Abstract
To find the genetic markers related to the antipsychotic-induced weight gain (AIWG), we analyzed associations among candidate gene single-nucleotide polymorphisms (SNPs) and quantitative traits of weight changes and lipid profiles in a Chinese Han population. A total of 339 schizophrenic patients, including 86 first-episode patients (FEPs), meeting the entry criteria were collected. All patients received atypical antipsychotic drug monotherapy and hospitalization and were followed for 12 weeks. Forty-three SNPs in 23 candidate genes were calculated for quantitative genetic association with AIWG, performed by PLINK. The TOX gene SNP rs11777927 (P = 0.009) and the ADIPOQ gene SNP rs182052 (P = 0.019) were associated with AIWG (in body mass index, BMI). In addition, the BDNF SNP rs6265 (P = 0.002), BDAF SNP rs11030104 SNP (P = 0.001), and ADIPOQ SNPs rs822396 (P = 0.003) were significantly associated with the change of waist-to-hip ratio (WHR) induced by atypical antipsychotics. These results were still significant after age and gender adjustments. These findings provide preliminary evidence supporting the role of TOX, ADIPOQ and BDNF in weight and WHR gain induced by atypical antipsychotics.
Similar content being viewed by others
Introduction
Schizophrenia is a complex and devastating psychiatric disorder that occurs worldwide, and for decades it was generally thought to have a uniform lifetime risk of approximately 1% across time, geography and sex1. Obesity and overweight affect 30–70% of patients with schizophrenia spectrum disorders. They have a 2.8–3.5 increased likelihood of being obese2. One of the many reasons that psychiatric patients gain weight and become overweight and obese is atypical antipsychotic drugs (AAPDs) which may affect the recovery process and quality of life3,4.
AAPDs have become the first-line medication treatment for patients with schizophrenic and schizoaffective disorders. Although AAPDs appear to be a better choice for the treatment of schizophrenic patients, they are often associated with some common and debilitating side effects, such as accelerated weight gain, insulin resistance, diabetes, hyperlipidemia, and coronary heart disease5,6,7. These metabolic alterations can develop as quickly as six months after the initiation of pharmacotherapy8. Antipsychotic-induced weight gain (AIWG) is quite commonly associated with AAPDs; more-so than typical antipsychotic drugs (TAPDs)9.
Although the phenomenon of antipsychotic-induced weight gain (AIWG) is well recognized, some individuals actually lose weight or their weight remains unchanged during single AAPD treatment, which shows that not all individuals are equally prone to this adverse side effect10. Although many variables contribute to the heterogeneity of AIWG, genetic factors may plan a significant role. Twin and sibling genetic studies have demonstrated similar degrees in weight gain profiles upon receiving AAPD11,12, and the heritability (h2) is 0.6–0.813.
With the rapid development of molecular genetics, numerous pharmacogenetics studies have focused on identifying specific gene variants contributing to AIWG. Most studies have been based on the candidate gene approach, such as HTR2C14, MC4R15, Leptin16, FTO17 and BDNF18. So far, three genome-wide association studies (GWAS) of AIWG were carried out, associations were found on pro-melanin-concentrating (PMCH) and MC4R gene polymorphisms19,20. A recent GWAS for AIWG found that PTPRD polymorphisms might modulate AIWG21. Replications of these findings need to be carried out in independent cohorts. Compared with the GWAS design that requires very large sample sizes to achieve sufficient power, the candidate gene approach allowed us to perform an association study with increased statistical power for prioritized genes22.
Previous studies are mainly categorical correlation analyses. Patients with clinically significant weight gain (7% increases from baseline) were in the case group, and the others were controls. With such a research design, it is difficult to dynamically observe the quantitative traits related to AIWG. In this study, we aimed to find AIWG candidate genes in Han Chinese schizophrenia patients by studying quantitative associations between single-nucleotide polymorphisms (SNPs) of candidate genes and AIWG and other antipsychotic-treatment-related phenotypes.
Results
Sample demographics
Demographic characteristics of the 339 individuals, including 86 first-episode patients (FEPs), from this study are provided in Table 1. Patients received 12 weeks of treatment with single AAPDs: olanzapine (OLZ), risperidone (RIS), clozapine, quetiapine, aripiprazole, or ziprasidone.
After 12-week AAPD intervention, the average weight gain was 4.21 kg in all patients. The 12-week BMI change in our subjects (ΔBMI12w) was 1.52 ± 1.51 kg/m2, ranged from −0.78–8.36 kg/m2. The 12-week change in waist-to-hip ratio (ΔWHR12w) was 0.04 ± 0.05, ranged from −0.15–0.18. The definition of a weight gain “case” was that a patient who gained 7% or more of baseline body weight in a short-term trial23. The percentage of all patients meeting this criterion was 41%; 57% in OLZ group; and 24% in the RIS group. OLZ-treated patients demonstrated a markedly different distribution, with the majority of patients experiencing extreme AIWG. RIS treatment showed moderate effects on weight gain (Supplemental Figure 1).
Quantitative association analysis
After HWE tests, SNP rs1526167 failed. In our candidate association study, the TOX SNP rs11777927 (P = 0.009) and the ADIPOQ rs182052 (P = 0.019) were significantly associated with AIWG. The CDKN2A/B SNPs rs3731245 (P = 0.04) and rs2811708 SNP (P = 0.039) were also associated with the AIWG; CDKN2A/B associations with AIWG remained significant in the FEP and RIS groups. The BDNF SNP rs6265 (P = 0.002), BDAF SNP rs11030104 SNP (P = 0.001), and ADIPOQ SNPs rs822396 (P = 0.003) and rs1501299 (P = 0.040) were associated with the change of WHR; consistent results were found for those SNPs in the OLZ group. These results were still significant after age adjustments (Table 2). The BDNF SNP rs6265 and the BDAF SNP rs11030104 associations remained significant after Bonferroni corrections.
Changes of lipid profiles (high- and low-density lipoprotein and total cholesterol) after AAPD treatment were linked to a PKHD1 gene SNP rs9395706. The CDKN2A/B gene SNP rs10811661 was associated with the FPG changes induced by OLZ (P = 0.009). Results remained significant after age adjustments (Supplemental Table 1).
Gene-gene interaction analysis
Gene × gene interaction analyses showed significant epistasis between MTHFR and PCAF, EPB41L4A and LEPR, ADIPOQ and CDKN2A/B, ADIPOQ and NRXN3, TOX and PKHD1, TOX and RPTOR, and MC4R and COMT gene SNPs for AIWG (ΔBMI) in all patients (Table 3).
Genotypic association analysis
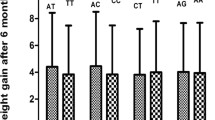
Compared with patients that carried the TOX rs11777927 T allele, the BMI of the patients with the TOX rs11777927 AA genotype increased significantly after AAPD treatment (P < 0.05); compared with patients that carried ADIPOQ rs182052 genotypes GG and AG, BMI of patients with the AA genotype increased significantly (P < 0.05) (Fig. 1).
(a) ΔBMI at 12 weeks of TOX rs11777927 genotypes (AA, AT, TT) in all patients and by treatment group: olanzapine (OLZ) and risperidone (RIS). ΔBMI was compared across the three genotypes in each group (*P < 0.05, one-way ANOVA). (b) ΔBMI over a 12-week single AAPD treatment by TOX rs11777927 genotype (AA, AT, TT) in all patients. BMI was measured at baseline and at weeks 2, 4, 6, 8, and 12. ΔBMI was compared across the three genotypes at each time point (*P < 0.05, **P < 0.01, one-way-ANOVA). (c) ΔBMI at 12 weeks of ADIPOQ rs182052 genotypes (GG, AG, AA) in all patients and by treatment group: OLZ and RIS. ΔBMI was compared across the three genotypes in each group (*P < 0.05, one-way-ANOVA). (d) ΔBMI over a 12-week single AAPD treatment by ADIPOQ rs182052 genotype (GG, AG, AA) in all patients. BMI was measured at baseline and at weeks 2, 4, 6, 8, and 12. ΔBMI was compared across the three genotypes at each time point (*P < 0.05, one-way-ANOVA).
Compared with patients that carried CDKN2A/B rs3731245 genotypes CC and CT, BMI of patients with the TT genotype increased significantly (P < 0.05) (Supplemental Figure 2); compared with the patients that carried the CDKN2A/B rs2811708 G allele, the BMI of the patients with the CDKN2A/B rs2811708 TT genotype increased significantly after the treatment of AAPD (P < 0.05) (Supplemental Figure 3).
In binary association studies for BMI changes, the TOX gene SNP rs11777927 (P = 0.004), the ADIPOQ gene SNP rs1501299 (P = 0.016), the CDKN2A/B gene SNP rs3731245 (P = 0.032), rs2811708 (P = 0.017), the MC4R gene SNP rs6567160 (P = 0.016), and the NRXN3 gene SNP rs12891144 (P = 0.023) were associated with AIWG.
Discussion
With the development of molecular genetics during the past decade, intensive research has examined the influence of genetic variations on AIWG. However, as yet no genetic tests for AIWG are endorsed for clinical application. Although there are significant findings for AIWG associations with many other genes, the most consistently replicated findings are with HTR2C, MC4R, and leptin genes24,25. A recent meta-analysis reported that 11 SNPs from 8 genes were associated with weight or BMI change, and 4 SNPs from 2 genes were significantly related to categorical weight or BMI increase14. Many previous studies have not been confirmed because of varying criteria, differences in frequencies across different populations, or poor statistical power. In addition, compared with the candidate-gene-based approach, GWAS of AIWG are relatively limited19,20,21. Genes PTPRD, MC4R, and PMCH were discovered having the strongest associations with AIWG.
AIWG is thought to be multifactorial and polygenic, which may be different from simple obesity per se. In recent years research has pointed to a broader array of genes and pathways hypothesized to underpin AIWG. Attention has turned to genes and pathways harboring variants that might make the risk of energy unbalance increased by the influence of antipsychotics, leading to AIWG26. Thus, seeking AIWG genes in Han Chinese would promote the development of predictive genetic tests and help us better understand and guide treatment.
In this study, we analyzed 43 SNPs in 23 candidate genes for quantitative association with AIWG for the first time. Here, we decided to use the candidate gene approach rather than performing a GWAS to select a small set of variants for a more focused study, providing sufficient power for these selected variants given the small sample size. As candidate genes, we chose (a) genes mainly from a previous GWAS for Caucasian obesity-AIWG subjects and (b) genes associated with simple obesity, type 2 diabetes, microvascular complications of diabetes, and AIWG.
In the present study, SNPs in TOX and ADIPOQ yielded the most significant associations for AIWG, and CDKN2A/B showed light to strong associations. We did not replicate associations with several well-known AIWG genes, including FTO, MTHFR, and COMT, in our Han Chinese population. Potential reasons for this include genetic heterogeneity, less covered genes (i.e., too few SNPs were genotyped for certain candidate genes), entry criteria, and the relatively small sample size. Although we were unable to test genome-wide association for all genes, the spectrum of AIWG-associated genes differed between Han Chinese in our study and European populations in the previous study15,16,17,27.
The TOX quantitative association with AIWG was first discovered in this study. TOX (thymocyte selection-associated HMG box) is a member of a novel gene family and encodes a novel nuclear DNA-binding protein belonging to a large superfamily of HMG (high-mobility-group) proteins28,29. TOX may play a role in regulating expression of genes involved in cell cycle progression, such as the cell division cycle gene and oncogenes30. In recent years, emerging studies have found that TOX is aberrantly expressed or mutated in various diseases, such as leukemia29 or cardiovascular diseases31. A previous study by our group first found the TOX gene association with type 2 diabetes32. In the present study, we tested 3 SNPs of the TOX gene, rs1526167, rs2726557, and rs11777927 (Supplemental Figure 4). Unfortunately, the SNP rs1526167, which was located in a separate haplotype block, not in linkage disequilibrium with SNPs in the TOX gene coding region and introns, failed the HWE test.
Our results showed that the TOX SNP rs11777927 was significantly associated with AIWG (P = 0.009) and that the A allele contributed to the increased risk for AIWG. And gene × gene interaction analyses showed significant epistasis between TOX and PKHD1 and between TOX and RPTOR for AIWG (ΔBMI) in all patients. We found some associations of TOX SNPs with obesity and metabolic-syndrome-related phenotypes. It is stated by Cox et al. in a published US patent application (US 2006/0177847 A1, August 10, 2006), that they found the TOX polymorphism and other 27 DNA sequence variations to be related to OLZ-treatment-emergent weight gain and “metabolic syndrome” in a 1.7 million SNP genome association study.
However, no rs11777927 association has been reported for AIWG. To the best of our knowledge, only one association study of rs11777927 and disease—intracranial aneurysm— has been reported33. The biological connections between TOX and AIWG are poorly understood, perhaps owing to the inflammatory responses mediated by immune cells, developmentally regulated by TOX gene, contributing to AIWG.
The ADIPOQ locus has been shown to be the only major gene for plasma adiponectin, which is exclusively expressed in adipose tissue34. The ADIPOQ SNP rs1501299 has been reported to be associated with the risk of obesity35,36 and cardiovascular diseases37,38,39. Four candidate SNPs in ADIPOQ in this study were selected from four different haplotype blocks: rs182052, rs822396, rs7649121, and rs1501299 (Supplemental Figure 5). The results showed that the ADIPOQ SNP rs182052 (P = 0.019) was significantly associated with AIWG, and that the ADIPOQ SNPs rs822396 (P = 0.003) and rs1501299 (P = 0.040) were associated with changes in WHR.
Other studies have not delivered consistent findings for ADIPOQ and AIWG. An association of rs1501299 with significant weight gain (>7% of the baseline weight) was reported in Chinese patients40. Another study reported genotypic or allelic association of 6 ADIPOQ variants with AIWG in European patients41. However, the latest two studies did not support a major role of ADIPOQ rs1501299 in the regulation of AIWG42,43, and the association of rs1501299 with AIWG was not present in Japanese patients44. The SNP rs1501299 was a hot spot that had attracted many researchers’ interest. Interestingly, our results showed rs1501299 was associated with changes in WHR but not in AIWG, and that rs182052 and rs822396 were significantly associated with AIWG and changes in WHR induced by AAPD, which haven’t previously been reported. ADIPOQ is most likely closely related to AIWG, which needs further research.
BDNF can encode the BDNF precursor protein located in the chromosome 11p13 region45. And BDNF crossing the blood-brain barrier is the most abundant neurotrophin that modulates synaptic transmission and neuroplasticity in the central nervous system46,47. Previous researches suggested that the BDNF rs6265 was associated with many eating disorders48,49. In addition, several SNPs in the BDNF and BDAF were strongly associated with obesity in GWAS studies50,51.
In the field of the genetics of AIWG, the recent two studies showed BDNF rs6265 was associated with the increased BMI in the psychiatric patients receiving AAPD18,52. Moreover, the BDNF haplotypes including SNP rs6265 were associated with AIWG53,54. However, a study reported that BDNF rs6265 was not associated with AIWG performed by Tsai et al.55. In the present study, we failed to find the association between BDNF rs6265 and AIWG, which was consisted with the results of Tsai’s. Interestingly, we found the BDNF SNP rs6265 (P = 0.002) and BDAF SNP rs11030104 SNP (P = 0.001) were significantly associated with the change of WHR induced by AAPD. The WHR is a alternative measure which have been found to be superior to BMI to reflect abdominal obesity in the World Health Organization (WHO) guidelines56.
All the above findings, including ours, indicate that the SNPs of BDNF have a significant impact on the obesity schizophrenic patients induced by AAPD. The associated genetic markers from BDNF may have different effects, and accumulated mutations may provide a whole contribution to the obesity induced by AAPD. The SNP BDNF rs6265 may play an important role in this process. Hence, the following study about the function of the genetic variants will be necessary to elucidate the mechanism how the genetic variants in BDNF medicate signaling pathway and lead to obesity.
CDKN2A/B is located in the chromosome 9p21 region, has been highlighted as the strongest genetic susceptibility locus for cardiovascular disease57,58 and type 2 diabetes58,59. CDKN2A/B encodes the CDK inhibitor proteins involved in cell cycle regulation, aging, senescence, and apoptosis. In the present study, we selected 3 SNPs of CDKN2A/B: rs3731245, rs2811708 and rs10811661 (Supplemental Figure 6); rs3731245 and rs2811708 are in a haplotype block. SNPs rs3731245 (P = 0.04) and rs2811708 SNP (P = 0.039) were associated with the AIWG but with low statistical power. We found no association between rs10811661 and AIWG, but this SNP was associated with the FPG changes that induced by OLZ (P = 0.009). These results suggest that glucose and lipid metabolism abnormalities and weight gain are influenced by AAPD through different pathways. Previous studies have found that AAPD may not affect glucose and lipid metabolism directly through weight gain10,60. The mechanism by which the CDKN2A/B gene affects susceptibility for AIWG remains to be investigated.
In the future, we will further substantiate our gene results and explore more genetic factors underlying AIWG. However, the heterogeneity of medications and psychiatric disorder factors still exist. We analyzed the genetic association in FEPs, OLZ-treated patients, and RIS-treated patients, but the subgroups were too small for strong statistical power. For this reason, a better designed trial with different AAPDs in larger samples and different populations should also be implemented to validate the genes associated with AIWG.
Given the sample size of our study, we have moderate power to detect major AIWG associations. We performed power calculations by GPC (Genetic Power Calculator61), given a total QTL (quantitative trait locus) variance = 0.05, QTL increaser allele frequency = 0.25, marker allele frequency = 0.25, dominant model, total LD (linkage disequilibrium, D’ = 0.95), case trait threshold (0–4 standard deviations), control threshold (0 to −1.5), type I error α = 0.05, we have more than 80% (84%) power.
We also performed binary association studies for AIWG, the discrete analyses showed similar associations with quantitative BMI changes. Replication is essential for association studies, although GWASs for AIWG were relatively limited. We compared our associations with published GWAS for body weight related traits (GWASdb, v2. http://jjwanglab.org/gwasdb), ADIPOQ (rs182052), BDNF (rs6265), and BDAF (rs11030104) gene SNPs all yielded genome-wide associations.
In our study, we used quantitative associations between SNPs of candidate genes and AIWG and other antipsychotic-related phenotypes to find AIWG candidate genes in Han Chinese schizophrenia patients. We measured not only weight but also the WHR and lipid and glycemic profiles, which are a common index of metabolic syndrome. In addition, for the purpose of excluding environmental factors that may affected AIWG62, we selected a sample of inpatients with lifestyle, exercise, and diet under unified management by the hospital in order to reduce the heterogeneity. Our comprehensive study of TOX and ADIPOQ is an important contribution to understanding the biology of AIWG.
Patients and Materials
Study participants and design
A total of 339 patients hospitalized with schizophrenia or schizoaffective disorders were recruited. All the subjects were unrelated Han Chinese collected from the Tianjin Metal Health Centre, which is Asia’s largest independent psychiatric hospital. The inclusion criteria for this study were as follows: (a) Clinical diagnoses were independently confirmed by two psychiatrists according to the Diagnostic and Statistical Manual of Mental Disorders, fourth edition, text revision. (b) All patients were first-episode drug-naive patients or without any antipsychotic drugs at least 4 weeks before enrollment. (c) The patients were physically healthy with normal hematological and biochemical parameters. (d) All patients were 18–60 years old. Subjects with neurological disorders, eating disorders, and thyroid diseases were excluded from the study. Clinical characteristics of patients were shown in Table 1. All subjects provided written informed consent prior for this study, and the protocol was approved by the Committee on Studies Involving Human Beings at Tianjin Medical University. All experiments were performed in accordance with relevant guidelines and regulations.
All patients received single AAPD intervention on the basis of clinical treatment need. Only trihexyphenidyl for extrapyramidal symptoms and lorazepam for insomnia or agitation were allowed as needed during the study period as concomitant medications.
Patients were followed up during the 12-week treatment course. All patients were measured for body weight and for waist, abdominal, and hip circumferences at baseline and at weeks 2, 4, 6, 8 and 12 after treatment initiation. Body mass index (BMI) and waist-to-hip ratio (WHR) were calculated. Before and after treatment for 4, 8, and 12 weeks, triglyceride, high- and low-density lipoprotein, total cholesterol, total protein, albumin, fasting plasma glucose (FPG), blood creatinine, urea nitrogen (urea), urea/creatinine ratio, and serum C reactive protein were measured for all patients.
Candidate gene and SNP selection
Candidate genes mainly came from a comparative GWAS for Caucasian obesity-AIWG subjects performed by our project team members and colleagues. Genes associated with simple obesity, type 2 diabetes, or microvascular complications of diabetes in our previous studies were also included32,63, in a GWAS of simple obesity, which was the largest sample size by far (340,000 persons)51, and in other previous candidate gene associations for AIWG that gave inconsistent or controversial results15,16,17,18,27. Forty-three (43) SNPs in 23 candidate genes were selected in our study (Table 4).
For some genes, tagging SNPs were selected using the HapMap database (phase2 + phase3, release #28, CEU population, Build36; www.hapmap.org) and Tagger in Haploview64. Minor allele frequencies of Han Chinese were taken from dbSNP (http://www.ncbi.nlm.nih.gov/snp/). For previously reported associations, we selected SNPs with the most significant association rather than genotyping the whole gene. For less studied genes, multiple SNPs were chosen based on the LD pattern of the gene (r2 > 0.8).
Genotyping and quality control
Genomic DNA samples were extracted from 5 ml of peripheral whole blood samples using the high-salt method. Samples were stored and processed by the Center for Molecular and Population Genetics at Tianjin Medical University. Genotyping was performed by primer extension of multiplex products with detection by matrix-assisted laser desorption time-of-flight mass spectrometry. For quality control, 10% of the sample was randomly re-genotyped, with a 100% concordant rate. All genotyping was done blind to knowledge of subjects’ clinical data.
Statistical analysis
The Hardy-Weinberg equilibrium (HWE) test was carried out before the association analysis (Table 4). All phenotypes were documented in a Filemaker Pro database. Statistical analyses for phenotypes were performed by SPSS software (version 20.0). We tested for recessive effects for three significant SNPs by comparing the three genotypes or different subgroups using ANOVA, with change in BMI across the 12-week trial as the dependent measure. The level of statistical significance for the above tests was set a priori at P < 0.05.
The linear regression model in the PLINK65 was used to test the association between the chosen SNPs and several phenotypes, where the changes of weight, BMI, WHR, lipid and glycemic profiles were used as phenotypes for the quantitative trait locus analyses. Linear regressions were performed for each quantitative trait against age within sexes, standardized residuals were saved to make mean = 0 and standard deviation = 1 for each phenotype. Outliers (more than 4 standard deviations) were deleted from this study. As 43 SNPs in 23 candidate genes were analyzed, we used Bonferroni corrections for multiple testing, and the nominal P-value should be at least 0.002 to be significant after multiple test corrections.
Pairwise gene-gene interaction analyses (epistasis) were carried out by PLINK65 among candidate gene SNPs in all patients, the FEP group, the OLZ treatment group, and the RIS treatment group, Bonferroni corrections were also employed for multiple testing.
In addition, we also performed a discrete association study for AIWG by PLINK. A weight gain “case” was defined as a patient who gained 7% or more of his or her baseline body weight in a 12-week trial.
Conclusions
In Conclusion, our findings suggest the role of TOX, ADIPOQ and BDNF in weight and WHR gain induced by atypical antipsychotics in schizophrenia subjects. This study provides a better understanding of genetic factors predisposing individuals to AAPD-induced obesity, dyslipidemia, and abnormal glucose metabolism that may help guide clinical medication intervention and also reveal the pathogenesis of AIWG.
Additional Information
How to cite this article: Li, S. et al. TOX and ADIPOQ Gene Polymorphisms Are Associated with Antipsychotic-Induced Weight Gain in Han Chinese. Sci. Rep. 7, 45203; doi: 10.1038/srep45203 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Owen, M. J., Sawa, A. & Mortensen, P. B. Schizophrenia. Lancet 388, 86–97, doi: 10.1016/S0140-6736(15)01121-6 (2016).
Coodin, S. Body mass index in persons with schizophrenia. Canadian journal of psychiatry. Revue canadienne de psychiatrie 46, 549–555 (2001).
Lett, T. A. et al. Pharmacogenetics of antipsychotic-induced weight gain: review and clinical implications. Molecular psychiatry 17, 242–266, doi: 10.1038/mp.2011.109 (2012).
Mas, S., Llerena, A., Saiz, J., Bernardo, M. & Lafuente, A. Strengths and weaknesses of pharmacogenetic studies of antipsychotic drugs: the potential value of the PEPs study. Pharmacogenomics 13, 1773–1782, doi: 10.2217/pgs.12.159 (2012).
De Hert, M., Dobbelaere, M., Sheridan, E. M., Cohen, D. & Correll, C. U. Metabolic and endocrine adverse effects of second-generation antipsychotics in children and adolescents: A systematic review of randomized, placebo controlled trials and guidelines for clinical practice. European psychiatry: the journal of the Association of European Psychiatrists 26, 144–158, doi: 10.1016/j.eurpsy.2010.09.011 (2011).
Rojo, L. E. et al. Metabolic syndrome and obesity among users of second generation antipsychotics: A global challenge for modern psychopharmacology. Pharmacological research 101, 74–85, doi: 10.1016/j.phrs.2015.07.022 (2015).
Manu, P. et al. Weight gain and obesity in schizophrenia: epidemiology, pathobiology, and management. Acta psychiatrica Scandinavica 132, 97–108, doi: 10.1111/acps.12445 (2015).
Kelly, D. L. et al. Cardiovascular disease mortality in patients with chronic schizophrenia treated with clozapine: a retrospective cohort study. The Journal of clinical psychiatry 71, 304–311, doi: 10.4088/JCP.08m04718yel (2010).
Datta, S. S., Kumar, A., Wright, S. D., Furtado, V. A. & Russell, P. S. Evidence base for using atypical antipsychotics for psychosis in adolescents. Schizophrenia bulletin 40, 252–254, doi: 10.1093/schbul/sbt196 (2014).
Correll, C. U. et al. Cardiometabolic risk of second-generation antipsychotic medications during first-time use in children and adolescents. Jama 302, 1765–1773, doi: 10.1001/jama.2009.1549 (2009).
Theisen, F. M. et al. Clozapine-induced weight gain: a study in monozygotic twins and same-sex sib pairs. Psychiatric genetics 15, 285–289 (2005).
Wehmeier, P. M. et al. Clozapine: weight gain in a pair of monozygotic twins concordant for schizophrenia and mild mental retardation. Psychiatry research 133, 273–276, doi: 10.1016/j.psychres.2004.02.018 (2005).
Gebhardt, S. et al. Body weight gain induced by atypical antipsychotics: an extension of the monozygotic twin and sib pair study. Journal of clinical pharmacy and therapeutics 35, 207–211, doi: 10.1111/j.1365-2710.2009.01084.x (2010).
Zhang, J. P. et al. Pharmacogenetic Associations of Antipsychotic Drug-Related Weight Gain: A Systematic Review and Meta-analysis. Schizophrenia bulletin, doi: 10.1093/schbul/sbw058 (2016).
Czerwensky, F., Leucht, S. & Steimer, W. MC4R rs489693: a clinical risk factor for second generation antipsychotic-related weight gain? The international journal of neuropsychopharmacology/official scientific journal of the Collegium Internationale Neuropsychopharmacologicum 16, 2103–2109, doi: 10.1017/S1461145713000849 (2013).
Shen, J., Ge, W., Zhang, J., Zhu, H. J. & Fang, Y. Leptin −2548g/a gene polymorphism in association with antipsychotic-induced weight gain: a meta-analysis study. Psychiatria Danubina 26, 145–151 (2014).
Song, X. et al. Fat-mass and obesity-associated gene polymorphisms and weight gain after risperidone treatment in first episode schizophrenia. Behavioral and brain functions: BBF 10, 35, doi: 10.1186/1744-9081-10-35 (2014).
Fonseka, T. M. et al. The role of genetic variation across IL-1beta, IL-2, IL-6, and BDNF in antipsychotic-induced weight gain. The world journal of biological psychiatry: the official journal of the World Federation of Societies of Biological Psychiatry 16, 45–56, doi: 10.3109/15622975.2014.984631 (2015).
Chagnon, Y. C. et al. A genome wide linkage study of obesity as secondary effect of antipsychotics in multigenerational families of eastern Quebec affected by psychoses. Molecular psychiatry 9, 1067–1074, doi: 10.1038/sj.mp.4001537 (2004).
Malhotra, A. K. et al. Association between common variants near the melanocortin 4 receptor gene and severe antipsychotic drug-induced weight gain. Archives of general psychiatry 69, 904–912, doi: 10.1001/archgenpsychiatry.2012.191 (2012).
Yu, H. et al. Genome-Wide Association Study Suggested the PTPRD Polymorphisms Were Associated With Weight Gain Effects of Atypical Antipsychotic Medications. Schizophrenia bulletin, doi: 10.1093/schbul/sbv179 (2015).
Sun, L. et al. Multiple apical plasma membrane constituents are associated with susceptibility to meconium ileus in individuals with cystic fibrosis. Nature genetics 44, 562–569, doi: 10.1038/ng.2221 (2012).
Lieberman, J. A. et al. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. The New England journal of medicine 353, 1209–1223, doi: 10.1056/NEJMoa051688 (2005).
Shams, T. A. & Muller, D. J. Antipsychotic induced weight gain: genetics, epigenetics, and biomarkers reviewed. Current psychiatry reports 16, 473, doi: 10.1007/s11920-014-0473-9 (2014).
Chowdhury, N. I., Remington, G. & Kennedy, J. L. Genetics of antipsychotic-induced side effects and agranulocytosis. Current psychiatry reports 13, 156–165, doi: 10.1007/s11920-011-0185-3 (2011).
McCracken, J. T. Antipsychotic-induced weight gain: can the energy balance gene variants help us tip the scales? Pharmacogenomics 15, 403–406, doi: 10.2217/pgs.13.209 (2014).
Kao, A. C. et al. Methylenetetrahydrofolate reductase gene variants and antipsychotic-induced weight gain and metabolic disturbances. Journal of psychiatric research 54, 36–42, doi: 10.1016/j.jpsychires.2014.03.012 (2014).
Wilkinson, B. et al. TOX: an HMG box protein implicated in the regulation of thymocyte selection. Nature immunology 3, 272–280, doi: 10.1038/ni767 (2002).
Yu, X. & Li, Z. TOX gene: a novel target for human cancer gene therapy. American journal of cancer research 5, 3516–3524 (2015).
Eymin, B. & Gazzeri, S. Role of cell cycle regulators in lung carcinogenesis. Cell adhesion & migration 4, 114–123 (2010).
Shiffman, D. et al. Association of gene variants with incident myocardial infarction in the Cardiovascular Health Study. Arteriosclerosis, thrombosis, and vascular biology 28, 173–179, doi: 10.1161/ATVBAHA.107.153981 (2008).
Wei, F. et al. TOX and CDKN2A/B Gene Polymorphisms Are Associated with Type 2 Diabetes in Han Chinese. Scientific reports 5, 11900, doi: 10.1038/srep11900 (2015).
Zhang, L. T. et al. Intracranial aneurysm risk factor genes: relationship with intracranial aneurysm risk in a Chinese Han population. Genetics and molecular research: GMR 14, 6865–6878, doi: 10.4238/2015.June.18.30 (2015).
Heid, I. M. et al. Clear detection of ADIPOQ locus as the major gene for plasma adiponectin: results of genome-wide association analyses including 4659 European individuals. Atherosclerosis 208, 412–420, doi: 10.1016/j.atherosclerosis.2009.11.035 (2010).
Siitonen, N. et al. Association of ADIPOQ gene variants with body weight, type 2 diabetes and serum adiponectin concentrations: the Finnish Diabetes Prevention Study. BMC medical genetics 12, 5, doi: 10.1186/1471-2350-12-5 (2011).
Yu, Z. et al. Genetic polymorphisms in adipokine genes and the risk of obesity: a systematic review and meta-analysis. Obesity 20, 396–406, doi: 10.1038/oby.2011.148 (2012).
Leu, H. B. et al. Adiponectin gene polymorphism is selectively associated with the concomitant presence of metabolic syndrome and essential hypertension. PloS one 6, e19999, doi: 10.1371/journal.pone.0019999 (2011).
Zhang, H., Mo, X., Hao, Y. & Gu, D. Association between polymorphisms in the adiponectin gene and cardiovascular disease: a meta-analysis. BMC medical genetics 13, 40, doi: 10.1186/1471-2350-13-40 (2012).
Gui, M. H. et al. Association of the adiponectin gene rs1501299 G > T variant, serum adiponectin levels, and the risk of coronary artery disease in a Chinese population. Diabetes research and clinical practice 97, 499–504, doi: 10.1016/j.diabres.2012.05.011 (2012).
Wu, R., Zhao, J., Shao, P., Ou, J. & Chang, M. Genetic predictors of antipsychotic-induced weight gain: a case-matched multi-gene study. Zhong nan da xue xue bao. Yi xue ban = Journal of Central South University. Medical sciences 36, 720–723, doi: 10.3969/j.issn.1672-7347.2011.08.003 (2011).
Jassim, G. et al. Association study of energy homeostasis genes and antipsychotic-induced weight gain in patients with schizophrenia. Pharmacopsychiatry 44, 15–20, doi: 10.1055/s-0030-1263174 (2011).
Brandl, E. J. et al. No evidence for a role of the peroxisome proliferator-activated receptor gamma (PPARG) and adiponectin (ADIPOQ) genes in antipsychotic-induced weight gain. Psychiatry research 219, 255–260, doi: 10.1016/j.psychres.2014.05.031 (2014).
Klemettila, J. P. et al. Association study of the HTR2C, leptin and adiponectin genes and serum marker analyses in clozapine treated long-term patients with schizophrenia. European psychiatry: the journal of the Association of European Psychiatrists 30, 296–302, doi: 10.1016/j.eurpsy.2014.08.006 (2015).
Ujike, H. et al. Multiple genetic factors in olanzapine-induced weight gain in schizophrenia patients: a cohort study. The Journal of clinical psychiatry 69, 1416–1422 (2008).
Seidah, N. G., Benjannet, S., Pareek, S., Chretien, M. & Murphy, R. A. Cellular processing of the neurotrophin precursors of NT3 and BDNF by the mammalian proprotein convertases. FEBS letters 379, 247–250 (1996).
Egan, M. F. et al. The BDNF val66met polymorphism affects activity-dependent secretion of BDNF and human memory and hippocampal function. Cell 112, 257–269 (2003).
Adachi, N., Numakawa, T., Richards, M., Nakajima, S. & Kunugi, H. New insight in expression, transport, and secretion of brain-derived neurotrophic factor: Implications in brain-related diseases. World journal of biological chemistry 5, 409–428, doi: 10.4331/wjbc.v5.i4.409 (2014).
Gamero-Villarroel, C. et al. BDNF genetic variability modulates psychopathological symptoms in patients with eating disorders. European child & adolescent psychiatry 23, 669–679, doi: 10.1007/s00787-013-0495-6 (2014).
Notaras, M., Hill, R. & van den Buuse, M. The BDNF gene Val66Met polymorphism as a modifier of psychiatric disorder susceptibility: progress and controversy. Molecular psychiatry 20, 916–930, doi: 10.1038/mp.2015.27 (2015).
Thorleifsson, G. et al. Genome-wide association yields new sequence variants at seven loci that associate with measures of obesity. Nature genetics 41, 18–24, doi: 10.1038/ng.274 (2009).
Locke, A. E. et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 518, 197–206, doi: 10.1038/nature14177 (2015).
Bonaccorso, S. et al. The brain-derived neurotrophic factor (BDNF) Val66Met polymorphism is associated with increased body mass index and insulin resistance measures in bipolar disorder and schizophrenia. Bipolar disorders 17, 528–535, doi: 10.1111/bdi.12294 (2015).
Zai, G. C. et al. The role of brain-derived neurotrophic factor (BDNF) gene variants in antipsychotic response and antipsychotic-induced weight gain. Progress in neuro-psychopharmacology & biological psychiatry 39, 96–101, doi: 10.1016/j.pnpbp.2012.05.014 (2012).
Zhang, X. Y. et al. BDNF levels and genotype are associated with antipsychotic-induced weight gain in patients with chronic schizophrenia. Neuropsychopharmacology: official publication of the American College of Neuropsychopharmacology 33, 2200–2205, doi: 10.1038/sj.npp.1301619 (2008).
Tsai, A. et al. Association study of brain-derived neurotrophic factor gene polymorphisms and body weight change in schizophrenic patients under long-term atypical antipsychotic treatment. Neuromolecular medicine 13, 328–333, doi: 10.1007/s12017-011-8159-5 (2011).
Nishida, C., Ko, G. T. & Kumanyika, S. Body fat distribution and noncommunicable diseases in populations: overview of the 2008 WHO Expert Consultation on Waist Circumference and Waist-Hip Ratio. European journal of clinical nutrition 64, 2–5, doi: 10.1038/ejcn.2009.139 (2010).
Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 447, 661-678, doi: nature05911 [pii]10.1038/nature05911 (2007).
Broadbent, H. M. et al. Susceptibility to coronary artery disease and diabetes is encoded by distinct, tightly linked SNPs in the ANRIL locus on chromosome 9p. Hum Mol Genet 17, 806–814, doi: ddm352 [pii]10.1093/hmg/ddm352 (2008).
Cugino, D. et al. Type 2 diabetes and polymorphisms on chromosome 9p21: a meta-analysis. Nutr Metab Cardiovasc Dis 22, 619–625, doi: 10.1016/j.numecd.2010.11.010S0939-4753(10)00294-2 [pii] (2012).
Newcomer, J. W. et al. Abnormalities in glucose regulation during antipsychotic treatment of schizophrenia. Archives of general psychiatry 59, 337–345 (2002).
Purcell, S., Cherny, S. S. & Sham, P. C. Genetic Power Calculator: design of linkage and association genetic mapping studies of complex traits. Bioinformatics 19, 149–150 (2003).
Correll, C. U., Lencz, T. & Malhotra, A. K. Antipsychotic drugs and obesity. Trends in molecular medicine 17, 97–107, doi: 10.1016/j.molmed.2010.10.010 (2011).
Wang, K. et al. A genome-wide association study on obesity and obesity-related traits. PloS one 6, e18939, doi: 10.1371/journal.pone.0018939 (2011).
Barrett, J. C., Fry, B., Maller, J. & Daly, M. J. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21, 263–265, doi: 10.1093/bioinformatics/bth457 (2005).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. American journal of human genetics 81, 559–575, doi: 10.1086/519795 (2007).
Acknowledgements
This work was supported by grant 81070576 from the National Natural Science Foundation of China and grant 12JCZDJC24700 from Natural Science Foundation of Tianjin City to W.-D.L. We thank all of the study participants for their cooperation.
Author information
Authors and Affiliations
Contributions
W.D.L. and J.L. contributed to the design and execution of the study. S.L. and W.D.L. were responsible for the primary drafting of the manuscript. S.L., C.X., X.W., Y.T., C.S., Y.H. and M.Z. performed the experiments. S.L., C.X., R.J., X.W., G.Y., L.W., Y.G. and Y.Z. were responsible for the acquisition of data. S.L., Y.T., M.Z. and W.D.L. performed the statistical analysis. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Li, S., Xu, C., Tian, Y. et al. TOX and ADIPOQ Gene Polymorphisms Are Associated with Antipsychotic-Induced Weight Gain in Han Chinese. Sci Rep 7, 45203 (2017). https://doi.org/10.1038/srep45203
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep45203
- Springer Nature Limited
This article is cited by
-
Genome-wide association studies for economically important traits in mink using copy number variation
Scientific Reports (2024)
-
Dance/movement therapy for improving metabolic parameters in long-term veterans with schizophrenia
Schizophrenia (2024)
-
Mechanism and treatments of antipsychotic-induced weight gain
International Journal of Obesity (2023)
-
Dance/movement therapy for improving balance ability and bone mineral density in long-term patients with schizophrenia: a randomized controlled trial
Schizophrenia (2023)
-
The association between BDNF levels and risperidone-induced weight gain is dependent on the BDNF Val66Met polymorphism in antipsychotic-naive first episode schizophrenia patients: a 12-week prospective study
Translational Psychiatry (2021)