Abstract
Controlling oxygen content in perovskite oxides with ABO3 structure is one of most critical steps for tuning their functionality. Notably, there have been tremendous efforts to understand the effect of changes in oxygen content on the properties of perovskite thin films that are not composed of cations with multiple valance states. Here, we study the effect of oxygen vacancies on structural and electrical properties in epitaxial thin films of SrFeO3−δ (SFO), where SFO is a compound with multiple valance states at the B site. Various annealing treatments are used to produce different oxygen contents in the films, which has resulted in significant structural changes in the fully strained SFO films. The out-of-plane lattice parameter and tetragonality increase with decreasing oxygen concentration, indicating the crystal structure is closely related to the oxygen content. Importantly, variation of the oxygen content in the films significantly affects the dielectric properties, leakage conduction mechanisms, and the resistive hysteresis of the materials. These results establish the relationship between oxygen content and structural and functional properties for a range of multivalent transition metal oxides.
Similar content being viewed by others
Introduction
The functionality for a range of complex metal oxides is controlled by the interplay between lattice, spin, charge, and orbital degrees of freedom. Experimentally, it has been demonstrated that the oxygen content is one of the key parameters in controlling this interplay and thus significantly influence the physical properties; for example, Mn-O-Mn chains in manganite perovskite oxides determine the ferromagnetism and magnetotransport properties1,2. In semiconducting oxides, oxygen vacancies usually serve as dopants (since the energy level is above the middle of the bandgap) and impact the electrical behavior. Because the oxygen content in complex metal oxides allows the manipulation of desired physical properties3,4,5,6,7,8, it is a crucial aspect of consideration in the design, synthesis, and application of functional oxide thin films. In perovskite oxides with ABO3 structure, oxygen content-driven A-site and B-site vacancies have been reported to significantly alter the physical properties of thin films. There have been tremendous efforts to understand the effect of changes in oxygen content on the properties of perovskite thin films that are not composed of cations with multiple valance states. While oxygen vacancies have been recently been reported to act as desired defects9,10,11,12,13,14,15,16,17, the oxygen content effect in perovskite compounds with multiple valance states at the B site can provide a deeper understanding of the underlying mechanics of the functional properties observed in these materials18.
During thin film growth, oxygen content in oxide thin films varies depending on the growth conditions or post-treatments. Three main approaches have been commonly used to accommodate oxygen vacancies: (I) by generating corresponding vacancies in cation sites; (II) by altering the valence state of cations without cation non-stoichiometry; and (III) by incorporating both cation vacancies and change of valence state. Generation of vacancies in cation sites or forming cation-anion vacancy pairs usually occurs in metal oxides without multiple valence states such as ZnO, SrTiO3, BaTiO3, etc19. For example, electronic conduction and superconductivity in SrTiO3 as well as ferroelectricity in BaTiO3 are influenced by oxygen vacancies induced cation stoichiometry20,21,22. It was reported that the cation stoichiometry is strongly affected by the oxygen pressure during synthesis. In both SrTiO3 and BaTiO3, lower oxygen pressure during film deposition results in larger out-of-plane lattice parameter23.
Altering the valence state of cations without cation non-stoichiometry usually occurs in compounds with multiple valance states, where oxygen vacancies are often charge-compensated by the change of cation valence state24. Perovskite oxides with transition metals such as V, Co, Fe, and Mn belong to this category. SrFeO3−δ is an example of this type. The existence of multiple valence states of Fe allows for various stable states of oxygen occupancy in the lattice. Much of the recent attention in SFO thin films stems from the strong dependence of the crystal structure, magnetic and electrical properties of this material on the oxygen content (0 ≤ δ ≤ 0.5). SrFeO2.5 can exhibit semiconducting behavior and brownmillerite structure, while cubic perovskite SrFeO3 has been shown to exhibit metallic behavior with existence of helical antiferromagnetic-ordered spin structure25,26,28.
The control of vacancy state behavior in compounds with multiple valance states represents great promise for the malleability of multifunctional oxide thin films, but more investigation is still required to establish and understand the underlying mechanisms. Here, we report on the effect of various processing parameters on the oxygen content of SFO thin films and correlate oxygen content to the transport and dielectric properties. Studying the effect of oxygen vacancies in compounds with multiple valance states such as SFO provides an avenue to better understand the relationship between lattice, charge and structure. We propose that the metastable oxygen deficient states of SFO thin films offer an opportunity to accomplish highly tunable electronic properties applicable to a wide range of technological applications that leverage the variability in structural, magnetic and electrical properties of SFO.
Results and Discussion
SFO thin films with a thickness of 75 nm were deposited by PLD on Nb-doped (0.7% wt) STO (001) substrates (Nb:STO) at 800 °C in three different post-growth annealing conditions. O2 and vacuum anneals were performed by holding the films at 600 °C for 1 hour in O2 of 500 Torr and <10−6 Torr, respectively. The films were then cooled down to room temperature at 5 °C/min at the same oxygen pressure. SFO with no anneal was also investigated, in which the heater was promptly shut off after deposition and films cooled freely in the growth pressure (250 mTorr O2) environment.
Similar to well-studied transition metal oxide systems such as SrCoO3−δ29, bulk SFO exhibits two topotactic phases: the cubic perovskite SrFeO3 and the brownmillerite SrFeO2.530,31. Perovskite SrFeO3 has a bulk lattice parameter of 3.851 Å. The oxygen-deficient brownmillerite SrFeO2.5 structure has unit cell parameters of a = 5.672 Å, b = 15.59 Å, and c = 5.527 Å. This structure can be reduced to pseudotetragonal with unit cell parameters of  = 4.011 Å,
= 4.011 Å,  = 3.898 Å, and
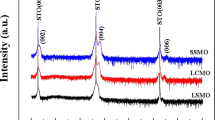
= 3.898 Å, and  = 3.908 Å31,32. Figure 1 shows the X-ray diffraction (XRD) patterns of the films treated with different conditions. The out-of-plane lattice parameters of SFO thin films are shown to increase with decreasing oxygen pressure during anneal, as evidenced by the progressively smaller 2θ values of the SFO film peak when comparing O2 anneal, no anneal and vacuum anneal samples, respectively. A broad XRD 2θ scan indicates that films are c-axis oriented with no detectable mixed phases present (not shown here). The out-of-plane SFO film lattice parameter is ~3.83 Å for the O2 annealed film, ~3.85 Å for the film with no anneal, and ~3.97 Å for the vacuum annealed film. Reciprocal space mapping (RSM) scans around the (103) peak of the SFO films are compared in Fig. 1b–d. In all three films, it can be seen that the in-plane lattice parameter of SFO remains strained to the Nb:STO substrate, indicating a = b ~3.905 Å. The out-of-plane lattice parameters progressively increase from 3.83 Å to 3.97 Å with reducing oxygen content in annealing. This corresponds to a relative unit cell volume increase of approximately 3.6% when comparing anneal in vacuum vs. oxygen. Oxygen vacancies donate electrons to the empty 3d-orbitals of Fe, reducing the ion from Fe4+ to Fe3+ and increasing the ionic radius of the Fe ion14. Hence, the lattice parameter is directly related with the oxygen content. These results agree with SFO materials reported by Yamada et al.32.
= 3.908 Å31,32. Figure 1 shows the X-ray diffraction (XRD) patterns of the films treated with different conditions. The out-of-plane lattice parameters of SFO thin films are shown to increase with decreasing oxygen pressure during anneal, as evidenced by the progressively smaller 2θ values of the SFO film peak when comparing O2 anneal, no anneal and vacuum anneal samples, respectively. A broad XRD 2θ scan indicates that films are c-axis oriented with no detectable mixed phases present (not shown here). The out-of-plane SFO film lattice parameter is ~3.83 Å for the O2 annealed film, ~3.85 Å for the film with no anneal, and ~3.97 Å for the vacuum annealed film. Reciprocal space mapping (RSM) scans around the (103) peak of the SFO films are compared in Fig. 1b–d. In all three films, it can be seen that the in-plane lattice parameter of SFO remains strained to the Nb:STO substrate, indicating a = b ~3.905 Å. The out-of-plane lattice parameters progressively increase from 3.83 Å to 3.97 Å with reducing oxygen content in annealing. This corresponds to a relative unit cell volume increase of approximately 3.6% when comparing anneal in vacuum vs. oxygen. Oxygen vacancies donate electrons to the empty 3d-orbitals of Fe, reducing the ion from Fe4+ to Fe3+ and increasing the ionic radius of the Fe ion14. Hence, the lattice parameter is directly related with the oxygen content. These results agree with SFO materials reported by Yamada et al.32.
The oxygen content and associated structural changes in SFO films significantly modify the electrical properties. The room temperature dielectric and leakage current properties of SFO films with various anneal treatments are shown in Fig. 2. A metal-insulator-metal (MIM) structure was employed with Au top circular electrodes of area approximately 0.3 mm2 sputtered on the SFO/Nb:STO samples. In samples with O2 anneal and no anneal, the dielectric constant is higher than that of the vacuum annealed samples, but the loss is also significantly greater, with dissipation factor, D, of 0.547 and 0.409 for SFO at 100 kHz without anneal and O2 annealed, respectively (see Fig. 2a,c and e). The dissipation factor of SFO annealed in vacuum is a magnitude of order lower, with a value of 0.064 at 100 kHz. The lower loss is due to the increased formation of the semiconducting SrFeO2.5 brownmillerite phase in the vacuum annealed SFO, which can also be seen in the trend of leakage current behavior in Fig. 2b,d and f. The phase transition between the semiconducting SrFeO2.5 phase and metallic SrFeO3 phase has been used to design filament type resistive switching devices31. The formation of conducting filaments in SFO is possibly related to the migration of oxygen vacancies within SrFeO3−δ., under application of electric field, which allows metallic SrFeO3 channels to flow current within the semiconducting matrix. As a result, the hysteretic behavior related to this filament formation is directly related to the density and mobility of oxygen vacancies within the SFO bulk. The asymmetry of leakage behavior between positive and negative applied bias is a product of the difference between conduction activity at the Au/SFO and Nb:STO/SFO interfaces33,34. By applying fields in both directions in the MIM structure, the Schottky diode behavior allows for investigation in the dominant I-V characteristics at either of the metal-semiconductor contact interfaces35.
To understand the oxygen vacancies’ effect on electrical properties, the current conduction mechanisms with applied field in the range of 0–66.6 MV/m were analyzed by fitting to both bulk and interface-mediated emission models. Mathematical fitting functions were applied to the I-V curves in regions of applied field above which the majority of hysteresis was observed, since fitting in these regions must also account for the high density of oxygen migration. One possible mechanism of conduction that considers the injection of charge carriers from a metal into nearby oxygen vacancy sites as traps, with an associated barrier height that governs bulk conduction in a solid, is the trap-assisted tunneling current model:33

where A is a constant, e is the electronic charge,  is the corresponding tunneling barrier height,
is the corresponding tunneling barrier height,  is the effective electron mass, E is the electric field magnitude and h is the Planck constant. Based on this model, linear fitting was performed on the SFO thin films by plotting ln(J) vs. 1/E. A good linear fit was obtained for no anneal and O2 annealed SFO samples, suggesting a trap-assisted tunneling conduction mechanism, as seen by the fitting data in the insets of Fig. 2b and d, respectively. The fitting shows that
is the effective electron mass, E is the electric field magnitude and h is the Planck constant. Based on this model, linear fitting was performed on the SFO thin films by plotting ln(J) vs. 1/E. A good linear fit was obtained for no anneal and O2 annealed SFO samples, suggesting a trap-assisted tunneling conduction mechanism, as seen by the fitting data in the insets of Fig. 2b and d, respectively. The fitting shows that  is 132 meV and 136 meV for the SFO samples without annealing and annealed in O2, respectively. However, the trap-assisted tunneling model did not yield a good linear trend for the SFO film annealed in vacuum. On the other hand, the Schottky emission model is used to explain temperature-dependent leakage mechanisms that are dominated by injection of charge carriers at the metal-insulator interface, and is given by the following:
is 132 meV and 136 meV for the SFO samples without annealing and annealed in O2, respectively. However, the trap-assisted tunneling model did not yield a good linear trend for the SFO film annealed in vacuum. On the other hand, the Schottky emission model is used to explain temperature-dependent leakage mechanisms that are dominated by injection of charge carriers at the metal-insulator interface, and is given by the following:

where J is the field-dependent current density, T is the absolute temperature,  is the Schottky barrier height,
is the Schottky barrier height,  is the optical dielectric constant,
is the optical dielectric constant,  is the permittivity of free space, and k is the Boltzmann constant.
is the permittivity of free space, and k is the Boltzmann constant.  is known as the Richardson constant, represented as
is known as the Richardson constant, represented as

This model also takes into account mediation of leakage current by defects such as oxygen vacancies, which makes it a useful tool to analyze the behavior in SFO thin films34,36,37,38. Fitting of the data yields a good linear fit of ln(J/T2) vs.  for the vacuum annealed SFO thin film, as can be seen in the inset of Fig. 2f. An optical dielectric constant of 3.74 was extracted from the slope of the linear fitting curve. There are limited data on the optical dielectric constant of SFO, but similar studies on SrCoOx thin films suggest that this value is within reason39. Although the results suggest that the leakage current behavior in the vacuum annealed SFO thin film is well-explained by the Schottky equation, the SFO samples without annealing and annealed in O2 do not yield a linear trend of the ln(J/T2) vs.
for the vacuum annealed SFO thin film, as can be seen in the inset of Fig. 2f. An optical dielectric constant of 3.74 was extracted from the slope of the linear fitting curve. There are limited data on the optical dielectric constant of SFO, but similar studies on SrCoOx thin films suggest that this value is within reason39. Although the results suggest that the leakage current behavior in the vacuum annealed SFO thin film is well-explained by the Schottky equation, the SFO samples without annealing and annealed in O2 do not yield a linear trend of the ln(J/T2) vs.  data. Dielectric properties for all SFO films at 100 kHz are compared in Table 1. We can use these data to infer that as the oxygen content in SFO varies, not only the structure but also the nature of the conduction mechanisms in the film undergoes a notable change. Further understanding of the conduction mechanisms is critical to improving and tailoring SFO thin films for specific applications.
data. Dielectric properties for all SFO films at 100 kHz are compared in Table 1. We can use these data to infer that as the oxygen content in SFO varies, not only the structure but also the nature of the conduction mechanisms in the film undergoes a notable change. Further understanding of the conduction mechanisms is critical to improving and tailoring SFO thin films for specific applications.
There are several methods used to extract the Schottky barrier height,  , in the Schottky thermionic emission model, but one of the most widely used is to compare the conduction behavior against variation in temperature38,40. Investigation of the electronic properties of SFO thin film annealed in vacuum was therefore conducted in the temperature range of 120 K–300 K. Results for the temperature dependence of permittivity, dielectric loss and leakage current are shown in Fig. 3.
, in the Schottky thermionic emission model, but one of the most widely used is to compare the conduction behavior against variation in temperature38,40. Investigation of the electronic properties of SFO thin film annealed in vacuum was therefore conducted in the temperature range of 120 K–300 K. Results for the temperature dependence of permittivity, dielectric loss and leakage current are shown in Fig. 3.
Vacuum annealed SFO temperature dependence in the range of 120 K–300 K of (a) Dielectric constant and dissipation factor, (b) leakage current vs. applied voltage (J-V) behavior with forward bias from 0–66.6 MV/m, (c) linear fit of ln(J/T2) vs. 1/T from which the effective thermionic emission barrier height is calculated.
The dielectric constant and loss of the vacuum annealed SFO sample were observed to decrease with decreasing temperature while maintaining a similar frequency-dependent profile, as shown in Fig. 3a. The temperature dependence of the leakage current at the Au/SFO interface, shown in Fig. 3b, follows an exponential behavior with increasing temperature, which indicates a good fit to the thermionic emission model. An effective thermal barrier height,  , is extracted from the Schottky equation by using the relationship between ln(J/T2) vs. 1/T at a given E, which is presented in Fig. 3c33,38. By then extrapolating from the linear trend of
, is extracted from the Schottky equation by using the relationship between ln(J/T2) vs. 1/T at a given E, which is presented in Fig. 3c33,38. By then extrapolating from the linear trend of  vs.
vs.  to zero electric field, the intrinsic thermal barrier height,
to zero electric field, the intrinsic thermal barrier height,  , was calculated with a value of 225 meV for the Au/SFO interface.
, was calculated with a value of 225 meV for the Au/SFO interface.
Dielectric property data measured at 100 kHz is summarized in Fig. 4. As temperature is varied, the dielectric response depends on associated changes in volume and polarizability in the dielectric41. The linear increasing trend can be attributed to the increasing polarizability of the SFO thin film with temperature, as shown in Fig. 4a and 4b. Indeed, the temperature dependence of the tunability of the SFO thin film, as shown in Fig. 4c, follows an exponential trend in the investigated temperature range. The inset of Fig. 4c shows tunability curves at selected temperatures, where the maximum tunability was calculated at 66.6 MV/m. This behavior is suggestive of the suppression of polarizability in the SFO thin film with decreasing temperature, and indicates that the temperature dependence of polarizability is a dominant effect in governing the response observed in the vacuum annealed SFO thin film42. Similar dielectric response behavior has also been seen in LaFeO3 in studies reported by Gaikwad et al.43.
Conclusion
In summary, the effect of oxygen vacancies on structural and electrical properties in epitaxial thin films of SrFeO3−δ (SFO) is studied, where SFO is a compound with multiple valance states at the B site. Various annealing treatments are used to produce different oxygen contents in the films, which results in significant structural changes in the fully strained SFO films. The out-of-plane lattice parameter and tetragonality increase with decreasing oxygen concentration, indicating the crystal structure is closely related to the oxygen content. Importantly, variation of the oxygen content in the films significantly affects the dielectric properties, leakage conduction mechanisms, and the resistive hysteresis of the materials. Leakage current mechanisms are found to shift from dominantly bulk-mediated trap-assisted tunneling to interface-mediated Schottky thermionic emission depending on oxygen vacancy concentration. Temperature dependence of vacuum annealed SFO thin film is investigated in the temperature range of 120 K–300 K, and results suggested a suppression of polarizability of the SFO thin film with decreasing temperature, as indicated by a reduction in the dielectric constant and tunability of the SFO film. These results establish the relationship between oxygen content and structural and functional properties for a range of multivalent transition metal oxides.
Methods
SFO thin films were grown by PLD using a KrF excimer laser (Lambda Physik LPX 300, λ = 248 nm, 2 Hz). The laser beam, defined by the image beam method44, was focused onto the target with an energy density of 1.87 J/cm2. Prior to the deposition, the chamber was pumped down to a base pressure of 1 × 10−6 Torr. A substrate temperature of 800 °C and an oxygen pressure of 250 mTorr were maintained during all depositions. After deposition, various anneal treatments were used to modify the oxygen content in the SFO thin films. An O2 anneal was used with an oxygen pressure of 500 Torr and the films were held at 600 °C for 1 hour to allow more oxygen to enter the thin film. The films were then cooled down to room temperature at 5 °C/min. A vacuum anneal was performed by maintaining films in a pressure environment of < 10−6 Torr, held at 600 °C followed by cooling to room temperature at 5 °C/min. Samples were also grown with no anneal, in which the heater was promptly shut off after deposition and films cooled freely in the growth pressure (250 mTorr O2) environment.
X-ray diffraction (Panalytical X’Pert PRO MRD) 2θ-ω and reciprocal space mapping (RSM) scans were employed to obtain information on the orientation, lattice parameters and epitaxial quality of the thin films. Dielectric measurements (E4980A Precision LCR meter) were performed in the temperature range of 120 K–300 K by using a physical property measurement system to produce the cryogenic environment. An MIM (Au/SFO/Nb:STO) structure was used for dielectric measurements, with positive bias applied indicating the high potential applied to the Nb:STO substrate and low potential at the Au electrode.
Additional Information
How to cite this article: Enriquez, E. et al. Oxygen Vacancy-Tuned Physical Properties in Perovskite Thin Films with Multiple B-site Valance States. Sci. Rep. 7, 46184; doi: 10.1038/srep46184 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Chen, A. P. et al. Microstructure, Magnetic, and Low-Field Magnetotransport Properties of Self-Assembled (La0.7Sr0.3MnO3)0.5:(CeO2)0.5 Vertically Aligned Nanocomposite Thin Films. Nanotechnology 22, 315712 (2011).
Hwang, H. Y. et al. Lattice Effects on the Magnetoresistance in Doped LaMnO3, Phys. Rev. Lett. 75, 914 (1995).
Keeble, D. J. et al. Identification of Vacancy Defects in a Thin Film Perovskite Oxide. Phys. Rev. B 81, 064102 (2010).
Xiao, G. et al. High-Temperature Superconductivity in Tetragonal Perovskite Structures - Is Oxygen-Vacancy Order Important. Phys. Rev. Lett. 60, 1446–1449 (1988).
Warren, W. L., Vanheusden, K., Dimos, D., Pike, G. E. & Tuttle, B. A. Oxygen Vacancy Motion in Perovskite Oxides. J. Am. Ceram. Soc. 79, 536–538 (1996).
Raveau, B. & Seikh, M. M. in Cobalt Oxides 10.1002/9783527645527.ch1 Ch. 1. Crystal Chemistry of Cobalt Oxides, 3–70 (Wiley-VCH Verlag GmbH & Co. KGaA, 2012).
Anderson, M. T., Vaughey, J. T. & Poeppelmeier, K. R. Structural Similarities Among Oxygen-Deficient Perovskites. Chem. Mater. 5, 151–165 (1993).
Grenier, J. C., Pouchard, M. & Hagenmuller, P. Vacancy Ordering in Oxygen-Deficient Perovskite-Related Ferrites. Struct. Bond. 47, 1–25 (1981).
Kalinin, S. V. & Spaldin, N. A. Functional Ion Defects in Transition Metal Oxides. Science 341, 858–859 (2013).
Kalinin, S. V., Borisevich, A. & Fong, D. Beyond Condensed Matter Physics on the Nanoscale: The Role of Ionic and Electrochemical Phenomena in the Physical Functionalities of Oxide Materials. ACS Nano 6, 10423–10437 (2012).
Kim, Y. M. et al. Probing Oxygen Vacancy Concentration and Homogeneity in Solid-Oxide Fuel-Cell Cathode Materials on the Subunit-Cell Level. Nature Mater. 11, 888–894 (2012).
Biskup, N. et al. Insulating Ferromagnetic LaCoO3−delta Films: A Phase Induced by Ordering of Oxygen Vacancies. Phys. Rev. Lett. 112, 087202 (2014).
Salluzzo, M. et al. Origin of Interface Magnetism in BiMnO3/SrTiO3 and LaAlO3/SrTiO3 Heterostructures. Phys. Rev. Lett. 111, 087204 (2013).
Kormondy, K. J. et al. Quasi-Two-Dimensional Electron Gas at the Epitaxial Alumina/SrTiO3 Interface: Control of Oxygen Vacancies. J. Appl. Phys. 117, 095303 (2015).
Liu, Z. Q. et al. Bandgap Control of the Oxygen-Vacancy-Induced Two-Dimensional Electron Gas in SrTiO3 . Adv. Mater. Interfaces 1, 1400155 (2014).
Enriquez, E. et al. Oxygen Vacancy-Driven Evolution of Structural and Electrical Properties in SrFeO3−δ Thin Films and a Method of Stabilization. Appl. Phys. Lett. 109, 141906 (2016).
Harrell, Z. et al. Oxygen Content Tailored Magnetic and Electronic Properties in Cobaltite Double Perovskite Thin Films. Appl. Phys. Lett. 110, 093102 (2017).
Jeen, H. et al. Reversible Redox Reactions in an Epitaxially Stabilized SrCoOx Oxygen Sponge. Nat. Mater. 12, 1057–1063 (2013).
Lee, C. H. et al. Effect of Stoichiometry on the Dielectric Properties and Soft Mode Behavior of Strained Epitaxial SrTiO3 Thin Films on DyScO3 Substrates. Appl. Phys. Lett. 102, 082905 (2013).
Schooley, J. F., Hosler, W. R. & Cohen, M. L. Superconductivity in Semiconducting SrTiO3 . Phys. Rev. Lett. 12, 474–475 (1964).
Hou, Y. S., Xiang, H. J. & Gong, X. G. Intrinsic Insulating Ferromagnetism in Manganese Oxide Thin Films. Phys. Rev. B 89, 064415 (2014).
Lee, S. A. et al. Phase Transitions via Selective Elemental Vacancy Engineering in Complex Oxide Thin Films. Sci. Rep. 6, 23649 (2016).
Chen, A. P. et al. Strong Oxygen Pressure Dependence of Ferroelectricity in BaTiO3/SrRuO3/SrTiO3 Epitaxial Heterostructures. J. Appl. Phys. 114, 124101 (2013).
Jeen, H. et al. Reversible Redox Reactions in an Epitaxially Stabilized SrCoOx Oxygen Sponge. Nat. Mater. 12, 1057–1063 (2013).
Tofield, B. C., Greaves, C. & Fender, B. E. F. SrFeO2.5 - SrFeO3.0 System - Evidence of a New Phase Sr4Fe4O11 (SrFeO2.75). Mater. Res. Bull. 10, 737–746 (1975).
Adler, P. et al. Magnetoresistance Effects in SrFeO3−δ: Dependence on Phase Composition and Relation to Magnetic and Charge Order. Phys. Rev. B 73, 094451 (2006).
Chakraverty, S. et al. Multiple Helimagnetic Phases and Topological Hall Effect in Epitaxial Thin Films of Pristine and Co-doped SrFeO3 . Phys. Rev. B 88, 220405 (2013).
Oda, H., Yamaguchi, Y., Takei, H. & Watanabe, H. Single-Crystal Neutron-Diffraction Study of SrFeO3−x (x = 0.1). J. Phys. Soc. Jpn. 42, 101–106 (1977).
Jeen, H. et al. Topotactic Phase Transformation of the Brownmillerite SrCoO2.5 to the Perovskite SrCoO3−δ. Adv. Mater. 25, 3651–3656 (2013).
Chakraverty, S., Ohtomo, A., Okude, M., Ueno, K. & Kawasaki, M. Epitaxial Structure of (001)- and (111)-Oriented Perovskite Ferrate Films Grown by Pulsed-Laser Deposition. Cryst. Growth Des. 10, 1725–1729 (2010).
Acharya, S. K . et al. Epitaxial Brownmillerite Oxide Thin Films for Reliable Switching Memory. ACS Appl. Mater. Interfaces 8, 7902–7911 (2016).
Yamada, H., Kawasaki, M. & Tokura, Y. Epitaxial Growth and Valence Control of Strained Perovskite SrFeO3 Films. Appl. Phys. Lett. 80, 622–624 (2002).
Mojarad, S. A. et al. A Comprehensive Study on the Leakage Current Mechanisms of Pt/SrTiO3/Pt Capacitor. J. Appl. Phys. 111, 014503 (2012).
Hudec, B. et al. Atomic Layer Deposition Grown Metal-Insulator-Metal Capacitors with RuO2 Electrodes and Al-doped Rutile TiO2 Dielectric Layer. J. Vac. Sci. Technol. B 29, 01AC09 (2011).
Elhadidy, H., Sikula, J. & Franc, J. Symmetrical Current-Voltage Characteristic of a Metal-Semiconductor-Metal Structure of Schottky Contacts and Parameter Retrieval of a CdTe Structure. Semicond. Sci. Technol. 27, 015006 (2012).
Wang, M. T., Wang, T. H. & Lee, J. Y. M. Electrical Conduction Mechanism in High-Dielectric-Constant ZrO2 Thin Films. Microelectron. Reliab. 45, 969–972 (2005).
Chakraborty, S., Bera, M. K., Bhattacharya, S. & Maiti, C. K. Current Conduction Mechanism in TiO2 Gate Dielectrics. Microelectron. Eng. 81, 188–193 (2005).
Lee, S. W., Han, J. H. & Hwang, C. S. Electronic Conduction Mechanism of SrTiO3 Thin Film Grown on Ru Electrode by Atomic Layer Deposition. Electrochem. Solid State Lett. 12, G69–G71 (2009).
Choi, W. S. et al. Reversal of the Lattice Structure in SrCoOx Epitaxial Thin Films Studied by Real-Time Optical Spectroscopy and First-Principles Calculations. Phys. Rev. Lett. 111, 097401 (2013).
Jeong, H. Y., Lee, J. Y., Ryu, M. K. & Choi, S. Y. Bipolar Resistive Switching in Amorphous Titanium Oxide Thin Film. Phys. Status Solidi RRL 4, 28–30 (2010).
Havinga, E. E. The Temperature Dependence of Dielectric Constants. J. Phys. Chem. Solids 18, 253–255 (1961).
Tagantsev, A. K., Sherman, V. O., Astafiev, K. F., Venkatesh, J. & Setter, N. Ferroelectric materials for microwave tunable applications. J. Electroceram. 11, 5–66 (2003).
Gaikwad, V. M., Sheikh, J. R. & Acharya, S. A. Investigation of Photocatalytic and Dielectric Behavior of LaFeO3 Nanoparticles Prepared by Microwave-Assisted Sol-Gel Combustion Route. J. Sol-Gel Sci. Technol. 76, 27–35 (2015).
Dowden, P. C. & Jia, Q. X. Stabilizing laser energy density on a target during pulsed laser deposition of thin films. United States patent 9353435 (2016).
Acknowledgements
The work at Los Alamos National Laboratory was supported by the NNSA’s Laboratory Directed Research and Development Program and was performed, in part, at the Center for Integrated Nanotechnologies, an Office of Science User Facility operated for the U.S. Department of Energy (DOE) Office of Science. Los Alamos National Laboratory, an affirmative action equal opportunity employer, is operated by Los Alamos National Security, LLC, for the National Nuclear Security Administration of the U.S. Department of Energy under contract DE-AC52-06NA25396.
Author information
Authors and Affiliations
Contributions
E.E. conducted thin film growth, characterization and electrical properties measurements. A.P.C., M.J. and Q.X.J. supervised the project. Z.H., N.K., J.R. participated in analysis of characterization and electrical properties data. P.D. and C.L.C. participated in discussions on thin film growth. All authors participated in discussions, analysis and revision of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Enriquez, E., Chen, A., Harrell, Z. et al. Oxygen Vacancy-Tuned Physical Properties in Perovskite Thin Films with Multiple B-site Valance States. Sci Rep 7, 46184 (2017). https://doi.org/10.1038/srep46184
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep46184
- Springer Nature Limited
This article is cited by
-
Influence of post-annealing on Mn-doped (K0.5Na0.5)NbO3 thin films
Journal of the Korean Physical Society (2022)
-
Competing ferro- and antiferromagnetic exchange drives shape-selective \(\hbox{Co}_3\hbox{O}_4\) nanomagnetism
Scientific Reports (2020)
-
Proposal for an electrostrictive logic device with the epitaxial oxide heterostructure
Scientific Reports (2020)
-
The effects of the oxygen content on the photoelectrochemical properties of LaFeO3 perovskite thin films obtained by pulsed laser deposition
Applied Physics A (2019)
-
Electronic and Bandgap Tuning of Hydrogenated Ti-doped CdO Semiconductor
Journal of Electronic Materials (2019)