatability. For intersession reproducibility, the Sw and ICC of TonoVet and Tono-Pen were 1.42 mmHg/1.66 mmHg and 0.73/0.67, respectively. For interoperator reproducibility, the Sw and ICC of the TonoVet and Tono-Pen were 0.72 mmHg/1.11 mmHg and 0.91/0.82 respectively. Both TonoVet and Tono-Pen underestimated the IOP measured by manometry. The regression function was: y = 0.8249x + 0.1011 and y =0.6881x + 2.2290 for TonoVet and Tono-Pen, respectively. Our study suggests that both TonoVet and Tono-Pen had excellent intrasession repeatability and inter-operator reproducibility, but good intersession reproducibility. Both TonoVet and Tono-Pen correlated well with manometry, but underestimated the manometric IOP with presence of fixed and proportional biases. These factors should be considered when measuring IOP with Tono-Pen or TonoVet in rabbit eyes.
Similar content being viewed by others
Introduction
Intraocular pressure (IOP) is the fluid pressure of aqueous humor inside the eyeball. It is an essential parameter for diagnosis, follow up and treatment of glaucoma and other ocular diseases. Currently, several different methods have been developed to measure IOP, including indentation1, applanation2, rebound3, ocular response analyzer4 and dynamic contour tonometery5. Generally, these tonometers are designed for human use, or more precisely for use in white populations6,7. However, novel therapies aiming at lowering intraocular pressure must undergo their initial tests in animal models and demonstrate their effectiveness prior to clinical trials. Many investigations were performed on non-primate animal models, among which, rabbits are becoming a popular model, for evaluation of new drugs and surgical procedures for glaucoma, owing to their resemblance to the human eye in size and a better understanding of their eye anatomy and physiology. However, there are distinct differences between human and rabbit eyes in terms of corneal thickness and composition8. Therefore, it is of great essence and importance to evaluate the precision and accuracy of IOP measuring devices to validate whether they are suitable and reliable for estimation of IOP in rabbits.
Currently, the portable and non-invasive tonometers, including TonoVet and Tono-Pen, are widely used. The TonoVet, based on rebound tonometry, is equipped with a magnetic probe that, when electromagnetically propelled against the cornea, produces a resultant rebound from the cornea to induce a voltage change, which is converted an electric signal representing the IOP9. In contrast, the Tono-Pen is based on the principle of applanation tonometry, and has been used for a wide range of species. It measures IOP indirectly by applying the force needed to flatten a certain area of cornea surface, which can be converted into a pressure reading that is equivalent the pressure in the eye10.
Previous articles8,11,12,13,14,15,16 have reported the use of TonoVet or Tono-Pen in rabbits. The accuracy of Tonometer has been measured in terms of its agreement with direct measurement of intracameral pressure12. However, their results are inconsistent. Furthermore, repeatability and reproducibility are important parameters to evaluate the variability of a measurement. To the best of our knowledge, there is no such a report comparing the repeatability and reproducibility of TonoVet and Tono-Pen.
This study was undertaken to assess the intrasession repeatability, intersession and inter-operator reproducibility of the Tono-Pen and TonoVet in measurement of IOP in rabbit eyes. Additionally, the accuracy of the tonometers was determined by comparing the measured IOPs, taken via Tono-Pen and TonoVet, with IOPs obtained manometrically.
Results
Intrasession Repeatability
Table 1 presents the mean IOP values, within-subject standard deviation (Sw), coefficient of variation (CVw), precision and intraclass correlation coefficient (ICC) values for intrasession repeatability when using the TonoVet and Tono-Pen. The Sw, CVw, precision and ICC values for the TonoVet were 0.61 mmHg, 5.50%, 1.19 mmHg and 0.97, respectively. Likewise, the Sw, CVw, precision and ICC values for the Tono-Pen were 0.86 mmHg, 7.72%, 1.63 mmHg and 0.94, respectively. The very high ICC values (ICC = 0.97 for TonoVet while ICC = 0.94 for Tono-Pen) together with quite low Sw and small precision range indicated excellent intrasession repeatability of TonoVet and Tono-Pen for IOP measurement. Furthermore, the IOP on average measured by TonoVet (11.02 ± 1.65 mmHg) was marginally different from that by Tono-Pen (10.78 ± 1.69 mmHg) (p = 0.026, Student’s paired t-test).
Intersession and interoperator reproducibility
Tables 2 and 3 show the mean IOP values, Sw, CVw, precision and ICC values for intersession and interoperator reproducibility, respectively. Both TonoVet and Tono-Pen have higher values of Sw, CVw and precision in intersession reproducibility than intrasession variation. In contrast, regarding interoperator reproducibility, TonoVet and Tono-Pen possess low Sw but small to moderate CVw and precision. The moderate intersessional ICC values (ICC = 0.73 and 0.67 for TonoVet and Tono-Pen, respectively) and high interoperator ICC values (ICC = 0.91 and 0.82 for TonoVet and Tono-Pen, respectively) indicated that there was good intersession reproducibility and excellent interoperator reproducibility of TonoVet and Tono-Pen for IOP measurements. It should be noted that the average IOP obtained by TonoVet (11.06 ± 1.16 mmHg) in intersession reproducibility marginally differed from that by Tono-Pen (10.80 ± 1.19 mmHg) (p = 0.047, Student’s paired t-test) while in interoperator reproducibility the mean IOP read by TonoVet (11.06 ± 1.62 mmHg) was not significantly different by Tono-Pen (10.80 ± 1.69 mmHg) (p = 0.099, Student’s paired t-test).
Diurnal change of IOP
The profile of the mean rabbit IOP over a 24-h period measured by TonoVet and Tono-Pen is shown in Fig. 1. The fluctuation profile of the IOP obtained with TonoVet and Tono-Pen showed a similar pattern; the IOP gradually decreased from 6 AM to 9 AM, and then appeared to gradually increase from 9 AM to 12 PM. The lowest IOP was recorded at 9 AM (10.63 and 10.13 mmHg for TonoVet and Tono-Pen, respectively) while the highest one was recorded at around 12 PM (14.13 and 13.03 mmHg for TonoVet and Tono-Pen, respectively). Generally, the IOP in the daytime was lower than that in the nighttime.
Agreement with manometry
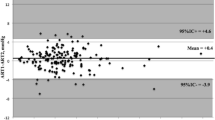
There was high correlation between real IOP and IOP measured with either the TonoVet (r2 = 0.9949, p < 0.0001) or Tono-Pen (r2 = 0.9927, p < 0.0001). However, significant difference was observed between the TonoVet IOP value (p = 0.000, Student’s paired t-test) and the Tono-Pen IOP value (p = 0.000, Student’s paired t-test) vs. the manometric IOP value at all levels except at 5 mmHg for Tono-Pen. Both the TonoVet and the Tono-Pen underestimated the real IOP and the difference increased with the increase in IOP (Fig. 2A–D). The 95% regression-based limits of agreement are shown in Table 4.
Correlation and Bland-Altman plots of agreement between manometry vs. TonoVet and Tono-Pen.
(A) Correlation between manometry and TonoVet. Solid and dotted lines represent best-fit regression line and the ideal regression line (i.e. y = x), respectively. (B) Correlation between manometry and TonoVet. The solid and dash-dot lines represent the regression line of difference on average and regression-based 95% limit of agreement, respectively. (C) Correlation between manometery Tono-Pen. (D) Correlation between manometry and Tono-Pen. (E) Correlation between Tono-Pen and TonoVet. (F) Agreement between Tono-Pen and TonoVet.
Except at pressure between 5 and 15 mmHg, IOP read by TonoVet was significantly higher than that by Tono-Pen (p ≤ 0.005, independent t-test) (Table 5). The correlation between the TonoVet and Tono-Pen reading (r2 = 0.9906, p < 0.0001) was high (Fig. 2E). The mean difference in IOP measured by TonoVet and Tono-Pen was 2.29 mmHg, being distinguishable from zero (p = 0.000, one-sample t-test), which indicated that there was a fixed bias. Additionally, a Bland-Altman test revealed a proportional bias, since the slope of regression of differences on averages was distinguishable from zero (p < 0.0001). Nevertheless, the TonoVet apparently tended to give a higher IOP relative to Tono-Pen at manometric pressure more than 20 mmHg and tended to give a lower IOP relative to Tono-Pen at manometric pressure no more than 5 mmHg.
Discussion
In this study, we assessed the repeatability and reproducibility of TonoVet and Tono-Pen for IOP measurement in rabbits, and determined their agreement with manometric IOP. We found that IOP measurements with TonoVet and Tono-Pen have excellent intrasession repeatability, as evidenced by a low variability (Sw ≤ 0.83 mmHg) and small precision range within 1.63 mmHg, as well as high ICC values ranging between 0.94 and 0.97. Furthermore, the two devices had excellent interoperator reproducibility as indicated by low variability (Sw ≤ 1.11 mmHg) and high ICC values between 0.82 and 0.91. Both devices displayed good intersession reproducibility, as demonstrated by medium variability (Sw ≤ 1.66 mmHg) with wider precision range beyond 2.78 mmHg and moderate ICC values varying from 0.67 to 0.73. The lower intersession reproducibility may be due to the variation of IOP measured on different days.
Our study is the first one to compare the repeatability, intersession and interoperator reproducibility of TonoVet and Tono-Pen in rabbit. Our results found that TonoVet was superior to Tono-Pen in terms of intrasession repeatability, intersession and interoperator reproducibility as indicated by less variability and higher ICC, suggesting that TonoVet was more precise than Tono-Pen. Nevertheless, our repeatability result of Tono-Pen was better than that reported by Acosta et al. who found that the ICC value for interobserver varied from 0.48 to 0.69 at normal rabbit IOP levels8. The difference might be likely attributed to their small sample size with only 4 rabbit eyes being measured.
The comparison of TonoVet and Tono-Pen against the manometric method for measuring IOP in rabbits has been investigated previously8,11,12,13,14,15,16. Lim et al. reported that Tono-Pen underestimates the actual IOP and is apt to error at high IOP levels, but it has been found to be the most accurate tonometer among the Tono-Pen, Perkins and pneumatonometer11. Acosta et al. found that Tono-Pen underestimated rabbit IOP at all pressure ranges from 5 to 50, and the low prediction was more notable at high pressures8. Zhang et al. reported that TonoVet tended to consistently display lower IOP values than manometer values in all pressure ranges, regardless of using a ‘d’ (dogs or cats) or ‘p’ (other species) mode of calibration15. In agreement with previous studies, our findings indicated that both TonoVet and Tono-Pen consistently underestimated the actual IOP except at 5 mmHg for the Tono-Pen and the underestimation increased with the increase in IOP. The increasing inaccuracy might be attributed to the measurement error, which is increased for both devices as the pressure increases. Kalesnykas et al. reported that Tono-Pen overestimated IOP at reservoir pressure no more than 10 mmHg and underestimated IOP otherwise12. The inconsistency might be explained by the fact that their data were based on small sample size (n = 2).
To assess whether TonoVet and Tono-Pen had a similar degree of low prediction, we determined the difference of TonoVet and Tono-Pen measurements. As shown in Table 4, TonoVet had a tendency to measure a significantly higher IOP (p = 0.000, independent t-test) relative to Tono-Pen at real pressure greater than 20 mmHg, and gave a markedly lower IOP (p = 0.000, independent t-test) relative to Tono-Pen at pressure lower than 5 mmHg. However, at a pressure level between 10 and 15 mmHg, TonoVet was comparable with Tono-Pen in IOP estimation. These results suggest that Tono-Pen could be more accurate at IOP no more than 5 mmHg, whereas TonoVet could be more accurate at IOP higher than 20 mmHg. Overall, TonoVet was significantly more accurate than Tono-Pen, which suggested that TonoVet could be a more predictable device for evaluation of effectiveness of drugs or surgical treatments in rabbit eyes.
Differences in measurements can be attributed not only to the instruments and topical anesthesia, but also to the fluctuation of IOP in rabbit’s eyes. In our study, the mean IOP gradually decreased from 6 AM to 9 AM, and then gradually increased from 9 AM to 12 PM. Our results are in good agreement with a previous study, conducted in rabbits, which showed that the IOP during the daylight period (7 AM to 7 PM) was lower than that during the night (8 PM to 6 AM)17. The fluctuation pattern of IOP is likely linked to the variation of circadian rhythms and β-adrenergic systems during day-night alternation, which has an impact on the formation and regulation of aqueous humor13,18,19.
We recognized some limitations of the current study. First, measurement of IOP using rebound or applanation may be affected by the biomechanical characteristics of cornea. The tonometers were designed based on the characteristics of normal cornea. Therefore, we need to be careful when proposing the use of these instruments for assessing the treatment efficacy parameters in animal models of glaucoma where the corneal status of these animals remains unknown. Secondly, the performance of IOP measurement depends on the operator. Proper operation and experience are needed in clinical practice of IOP measurement.
In summary, we find that both TonoVet and Tono-Pen exhibit good precision in terms of excellent intrasession repeatability, excellent interoperator reproducibility and good intersession reproducibility. Additionally, progressive underestimation of the true IOP by TonoVet and Tono-Pen was observed with fixed and proportional biases, but overall the TonoVet was superior to Tono-Pen in both precision and accuracy. Altogether, the hand-held Tono-Pen could be expected to be a reliable tonometer for measurement of normal rabbit IOP, while the TonoVet could be applied for the measurement of IOP in both normal and hypertensive eyes.
Materials and Methods
Animals
The Ethics Committee for Animal Study of the Joint Shantou International Eye Center approved this study. The experiments were designed and conducted in accordance with the Association of Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Visual Research. Thirty female New Zealand albino rabbits were used with an average age of 5 months (range, 4–6 months) and mean weight of 2.8 kg (range, 2.5 to 3.1 kg). Rabbits were handled gently and were required to be calm with gentle touch at least 3 minutes before IOP measurement. If any sign of stress was found, IOP measurements were postponed for at least another 3 minutes. We used the right eyes only for IOP measurement to avoid bias due to bilateral correlation. All the measurement was performed between 8 to 10 am except in the diurnal study.
IOP measurements by TonoVet
IOP measurements were first performed with the TonoVet (Helsinki, Finland). Since there was no calibration mode programmed for rabbit in the TonoVet, a dog calibration mode was chosen instead. After securing the rabbit, the TonoVet was brought near the rabbit’s eye and fixed at the rabbit’s nose. The central groove of the tonometer was kept in a horizontal position and the distance was maintained at 4–8 mm between the tip of the probe and the cornea. The measurement button was pressured lightly to enable the tip of the probe to hit the central cornea. Six consecutive measurements were taken and the resulting IOP was displayed.
IOP measurements by Tono-Pen XL
The Tono-Pen XL (Reichert, Depew, NY) was equipped with an Ocu-Film tip cover and calibrated as instructed in the manufacturer’s manual prior to the first use each day. A drop of Alcaine ophthalmic solution (Alcon, Fort Worth, TX) was instilled onto the rabbit’s eye before the measurement. To reduce hand-held tonometer movement, the elbow of the operator was braced on the table for stability. The Tono-Pen transducer was held perpendicular to and within 1/2 an inch of the rabbit’s cornea. After pressing the operator’s button to initiate an IOP measurement and following the beep tone, the probe tip was touched lightly with the central cornea without indentation and then withdrawn. After four valid measurements, a final beep sounded and the averaged measurement displayed on the LCD, along with the single bar denoting statistical reliability. Measurements were repeated until the displayed coefficient of variation was less than 5%. The Ocu-Film tip cover was replaced before using the Tono-Pen XL unit on another rabbit.
Repeatability and reproducibility study
The precision of tonometry was characterized by its repeatability and reproducibility. Repeatability is defined as the intrasession variability for repeated measurements, which are performed by the same operator using the same instrument during a short period. Reproducibility refers to the variability in repeated measurements in which time or the operator is varied, characterized as intersession variability and interoperator variability, respectively. According to the assumption20 that the 95% confidence interval of within-subject standard deviation (Sw) to be estimated within 15% of Sw, 1.96 = 15% × Sw, sample size can be calculated using the following formula: n = 1.962/[2(m-1) × 0.152], where n and m represent as the number of subjects and observations (m = 4, n = 30 on this study), respectively. When measuring, IOP of the same eyes was determined with TonoVet and subsequently with Ton-Pen by two respective blinded operators consecutively for 4 days. The operators were blind to the reading of the other operator. In the repeatability analysis, four measurements from each operator measured every day were used while in the reproducibility analysis, the second measurement from each operator on each day was used. Parameters were calculated with Excel and SPSS, and included the intrasession, intersession and interoperator within-subject standard deviation (Sw), precision (repeatability coefficient) (1.96 × Sw), coefficient of variation (CVw) (100 × Sw/overall mean) and intraclass correlation coefficient (ICC). ICC values greater than 0.75 were considered as excellent reproducibility, between 0.40 and 0.59 as fair, between 0.6 and 0.75 as good reproducibility, and lower than 0.4 poor reproducibility8.
= 15% × Sw, sample size can be calculated using the following formula: n = 1.962/[2(m-1) × 0.152], where n and m represent as the number of subjects and observations (m = 4, n = 30 on this study), respectively. When measuring, IOP of the same eyes was determined with TonoVet and subsequently with Ton-Pen by two respective blinded operators consecutively for 4 days. The operators were blind to the reading of the other operator. In the repeatability analysis, four measurements from each operator measured every day were used while in the reproducibility analysis, the second measurement from each operator on each day was used. Parameters were calculated with Excel and SPSS, and included the intrasession, intersession and interoperator within-subject standard deviation (Sw), precision (repeatability coefficient) (1.96 × Sw), coefficient of variation (CVw) (100 × Sw/overall mean) and intraclass correlation coefficient (ICC). ICC values greater than 0.75 were considered as excellent reproducibility, between 0.40 and 0.59 as fair, between 0.6 and 0.75 as good reproducibility, and lower than 0.4 poor reproducibility8.
Diurnal change of IOP
Since intraocular pressure is dynamic and fluctuates with the change of physiological state, IOP in 10 rabbits of both eyes was measured every 3 hours, beginning at 6 AM and ending at midnight, by a single operator under same conditions as stated in repeatability measurement.
Agreement with manometric measurements
Sample size was calculated for the agreement study according to the formula n ≥  21, where n, α and β represent the sample size, discordance rate and tolerance probability, respectively (When α = 0.15 and β = 80%, n ≥ 10). The experiment was performed under general anesthesia using ketamine (30 mg/kg) and xylazine (5 mg/kg) intramuscularly. Two 25-gauge needles were inserted into the anterior chamber of the right eye via a self-sealing limbal incision at the 3 and 9 o’clock positions. One of them was connected via polyethylene tubing to a bottle filled with balanced salt solution under open stopcock condition, while the other was connected to a vertical water column for measurement of IOP. The height of the balanced salt solution bottle was vertically altered to achieve various IOPs, ranging from 60 to 5 mmHg by 5-mmHg decrements, based on the reading of the water column. When the reading of the water column reached a stable value for more than 2 minutes, four sets of IOP readings were taken blindly with TonoVet, and subsequently with Tono-Pen, at each bottle height. IOP readings of the two tonometers at each pressure level were compared with the actual IOP to determine the accuracy. Pearson’s correlation and linear regression analyses were used to determine a best-fit line for assessing the variation of measurement error. Bland-Altman bias plots22,23 were drawn to evaluate the level of agreement between IOP values measured by the two tonometers and manometer.
21, where n, α and β represent the sample size, discordance rate and tolerance probability, respectively (When α = 0.15 and β = 80%, n ≥ 10). The experiment was performed under general anesthesia using ketamine (30 mg/kg) and xylazine (5 mg/kg) intramuscularly. Two 25-gauge needles were inserted into the anterior chamber of the right eye via a self-sealing limbal incision at the 3 and 9 o’clock positions. One of them was connected via polyethylene tubing to a bottle filled with balanced salt solution under open stopcock condition, while the other was connected to a vertical water column for measurement of IOP. The height of the balanced salt solution bottle was vertically altered to achieve various IOPs, ranging from 60 to 5 mmHg by 5-mmHg decrements, based on the reading of the water column. When the reading of the water column reached a stable value for more than 2 minutes, four sets of IOP readings were taken blindly with TonoVet, and subsequently with Tono-Pen, at each bottle height. IOP readings of the two tonometers at each pressure level were compared with the actual IOP to determine the accuracy. Pearson’s correlation and linear regression analyses were used to determine a best-fit line for assessing the variation of measurement error. Bland-Altman bias plots22,23 were drawn to evaluate the level of agreement between IOP values measured by the two tonometers and manometer.
Additional Information
How to cite this article: Ma, D. et al. Repeatability, reproducibility and agreement of intraocular pressure measurement in rabbits by the TonoVet and Tono-Pen. Sci. Rep. 6, 35187; doi: 10.1038/srep35187 (2016).
References
Estrovich, I. E. et al. Schiotz tonometry accurately measures intraocular pressure in Boston type 1 keratoprosthesis eyes. Cornea 34, 682–685 (2015).
Feltgen, N., Leifert, D. & Funk, J. Correlation between central corneal thickness, applanation tonometry, and direct intracameral IOP readings. Br J Ophthalmol 85, 85–87 (2001).
Arribas-Pardo, P. et al. Measuring Intraocular Pressure After Intrastromal Corneal Ring Segment Implantation With Rebound Tonometry and Goldmann Applanation Tonometry. Cornea 34, 516–520 (2015).
Luce, D. A. Determining in vivo biomechanical properties of the cornea with an ocular response analyzer. J Cataract Refract Surg 31, 156–162 (2005).
Kanngiesser, H. E., Kniestedt, C. & Robert, Y. C. A. Dynamic contour tonometry - Presentation of a new tonometer. J Glaucoma 14, 344–350 (2005).
Whitacre, M. M., Emig, M. & Hassanein, K. The effect of Perkins, Tono-Pen, and Schiotz tonometry on intraocular pressure. Am J Ophthalmol 111, 59–64 (1991).
Davey, P. G., Elsheikh, A. & Garway-Heath, D. F. Clinical evaluation of multiparameter correction equations for Goldmann applanation tonometry. Eye 27, 621–629 (2013).
Acosta, A. C. et al. Estimation of intraocular pressure in rabbits with commonly used tonometers. Ophthalmic Surg Lasers Imaging 38, 43–49 (2007).
Wang, W. H., Millar, J. C., Pang, I. H., Wax, M. B. & Clark, A. F. Noninvasive measurement of rodent intraocular pressure with a rebound tonometer. Invest Ophthalmol Vis Sci 46, 4617–4621 (2005).
Görig, C., Coenen, R. T. I., Stades, F. C., Djajadiningrat-Laanen, S. C. & Boevé, M. H. Comparison of the use of new handheld tonometers and established applanation tonometers in dogs. Am J Vet Res 67, 134–144 (2006).
Lim, K. S., Wickremasinghe, S. S., Cordeiro, M. F., Bunce, C. & Khaw, P. T. Accuracy of intraocular pressure measurements in New Zealand white rabbits. Invest Ophthalmol Vis Sci 46, 2419–2423 (2005).
Kalesnykas, G. & Uusitalo, H. Comparison of simultaneous readings of intraocular pressure in rabbits using Perkins handheld, Tono-Pen XL, and TonoVet tonometers. Graefes Arch Clin Exp Ophthalmol 245, 761–762 (2007).
Pereira, F. Q., Bercht, B. S., Soares, M. G., da Mota, M. G. B. & Pigatto, J. A. T. Comparison of a rebound and an applanation tonometer for measuring intraocular pressure in normal rabbits. Vet Ophthalmol 14, 321–326 (2011).
Setogawa, A. & Kawai . Measurement of intraocular pressure by both invasive and noninvasive techniques in rabbits exposed to head-down tilt. Jpn J Physiol 48, 25–31 (1998).
Zhang, H. et al. Validation of rebound tonometry for intraocular pressure measurement in the rabbit. Exp Eye Res 121, 86–93 (2014).
Abrams, L. S., Vitale, S. & Jampel, H. D. Comparison of three tonometers for measuring intraocular pressure in rabbits. Invest Ophthalmol Vis Sci 37, 940–944 (1996).
Wang, X. G., Dong, J. & Wu, Q. Twenty-four-hour measurement of IOP in rabbits using rebound tonometer. Vet Ophthalmol 16, 423–428 (2013).
Bromberg, B. B., Gregory, D. S. & Sears, M. L. Beta-adrenergic receptors in ciliary processes of the rabbit. Invest Ophthalmol Vis Sci 19, 203–207 (1980).
McCannel, C. A., Heinrich, S. R. & Brubaker, R. F. Acetazolamide but not timolol lowers aqueous humor flow in sleeping humans. Graefes Arch Clin Exp Ophthalmol 230, 518–520 (1992).
Bland, J. M. How can I decide the sample size for a repeatability study? Available at:http://www-users.york.ac.uk/~mb55/meas/sizerep.htm (2010).
Liao, J. J. Z. Sample size calculation for an agreement study. Pharm Stat 9, 125–132 (2010).
Bland, J. M. & Altman, D. G. STATISTICAL METHODS FOR ASSESSING AGREEMENT BETWEEN TWO METHODS OF CLINICAL MEASUREMENT. Lancet 327, 307–310 (1986).
Bland, J. M. & Altman, D. G. Measuring agreement in method comparison studies. Stat Methods Med Res 8, 135–160 (1999).
Acknowledgements
This study was supported in part by the National Nature Science Foundation of China (81170853 and 81470636) and the Specialized Research Fund for the Doctoral Program of Higher Education (20114402110005). The authors thank Dr. Stanley Lin, Department of Pathophysiology, The Key Immunopathology Laboratory of Guangdong Province, Shantou University Medical College, for the assistance in revising the paper.
Author information
Authors and Affiliations
Contributions
H.C. designed the study. D.M., C.B.C., J.L. and Z.L. conducted the experiments, D.M. and C.B.C. wrote the main manuscript and prepared all figures. H.C. and M. Z. revised the manuscript. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Ma, D., Chen, CB., Liang, J. et al. Repeatability, reproducibility and agreement of intraocular pressure measurement in rabbits by the TonoVet and Tono-Pen. Sci Rep 6, 35187 (2016). https://doi.org/10.1038/srep35187
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep35187
- Springer Nature Limited
This article is cited by
-
Effect of SMILE-derived decellularized lenticules as an adhesion barrier in a rabbit model of glaucoma filtration surgery
BMC Ophthalmology (2021)
-
In vivo biocompatibility evaluation of in situ-forming polyethylene glycol-collagen hydrogels in corneal defects
Scientific Reports (2021)
-
Repeatability, reproducibility, and agreement of three tonometers for measuring intraocular pressure in rabbits
Scientific Reports (2021)
-
Pluripotent epigenetic regulator OBP-801 maintains filtering blebs in glaucoma filtration surgery model
Scientific Reports (2020)