Abstract
Background
The reflux of noxious contents of the stomach may cause oesophageal and extra-oesophageal complications either by direct contact of aspirated gastric refluxate with the upper airway or by a vago-vagal reflex. This study aimed to determine the prevalence of gastroesophageal disease (GERD) and extraesophageal manifestations among undergraduate students in a tertiary institution in Nigeria.
Methods
This is a cross-sectional study involving undergraduate students in a private University in Nigeria. Study proforma had three parts. Part A consisted of self-administered questionnaire designed to obtain students biodata. Part B consisted of standard Carlsson-Dent questionnaire. A score of 4 and above on Carlsson- Dent questionnaire was considered diagnostic of gastroesophageal reflux symptoms (GERD). Thereafter those who had GERD were further questioned and examined for extra-oesophageal symptoms of GERD.
Results
The total number of the study participants was 647, out of which 212 (32.8%) had GERD. One hundred and forty-four (67.9%) and 86 (32.1%) females and male had GERD respectively (p = 0.13).
The extraesophageal symptoms found in those with GERD were, dysphagia, coated tongue, nocturnal cough, xerostomia, lump in the throat, asthma-like symptoms, recurrent sore throat, frequent throat clearing, halithosis and dental erosion among others.
Conclusion
GERD is common among this study population, with a prevalence rate of 32.8%. Only age showed significant predictor for GERD. Varying extra-oesophageal manifestations were found in those with GERD.
Similar content being viewed by others
Background
Gastroesophageal reflux disease (GERD) occurs due to reflux of gastric contents into the oesophagus leading to persistent symptoms and or complications [1]. Oesophageal manifestations of GERD include heartburns, regurgitation, oesophagitis, Barrett’s oesophagus and oesophageal adenocarcinoma. In addition, GERD has an array of manifestations and complications beyond the oesophagus and these are called extra-oesophageal or supra oesophageal manifestations or features of GERD [2]. The organs that are mainly involved in these extra oesophageal manifestations of GERD include lungs, Ear, Nose, Throat (ENT) and the mouth [3]. GERD manifestations in the ENT system include chronic laryngitis, chronic sinusitis, otitis media, hoarseness, cough, globus, sore throat, post nasal drip, nasal congestion, halitosis, paroxysmal laryngospasm, laryngeal and subglottic stenosis and laryngeal and pharyngeal carcinoma [4,5,6,7]. Respiratory features of GERD include chronic cough, choking episodes, aspiration pneumonitis, asthma-like disease among others. GERD is also a common cause of dental erosion which occurs as a result of long-term exposure to acid [8]. It is usually a slow process occurring over many years and its subtle appearances may be easily missed. Therefore, dental erosion is usually detected only after significant damage has occurred to the dentition and the masticatory system [9].
There is paucity of information in Nigeria regarding the GERD especially, its extraesophageal manifestations. There is therefore the need to find out the prevalence of GERD and its extraesophageal manifestations in our population.
The main objective was to determine the prevalence of gastroesophageal reflux symptoms and its extraesophageal manifestations among undergraduate students of Babcock University. Our specific objectives were to determine ear, nose, throat, respiratory and dental manifestations of GERD and the relationship between it and obesity among this study population.
Methods
This cross-sectional study was carried out in Babcock University, a private university located in Ilishan Remo, Ogun State, South west Nigeria with a students’ population of 8000 as at the time of this study.
Study participants
This study involved all undergraduate students at Babcock University residing in school halls of residency within the study period, who agreed to participate in the study.
Sample size
The Sample size for this study was determined using Leslie- Kish formula for population-based study at 95% confidence level with 50.0% prevalence and degree of freedom put at 0.05%. Using the formula, the sample size was 384 including 10% attrition rate. However, for this study 800 students were targeted but 668 students participated. Others did not give consent to participate.
Sampling technique
All the 8 students’ hostels were used for the study. Using the register of the students in each hostel, 100 students were randomly selected per hostel. The rooms of the randomly selected students were obtained from the register and they were approached in their respective rooms. Male Assistants were engaged for male hostels while female Assistants for the female hostels.
Instrument for the study
The instrument for this study consisted of three parts: A, B and C
Part A was a proforma questionnaires for students’ bio data, weight, height and waist circumference while Part B consisted of Carlsson- Dent questionnaire for diagnosis of GERD. Carlsson-Dent questionnaire is a 7-item questionnaire on typical symptoms of GERD and their relationship to meal, antacid use, posture, straining and effect of regurgitation. Positive or negative marks were awarded to the answers to the questions. A summation score of 4 and above was considered diagnostic of GERD [10]. Part C consisted of questionnaire on extraoesophageal symptoms developed the researchers and the Smith and Knight tooth wear index to assess dental erosion [11].
Data collection procedure
Trained research Assistants administered part A and B of the research instrument. Each subject had weight, height, and waist circumference taken. Weight (Kg) was taken using bathroom scales while heights were taken with stadiometers. Stretch -resistant tapes were used to measure the waist circumference at a point mid- way between the last rib and the iliac crest. The Subjects who had GERD based on the Carlson-Dent questionnaire score of 4 and above were recruited for the part C of the study (extra-oesophageal symptoms and examination). They were questioned and examined by the ENT surgeon, Orthodontist and a Chest Physician.
Data analysis
The age of the participants were grouped into 15–18 years, 19–22 years and greater or equal to 23 years, the BMI was classified as underweight (< 18 kg/m2), normal (18–24.9 kg/ m2), over-weight(25–29.9 kg/m2), and obese (40 > kg/m2). Waist circumference was also classified into normal (< 94 cm for male, < 80 cm for female), risky (94–101 cm for male, 81-88 cm for female) and highly risky (≥ 102 cm for male, ≥ 88 cm for female) waist circumference. Descriptive analysis was used to describe the demographic variables, the prevalence of GERD.
Pearson’s Chi square test was used to establish the association between GERD, age, sex, BMI and waist circumference. Inferential analysis using logistic regression was done to determine predictors of GERD. Values was be taken to be significant when P value was < 0.05.
Ethical consideration
The protocol to the study was submitted to the peer review committee of Babcock University Health Research Ethics Committee-BUHREC and was subsequently approved (BUHREC 002/17). Written informed consent was taken from all the Subjects that participated in the study.
Results
A total number of 668 students participated in the study but only 647 questionnaires were analysed. The remaining 21 questionnaires were not properly filled. Gender distribution of the study participants showed that 413 (63.8%) were females while 234 (36.2%) were males. The number of Participants with gastroesophageal reflux symptoms was 212 giving a prevalence of 32.8%.
Table 1 showed age and gender distribution of the study participants. The age group 15–18 years had the highest number of Participants with GERD (38.7%) while aged 19–22 has the least number of Participants with GERD (29.3%). The association between the age of the Participants and GERD was not statistically significant (p = 0.06). This study showed that there were more females with GERD (34.9%) when compared with their male counterpart (29.1%) (p = 0.13).
In addition, participants with BMI ≥30 had higher percentage of GERD (41.2%), followed by those with BMI of ≤18.4 (36.1%), then BMI of 18.5–24.9 (32.5%) while participants with BMI of 25–30 had the least GERD (28.8%). The association between BMI and GERD was also not statistically significant (p = 0.41). Also, there was no statistically significant association between GERD and waist circumference in both males and females (p = 0.51 and p = 0.23).
Table 2 shows the result of multivariate logistic regression to determine the predictors of GERD in the study population. Only age shows significant association with GERD, participants aged 19–22 years showed reduced odds of having GERD compared with other age groups (p = 0.04). Participants who were obese and underweight showed increased odds of having GERD (AOR 1.67; 95% C. I 0.84–3.33; P = 0.15) and (AOR 1.14; 95% C. I 0.65–2.01; P = 0.65) respectively. Male participants showed reduced odds of having GERD (AOR 0.77; 95% C. I 1.53–1.13; P = 0.19), then participants from age group greater than 23 year (AOR 0.91; 95% C. I 0.45–0.88; P = 0.81), overweight participants (AOR 0.90; 95% C. I 0.57–1.42; P = 0.66). The waist circumference was not a significant predictor of GERD in both male and female Participants.
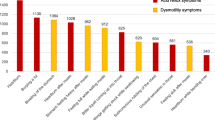
Table 3 shows various extraesophageal symptoms seen among those with GERD. Out of 212 with GERD, 69 (32.0%) reported at the clinic for assessment on extraesophageal manifestation of GERD.
Out of 69 students who reported in the clinic, 8.7% had chronic or recurrent cough, 10.1% had halitosis, 10.1% had excessive phlegm, 10.1% had dysphagia, 14.5% had coated tongue, 15.9% had nocturnal cough, 17.4% had xerostomia, 17.4% had feeling of lump in the throat, 17.4% had difficulty in breathing especially at night, 17.4% had asthma-like symptoms, 20.3% had chocking sensation on taking fluid, 23.2% had recurrent sore throat, 26.1% had frequent throat clearing and 37.7% had hoarseness. Dental erosion was found 36.2% out of which 19% were mild erosion, 7.2% moderate erosion and 1.4% had severe dental erosion. See Table 4.
Discussion
In Nigeria most studies on GERD were carried out on patients referred for upper gastrointestinal endoscopy where diagnosis was limited to patients with endoscopically defined lesions [12]. However, only about 50% of patients with GERD will have endoscopically detectable lesions [13, 14]. Therefore, the use of a patient-centred, symptom driven approach to diagnosis which is independent of endoscopic findings is more appropriate. The prevalence of GERD in a similar community-based study carried out in Nigeria was 26.34% which is comparable to 32.8% reported in this study though the studies were from different geo-political zones of Nigeria [15]. Even though the use of symptom-based questionnaire is advocated in the diagnosis of GERD, there are many of such questionnaires, each with its strength and weakness [16]. We decided to use Carlsson- Dent questionnaire though not validated but has been used by two previous studies in Nigeria [15, 17]. When compared with GERD Q, another commonly used GERD questionnaire. Carlsson-Dent questionnaire was found to be easier for patients to understand and answer although it detected less GERD in patients that were overweight and obese [18]. A Thai study found Carlsson-Dent questionnaire diagnosed GERD more than PH monitoring and endoscopy although a prospective, open label multi-centre Dutch study reported a poor diagnostic performance of Carlsson-Dent [19, 20].
GERD has been associated with many factors [21]. In this study, age had a significant association with GERD with participants between the age group of 19–22 years having reduced odds of having GERD compared with age groups 15–18 years and age group greater than 23 years.
Song et al. [22] reported an increased risk for GERD in overweight and obese subjects. However, in this study we found that there are increased odds of having GERD in both underweight and obese subjects. The association between GERD and underweight is surprising and we have no explanation for it. It may however be related to spices contents of food as suggested by Song et al. [22] .
Gastroesophageal reflux disease causes extra-oesophageal symptoms by direct and indirect mechanisms. The direct is by aspiration while the indirect is vagally mediated [23, 24].
Chronic cough is a known extra-oesophageal manifestation of GERD. The most common causes of chronic cough in non-smoking patients with normal chest radiographs, who are not on angiotensin converting enzyme (ACE) inhibitors are post nasal drip syndrome (PNDS), asthma, gastroesophageal reflux and chronic bronchitis [25]. In our study, 8.7% of the study population had chronic or recurrent cough while 15.9% had nocturnal cough. Poe et- al found that GERD alone accounted for cough in 13% of their study population, while in 56% of patients, it was a contributing factor to persistence of cough [26].
Gastroesophageal reflux disease is a known aetiology of laryngeal inflammation otherwise called laryngopharyngeal reflux (LPR) [27]. Symptoms of LPR include hoarseness, throat pain, cough, hawking, dysphagia, odynophagia and voice fatigue. However, these symptoms are nonspecific and can also be seen in other patients with postnasal drip and those exposed to allergens and smoke. In our study population, 37.7% of students with GERD had hoarseness and 10.1% of them had dysphagia. Dysphagia in these group of patients is likely due to GERD as there are no demographic or clinical features suggestive of other oesophageal diseases like carcinoma.
Asthma has a strong correlation with GERD and the conditions seem to induce each other. Both epidemiologic studies and physiologic testing with ambulatory 24-h pH monitoring have established association between GERD and asthma [27, 28]. In our study we found asthma-like symptoms in 17.4% of students with GERD. In studying the prevalence of GERD in asthma patients, Kijander et al. found that though 35% of GERD related patients did not express the typical reflux symptoms, they had abnormal oesophageal acid exposure by pH monitoring [29] Similarly, Legget et al. conducted a study assessing GERD in patients whose asthma was difficult to control using 24-h ambulatory pH probes [30] .They reported that 55% had reflux at the distal probe while 35% had proximal probe reflux [31]. They therefore submitted that reflux occurs commonly in asthma patients.
This study also showed that dental erosion is a significant finding in subjects with GERD. A larger percentage of study participants in this study had dental erosion which is due to the gastric acidity [32]. . A study by Oginni et al. [33] reported that tooth wear index (TWI) scores were higher in patients with GERD than in control subjects. The frequency of regurgitation and duration of gastroesophageal reflux directly influence the severity of dental erosion.
In addition, other oral features such coated tongue, Xerostomia and halitosis were reported in some of the study participants with GERD. Impaired lower esophageal sphincter function also results in gas and stomach contents entering the esophagus resulting in halitosis.
Our study has some limitations. Information on the lifestyles of the Participants were not collected in this study because two other studies had focused on GERD, diet and lifestyles in our population [15, 17] and we felt there was no need for repetition. Secondly, Participants in this study, being University students, cannot be said to be representative of the Nigerian population. Lastly, we studied extra-oesophageal manifestations in those diagnosed with GERD only. Extra oesophageal symptoms of GERD have been reported without the typical symptoms of heartburns and or GERD [34]. Though there is no gold standard test for the association between extraoesophageal symptom and GERD, it is necessary, in our opinion, to do oesophageal PH monitoring and or GI endoscopy with oesophageal biopsy to be able to attribute those features to GERD since these extra oesophageal symptoms are not pathognomonic of GERD [28]. In this study, we did not do either of oesophageal PH monitoring or upper GI endoscopy for the Participants.
Conclusion
This study reported GERD prevalence of 32.8% and it identified age as the only predictor of GERD among the participants. Extra oesophageal features are quite common among the participants with GERD.
Availability of data and materials
The data sets used and /or analysis during the current study are available from the corresponding author on request.
Abbreviations
- GERD:
-
Gastro Esophageal Reflux Disease
- BMI:
-
Body Mass Index
- LPR:
-
Laryngopharyngeal Reflux
- ACE:
-
Angoitensin Converting Enzyme
- PNDS:
-
Post Nasal Drip Syndrome
- BUHREC:
-
Babcock University Health Research Ethics Committee
References
Vankil N, Veldhuyzen van Zanten S, Kahrilas P, et al. The montreal definition and classification of gastroesophageal reflux disease (GERD)- a global evidence based consensus. Am J Gastroenterol. 2006;101:1900–20.
Shaheen N, Ransohoff D. F. Gastroesophageal reflux, Barret oesophagus and oesophageal cancer: scientific review. JAMA. 2002;287(15):1972–81.
Hungin AP, Raghunath A, Wiklund I. Beyond heartburn: a review of the spectrum of reflux- induced disease. Fam Pract. 2005;6:591–603 Epub 2005 Jul 15.
Etraoesophageal presentation of gastroesophageal reflux disease. Semin Gastrointest Dis. 1997;(4):210.
Wong RK, Hanson DG, Waring PJ, et al. ENT manifestations of gastroesophageal reflux. AMJ Gastroenterol. 2000;95(8 suppl):S15–22.
Harding SM. Recent clinical investigations examining the association of asthma and gastroesophageal reflux. AMJ med. 2003;115(Suppl 3A):395–44S.
Vaezi MF. Sensitivity and Specificity of reflux- attributed laryngeal lessions: experimental and clinical evidence. AMJ. 2003;115(Suppl 3 A):975–104S.
Imfeld T. Dental erosion. Definition, classification and links. Eur J Oral Sci. 1996;2(pt2):151–5.
Kenneth RD. Extraesophageal symptoms of GERD. Cleve Clin J Med. 2003;70(Suppl. 5):S20–32.
Carlsson R, Dent J, Boiling- Stervenald E, Johnson F, Junghard O, Lauristsen K, Lundell L. The usefulness of a structured questionnaire in the assessment of a symptomatic gastroesophageal reflux disease [research support, Non US Gov’t]. Scand J Gastroenterol. 1998;33(10):1023–9.
Smith BG, Knight JK. An index for measuring the wear of teeth. Br Dent J. 1984;156:435–8.
Danbauchi SS, Keshinro IB, Abdu-Gasua K. Fifteen years of upper gastrointestinal endoscopy in Zaria (1978–1993). Afr J Med Sci. 1999;28(1–2):87–90.
Katz PO. Pathogenesis and management of gastroesophageal reflux disease. J ClinGastroenterol. 1991;13(Suppl2):S6–15.
Fass R, Fennerty MB, Vakil N. Nonerosive reflux disease current concepts and dilemmas. Am J Gastroenterol. 2001;96(2):303–14.
Nwokediuko S. Gatroesophageal Reflux Disease: a population based study. Gastroenterology Res. 2009;2(3):152–6 Epub 2009 May20.
Fass R. Symptom assessment tools for gastroesophageal reflux disease (GERD) treatment. J Clin GAastroenterol. 2007;41(5):437–44.
Akande KO, Fadupin GT, Akinola MA. The prevalence of gastro-oesophageal reflux disease and its relationship with diet and obesity among public school teachers in Abeokuta, south-west Nigeria. Tanzan J Health Res. 2018;20(4):1 [cited 2020 Mar 5]Available from: https://www.ajol.info/index.php/thrb/article/view/173050.
Contreras-Omaña R, Sánchez-Reyes O, Ángeles-Granados E. Comparación de los cuestionarios Carlsson-Dent y GERD-Q para detección de síntomas de enfermedad por reflujo gastroesofágico en población general. Rev Gastroenterol Mex. 2017;82(1):19–25.
Netinatsunton N, Attasaranya S, Ovartlarnporn B, Sangnil S, Boonviriya S, Piratvisuth T. The value of Carlsson-Dent questionnaire in diagnosis of Gastroesophageal reflux disease in area with low prevalence of Gastroesophageal reflux disease. J Neurogastroenterol Motil. 2011;17(2):164–8.
Numans ME, Wit NJD. Reflux symptoms in general practice: diagnostic evaluation of the Carlsson–Dent gastro-oesophageal reflux disease questionnaire. Aliment Pharmacol Ther. 2003;17(8):1049–55.
Ghanaei FM, Joukar F, Atshani SM, Chagharvand S, Souti F. The Epidemiology of GERD:a survey on the prevalence and the associated factors in a random sample of the general population in the Northern part of Iran. Int J Mol Epidemiol Genet. 2013;4(3):175–82 Published Sep12, 2013.
Song JH, Chung SJ, Lee JH, Kim YH, Chang DK, Son HJ, Kim JJ, Rhee JC, Rhee PL. Relationship between gastroesophageal reflux symptoms and dietary factors in Korea. J Neurogastroenterol Moti. 2011;17(1):54–60.
Agreus L, Svardsudd K, Talley NJ, Jones MP, Tibblin G. Natural history of gastroesophageal reflux disease and functional abdominal disorders: a population-based study. Am J Gastroenterol. 2001;96(10):2905–14.
Tuchman DN, Boyle JT, Pack AI, Scwartz J, Kokonos M, Spitzer AR, et al. Comparism of airway responses following tracheal or esophageal acidification in the cat. Gastroenterology. 1984;87(4):872–81 Epub 1984/10/01.
Poe RH, Kallay MC. Chronic cough and gastroesophageal reflux disease; experience with specific therapy for diagnosis and treatment. Chest. 2003;123(3):679–84 Epub 2003/03/12.
Baldi F, Cappiello R, Cavoli C, Ghersi S, Torresan F, Roda E. Proton pump inhibitor treatment of patients with gastroesophageal reflux related chronic cough: a comparison between two different daily doses of lansoprazole. World J Gastroenterol. 2006;12(1):82–8 Epub 2006/01/28.
Vaezi MF. Laryngitis and gastroesophageal reflux disease: increasing prevalence or diagnostic tests? Am JGatroenterol. 2004;99(5):786–8 Epub 2004/05/07.36. Vavricka SR,Storck CA, Wildi SM.
Kiljander TO, Salomaa ER, Hietanen EK, Terho EO. Gastroesophageal reflux in asthmatics: A double-blind, placebo-controlled crossover study with omeprazole. Chest. 1999;116(5):1257–64 Epub 1999/11/13.
Ahmed T, Vaezi MF. The role of pH monitoring in extraesophageal gastroesophageal reflux disease. Gastrointest Endosc Clin N Am. 2005;15(2):319–31 Epub 2005/02/22.
Leggett JJ, Johnson BT, Mills M, Gamble J, Heaney LG. Prevalence of gastroesophageal reflux in difficult asthma: relationship to asthma outcome. Chest. 2005;127(4):1227–31.
Meir JH, McNally PR, Punja M, Freeman SR, Sudduth RH, Stocker N, et al. Does omeprazole (Prilosec) improve respiratory function in asthmatics with gastroesophageal reflux ? A double-blind, placebo-controlled crossover study. Dig Dis Sci. 1994;39(10):2127–33 Epub 1994/10/01.
Barlett DW, Evans DF, Smith BGN. The relationship between gastro-esophageal reflux disease and dental erosion. J Oral Rehabil. 1996;23(5):289–97.
Oginni A, Agbakwuru E, Ndububa D. The prevalence of dental erosion in Nigeria patients with gastro-esophageal disease. BMC Oral Health. 2005;5:1–6.
Oh JH. Atypical symptoms are related to typical symptoms rather than histologic and endoscopic esophagitis. J Neurogastroenterol Motil. 2012;18(3):233–5.
Acknowledgements
We wish to acknowledge Dr. Adeyeye Rachael Adetola who contributed to proof reading and editing of the final manuscript.
This work is original and all the authors have given consent for submission and publication in this journal.
This manuscript has not been submitted or published anywhere else.
Funding
The research was funded solely by financial contributions made by the authors.
Author information
Authors and Affiliations
Contributions
AMA: Development of the research protocol, securing of ethical approval and collection of data. OTA: Collection and analysis of data from participants. AK: Initial write up of research protocol and correction of research proposal and manuscript. OOY: collection of data and initial write up of the manuscript. SOF: Collection of data and initial write up of research proposal. AAM: Data collection. AAD: Mobilization of students and data collection. All authors have read and approve the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Ethical approval for this study was obtained from Babcock University Health Research and Ethics committee (BUHREC 002/2017). Written informed consent for the study was also obtained from all the participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Akinola, M.A., Oyedele, T.A., Akande, K.O. et al. Gastroesophageal reflux disease: prevalence and Extraesophageal manifestations among undergraduate students in South West Nigeria. BMC Gastroenterol 20, 160 (2020). https://doi.org/10.1186/s12876-020-01292-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12876-020-01292-1