Abstract
Eight heterogeneous vancomycin-intermediate S. aureus (h-VISA) and seven VISA clinical isolates confirmed by the population analysis profile/area under the curve ratio (PAP/AUC) were collected. We further performed the PAP/AUC, time-killing methods and MIC tests using vancomycin/teicoplanin alone or combination with susceptible breakpoint concentrations of cefazolin, cefmetazole, cefotaxime, and cefepime for these isolates. The PAP/AUC MIC curve shifted left after addition of cephalosporins with vancomycin or teicoplanin for both h-VISA and VISA isolates. With the combination of different cephalosporins with vancomycin or teicoplanin, the AUC/Mu3 AUC ratio decreased to <0.9 for the standard Mu3 isolate which are compatible with the definition of vancomycin susceptible S. aureus. These decreases ranged between 1.81–2.02 and 2.37–2.85-fold for h-VISA treated with cephalosporins and vancomycin or teicoplanin, and 2.05–4.59, and 2.93–4,89-fold for VISA treated with cephalosporins with vancomycin or teicoplanin. As measured by time-killing assays, the combinations of different cephalosporins with vancomycin concentrations at 1/2 and 1/4 MIC, exhibited a bactericidal and bacteriostatic effect in VISA. The mean fold of MIC decline for vancomycin base combinations ranged from 1.81–3.83 and 2.71–9.33 for h-VISA and VISA, respectively. Overall, this study demonstrated the enhanced antibacterial activity of vancomycin/teicoplanin after adding cephalosporins against clinical h-VISA/VISA isolates.
Similar content being viewed by others
Introduction
Methicillin-resistant Staphylococcus aureus (MRSA) is a prevalent pathogen that causes human infections in community and hospital settings globally1,2,3. Moreover, MRSA infection can be associated with high morbidity and mortality4. Vancomycin is the most commonly used antibiotic to treat MRSA infections4,5,6. However, due to the increasing use of vancomycin, clinical MRSA isolates with reduced susceptibility to vancomycin have emerged recently7,8,9. Additionally, the clinical outcomes of heterogeneous vancomycin-intermediate S. aureus (h-VISA) and vancomycin-intermediate S. aureus (VISA) infections are poor10,11,12. Most important of all, the treatment options for h-VISA/VISA infections are limited13.
Aside from vancomycin, there are some viable alternative antimicrobial agents, such as daptomycin, linezolid, ceftaroline, trimethoprim/sulfamethoxazole, tigecycline, and quinupristin/dalfopristin13. Currently, these alternative choices with single antibiotics have not proved to be superior to vancomycin. Therefore, combination therapy may provide another option for combating this critical condition caused by h-VISA/VISA14,15. Werth et al.15 demonstrated vancomycin plus oxacillin or ceftaroline may improve the activity of vancomycin against h-VISA/VISA by enhancing vancomycin-cell wall interaction. Dilworth et al.14 showed the synergistic activity of vancomycin with piperacillin-tazobactam or oxacillin against VISA. However, no investigation has assessed the activity of combination of any of the four generations of cephalosporins with either vancomycin or teicoplanin against h-VISA/VISA. In this report, we conducted a comparative study of the combination of cephalosporins of all generations with either vancomycin or teicoplanin against h-VISA/VISA isolates by three laboratory methods to evaluate the in vitro antibacterial activity among different glycopeptide/cephalosporin (G/C) combinations.
Material and Method
Bacterial isolates
Eight h-VISA and seven VISA clinical isolates were collected from the Tigecycline In Vitro Surveillance in Taiwan (TIST) study, which collected clinical isolates from 22 hospitals between 2006 and 201016. Staphylococci were identified by colony morphology, Gram stain, and coagulase test. MRSA isolates were further confirmed by the tube coagulase test and growth on 6 μg/ml oxacillin salt agar screen plates. Vancomycin MIC was measured by the agar dilution method. All h-VISA or VISA isolates were confirmed by calculating the population analysis profile/area under the curve ratio (PAP/AUC). Isolates were stored at −70 °C in Protect Bacterial Preservers (Technical Service Consultants Limited, Heywood, UK) until use. All eight h-VISA and seven VISA isolates were selected from different PFGE types, as previously described17,18.
Antibiotics and MIC measurement
The MICs of cefazolin (CF), cefmetazole (CMZ), cefotaxime (CTX), cefepime (CPO), vancomycin (Sigma, St Louis, MO), and teicoplanin (Sanofi-Aventis, Bridgewater, NJ) were determined by agar dilution method, and interpretation criteria were based on the recommendations of the Clinical and Laboratory Standards Institute (CLSI)19. S. aureus ATCC 29213 was used as a control strain for MIC measurements.
PAP/AUC for vancomycin and teicoplanin
The PAP/AUC was measured for all isolates by inoculating serial 10-fold dilutions of the test organism onto increasing concentrations of vancomycin- or teicoplanin-containing brain heart infusion (BHI) agar (Becton Dickinson, Sparks, MD, USA). The BHI agar plates contained vancomycin at the following concentrations 0, 0.5, 1, 1.5, 2, 3, 4, 6, and 8 mg/L or teicoplanin at the following concentrations 0, 1, 2, 4, 8, 16, and 32 mg/liter. Colony growth at 48 h was measured and graphed as log10 CFU/ml to obtain vancomycin PAPs (v-PAP) and teicoplanin PAPs (t-PAP). The v-PAP graph was used to calculate the AUC of each isolate, and the ratio of the AUC of the test isolate to the AUC of S. aureus Mu3 (ATCC 700698) was calculated. The AUC of tested isolate/Mu3 ratios were calculated. Ratios less than 0.9 were considered as VSSA and ratios of 0.9 to 1.3 and >1.3 were considered h-VISA and VISA, respectively20. Additionally, vancomycin criteria were extended to teicoplanin for this study.
v-PAP/AUC and t-PAP/AUC reducing test
The PAP/AUC were detected with BHI containing vancomycin or teicoplanin alone and combination with susceptible breakpoint concentration of cephalosporins of all generations against eight h-VISA and seven VISA clinical isolates with the ratios of tested isolate/Mu3 were calculated. The fold decrease of vancomycin or teicoplanin alone with the G/C combinations were also calculated.
Time-kill method
Eight h-VISA and seven VISA clinical isolates were selected for another in vitro measurement of inhibitory effect of combination regimens as recommended by the CLSI21. In brief, bacterial suspensions were diluted to 5.0 × 105 colony-forming units (CFU)/mL in fresh Mueller–Hinton broth. Drug concentrations of vancomycin or teicoplanin were adjusted to 1/2xMIC, and 1/4xMIC. Each cephalosporin was used at susceptible breakpoint concentrations when in combination with a glycopeptide. Bacterial counts were measured at 8 h and 24 h by enumerating the colonies in 10-fold serially diluted specimens of 100 μL aliquots plated on the nutrient agar (Difco Laboratories, Sparks, MD) at 37 °C. All experiments were performed in duplicate.
Synergism was defined as a ≥2 log10 decrease in CFU/mL between the combination regimen and its most active constituent after 24 h and the number of surviving organisms in the combination regimen must be ≥2 log10 CFU/mL below the starting inoculum. In addition, at least one of the combination drugs must be present at a concentration that does not affect the growth of the test organism. Bacteriostatic and bactericidal activities were defined as <3 log10 and ≥3 log10 reductions in CFU/ml at 24 h, respectively, relative to the starting inoculum21.
MIC change ratios of glycopeptide MICs
The MICs of vancomycin or teicoplanin alone and combined with 1x susceptible breakpoint concentration of a cephalosporin against eight h-VISA and seven VISA clinical isolates were determined by agar dilution method. A MIC ratio indicates the fold of the MIC decline of G/C combination versus a glycopeptide alone.
Results
The results of MIC tests
Table 1 shows the MICs of each cephalosporin, teicoplanin and vancomycin against eight h-VISA and seven VISA isolates. The MIC ranges of vancomycin and teicoplanin against h-VISA isolates were 1–2 mg/L, and 2–4 mg/L, respectively. For VISA isolates, the vancomycin MIC were all 4 mg/L, and teicoplanin MIC ranged from 4–16 mg/L. All of the h-VISA and VISA isolates were resistant to every cephalosporins based on the MIC level.
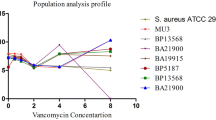
Figure 1 shows that MIC curve shifts left after addition of different cephalosporins with vancomycin for both h-VISA and VISA isolates. Figure 2 shows the change of teicoplanin MIC against h-VISA/VISA after the addition of various cephalosporins. A similar phenomenon with vancomycin MIC was noted.
PAU/AUC methods
The AUC/Mu3 AUC ratio of the eight h-VISA isolates were all approximately 0.95–1.14, which is compatible with the definition of h-VISA. In both hVISA and VISA and for all combinations of cephalosporins and vancomycin the PAP/AUC ratios decreased to below 0.9, compatible with VSSA. The v-PAP/AUC decreased between 1.81–2.02 and 2.37–2.85-fold after the combinations of cephalosporins for h-VISA and VISA isolates, respectively. For teicoplanin, the results were also the similar. With the combination of different cephalosporins with teicoplanin, all ratios decreased (<0.9), including the standard Mu3 isolate, and all of the tested h-VISA/VISA isolates are also compatible with the definition of VSSA. The t-PAP/AUC decreased between 2.05–4.59 and 2.93–4.89-fold after the combinations of cephalosporins for h-VISA and VISA isolates, respectively.
Time-killing methods
Time-killing assays against each isolate are shown in Table 2. For h-VISA isolates, only one of the combination of vancomycin with CMZ exhibited bactericidal synergistic effect. One or two of the isolates disclosed bacteriostatic synergistic effect between other cephalosporins with vancomycin. Neither bactericidal nor bacteriostatic effect for the combinations of CTX was observed. For VISA isolates, such combination exhibited bactericidal synergistic effect against one to five of the seven tested isolates, respectively. Such combination also exhibited bacteriostatic synergistic effect against one to four of the tested isolates, respectively.
The in vitro activities of combination of CF, CMZ, CTX, CPO and teicoplanin were shown in Table 3. For h-VISA isolates, one to five of such combination exhibited bactericidal synergistic effect. One to four of the isolates disclosed bacteriostatic synergistic effect. For VISA isolates, such combinations exhibited bactericidal synergistic effect against five to six of the seven tested isolates. However, such combinations disclosed bacteriostatic synergistic effect against one to five of tested isolates, respectively.
The changes of MIC levels
The MICs of vancomycin and teicoplanin with and without susceptible breakpoint concentration of cephalosporins for h-VISA/VISA isolates were shown in Table 4. The mean fold of MIC decline for vancomycin base combinations range from 1.81–3.83 and 2.71–9.33 for h-VISA and VISA, respectively. For teicoplanin base combinations, the mean fold of MIC decline range from 2.25–7.5 and 5.43–21.71 for h-VISA and VISA, respectively. The MICs of vancomycin and teicoplanin against h-VISA/VISA isolates lowered after combination with cephalosporins compared with single use. The decreases of MICs against h-VISA/VISA isolates seems to be highest for the combinations of CMZ and vancomycin or teicoplanin, followed by the CF regimen.
Discussion
This study assesses the effect of addition of different cephalosporins with glycopeptides against h-VISA/VISA isolates and has several significant findings. First of all, we demonstrated the synergistic effect of glycopeptides and cephalosporins against h-VISA/VISA isolates by different methods based on this in vitro study. Initially, we used the PAP/AUC method to confirm both of the h-VISA and VISA isolates, and demonstrated the change of PAP/AUC curve to fit the criteria of VSSA from h-VISA/VISA after the combination of different cephalosporins. This study describes experiments using both vancomycin and teicoplanin. Secondly, the MICs of cephalosporins to h-VISA/VISA remained very high. However, when we used susceptible breakpoint concentrations which were far lower than MIC, the enhanced synergistic effect persisted even at 1/2, 1/4xMIC vancomycin or teicoplanin. Finally, via such combinations, we can also find the mean vancomycin and teicoplanin MIC decreases many folds. All of these findings indicated that even though h-VISA/VISA is insensitive to cephalosporins alone, combinations of antimicrobials and very low concentrations of cephalosporins still play important roles in the glycopeptide therapy of h-VISA/VISA.
However, previous studies14,15,22,23,24,25 had conflicting findings regarding the combined effect of glycopeptides and various antibiotics against h-VISA/VISA isolates. In Kim et al.’s report22, an antagonistic effect was observed in combinations with less than 1 μg/ml β-lactam antibiotics – oxacillin and cefotaxime combined with vancomycin, whereas synergistic effects were noticed in combinations with more than 4 μg/ml β-lactams antibiotics. In Aritaka et al.’s study23, they noted that when combined with vancomycin, four of the seven tested β-lactams, including ampicillin, oxacillin, imipenem, and cefmetazole, exhibited an additive effect at or near their MICs; however, in contrast, all of the tested β-lactams showed an antagonistic effect at lower sub-MIC levels. Therefore, their findings indicated that β-lactam antibiotics may not provide a significant advantage in combination with vancomycin against Mu3-like hetero-VISA strains. In contrast, we find the synergistic effect exist between cephalosporins and glycopeptides even at very low cephalosporin concentrations. The differences between the present work and previous investigations may be due to the different antibiotics we used for combination and different concentrations of antibiotics we used in this study.
In the report by Dilworth et al.14, four bloodstream VISA strains, the mean 24-h reductions in VISA inoculum for piperacillin-tazobactam, piperacillin-tazobactam with vancomycin, and oxacillin with vancomycin were 2.85, 2.93, and 3.45 log10 CFU/ml, respectively, and indicated the synergistic activity against VISA by vancomycin with piperacillin-tazobactam or oxacillin. In Leonard’s in vitro pharmacokinetic(PK)/pharmacodynamics(PD) study25, 23 (92%) of 25 h-VISA strains showed synergy with the combination of vancomycin and nafcillin in time-killing method, and among five selected strains demonstrated an improvement in overall activity and organism burden at 72 hours in PK/PD model. In Werth et al.’s study15 using time-kill assays, vancomycin plus oxacillin was synergistic against 3 of 5 VISA and 1 of 5 h-VISA isolates and vancomycin plus ceftaroline demonstrated a synergistic effect against 5 of 5 VISA and 4 of 5 h-VISA isolates. In Hanaki et al.’s study24, they found that teicoplanin showed the strongest synergistic effect in combination with imipenem, followed by, in decreasing order, panipenem, meropenem, flomoxef, sulbactam/ampicillin, cefoselis, and the average FIC indexes of the beta-lactam antibiotics against these strains were 0.113, 0.124, 0.163, 0.230, 0.264 and 0.388, respectively. All of the above findings suggest the synergistic effect of glycopeptide and beta-lactam combinations and are consistent with our result.
Although the different cephalosporin MIC values of tested h-VISA/VISA isolates remains high, the antibacterial effect was evident by a significant reduction of glycopeptide MICs, if serum achievable concentrations (1/2x or 1x SBC) of a cephalosporin were combined with sub-inhibitory concentrations of vancomycin or teicoplanin. Such an in vitro effect of glycopeptide MIC reduction was most obvious for cefmetazole among four tested cephalosporins. Therefore, although high cephalosporin resistance in clinical h-VISA/VISA isolates, we showed the strong evidence that cephalosporins can enhance the antibacterial activity of two commonly prescribed glycopeptides.
Several studies and meta-analyses have shown the significant association between vancomycin MIC and the outcomes of MRSA infections26,27,28,29. In Sakoulas et al.’s study28, vancomycin can successful treated for 55.6% of MRSA bacteremia while the MRSA isolates with vancomycin MICs < 0.5 μg/ml, but vancomycin was only effective for 9.5% of cases in which vancomycin MICs for MRSA were 1 to 2 μg/ml. For MRSA pneumonia, Haquel et al.27 found that the 28-day mortality could significantly increase while vancomycin MIC increased from 0.75 to 3 mg/mL (P ≤ 0.001). One recent meta-analysis of 2439 patients with MRSA infections (1492 high MIC (defined as ≥1 mg/l by BMD or ≥1.5 mg/l by E-test) and 947 low MIC) concluded that a high MIC to vancomycin is associated with increased mortality and treatment failure26. The similar influence of teicoplanin MICs on treatment outcomes among patients with teicoplanin-treated MRSA infection were shown in previous investigations and teicoplanin MIC ≥4 μg/mL (P = 0.0123) had a significant association with treatment failure in such patients30,31,32. In this in vitro study, we showed that vancomycin MICs of h-VISA and VISA isolates could be reduced to ≤0.75 μg/ml after combination with different cephalosporins, and even more, these combinations could make h-VISA or VISA become VSSA. For teicoplanin, its MIC of h-VISA and VISA isolates could also be reduced to ≤2 μg/ml after combining with cephalosporins. Although in vitro activity cannot represent in vivo effect, we can expect that the outcome improvement of h-VISA and VISA infections is possible to achieve after introduction of the glycopeptide/cephalosporin combination therapy based on our findings and previous investigations26,27,28. Of course, the application of such combinations to treat h-VISA/VISA infections needs more clinical investigations.
In conclusions, this is the first in vitro study by different methods to evaluate G/C combinations, and it showed the enhanced antibacterial activity against clinical h-VISA/VISA isolates, irrespective of cephalosporin MICs. Further comprehensive including in vivo study is warranted to investigate the combination effect between each class of antibiotics and glycopeptides.
Additional Information
How to cite this article: Lai, C.-C. et al. Combination of cephalosporins with vancomycin or teicoplanin enhances antibacterial effect of glycopeptides against heterogeneous vancomycin-intermediate Staphylococcus aureus (hVISA) and VISA. Sci. Rep. 7, 41758; doi: 10.1038/srep41758 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
DeLeo, F. R., Otto, M., Kreiswirth, B. N. & Chambers, H. F. Community-associated meticillin-resistant Staphylococcus aureus. Lancet 375, 1557–1568, doi: 10.1016/s0140-6736(09)61999-1 (2010).
Otter, J. A. & French, G. L. Molecular epidemiology of community-associated meticillin-resistant Staphylococcus aureus in Europe. Lancet Infect. Dis. 10, 227–239, doi: 10.1016/s1473-3099(10)70053-0 (2010).
Tacconelli, E. et al. Rapid screening tests for meticillin-resistant Staphylococcus aureus at hospital admission: systematic review and meta-analysis. Lancet Infect. Dis. 9, 546–554, doi: 10.1016/s1473-3099(09)70150-1 (2009).
Liu, C. et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: executive summary. Clin. Infect. Dis. 52, 285–292, doi: 10.1093/cid/cir034 (2011).
VanEperen, A. S. & Segreti, J. Empirical therapy in methicillin-resistant Staphylococcus aureus infections: An up-to-date approach. J. Infect. Chemother. 22, 351–359, doi: 10.1016/j.jiac.2016.02.012 (2016).
Rodvold, K. A. & McConeghy, K. W. Methicillin-resistant Staphylococcus aureus therapy: past, present, and future. Clin. Infect. Dis. 58 Suppl 1, S20–27, doi: 10.1093/cid/cit614 (2014).
Hiramatsu, K. et al. Methicillin-resistant Staphylococcus aureus clinical strain with reduced vancomycin susceptibility. J. Antimicrob. Chemother. 40, 135–136 (1997).
Jones, R. N. Microbiological features of vancomycin in the 21st century: minimum inhibitory concentration creep, bactericidal/static activity, and applied breakpoints to predict clinical outcomes or detect resistant strains. Clin. Infect. Dis. 42 Suppl 1, S13–24, doi: 10.1086/491710 (2006).
Martin, J. H. et al. Therapeutic monitoring of vancomycin in adult patients: a consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society Of Infectious Diseases Pharmacists. Clin. Biochem. Rev. 31, 21–24 (2010).
Casapao, A. M. et al. Clinical outcomes in patients with heterogeneous vancomycin-intermediate Staphylococcus aureus (hVISA) bloodstream infection. Antimicrob. Agents Chemother. doi: 10.1128/aac.00380-13 (2013).
Charles, P. G., Ward, P. B., Johnson, P. D., Howden, B. P. & Grayson, M. L. Clinical features associated with bacteremia due to heterogeneous vancomycin-intermediate Staphylococcus aureus. Clin. Infect. Dis. 38, 448–451, doi: 10.1086/381093 (2004).
Howden, B. P. et al. Treatment outcomes for serious infections caused by methicillin-resistant Staphylococcus aureus with reduced vancomycin susceptibility. Clin. Infect. Dis. 38, 521–528, doi: 10.1086/381202 (2004).
Gomes, D. M., Ward, K. E. & LaPlante, K. L. Clinical implications of vancomycin heteroresistant and intermediately susceptible Staphylococcus aureus. Pharmacotherapy 35, 424–432, doi: 10.1002/phar.1577 (2015).
Dilworth, T. J., Sliwinski, J., Ryan, K., Dodd, M. & Mercier, R. C. Evaluation of vancomycin in combination with piperacillin-tazobactam or oxacillin against clinical methicillin-resistant Staphylococcus aureus Isolates and vancomycin-intermediate S. aureus isolates in vitro . Antimicrobial. Agents Chemother. 58, 1028–1033, doi: 10.1128/aac.01888-13 (2014).
Werth, B. J. et al. Novel combinations of vancomycin plus ceftaroline or oxacillin against methicillin-resistant vancomycin-intermediate Staphylococcus aureus (VISA) and heterogeneous VISA. Antimicrobial. Agents Chemother. 57, 2376–2379, doi: 10.1128/aac.02354-12 (2013).
Hsueh, P. R. Tigecycline In-vitro Surveillance in Taiwan (TIST). Int. J. Antimicrobial. Agents 32 Suppl 3, S173, doi: 10.1016/s0924-8579(08)70022-0 (2008).
Chung, M. et al. Molecular typing of methicillin-resistant Staphylococcus aureus by pulsed-field gel electrophoresis: comparison of results obtained in a multilaboratory effort using identical protocols and MRSA strains. Microb. Drug Resist. 6, 189–198, doi: 10.1089/mdr.2000.6.189 (2000).
Tenover, F. C. et al. Interpreting chromosomal DNA restriction patterns produced by pulsed-field gel electrophoresis: criteria for bacterial strain typing. J. Clin. Microbiol. 33, 2233–2239 (1995).
Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. Twenty-second informational supplement M100–S22 (Wayne, PA, 2012).
Wootton, M. et al. A modified population analysis profile (PAP) method to detect hetero-resistance to vancomycin in Staphylococcus aureus in a UK hospital. J. Antimicrob. Chemother. 47, 399–403 (2001).
National Committee for Clinical Laboratory Standards . Methods for determining bactericidal activity of antimicrobial agents. Approved guideline M26-A NCCLS, W. PA (1999).
Kim, Y. S. et al. Efficacy of vancomycin-beta-lactam combinations against heterogeneously vancomycin-resistant Staphylococcus aureus (hetero-VRSA). J. Korean Med. Sci. 18, 319–324, doi: 10.3346/jkms.2003.18.3.319 (2003).
Aritaka, N., Hanaki, H., Cui, L. & Hiramatsu, K. Combination effect of vancomycin and beta-lactams against a Staphylococcus aureus strain, Mu3, with heterogeneous resistance to vancomycin. Antimicrobial. Agents Chemother. 45, 1292–1294, doi: 10.1128/aac.45.4.1292-1294.2001 (2001).
Hanaki, H. & Hiramatsu, K. Combination effect of teicoplanin and various antibiotics against hetero-VRSA and VRSA. Kansenshogaku Zasshi 73, 1048–1053 (1999).
Leonard, S. N. Synergy between vancomycin and nafcillin against Staphylococcus aureus in an in vitro pharmacokinetic/pharmacodynamic model. PLOS ONE 7, e42103, doi: 10.1371/journal.pone.0042103 (2012).
Jacob, J. T. & DiazGranados, C. A. High vancomycin minimum inhibitory concentration and clinical outcomes in adults with methicillin-resistant Staphylococcus aureus infections: a meta-analysis. Inter. J. Infect. Dis. 17, e93–e100, doi: 10.1016/j.ijid.2012.08.005 (2013).
Haque, N. Z. et al. Relationship of vancomycin minimum inhibitory concentration to mortality in patients with methicillin-resistant Staphylococcus aureus hospital-acquired, ventilator-associated, or health-care-associated pneumonia. Chest 138, 1356–1362, doi: 10.1378/chest.09-2453 (2010).
Sakoulas, G. et al. Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin-resistant Staphylococcus aureus bacteremia. J. Clin. Microbiol. 42, 2398–2402, doi: 10.1128/jcm.42.6.2398-2402.2004 (2004).
Maclayton, D. O., Suda, K. J., Coval, K. A., York, C. B. & Garey, K. W. Case-control study of the relationship between MRSA bacteremia with a vancomycin MIC of 2 microg/mL and risk factors, costs, and outcomes in inpatients undergoing hemodialysis. Clin. Ther. 28, 1208–1216, doi: 10.1016/j.clinthera.2006.08.003 (2006).
Chang, H. J. et al. Influence of teicoplanin MICs on treatment outcomes among patients with teicoplanin-treated methicillin-resistant Staphylococcus aureus bacteraemia: a hospital-based retrospective study. J. Antimicrobial. Chemother. 67, 736–741, doi: 10.1093/jac/dkr531 (2012).
Chen, K. Y. et al. Relationship of teicoplanin MICs to treatment failure in teicoplanin-treated patients with methicillin-resistant Staphylococcus aureus pneumonia. J. Microbiol. Immunol. Infect. 46, 210–216, doi: 10.1016/j.jmii.2012.06.010 (2013).
Koh, Y. R., Kim, K. H., Chang, C. L. & Yi, J. Prevalence and clinical impact of heterogeneous Vancomycin-intermediate Staphylococcus aureus isolated from hospitalized patients. Ann. Lab. Med. 36, 235–243, doi: 10.3343/alm.2016.36.3.235 (2016).
Acknowledgements
This study was supported by a grant from the Ministry of Science and Technology of Taiwan (MOST 105–2314-B-384–007-MY3) and Chi-Mei Medical Center Research Foundation (CMFHT10501, CMNCKU10509).
Author information
Authors and Affiliations
Contributions
H.J. Tang is the guarantor of this manuscript, C.C. Lai, C.C. Chen, and Y.C. Chuang contributed to the conception and design of the study, C.C. Chen and H.J. Tang analysed and interpreted the data, C.C. Lai, C.C. Chen and H.J. Tang drafted the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lai, CC., Chen, CC., Chuang, YC. et al. Combination of cephalosporins with vancomycin or teicoplanin enhances antibacterial effect of glycopeptides against heterogeneous vancomycin-intermediate Staphylococcus aureus (hVISA) and VISA. Sci Rep 7, 41758 (2017). https://doi.org/10.1038/srep41758
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep41758
- Springer Nature Limited
This article is cited by
-
Ceftaroline Fosamil for the Treatment of Methicillin-Resistant Staphylococcus Aureus Bacteremia: A Real-World Comparative Clinical Outcomes Study
Drugs - Real World Outcomes (2024)
-
Systematic analysis of drug combinations against Gram-positive bacteria
Nature Microbiology (2023)