Abstract
Background
We assess whether short-term recovery of urinary incontinence following robot-assisted laparoscopic radical prostatectomy (RARP) is associated with postoperative membranous urethral length (MUL) and position of vesico-urethral anastomosis (PVUA).
Methods
Clinical variables including PVUA and pre-and postoperative MUL were evaluated in 251 patients who underwent RARP from August 2019 to February 2021. Continence recovery was defined as no pad or one security liner per day assessed by patient interview at least 6 months follow-up. Univariate and multivariate logistic regression analyses were used to assess variables associated with continence recovery at 3 months after the operation.
Results
Continence recovery rates at 3 and 6 months were 75% and 84%, respectively. Lower BMI (< 25 kg/m2) (p = 0.040), longer preoperative MUL (≥ 9.5 mm) (p = 0.013), longer postoperative MUL (≥ 9 mm) (p < 0.001), higher PVUA (< 14.5 mm) (p = 0.019) and shorter operating time (< 170 min) (p = 0.013) were significantly associated with continence recovery at 3 months in univariate analysis. Multivariate analysis revealed that postoperative MUL (OR 3.75, 95% CI 1.90–7.40, p < 0.001) and higher PVUA (OR 2.02, 95% CI 1.07–3.82, p = 0.032) were independent factors for continence recovery. Patients were divided into 3 groups based on the multivariate analysis, with urinary continence recovery rates found to have increased in turn with rates of 43.7% versus 68.2% versus 85.0% (p < 0.001) at 3 months.
Conclusions
PVUA and postoperative MUL were significant factors for short-term continence recovery. Preservation of urethral length might contribute to continence recovery after RARP.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Background
Radical prostatectomy (RP) is the principal surgical treatment for localized prostate cancer. The aim of RP is to achieve oncologic and functional outcomes, including urinary continence preservation [1]. Reported percentages of patients with post-operative incontinence range from 6 to 20% [2,3,4]. Although the recent introduction of robot-assisted radical prostatectomy (RARP) has been reported to allow for more accurate anatomical information on peri-prostatic structures, postoperative continence outcomes have not improved compared to open radical prostatectomy (ORP). According to a randomized clinical trial, there was no difference in functional outcomes between RARP and ORP, with a slight difference in biochemical recurrence rates in favor of RARP [5]. A Swedish LAPPRO trial also reported the slight benefit of RARP over ORP regarding erectile dysfunction but not urinary continence or oncologic outcomes [6]. Even in the era of RARP, urinary incontinence remains a problem that needs to be improved.
Several factors including clinical characteristics: obesity [7, 8], membranous urethral length (MUL) [7, 9], and surgical techniques; neurovascular bundle (NVB) preservation [10, 11], reconstruction techniques [12], have been addressed for early recovery of continence. Of these factors, MUL is an important factor that directly correlates with the functional sphincter mechanism. Preservation of maximal MUL is important for postoperative urinary continence [13]. Particularly, postoperative MUL is considered to affect continence directly. In addition, bladder neck descent was recently reported to be associated with early continence outcome in retzius-sparing (RS) RARP [14]. Position of vesico-urethral anastomosis (PVUA) is also important, suggesting the importance of the surgical technique used. In particular, the relatively early recovery of urinary continence in RS-RARP is noteworthy and several studies have been reported [15, 16], and the assessment of continence recovery in the relatively early postoperative period is important.
In this study, we aimed to investigate the impact of postoperative MUL and PVUA on urinary continence recovery at a relatively early time point after RARP.
Methods
Study population
All procedures were approved by the institutional review board. A total of 251 patients who underwent RARP from August 2019 to February 2021 in National Cancer Center Hospital East (NCCHE) were analyzed retrospectively. All study protocols were approved by the Ethical Committee of NCCHE. All patients provided informed consent before surgery.
Surgical technique
Surgery was conducted only in patients with localized and locally advanced prostate cancer without distant metastasis. Procedures were performed by 6 surgeons including 4 novices of < 20 RARP experiences, all of which were supervised by a senior surgeon (H. M.) using the da Vinci Xi robotic system (Intuitive Surgical, Sunnyvale, CA, USA), basically via transperitoneal anterior approach. The extraperitoneal approach was chosen only for patients with past open abdominal surgery. The decision to perform nerve preservation was made based on the wishes of the individual patient and the judgment of the attending physician after explaining that it would contribute to preservation of erectile function and urinary continence, assuming that the tumor was not in close proximity to the NVB on MRI. In all cases, puboprostatic ligament preservation was routinely performed. Dissection of the prostatic apex was performed with a sharp and direct division of the membranous urethra at the level of the urethroprostatic junction [17]. Rocco stitch was used for posterior reconstruction of the Denonvillier’s fascia [18, 19]. Vesico-urethral anastomosis was performed by using a 3/0 ‘‘barbed’’ running suture, starting at 5 o’clock on the urethra and then proceeding clockwise. The anterior reconstruction was performed by suturing visceral to parietal layers of the endopelvic fascia to recreate the pubovesical ligaments [12].
The outcome measured was continence recovery at 3 months after catheter removal, as previously repored [20, 21]. Urethral catheter was removed between postoperative day 5 to 7. Continence recovery was defined as no pad or 1 security liner per day by self-report, which was obtained during outpatient visit at 1, 3 and 6 months after operation.
Data collected
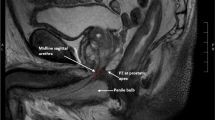
The following clinical variables were evaluated: age, preoperative serum prostate-specific antigen (PSA) level, prostate volume, biopsy Gleason score (GS), clinical T stage, N stage, pre- and postoperative MUL, preoperative androgen deprivation therapy (ADT). Operation approach, neurovascular bundle (NVB) sparing, pelvic lymph node dissection (PLND), operation time and estimated blood loss (EBL) were recorded to assess perioperative parameters. The cut-off points of age, BMI, prostate volume, pre- and postoperative MUL, PVUA, operation time and EBL were set at 70-year-old, 25 kg/m2, 50 ml, 9.5 mm, 9 mm, 14.5 mm, 170 min and 200 ml by calculating the maximum Youden index with the highest value of “sensitivity − (1 − specificity)” in the receiver operating characteristics (ROC) analysis using continence recovery at 3 month after RARP as an endpoint, respectively. Risk classification was defined according to the European Association of Urology guidelines, in which clinical tumor stage (cT) 3–4 or clinical node stage (cN) 1 disease was classified as locally advanced disease [22]. ADT was offered as a part of multimodal therapy to most patients with locally advanced disease by the discretion of the outpatient physician. Preoperative MUL was estimated by measuring the length from the prostatic apex to the level of the urethra at the penile bulb in the midline sagittal plane of T2‑weighted magnetic resonance imaging of the prostate [9]. The postoperative MUL and PVUA were evaluated by the length of the sphincter portion which is measured the distance from the beginning of the urethral sphincter contraction to the release area in the absence of abdominal pressure, and vertical distance from the superior border of the pubic symphysis to the vesico-urethral anastomosis of the cystourethrography at the time of catheter removal (Fig. 1).
Statistical analysis
Differences in the distribution of variables between groups were evaluated using a chi-square test or Fisher’s exact probability test. Uni- and multivariate logistic regression analyses were used to evaluate parameters associated with recovery of urinary continence after RARP at 3 months. A reduced multivariate model was developed using the stepwise backward method, in which the variable with the highest p-value was eliminated from each iteration of the multivariate analysis. Two-tailed p < 0.05 was regarded as significant. Statistical analyses were performed using JMP software version 13 (SAS Institute, Cary, NC, USA).
Results
Characteristics of all 251 patients are summarized in Table 1. Median (range) age at the time of RARP and PSA were 72 (51–81) years and 8.2 (1.9–148) ng/mL. The numbers of patients with low- or intermediate-risk, high-risk, and locally advanced disease were 105 (41.8%), 83 (33.1%) and 63 (25.1%), respectively. Preoperative ADT was given to 97 (38.6%) patients. Pre- and postoperative MUL were 11.2 (5.8–18.8) and 10.1 (4.2–15.8) mm. Pathological T3 or ypT3 or more and positive surgical margin (PSM) were 55 patients (21.9%) and 59 patients (23.5%), respectively.
Continence recovery rates at 3-month and 6-month were 76.5% and 84.8%, respectively.
Lower BMI (< 25 kg/m2) (p = 0.040), longer preoperative MUL (≥ 9.5 mm) (p = 0.013), longer postoperative MUL (≥ 9 mm) (p < 0.001), higher PVUA (< 14.5 mm) (p = 0.019) and shorter operating time (< 170 min) (p = 0.013) were significantly associated with continence recovery at 3 months in univariate analysis. Multivariate analysis revealed that postoperative MUL (OR 3.75, 95% confidence interval [CI] 1.90–7.40, p < 0.001) and PVUA (OR 2.02, 95% CI 1.07–3.82, p = 0.032) were independent factors for continence recovery at 3 months (Table 2). In addition, only MUL was an independent factor for continence recovery at 6 months after surgery (OR 6.3, 95% CI 3.01–13.39, p < 0.001, not shown in the table).
Based on the results of the multivariate analysis, we divided the patients into three groups according to the score predicting continence recovery at 3 months as follows: score = 1 (if PVUA < 14.5 mm) + 1 (if postoperative MUL > 9 mm). As shown in Fig. 2, the urinary continence recovery rates were increased in turn with rates of 43.7% versus 68.2% versus 85.0% (p < 0.001) at 3 months.
Discussion
Recent advances in our knowledge of surgical anatomy have led to a better understanding of the mechanisms of urinary incontinence [23]. In terms of MUL, a systematic review and meta-analysis reported that preoperative MUL measurement is important for postoperative urinary continence [9]. In the present study, we demonstrated that PVUA and postoperative MUL measurement by cystourethrography are independently associated with continence recovery among men undergoing RARP, and the presence or absence of these factors significantly changes the relatively short-term urinary continence recovery rate.
Postoperative MUL as measured on cystourethrography has been reported to be the most important factor for recovery of urinary continence in the early postoperative period after RARP. It is greater with preservation of the neurovascular bundle, allowing for early recovery of urinary continence [24]. Postoperative MUL as measured on MRI has also been reported to be the most important predictive factor for recovery of urinary continence in the early postoperative period after RARP [21]. MRI measurements are accurate, but costly to perform in all patients postoperatively. Cystourethrography can be performed at the same time as vesico-urethral anastomosis evaluation in the early postoperative period and can predict the recovery of continence in almost all individual patients. Since a recent retrospective study, in which preoperative MUL was assessed by MRI and postoperative MUL was assessed by cystourethrography for the patients who underwent both ORP and RARP, reported that the superiority of postoperative MUL measurement over preoperative MUL for continence recovery [25], we consider postoperative MUL assessment to be an important test. Kojima et al. reported that the urethral sphincter system and periurethral support system are two main mechanisms of preserving urinary continence [26]. These sphincter components consist of an inner smooth muscle layer (longitudinal and circular smooth muscle) and a striated urogenital sphincter muscle (rhabdosphincter). In the current study, the MUL mainly evaluated the condition of the rhabdosphincter status. However, this sphincter is originally located from the prostate apex to the urethra at the level of the penile bulb [27], preservation of maximal urethral length is preferable during operation.
Our data also revealed that the importance of the PVUA for the recovery of urinary incontinence after RARP. This is because the height of the bladder neck position plays a significant role in incontinence. Olgin et al., first described the measurement method, bladder neck to pubic symphysis ratio, calculated by measuring the distance from the superior edge of the pubic symphysis to the bladder neck and dividing it by the total pubic symphysis height, in prediction of postoperative continence in patient received RARP [28]. Similarly for the postoperative outcome of RS-RARP, less bladder neck descent at postoperative cystography has been reported to be associated with better early continence outcomes compared to standard RARP [14]. The retzius-sparing technique preserves the anatomy of the pelvic floor and therefore the position of the vesicourethral anastomosis is higher than standard RARP. According to a systematic review and analysis, RS-RARP reported a statistically significant advantage in terms of continence recovery including relatively early period after operation [15]. A randomized study which first underwent long-term clinical trial reported that RS-RARP achieved a higher continence rate at 3 months postoperatively than the standard technique, which was muted at 12 months follow-up [16].
In our results, NVBP was not associated with urinary continence recovery, partly due to the small number of NVBP cases. The benefit of NVBP on urinary continence remains controversial. A large multicenter study of more than 3000 patients who underwent ORP or RARP indicated that the degree of bilateral NVBP predicted recovery of urinary continence 1 year after surgery, and that bilateral NVBP was beneficial for urinary continence [29]. On the other hand, a meta-analysis revealed that NVBP improves urinary continence in the first 6 months after surgery, thereafter, there is no difference in continence between men who had these nerves removed and those who had them saved [10]. In contrast, it has also been reported that it is not the nerve preservation itself, but the careful surgical technique that accompanies the nerve preserving operation that is key in improving recovery of incontinence [30]. Our results on nerve preservation also support this theory of careful dissection in some sense.
The current study demonstrated that when both higher PVUA and longer postoperative MUL are present, the recovery rate of urinary continence reaches 85% even with standard RARP. We showed that there is a clear difference in the recovery of urinary continence regardless of whether these items are aligned. Although it is not directly comparable, our method of PVUA evaluation is simpler than Olgin’s method. In addition, it is possible to evaluate the length of the urethra at the same time, so that incontinence can be predicted from two important factors. Intraoperative maximal MUL preservation contributes to the recovery of urinary incontinence in terms of both position and length.
At our institution, the number of cT1c cases were low and the rate of PSM was high. The reason for the former is that many of low-risk patients desired active surveillance as initial treatment. The latter reason was that many of patients underwent RARP were classified as intermediate to high risk, and even when high-risk patients requested NVB sparing, intra or inter-fascial NVB spearing was performed unless MRI-positive lesion contacted the prostatic capsule.
The present study has several limitations. Firstly, this study has the weaknesses characteristic of a retrospective design. Secondly, the number of patients was not so large. Thirdly, urinary continence status was evaluated based on the self-reported number of pads patients used, which is subjective. Instead, 24-h pad weight is desirable for accurate measurement [3]. Fourthly, the two factors obtained in the results of this study are postoperative factors. However, they are factors that can be determined at the time of removal of the urinary catheter, which is relatively early after surgery, so they are highly useful in actual clinical practice. Fifthly, since the PVUA is measured vertically from the suprapubic margin, it basically does not affect the angle of rotation during cystourethrography, but the possibility of a slight error cannot be denied. Sixthly, it is possible that the results of the analysis did not reflect nerve preservation due to the small number of cases. Finally, there were multiple surgeons with varying years of experience. The surgeon's learning curve in RARP has been suggested to affect the recovery of incontinence [31]. However, all surgeons used the same technique with little variability. In particular, when inexperienced surgeons perform surgery, all procedures are performed under the supervision of a senior physician in all cases to minimize differences in skill at our institution, and we believe that evaluating the results of multiple surgeons is realistic.
Conclusions
PVUA and postoperative MUL were significant factors for short term continence recovery. Preservation of MUL for as long as possible might be important in determining favorable outcome of postoperative urinary continence.
Availability of data and materials
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
Abbreviations
- ADT:
-
Androgen deprivation therapy
- CI:
-
Confidence interval
- EBL:
-
Estimated blood loss
- GS:
-
Gleason score
- MUL:
-
Membranous urethral length
- NVB:
-
Neurovascular bundle
- ORP:
-
Open radical prostatectomy
- OR:
-
Odds ratio
- PSA:
-
Prostate-specific antigen
- PLND:
-
Pelvic lymph node dissection
- PVUA:
-
Position of vesico-urethral anastomosis
- RARP:
-
Robot-assisted laparoscopic radical prostatectomy
References
Orvieto MA, DeCastro GJ, Trinh QD, Jeldres C, Katz MH, Patel VR, et al. Oncological and functional outcomes after robot-assisted radical cystectomy: critical review of current status. Urology. 2011;78(5):977–84.
Majoros A, Bach D, Keszthelyi A, Hamvas A, Mayer P, Riesz P, et al. Analysis of risk factors for urinary incontinence after radical prostatectomy. Urol Int. 2007;78(3):202–7.
Bauer RM, Gozzi C, Hubner W, Nitti VW, Novara G, Peterson A, et al. Contemporary management of postprostatectomy incontinence. Eur Urol. 2011;59(6):985–96.
Sanda MG, Dunn RL, Michalski J, Sandler HM, Northouse L, Hembroff L, et al. Quality of life and satisfaction with outcome among prostate-cancer survivors. N Engl J Med. 2008;358(12):1250–61.
Coughlin GD, Yaxley JW, Chambers SK, Occhipinti S, Samaratunga H, Zajdlewicz L, et al. Robot-assisted laparoscopic prostatectomy versus open radical retropubic prostatectomy: 24-month outcomes from a randomised controlled study. Lancet Oncol. 2018;19(8):1051–60.
Nyberg M, Hugosson J, Wiklund P, Sjoberg D, Wilderang U, Carlsson SV, et al. Functional and oncologic outcomes between open and robotic radical prostatectomy at 24-month follow-up in the Swedish LAPPRO Trial. Eur Urol Oncol. 2018;1(5):353–60.
Matsushita K, Kent MT, Vickers AJ, von Bodman C, Bernstein M, Touijer KA, et al. Preoperative predictive model of recovery of urinary continence after radical prostatectomy. BJU Int. 2015;116(4):577–83.
Wolin KY, Luly J, Sutcliffe S, Andriole GL, Kibel AS. Risk of urinary incontinence following prostatectomy: the role of physical activity and obesity. J Urol. 2010;183(2):629–33.
Mungovan SF, Sandhu JS, Akin O, Smart NA, Graham PL, Patel MI. Preoperative membranous urethral length measurement and continence recovery following radical prostatectomy: a systematic review and meta-analysis. Eur Urol. 2017;71(3):368–78.
Reeves F, Preece P, Kapoor J, Everaerts W, Murphy DG, Corcoran NM, et al. Preservation of the neurovascular bundles is associated with improved time to continence after radical prostatectomy but not long-term continence rates: results of a systematic review and meta-analysis. Eur Urol. 2015;68(4):692–704.
Avulova S, Zhao Z, Lee D, Huang LC, Koyama T, Hoffman KE, et al. The effect of nerve sparing status on sexual and urinary function: 3-year results from the CEASAR study. J Urol. 2018;199(5):1202–9.
Vis AN, van der Poel HG, Ruiter AEC, Hu JC, Tewari AK, Rocco B, et al. Posterior, anterior, and periurethral surgical reconstruction of urinary continence mechanisms in robot-assisted radical prostatectomy: a description and video compilation of commonly performed surgical techniques. Eur Urol. 2019;76(6):814–22.
Song W, Kim CK, Park BK, Jeon HG, Jeong BC, Seo SI, et al. Impact of preoperative and postoperative membranous urethral length measured by 3 Tesla magnetic resonance imaging on urinary continence recovery after robotic-assisted radical prostatectomy. Can Urol Assoc J. 2017;11(3–4):E93–9.
Chang LW, Hung SC, Hu JC, Chiu KY. Retzius-sparing robotic-assisted radical prostatectomy associated with less bladder neck descent and better early continence outcome. Anticancer Res. 2018;38(1):345–51.
Checcucci E, Veccia A, Fiori C, Amparore D, Manfredi M, Di Dio M, et al. Retzius-sparing robot-assisted radical prostatectomy vs the standard approach: a systematic review and analysis of comparative outcomes. BJU Int. 2020;125(1):8–16.
Menon M, Dalela D, Jamil M, Diaz M, Tallman C, Abdollah F, et al. Functional recovery, oncologic outcomes and postoperative complications after robot-assisted radical prostatectomy: an evidence-based analysis comparing the Retzius sparing and standard approaches. J Urol. 2018;199(5):1210–7.
Bianchi L, Turri FM, Larcher A, De Groote R, De Bruyne P, De Coninck V, et al. A novel approach for apical dissection during robot-assisted radical prostatectomy: the “Collar” technique. Eur Urol Focus. 2018;4(5):677–85.
Rocco F, Carmignani L, Acquati P, Gadda F, Dell’Orto P, Rocco B, et al. Restoration of posterior aspect of rhabdosphincter shortens continence time after radical retropubic prostatectomy. J Urol. 2006;175(6):2201–6.
Rocco B, Gregori A, Stener S, Santoro L, Bozzola A, Galli S, et al. Posterior reconstruction of the rhabdosphincter allows a rapid recovery of continence after transperitoneal videolaparoscopic radical prostatectomy. Eur Urol. 2007;51(4):996–1003.
Koga F, Ito M, Kataoka M, Fukushima H, Nakanishi Y, Takemura K, et al. Novel anatomical apical dissection utilizing puboprostatic “open-collar” technique: impact on apical surgical margin and early continence recovery. PLoS ONE. 2021;16(4):e0249991.
Nakane A, Kubota H, Noda Y, Takeda T, Hirose Y, Okada A, et al. Improvement in early urinary continence recovery after robotic-assisted radical prostatectomy based on postoperative pelvic anatomic features: a retrospective review. BMC Urol. 2019;19(1):87.
Mottet N, Cornford P, van den Bergh RCN, Briers E, UOMO) EPAEPCCE, De Santis M, et al. EAU-EANM-ESTRO-ESUR-ISUP-SIOG guidelines on prostate cancer. 2021.
Walz J, Epstein JI, Ganzer R, Graefen M, Guazzoni G, Kaouk J, et al. A critical analysis of the current knowledge of surgical anatomy of the prostate related to optimisation of cancer control and preservation of continence and erection in candidates for radical prostatectomy: an update. Eur Urol. 2016;70(2):301–11.
Haga N, Ogawa S, Yabe M, Akaihata H, Hata J, Sato Y, et al. Factors contributing to early recovery of urinary continence analyzed by pre- and postoperative pelvic anatomical features at robot-assisted laparoscopic radical prostatectomy. J Endourol. 2015;29(6):683–90.
Cho DS, Choo SH, Kim SJ, Shim KH, Park SG, Kim SI. Postoperative membranous urethral length is the single most important surgical factor predicting recovery of postoperative urinary continence. Urol Oncol. 2020;38(12):930 e7-930 e12.
Kojima Y, Takahashi N, Haga N, Nomiya M, Yanagida T, Ishibashi K, et al. Urinary incontinence after robot-assisted radical prostatectomy: pathophysiology and intraoperative techniques to improve surgical outcome. Int J Urol. 2013;20(11):1052–63.
Hakimi AA, Faleck DM, Agalliu I, Rozenblit AM, Chernyak V, Ghavamian R. Preoperative and intraoperative measurements of urethral length as predictors of continence after robot-assisted radical prostatectomy. J Endourol. 2011;25(6):1025–30.
Olgin G, Alsyouf M, Han D, Li R, Lightfoot M, Smith D, et al. Postoperative cystogram findings predict incontinence following robot-assisted radical prostatectomy. J Endourol. 2014;28(12):1460–3.
Steineck G, Bjartell A, Hugosson J, Axen E, Carlsson S, Stranne J, et al. Degree of preservation of the neurovascular bundles during radical prostatectomy and urinary continence 1 year after surgery. Eur Urol. 2015;67(3):559–68.
Michl U, Tennstedt P, Feldmeier L, Mandel P, Oh SJ, Ahyai S, et al. Nerve-sparing surgery technique, not the preservation of the neurovascular bundles, leads to improved long-term continence rates after radical prostatectomy. Eur Urol. 2016;69(4):584–9.
Ficarra V, Novara G, Rosen RC, Artibani W, Carroll PR, Costello A, et al. Systematic review and meta-analysis of studies reporting urinary continence recovery after robot-assisted radical prostatectomy. Eur Urol. 2012;62(3):405–17.
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
YN, SM, NO, KT, MK, SY, HM, conception and design: YN, enrollment of patients and acquisition of data: YN, SM, NO, KT, MK and SY, drafting of the manuscript: YN and HM, statistical analysis: YN, analysis and interpretation of data: YN and HM, supervision: HM. We confirm that all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All patients were fully informed of the disease, examinations, operative procedures, and complications, and were required to sign a written informed consent form before undergoing any procedures. All study protocols were approved by the Ethical Committee of National Cancer Center Hospital East (approval no. 2018-159).
Consent for publication
Not applicable.
Competing interests
No potential competing interest are disclosed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Nakanishi, Y., Matsumoto, S., Okubo, N. et al. Significance of postoperative membranous urethral length and position of vesicourethral anastomosis for short-term continence recovery following robot-assisted laparoscopic radical prostatectomy. BMC Urol 22, 145 (2022). https://doi.org/10.1186/s12894-022-01097-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12894-022-01097-2