Abstract
Wound healing is complex and highly orchestrated. It is well appreciated that leukocytes, particularly macrophages, are essential for inducing the formation of new connective tissue, which requires the generation of signals that stimulate mesenchymal stem cells (MSC), myofibroblasts and fibroblasts. A key role for keratinocytes in this complex process has yet to be established. To this end, we investigated possible involvement of keratinocytes in connective tissue healing. By lineage-specific deletion of the forkhead box-O 1 (FOXO1) transcription factor, we demonstrate for the first time that keratinocytes regulate proliferation of fibroblasts and MSCs, formation of myofibroblasts and production of collagen matrix in wound healing. This stimulation is mediated by a FOXO1 induced TGFβ1/CTGF axis. The results provide direct evidence that epithelial cells play a key role in stimulating connective tissue healing through a FOXO1-dependent mechanism. Thus, FOXO1 and keratinocytes may be an important therapeutic target where healing is deficient or compromised by a fibrotic outcome.
Similar content being viewed by others
Introduction
Wound healing is complex and highly orchestrated. Normal wound healing is divided into overlapping phases of inflammation, cellular proliferation, matrix production and tissue remodeling1. New connective tissue provides a supporting bed for the epithelium and restores dermal function. The production of connective tissue matrix is carried out by fibroblasts and myofibroblasts that migrate from adjacent tissue or differentiate from mesenchymal stem cells (MSCs)2,3. Leukocytes, particularly macrophages are thought to be essential for inducing formation of new connective tissue4,5,6 by generating signals that stimulate MSCs and fibroblasts7. Interestingly, in some genetically modified mice and in fetal wounds there is substantially reduced numbers of macrophages without impairment of healing8,9, suggesting the existence of as yet unidentified critical mechanisms of wound healing.
Interactions between epidermal keratinocytes and dermal fibroblasts contribute to the organization of the epidermis and are thought to promote early wound healing10. Paracrine cross talk of cytokines between dermal fibroblasts and epidermal keratinocytes may play a role in this process11,12. During healing process re-epithelialization coincides with the recruitment of MSCs and proliferation of dermal fibroblasts13,14. The expression of growth factors, extracellular matrix constituents, proteases, and intracellular structural proteins in fibroblasts are modulated by keratinocytes when fibroblasts and keratinocytes are co-cultured15. In addition, myofibroblast differentiation is increased by co-culture of fibroblasts with keratinocytes16. This is useful to healing since myofibroblasts produce extracellular matrix and contract granulation tissue. Taken together these studies provide a framework to suggest that keratinocytes may modulate connective tissue healing but direct evidence for this concept is lacking.
We have recently shown that the forkhead box-O 1 (FOXO1) transcription factor plays a critical role in re-epithelialization in vitro, while FOXO3, a related FOXO transcription factor, had little to no effect17. FOXO1 modulates several aspects of keratinocyte behavior including resistance to oxidative stress, keratinocyte migration and is also an important regulator of broad gene expression programs, including metabolism (e.g. gluconeogenesis, amino acid catabolism, glycolysis, pentose phosphate shunt, and fatty acid/triglyceride/sterol synthesis), cell cycle, stress response, and longevity in Caenorhabditis elegans18,19. In this report, we tested the hypothesis that epithelial cell-specific FOXO1 is additionally involved in the regulation of connective tissue wound healing. Specifically, to investigate the role of cutaneous epithelium in connective tissue healing, we examined full thickness wounds in mice with keratin-14 driven FOXO1 deletion compared to littermate controls. The results demonstrate for the first time that FOXO1 activates epithelial cells to regulate proliferation of fibroblasts and MSCs, formation of myofibroblasts and production of collagen matrix. These studies provide a basis to further investigate the impact of epithelium on wound healing, which up to now has focused primarily on the effects of macrophages or the effect of connective tissue cells on epithelium. Furthermore, the results solidify the critical impact of the transcription factor FOXO1 in modulating the wound healing behavior of keratinocytes.
Results
Keratinocyte-specific deletion of FOXO1 impairs connective tissue healing responses in mice dermal wounds
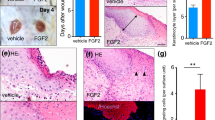
To explore the role of FOXO1 in dermal wound healing, experimental mice with keratinocyte-specific FOXO1 deletion (FOXO1 deleted in KC) were compared to littermate controls (wild type). FOXO1 expression was almost 10 fold higher in the epithelium than connective tissue and reduced in epithelium by more than 85% in experimental (p < 0.05) but not control littermates (p < 0.05) (Fig. 1a). We previously demonstrated that deleting FOXO1 in keratinocytes impaired re-eppithelialization17. Surprisingly, the effect on connective tissue was similarly important as FOXO1 ablation in keratinocytes significantly reduced connective tissue healing (Fig. 1b,c). We examined newly formed granulation tissue and collagen matrix by Masson’s trichrome staining 4 and 7 days after wounding, time points when granulation tissue formation is robust (day 4) and a later time point when granulation tissue is present but the wound is in the process of closing (day 7) (Fig. 1b,c). At 4 days keratinocyte-specific deletion of FOXO1 reduced the amount of granulation tissue by 44% and on day 7 by 40% compared to littermate control mice (p < 0.05) (Fig. 1d). FOXO1 deletion in keratinocytes also affected the formation of collagen matrix. On day 4 the amount of collagen matrix produced was reduced by 40% with a similar reduction on day 7 in experimental mice (Fig. 1e), demonstrating the influence of keratinocytes on connective tissue wound healing.
Dermal wounds were created in experimental mice with keratinocyte-specific FOXO1 deletion (FOXO1 deleted in KC) and littermate control (wild-type) mice. (a) FOXO1 expression as measured by fluorescence intensity in epithelium and connective tissue in 4 days post-wounding. (b,c) Representative images of Masson’s trichrome stained dermal wounds 4 and 7 days post-wounding. Bar represents 500 μm left panel and 100 μm right panel. Dotted blue lines represent healing edge of the connective tissue and the distance between them represents the connective tissue gap. Black arrows point to collagen bundles. (d) Area of granulation tissue formation was measured in Masson’s trichrome stained sections for day 4 and day 7 wounds. (e) Newly formed collagen was measured in Masson’s trichrome stained sections for day 4 and day 7 wounds. KC, keratinocytes; EP, epidermis; CT, connective tissue. n = 5–8 mice per group. *p < 0.05 vs. wild type.
Keratinocyte-specific deletion of FOXO1 reduces fibroblast and MSC proliferation
The effect of epithelial-specific FOXO1 deletion on myofibroblast was then assessed in vivo by immunofluorescent staining for α-smooth muscle actin (α-SMA)20 (Fig. 2a). Myofibroblasts play a crucial role in wound contraction during healing. The number of myofibroblasts was reduced 60% by FOXO1 deletion in epithelium (p < 0.05) (Fig. 2b). The impact of epithelial-specific FOXO1 deletion on fibroblast numbers was then assessed in vivo in histologic sections. Day 4 was chosen since it reflects a time point of robust connective tissue formation. Fibroblast density in connective tissue was reduced by 44% (p < 0.05) (Fig. 2b).
Dermal wounds were created in experimental (FOXO1 deleted in KC) and littermate control (wild-type) mice. (a) Representative images of α-SMA immunofluorescence with α-SMA specific antibody on day 4 wounds. (b) α-SMA immunopositive cells representative of myofibroblasts in newly formed connective tissue (left panel). Fibroblasts were identified by their characteristic fusiform-shaped nucleus in hematoxylin and eosin stained sections on 4-day wounds (right panel). (c) CD271 immunopositive cells were identified by immunofluorescence with CD271-secific antibody 4 days after wounding. (d) CD271 and Sca1 immunofluorescence analyses of MSCs in the connective tissue of day 4 wounds. (e) PCNA immunopositive fibroblastic cells were identified by immunofluorescence with PCNA specific antibody and by characteristic appearance of fibroblast nuclei in merged images with DAPI nuclear stain (left panel). Quantification of PCNA and Sca1 double immunopositive cells for analyses of proliferating MSCs (right panel). Scale bar, 100 μm; KC, keratinocytes; EP, epidermis; CT, connective tissue. White dashed lines were the boundary between the epidermis and the dermis. n = 5–8 mice per group, * p < 0.05 vs. wild type. Immunofluorescence was negative when a matched control antibody was used in lieu of specific antibody.
The impact of keratinocyte-specific FOXO1 deletion on cells that express MSC markers in connective tissue was then measured by assessing the number of CD271 and stem cell antigen1 (Sca1) immunopositive cells21. CD271 positive cells were reduced by 55% in keratinocyte-FOXO1 null mice compared to control littermates (p < 0.05) (Fig. 2c,d). Sca1 positive cells were reduced by 60% in keratinocyte-FOXO1 null mice (p < 0.05) (Fig. 2d). The number of proliferating fibroblasts was measured by assessment of proliferating cell nuclear antigen (PCNA) and were reduced by 42% in keratinocyte-FOXO1 null mice compared to littermate control mice (p < 0.05) (Fig. 2e), indicating impaired fibroblast proliferation with epithelial-specific FOXO1 deletion. The number of proliferating Sca-1 immunopositive cells was reduced by 60% in the connective of keratinocyte-FOXO1 null mice compared to littermate control (p < 0.05) (Fig. 2e), indicating that FOXO1 expression in keratinocytes promotes MSCs proliferation.
The effect of keratinocyte-specific FOXO1 deletion on connective tissue formation in vivo is mediated by TGFβ1
Connective tissue growth factor (CTGF also known as CCN2) plays an important role in fibroblast behavior during wound healing22 and is induced by transforming growth factor β1 (TGFβ1)17. Due to the importance of TGFβ123 and CTGF22 in healing we determined whether FOXO1 deletion in epithelial cells affected TGFβ1 and CTGF expression in the epithelium and connective tissue by standardized quantitative immunofluorescence measuring mean fluorescence intensity in each compartment (Fig. 3a). TGFβ1 expression was four-fold higher in the wounded epithelium than connective tissue (Fig. 3b). As expected, expression of TGFβ1 in epithelium was FOXO1 dependent based on our previous results. Surprisingly, TGFβ1 expression in the connective tissue compartment was reduced by ~60% when FOXO1 was absent in epithelia (Fig. 3b), indicating that keratinocytes influence TGFβ1 expression in connective tissue.
(a) CTGF expression was identified with specific CTGF antibody by immunofluorescence in 4-day wounds. Scale bar, 100 μm. (b) TGFβ1 and CTGF expression was identified with a specific antibody to each by immunofluorescence in day 4 wounds. Expression was determined by mean immunofluorescence intensity (MFI). (c) Quantification of new connective tissue formation by Masson’s trichrome stain of day 4 wounds with or without TGFβ1 treatment. (d) qRT-PCR analysis of TGFβ1 and CTGF expression in primary cultures of normal human epidermal keratinocytes (NHEK). (e) TGFβ1 expression was assessed in primary fibroblast cultures with specific antibody by measuring mean immunofluorescence intensity and CTGF by ELISA. Fibroblasts were incubated with keratinocyte-conditioned medium transfected in NHEK cells that had been transfected with FOXO1 siRNA (siFOXO1) or scrambled siRNA (siSCR). (f) TGFβ1 and CTGF immunofluorescence analyses of fibroblasts treated with keratinocyte-conditioned medium with TGFβ1 blocking antibody or matched control IgG. For all immunofluorescence experiments, the images were negative when a matched control antibody was used in lieu of specific antibody. MFI, mean fluorescence intensity; KC, keratinocytes; EP, epidermis; CT, connective tissues. White dashed lines were the boundary between the epidermis and the dermis. n = 5–8 mice per group. *p < 0.05 vs. matched wild type, **p < 0.05 vs. matched vehicle group, #p < 0.05 vs. scrambled siRNA, &p < 0.05 vs. control group, ##p < 0.05 vs. matched IgG control.
In contrast to TGFβ1, CTGF expression in connective tissue following wounding was 2 fold higher than epithelium (Fig. 3b). CTGF levels in the epithelium of keratinocyte-FOXO1 null mice were reduced by 61% in experimental mice. CTGF levels in connective tissue were also dependent upon cutaneous epithelium and were reduced by 66% when FOXO1 was ablated in these cells (Fig. 3b). Thus, keratinocytes have a significant role in regulating the expression of CTGF in connective tissue. The linkage between keratinocyte-specific FOXO1 deletion, TGFβ1 and connective tissue was reinforced by rescue of the deficit in connective tissue formation in experimental mice but not control mice treated with exogenous TGFβ1 (Fig. 3c). Thus, deficiency caused by keratinocyte-specific FOXO1 deletion is overcome by addition of TGFβ1 to wounds while this addition has little effect in normal littermate controls, indicating that keratinocyte-produced TGFβ1 is needed for optimal connective tissue healing.
The mechanisms through which keratinocytes may regulate fibroblasts and MSCs were examined in vitro. FOXO1 silencing in normal human epidermal keratinocytes (NHEK) in vitro significantly reduced TGFβ1 and CTGF mRNA levels (Fig. 3d), consistent with the data obtained from in vivo (Fig. 3b). Moreover, FOXO1 knockdown in keratinocytes reduced the capacity of epithelial cells to stimulate TGFβ1 expression in fibroblasts by almost 40% and CTGF production by 45% (p < 0.05) (Fig. 3e). Keratinocyte stimulation of TGFβ1 and CTGF expression in fibroblasts was blocked by antibody to TGFβ1 (p < 0.05) (Fig. 3f). Thus, keratinocytes stimulate CTGF expression via TGFβ1 that is mediated by FOXO1, consistent with reports that fibroblasts are an important source of CTGF in dermal wound healing22.
Inhibition of TGFβ1 and CTGF blocks fibroblast and MSC proliferation, myofibroblast differentiation, and collagen production in vitro
The role of keratinocytes in regulating fibroblasts and MSCs through FOXO1 was explored further. Keratinocytes stimulated fibroblast and MSC proliferation as determined by BrdU incorporation and this increase was blocked by FOXO1 knockdown (Fig. 4a,b). The increase stimulated by keratinocytes was inhibited by antibody to CTGF (Fig. 4a,b). The combination of TGFβ1 and CTGF blocking antibodies displayed a similar effect as CTGF antibody alone. Since TGFβ1 stimulates fibroblasts in part through CTGF24, the results indicate that CTGF is a critical factor by which keratinocytes regulate fibroblasts through a mechanism mediated by FOXO1.
Primary dermal fibroblasts (a,f) and MSCs (b–e) were incubated with conditioned medium collected from keratinocytes transfected with FOXO1 or scrambled siRNA, then incubated with TGFβ1 and/or CTGF blocking antibody or matched control IgG. (a,b) DNA synthesis of dermal fibroblasts (a) and MSCs (b) were determined by BrdU incorporation. (c) Representative images of α-SMA immunofluorescent staining for MSCs after treated with conditioned medium collected from keratinocytes transfected with FOXO1 or scrambled siRNA. (d) Representative images of α-SMA immunofluorescent staining for MSCs after treated with keratinocyte conditioned medium and TGFβ1 and/or CTGF blocking antibody. Scale bar, 100 μm. (e) The quantification of α-SMA protein expression by immunofluorescence in MSCs. Data is expressed as mean fluorescence intensity (MFI) per number of cells. (f) Collagen I levels in NHDF cells was measured by means of ELISA. * p < 0.05 compared to siSCR, #p < 0.05 vs. matched IgG group.
The effect of FOXO1 in modulating keratinocyte regulated MSC differentiation was also investigated. Keratinocytes induced a 4-fold increase in MSC differentiation to myofibroblasts determined by α-SMA expression and 70% of the induction effect was blocked by FOXO1 knockdown (Fig. 4c,e). Moreover, formation of myofibroblasts was dependent upon the TGFβ1/CTGF axis as it was blocked by antibody specific for CTGF or TGFβ1 (p < 0.05) (Fig. 4d,e). Cutaneous epithelial cells also stimulated a 4-fold increase in collagen I production examined by enzyme-linked immunosorbent assay (ELISA), which was reduced in half by FOXO1 knockdown compared to scrambled siRNA (p < 0.05) and dependent upon TGFβ1 and CTGF (p < 0.05) (Fig. 4f). These studies provide further evidence that FOXO1 regulated TGFβ1 and CTGF production represents an important mechanism through which epithelia modulate connective tissue healing.
Discussion
The cellular and molecular mechanisms that underlie tissue repair are incompletely understood and represent a significant health care issue1. Studies presented here establish for the first time in vivo that cutaneous epithelia play a key role in the repair of wounded connective tissue and that the transcription factor FOXO1 is essential for up-regulating this effect. When FOXO1 is specifically deleted in cutaneous epithelial cells there is a ~40–50% reduction in granulation tissue formation and production of extracellular matrix. This is equivalent to the substantial reduction reported for macrophage deletion and indicates the importance of activating cutaneous epithelial cells to promote connective tissue healing4,5,6.
It has been postulated that interactions between epidermal keratinocytes and dermal fibroblasts contribute to wound healing. The predominant line of thinking has been that cells and growth factors in connective tissue modulate proliferation and migration of keratinocytes to promote re-epithelialization25,26. This is supported by evidence that specific deletion of Rac1 in fibroblasts found in connective tissue in vivo negatively impacts re-epithelialization27. In contrast, less is known about the control of connective tissue by epithelial cells although in vitro studies suggest that factors produced by these cells may affect fibroblast activity15,16. Thus, our in vivo results provide the first direct evidence that keratinocytes modulate the repair of connective tissue and support previous in vitro studies that paracrine factors released by keratinocytes contribute to connective tissue healing28,29.
Mesenchymal cells such as fibroblasts are essential to dermal wound healing. In addition to producing connective tissue, fibroblasts provide signals to enhance repair30. The healing of a wound requires the generation of well-orchestrated signals that stimulate MSCs and fibroblasts31. Within the wound bed, fibroblasts produce collagen and other extracellular matrix constituents. We found that FOXO1 deletion in epithelium led to impaired healing that included decreased formation of new connective tissue. Fibroblast numbers and proliferation were both modulated by keratinocytes in a FOXO1 dependent mechanism, consistent with impaired production of extracellular matrix. In addition keratinocyte-specific FOXO1 deletion led to reduced MSCs numbers and proliferation, which may contribute to reduced myofibroblast numbers seen in our study, and have secondary effects on subsequent healing events32.
TGFβ1 is an important regulator of connective tissue healing stimulating the production of ECM proteins such as collagen and initiating granulation tissue formation33. TGFβ stimulates the expression of CTGF, which is thought to be responsible for many of the pro-fibrotic properties of TGFβ, particularly in the promotion of fibroblast proliferation and ECM production24,34. CTGF plays a physiological role in early wound healing35. Many different cell types including fibroblasts, epithelial cells, endothelial cells, and vascular smooth muscle cells express CTGF36. In wound repair in vivo there is a coordinated up-regulation of TGFβ1 followed by CTGF expression22. We found that TGFβ1 and CTGF expression in both epithelium and connective tissue were decreased by keratinocyte-specific deletion of FOXO1. In vitro knockdown of FOXO1 in keratinocytes also reduced TGFβ1 and CTGF expression in keratinocytes. Antibody blocking studies demonstrated that TGFβ1 produced by keratinocytes up-regulates CTGF in fibroblasts, and FOXO1-deletion in keratinocytes results in deficient connective tissue formation. In addition, we found that keratinocyte could induce MSCs to differentiate into myofibroblasts in vitro, which was blocked by inhibition of TGFβ1 and/or CTGF. This is consistent with the other report that CTGF could direct fibroblast differentiation from human mesenchymal stem/stromal cells in vitro2,34. It is also possible that keratinocyte produced CTGF contributes to connective tissue healing.
In summary, we found that keratinocytes played a significant role in regulating connective tissue healing through a mechanism that was dependent upon FOXO1. Moreover, a key component of this mechanism involved TGFβ1, which is produced in sub-optimal amounts when FOXO1 is deleted in keratinocytes as shown by improved healing when TGFβ1 is applied to experimental mice with keratinocyte-specific FOXO1 deletion but not in normal control littermates. Combined with our previous results17, we propose that FOXO1 activation induces release of TGFβ1 by keratinocytes to stimulate re-epithelialization17 and that TGFβ1 up-regulates CTGF expression to promote connective tissue healing by stimulating fibroblasts, myofibroblasts and cells that express mesenchymal stem cell markers (see Fig. 5). Our finding indicates that keratinocytes are also important as a cellular source of factors that drive connective tissue repair similar to macrophage-derived growth factors that stimulate fibroblast proliferation, myofibroblast differentiation and extracellular matrix synthesis4.
During wound healing FOXO1 binds to the TGFβ1 promoter in keratinocytes to up-regulate TGFβ1 expression, which promotes connective tissue wound healing directly and also induces CTGF production. TGFβ1 and CTGF stimulate differentiation of myofibroblasts and connective tissue formation. In addition, TGFβ1 stimulates keratinocyte migration to enhance re-epithelialization. Leukocytes, particularly macrophages also produce growth factors to enhance healing.
Material and Methods
Animal model
Experiments were approved by the University of Pennsylvania Institutional Animal Care and Use Committee and all methods were performed in accordance with guidelines and regulations relevant to the study. Mice with floxed FOXO1 were generously provided by Ronald A. DePinho (MD Anderson Cancer Center, Houston, TX) as previously described37. Mice expressing Cre recombinase [strain Tg (KRT14-cre)1Amc/J] were obtained from the Jackson Laboratory (Bar Harbor, ME). Keratinocyte-specific FOXO1 deletion was obtained by crossing the above two mentioned mice to generate experimental (FOXO1 deleted in KC) and control littermates. All the experiments were performed with adult mice age of 16–20 weeks.
Skin wounding experiment
Mice were anesthetized by i.p. administration of ketamine (80 mg/kg) and xylazine (5 mg/kg). The scalp was shaved and cleansed with isopropyl alcohol and prepared for surgery. Two excisional wounds were made with a 2mm sterile biopsy punch in the scalp to create a circular full-thickness wounds as described previously38. Wounds were harvested on days 4 and 7 post-wounding. Wound healing process was assessed by quantification of the calibrated digital photographs. The wound areas were calculated from the photographs using Nikon NIS-elements D image analysis software (Nikon). For TGFβ1 treatment experiment, 100ng of recombinant TGFβ1 (PeproTech) in 20 μl of 3% methylcellulose vehicle or vehicle alone was topically applied to the wounds every other day from the time of wounding. In most in vivo experiments, 6–8 mice were examined per group.
H&E staining and Masson’s trichrome staining
The scalp and attached calvarial bone were fixed in 4% paraformaldehyde overnight and decalcified in 10% EDTA solution. Five-micrometer sections were stained using hematoxylin & eosin (H&E) for microscopic evaluation of the epithelial gap and the granulation tissue gap using Nikon NIS-elements image analysis software (Nikon) at the center of each lesion. The connective tissue gap was measured by drawing parallel lines along the edge of the connective tissue adjacent to the open wound and the distance between them was measured. Granulation tissue formation was measured in Masson’s trichrome stained sections as highly cellular tissue which stained red. Recently formed collagen matrix stains light blue compared to dark blue mature collagen matrix. The wound edges were determined by use of histologic landmarks including the position of hair follicles and thickened epithelium and the floor of the wound was bordered by the osseous floor or muscle bundles. Newly formed collagen matrix in the wound area was measured with Nikon NIS-elements image analysis software. Collagen matrix was identified by its characteristic color in Masson’s trichrome stained sections.
Immunofluorescent staining in vivo
Immunofluorescent staining was performed on paraffin sections that were fixed with 4% paraformaldehyde sectioned at 4 microns. After blocking with nonspecific binding blocking buffer (Millipore), tissue sections were incubated with the appropriate primary antibodies followed by biotinylated secondary antibody (Vector Laboratories). Sections were subsequently incubated with avidin-biotin-peroxidase enzyme complex (Vector Laboratories) and followed by tyramide signal amplification (PerkinElmer) to enhance the fluorescent signal. Nuclei were stained with DAPI. Primary antibodies used were Sca1 (Abcam), CD271 (Abcam), CTGF (Santa Cruz Biotechnology Inc.), TGFβ1 (Abcam) and α-SMA (Abcam). Images were captured using a fluorescence microscope (ECLIPSE 90i, Nikon). Images for control antibodies gave negative immunostaining. Nikon NIS Elements AR image analysis software (Nikon) was used to do image analysis. Percent positive cells were calculated from the number of immunopositive cells divided by the number of DAPI positive cells. To examine expression levels mean fluorescence intensity was measured. In each case, a standardized region of interest was defined in each specimen and was of similar size. After the region of interest was selected the exposure time was set to obtain results in the linear range.
Cell culture
Primary normal human epidermal keratinocytes (NHEK) were purchased from Lonza and cultured in KGM-2 growth medium in the presence of human keratinocyte growth supplements (Lonza) and antibiotics (Life Technologies). For experiments, insulin was excluded from the culture media to prevent inactivation of FOXO1. NHDFs were maintained in DMEM medium containing 10% fetal bovine serum (FBS) (Gibco, life technologies), while hBMSCs were maintained in α-MEM medium supplemented with 10% FBS. All the cell cultures were maintained in 5% CO2/air and used between passages P1–P4.
Conditioned Media Experiments
The effect of keratinocyte-conditioned media (CM) on TGFβ1 and CTGF expression in normal human dermal fibroblasts (NHDF) was performed as follows. In cases where CM was collected from transfected cells, conditioned media was collected from transfected keratinocytes 24 hours after transfection with FOXO1 siRNA or scrambled control siRNA (Dharmacon) using GenMute siRNA Transfection Reagent (SignaGen Labs). Cells were incubated with 5–10 nM siRNA with transfection reagent for six hours and transferred to standard medium for 16 hours. Cells were transferred to fresh media for the collection of conditioned media. To assess the impact of conditioned media on fibroblasts or human bone marrow-derived mesenchymal stem cells (hBMSCs) the conditioned media was diluted 1:1 with α-MEM medium supplemented with 1% FBS. Therefore, the fibroblasts were incubated in low-serum (total 0.5% FBS) when stimulated with keratinocyte conditioned media. For antibody neutralization studies, anti-TGFβ139 (10 μg/ml, R&D Systems) or anti-CTGF40 blocking antibody (10 μg/ml, Santa Cruz) was added 1 hour prior to incubation of keratinocyte-conditioned media with NHDF or hBMSCs for 3 days.
Immunofluorescent staining in vitro
Cells were fixed briefly with 10% formalin, permeabilized with 0.5% Triton X-100 and incubated overnight at 4 °C with primary antibodies to TGFβ1 (Abcam), CTGF (Santa Cruz), α-SMA (Abcam) or an equal amount of matched negative control IgG, then with the corresponding secondary antibody. Nuclei were stained with DAPI. Images were captured with a fluorescence microscope (Nikon) and the mean fluorescence intensity (MFI) was determined using NIS-Elements AR software (Nikon).
BrdU incorporation Assay
The effect of keratinocyte-CM on the proliferation of fibroblasts and MSCs were determined with a BrdU Cell Proliferation Assay Kit (Cell Signaling Technology). CM were then collected from keratinocytes transfected with FOXO1 siRNA or scrambled control siRNA, and incubated with fibroblasts or MSCs in 96-well plates with/without TGFβ1 antibody (R&D Systems) or/and CTGF blocking antibody (Santa Cruz) for 72 hours, and incubated with 10 μM BrdU for 6 hours. Cells were fixed with cold methanol and incubated with anti-BrdU antibody. BrdU incorporation was determined by measuring absorbance at 450 nm. Experiments were repeated at least three times.
Real-time PCR
Total RNA was isolated from NHEK cells transfected with FOXO1 or scrambled siRNA using an RNeasy kit (Qiagen). After reverse transcription (Applied Biosystems), real-time quantitative PCR (qPCR) for was performed with Taqman system (Roche Diagnostics). Results were normalized with values obtained from ribosomal protein RPL32, a ribosomal protein. Experiments were repeated three to four times.
ELISA
CTGF levels in media from fibroblasts treated with keratinocyte-CM were determined by measuring the levels of CTGF in media using specific ELISA kit (PeproTech). Collagen levels in fibroblasts were measured as follows. Fibroblasts were first starved in 0.1% FBS for 24 hours, then treated with keratinocyte-CM with/without TGFβ1 (R&D Systems) or/and CTGF blocking antibody (Santa Cruz) for 72 hours. Supernatant was collected to measure collagen by ELISA (Biomatik).
Statistics
All data are reported as the mean ± standard deviation. Group mean values were compared, as appropriate, by Student’s two-tailed t-test or one-way ANOVA with Scheffe’s post-hoc test. A p-value < 0.05 was considered significant.
Additional Information
How to cite this article: Zhang, C. et al. FOXO1 expression in keratinocytes promotes connective tissue healing. Sci. Rep. 7, 42834; doi: 10.1038/srep42834 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Eming, S. A., Martin, P. & Tomic-Canic, M. Wound repair and regeneration: mechanisms, signaling, and translation. Sci Transl Med. 6(265), 265sr6 (2014).
Lee, C. H., Shah, B., Moioli, E. K. & Mao, J. J. CTGF directs fibroblast differentiation from human mesenchymal stem/stromal cells and defines connective tissue healing in a rodent injury model. J Clin Invest. 120, 3340–3349 (2010).
Wynn, T. A. & Ramalingam, T. R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. 18, 1028–1040 (2012).
Koh, T. J. & DiPietro, L. A. Inflammation and wound healing: the role of the macrophage. Expert Rev Mol Med. 13, e23 (2011).
Forbes, S. J. & Rosenthal, N. Preparing the ground for tissue regeneration: from mechanism to therapy. Nat Med. 20, 857–869 (2014).
Martins-Green, M., Petreaca, M. & Wang, L. Chemokines and their receptors are key players in the orchestra that regulates wound healing. Adv Wound Care (New Rochelle). 2, 327–347 (2013).
Novak, M. L. & Koh, T. J. Phenotypic transitions of macrophages orchestrate tissue repair. Am J Pathol. 183, 1352–1363 (2013).
Cowin, A. J., Brosnan, M. P., Holmes, T. M. & Ferguson, M. W. Endogenous inflammatory response to dermal wound healing in the fetal and adult mouse. Dev Dyn. 212, 385–393 (1998).
Martin, P. et al. Wound healing in the PU.1 null mouse–tissue repair is not dependent on inflammatory cells. Curr Biol. 13, 1122–1128 (2003).
Werner, S., Krieg, T. & Smola, H. Keratinocyte-fibroblast interactions in wound healing. J Invest Dermatol. 127, 998–1008 (2007).
Smola, H., Thiekotter, G. & Fusenig, N. E. Mutual induction of growth factor gene expression by epidermal-dermal cell interaction. J Cell Biol. 122, 417–429 (1993).
Szabowski, A. et al. c-Jun and JunB antagonistically control cytokine-regulated mesenchymal-epidermal interaction in skin. Cell. 103, 745–755 (2000).
Kim, W. S. et al. Wound healing effect of adipose-derived stem cells: a critical role of secretory factors on human dermal fibroblasts. J Dermatol Sci. 48, 15–24 (2007).
Kao, H. K. et al. Peripheral blood fibrocytes: enhancement of wound healing by cell proliferation, re-epithelialization, contraction, and angiogenesis. Ann Surg. 254, 1066–1074 (2011).
Fleischmajer, R., MacDonald, E. N., Contard, P. & Perlish, J. S. Immunochemistry of a keratinocyte-fibroblast co-culture model for reconstruction of human skin. J Histochem Cytochem. 41, 1359–1366 (1993).
Shephard, P. et al. Myofibroblast differentiation is induced in keratinocyte-fibroblast co-cultures and is antagonistically regulated by endogenous transforming growth factor-beta and interleukin-1. Am J Pathol. 164, 2055–2066 (2004).
Ponugoti, B. et al. FOXO1 promotes wound healing through the up-regulation of TGF-beta1 and prevention of oxidative stress. J Cell Biol. 203, 327–343 (2013).
Lettieri, B. D., Aquilano, K. & Ciriolo, M. R. FoxO1 at the nexus between fat catabolism and longevity pathways. Biochim Biophys Acta. 1841, 1555–1560 (2014).
Housley, M. P. et al. O-GlcNAc regulates FoxO activation in response to glucose. J Biol Chem. 283, 16283–16292 (2008).
Rao, K. B., Malathi, N., Narashiman, S. & Rajan, S. T. Evaluation of myofibroblasts by expression of alpha smooth muscle actin: a marker in fibrosis, dysplasia and carcinoma. J Clin Diagn Res. 8, ZC14–17 (2014).
Busser, H. et al. Isolation and Characterization of Human Mesenchymal Stromal Cell Subpopulations: Comparison of Bone Marrow and Adipose Tissue. Stem Cells Dev. 24, 2142–2157 (2015).
Igarashi, A., Okochi, H., Bradham, D. M. & Grotendorst, G. R. Regulation of connective tissue growth factor gene expression in human skin fibroblasts and during wound repair. Mol Biol Cell. 4, 637–645 (1993).
Pakyari, M., Farrokhi, A., Maharlooei, M. K. & Ghahary, A. critical role of transforming growth factor beta in different phases of wound healing. Adv Wound Care (New Rochelle). 2, 215–224 (2013).
Leask, A., Holmes, A., Black, C. M. & Abraham, D. J. Connective tissue growth factor gene regulation. Requirements for its induction by transforming growth factor-beta 2 in fibroblasts. J Biol Chem. 278, 13008–13015 (2003).
Geer, D. J., Swartz, D. D. & Andreadis, S. T. Biomimetic delivery of keratinocyte growth factor upon cellular demand for accelerated wound healing in vitro and in vivo . Am J Pathol. 167, 1575–1586 (2005).
El, G. A. et al. Fibroblasts facilitate re-epithelialization in wounded human skin equivalents. Lab Invest. 84, 102–112 (2004).
Liu, S., Kapoor, M. & Leask, A. Rac1 expression by fibroblasts is required for tissue repair in vivo . Am J Pathol. 174, 1847–1856 (2009).
Lacroix, M., Bovy, T., Nusgens, B. V. & Lapiere, C. M. Keratinocytes modulate the biosynthetic phenotype of dermal fibroblasts at a pretranslational level in a human skin equivalent. Arch Dermatol Res. 287, 659–664 (1995).
Walter, M. N. et al. Mesenchymal stem cell-conditioned medium accelerates skin wound healing: an in vitro study of fibroblast and keratinocyte scratch assays. Exp Cell Re. 316, 1271–1281 (2010).
Abraham, D. J., Eckes, B., Rajkumar, V. & Krieg, T. New developments in fibroblast and myofibroblast biology: implications for fibrosis and scleroderma. Curr Rheumatol Rep. 9, 136–143 (2007).
Reinke, J. M. & Sorg, H. Wound repair and regeneration. Eur Surg Res. 49, 35–43 (2012).
Proffen, B. L., Haslauer, C. M., Harris, C. E. & Murray, M. M. Mesenchymal stem cells from the retropatellar fat pad and peripheral blood stimulate ACL fibroblast migration, proliferation, and collagen gene expression. Connect Tissue Res. 54, 14–21 (2013).
Barrientos, S. et al. Growth factors and cytokines in wound healing. Wound Repair Regen. 16, 585–601 (2008).
Tong, Z., Sant, S., Khademhosseini, A. & Jia, X. Controlling the fibroblastic differentiation of mesenchymal stem cells via the combination of fibrous scaffolds and connective tissue growth factor. Tissue Eng Part A. 17, 2773–2785 (2011).
Alfaro, M. P. et al. A physiological role for connective tissue growth factor in early wound healing. Lab Inves 93, 81–95 (2013).
Moussad, E. E. & Brigstock, D. R., Connective tissue growth factor: what’s in a name? Mol Genet Metab. 71, 276–292 (2000).
Paik, J. H. et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell. 128, 309–323 (2007).
Siqueira, M. F. et al. Impaired wound healing in mouse models of diabetes is mediated by TNF-alpha dysregulation and associated with enhanced activation of forkhead box O1 (FOXO1). Diabetologia. 53, 378–388 (2010).
Sommer, A. & Fabri, M. Vitamin D regulates cytokine patterns secreted by dendritic cells to promote differentiation of IL-22-producing T cells. PLoS One. 10, e0130395 (2015).
Duncan, M. R. et al. Connective tissue growth factor mediates transforming growth factor beta-induced collagen synthesis: down-regulation by cAMP. FASEB J. 13, 1774–1786 (1999).
Acknowledgements
This work was supported by a grant R01DE019108 and R01 AR060055 from NIAMS from the NIDCR. We are particularly grateful to Dr. R.A. DePinho for providing the floxed FOXO1 mice. We thank Youde Liang for help in preparing images for several figures.
Author information
Authors and Affiliations
Contributions
D.G. designed the experiments, C.Z., J.L., J.L., B.P., C.T., S.A. and R.M. conducted the experiments and analyzed the results, C.Z. and D.G. prepared the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, C., Lim, J., Liu, J. et al. FOXO1 expression in keratinocytes promotes connective tissue healing. Sci Rep 7, 42834 (2017). https://doi.org/10.1038/srep42834
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep42834
- Springer Nature Limited
This article is cited by
-
Supraphysiological testosterone supplementation improves granulation tissue maturation through angiogenesis in the early phase of a cutaneous wound healing model in rats
Inflammation Research (2022)
-
FOXO1 deletion in keratinocytes improves diabetic wound healing through MMP9 regulation
Scientific Reports (2017)