Abstract
Background
The IL-23/IL-17 axis plays an important role in the immunopathogenesis of periodontal disease. A systematic review was conducted to synthesize all research reporting on the levels of the IL-23/IL-17 axis in gingival crevicular fluid (GCF) from subjects with gingivits, and periodontitis, compared to healthy controls.
Methods
The protocol followed the PRISMA, and Cochrane guidelines, and was registered with the Open Science Framework (OSF): https://doi.org/10.17605/OSF.IO/7495V. A search was conducted in the electronic databases PubMed/MEDLINE, Scopus, Google Schoolar, and Cochrane from November 15th, 2005, to May 10th, 2023. The quality of the studies was assessed using the JBI tool for cross-sectional studies.
Results
The search strategy provided a total of 2,098 articles, of which 12 investigations met the inclusion criteria. The total number of patients studied was 537, of which 337 represented the case group (subjects with gingivitis, and chronic periodontitis), and 200 represented the control group (periodontally healthy subjects). The ages of the patients ranged from 20 to 50 years, with a mean (SD) of 36,6 ± 4,2, of which 47% were men, and 53% were women. 75% of the investigations collected GCF samples with absorbent paper strips, and analyzed cytokine IL-17 levels individually. In addition, qualitative analysis revealed that there are differences between IL-23/IL-17 axis levels in subjects with chronic periodontitis, gingivitis and healthy controls.
Conclusions
Thus, IL-23/IL-17 axis levels could be used in the future as a diagnostic tool to distinguish between periodontal diseases.
Similar content being viewed by others
Introduction
Periodontal disease is an umbrella term for a group of diseases that affect the supporting tissues of the teeth (gingiva, root cementum, periodontal ligament and alveolar bone) [1]. The two most common forms of clinical presentation are gingivitis and periodontitis [2]. Gingivitis involves inflammation of the gingiva without apparent changes in clinical attachment levels. In fact, almost half of the world’s population suffers from this condition [3], and if gingivitis is not resolved, a part of gingivitis progresses and gives rise to periodontitis, which corresponds to an inflammatory and destructive process of the periodontium [4]. Both conditions occur in response to a dysbiotic polymicrobial challenge with a high prevalence of periodontopathogens such as Porphyromonas gingivalis, Tannerella forsythia, and Treponema denticola, accompanied by an aberrant immune response, in a genetically susceptible host [5]. Periodontitis is the sixth most common osteolytic disease affecting humans [6] and currently has a prevalence of 62.3%, and in its most severe form can affect up to 23.6% of the world’s population [7]. For didactic purposes, periodontitis is classified into chronic and aggressive [8, 9].
Periodontal probing and radiographic evaluation are considered the gold standard for establishing the diagnosis of periodontal disease [10]. However, clinical parameters and radiographic diagnosis only represent the sequelae of a previous bacterial challenge, and alone do not assess the onset and progression of periodontal destructive changes [11]. In this sense, biomarkers are host-derived molecules whose main purpose is to identify the state of health or disease [12]. In the oral cavity, they can be detected in saliva, tissue biopsies, supra- and subgingival plaque, peri-implant gingival crevicular fluid (PICF), as well as in gingival crevicular fluid (GCF) [13]. The GCF consists of a complex mixture of serum-derived substances such as leukocytes and their products (cytokines, chemokines, enzymes), inorganic ions, structural cells of the periodontium and oral bacteria [14,15,16,17,18].
Inflammatory mediators are the most commonly studied type of biomarkers in periodontal diseases, and their importance lies in the fact that these molecules reflect the dynamics of inflammation, highlighting the innate and adaptive immune activity of the host [19] in response to microbial-associated molecular patterns (MAMPS) in the proinflammatory microenvironment of the gingival sulcus [20]. Among them, interleukin-23 (IL-23), and interleukin-17 (IL-17), have been investigated individually and together to elucidate their role in the pathogenesis of periodontal diseases [21]. In this context, one of the main functions of IL-23 is to regulate the differentiation of CD4 and CD8 (+) T naive cells to T helper 17 (Th17) cells [22], in turn Th17 cells produce tumor necrosis factor alpha (TNF-α), which promotes the development of myeloid cells, induces osteoclastic activity, inhibits osteoblastic activity and up-regulates the production of other cytokines/chemokines such as interleukin-6 (IL-6), interleukin-1 beta (IL-1β), interleukin-8 (IL-8), and ligan 1 (C-X3-C motif) (CX3CL1), creating sustained feedback loop, which enhances disease development [23]. IL-17 regulates the migration of neutrophils which phagocytize bacteria, release their extracellular traps (NETs) and with it lysosomal enzymes to fight pathogens [24, 25]. In addition, IL-17 acts synergistically with TNF-α and IL-1 to induce the release of receptor activator of NF-κB ligand (RANKL) that binds with its receptor RANK on the surface of preosteoclasts, producing osteoclastogenesis and initiating the process of bone destruction in periodontitis [26, 27].
Numerous studies have reported differences in IL-23/IL-17 axis levels in GCF [28,29,30,31,32,33,34,35,36,37,38,39] and in other biological samples such as gingival tissue [40], serum [22] and saliva [41] from subjects with gingivitis and periodontitis, suggesting that this axis is involved in the progression and severity of periodontal diseases [21]. Moreover, it has been shown that, IL-1β is considered a remarkable inflammatory biomarker in the development and progression of gingivitis [42] and periodontitis [43]. While, on the other hand, a close association between elevated TNF-α levels in GCF with periodontal disease has also been demonstrated, supporting its use as a potential biomarker for its diagnosis [44]. However, to date, it has not been determined whether the IL-23/IL-17 axis could be a practical and accurate indicator based on GCF analysis to distinguish between periodontal diseases.
Therefore, the objectives of the present study were:
-
1.
To perform a comprehensive systematic review of the literature and compile the available evidence on IL-23/IL-17 axis levels in GCF of subjects with periodontal disease.
-
2.
To identify whether IL-23/IL-17 axis can be used as a diagnostic tool to distinguish between periodontal diseases.
Materials and methods
Protocol registration
The present study followed the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA), [45] and Cochrane Handbook for Systematic Reviews guidelines [46]. The protocol was recorded with the OSF enrollment (Registration DOI: https://doi.org/10.17605/OSF.IO/7495V).
PICO focus question
The central question was formulated considering the PICO elements (Population, Intervention, Comparison, and Outcome).
-
P: Subjects with periodontal disease (gingivitis, and chronic periodontitis).
-
I: IL-23, and IL-17 levels in GCF can be used to differentiate between healthy, gingivitis, chronic periodontitis subjects.
-
C: Changes in IL-23, and IL-17 levels in GCF of healthy, gingivitis, and chronic periodontitis subjects.
-
O: There is a difference in the level of IL-23, and IL-17 between (1) healthy versus gingivitis; (2) gingivitis versus chronic periodontitis; (3) healthy versus chronic periodontitis.
The question was the following: Are there differences in IL-23 and IL-17 levels in GCF between chronic periodontitis, gingivitis and periodontally healthy subjects?
Eligibility criteria
Inclusion criteria
For this systematic review, inclusion criteria were as follows:
-
Original cross-sectional, and longitudinal clinical studies that analyzing IL-23, and IL-17 levels in GCF of subjects with gingivitis, and chronic periodontitis diagnosed according to clinical parameters.
-
Studies that analyzing the proteins by ELISA technique.
-
Studies that included systemically healthy subjects, without any comorbidities. Non-smokers, no antibiotics and/or immunosuppressors, as well as without orthodontic appliances.
-
Articles in English language.
-
Articles published after 2000.
Exclusion criteria
Exclusion criteria were as follows:
-
Experimental studies with animal models or cell lines that analyzing the levels of proinflammatory cytokines.
-
Quantification of IL-23/IL-17levels in saliva, serum or gingival tissue.
-
Unreported exact numbers of cytokines levels.
-
Studies that analyzing proteins by other techniques such as western blott, flow cytometry using a bead array system and/or immunoprecipitation.
-
Articles in a language other than English.
-
Articles published before 2000.
Search strategy
Two researchers (M.A.A.S and C.G.V) performed a comprehensive electronic search in the following databases: PubMed/MEDLINE, Scopus, Google Schoolar, and Cochrane from November 15th, 2005, to May 10th, 2023, with the main purpose of finding the most relevant articles according to the research topic and that previously met the study criteria. Table 1 shows the search terms used. Additionally, the digital search was complemented with an iterative manual search in journals such as: Journal of Periodontology, Journal of Periodontal Research, Journal of Clinical Periodontology, Periodontology 2000, Journal of Periodontal & Implant Science, and International Journal of Periodontics & Restorative Dentistry.
Screening
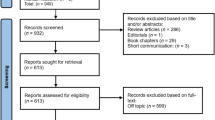
After retrieval of the articles, the studies were exported to the EndNote reference management tool, facilitating the elimination of duplicates. Next, two investigators (M.A.A.S and C.G.V) independently evaluated the titles and abstracts of each of the articles to determine their suitability for inclusion in the review. Any disagreement between the reviewers involved a third investigator (R.R.M) to resolve the debate, thus excluding irrelevant articles. Finally, the full texts of potentially eligible studies were thoroughly evaluated for inclusion. Cohen’s kappa coefficient was calculated to determine the inter-rater agreement or reproducibility corresponding to the literature selection. The kappa value (κ) was calculated based on the frequency of precise agreements between reviewers. Figure 1 shows the study review process.
Assessment of quality
The quality of the studies was evaluated with the Joanna Briggs Institute (JBI) critical appraisal tool, which was adapted for cross-sectional studies [47], in the form of a series of question that can be rated as: Yes, No, Unclear, or Not applicable.
The questions were as follows:
-
1.
Were the criteria for inclusion in the sample clearly defined?
-
2.
Were the study subjects and the setting described in detail?
-
3.
Was the exposure measured in a valid and reliable way?
-
4.
Were objective, standard criteria used for measurement of the condition?
-
5.
Was confounding factors identified?
-
6.
Were strategies to ideal with confounding factors stated?
-
7.
Were the outcomes measured in a valid and reliable way?
-
8.
Was appropriate statistical analysis used?
All included articles underwent quality assessment independently by two researchers (R.R.M and J.S.B.R). Finally, the articles were classified in terms of quality, and were placed in three levels: High bias, when the study reached up to 49% of the scores. Moderate bias, when it reached scores of 50 to 69%, and low bias, when it exceeded scores of 70%.
Data extraction
Data on the eligible articles for this research were extracted by the main reviewer (M.A.A.S), in a customized database in Excel software. All mean values and standard deviations were retrieved from the articles or calculated based on available data. The data extracted were: first author’s name, year, country, study design, title, journal of publication, gender, mean age (SD) of participants, number of cases with periodontal disease and healthy controls, sample size, definition of study groups, GCF sampling, type of biomarkers assessed, type of assay and corresponding kit, the mean levels of cytokines assessed (pg/ml) and their main findings, as well as the quality score for each article.
Results
Selection of studies
Initially 2,098 articles were found in the four databases, including PubMed (36 articles were found), Scopus (30 articles were found), Google Scholar (1,990 articles were found), Cochrane (40 articles were found), and manual searching (2 articles were found). Duplicates were removed and, based on title and abstract, the remaining 748 studies were reviewed. After analyzing the full text of the remaining articles, 733 records were excluded as irrelevant. A total of 15 articles were assessed for eligibility, of which 3 studies were excluded because cytokine expression was assessed in patients with other comorbidities (Diabetes mellitus, rheumatoid arthritis, and psoriasis). Therefore, a total of 12 articles were included for the qualitative and quantitative analysis of the present review. The details of study selection are shown in Fig. 1. Cohen’s Kappa coefficient (κ = 0.92) showed almost perfect agreement between reviewers, in the article selection process.
Description of the studies
Twelve articles with a cross-sectional design were reviewed in this study [28,29,30,31,32,33,34,35,36,37,38,39]. The total number of subjects studied in the included investigations was 537, of which 337 represented the case group (subjects with gingivitis and chronic periodontitis) and 200 represented the control group (periodontally healthy subjects). The ages of the subjects ranged from 20 to 50 years; the mean ± (SD) age of the subjects studied was 36.6 ± 4.2 years, of which 47% were male and 53% were female. Most of the articles were published after 2014 (8:67%) [28,29,30,31,32,33,34,35]. The oldest study was from 2005 [39], and the most recent from 2022 [28]. Five (41.6%) studies were conducted in India [28,29,30, 35, 36], two (16.6%) studies in Iran [31, 32], and other studies (8.3%) in Saudi Arabia [33], Japan [34], Egypt [37], Turkey [38], and Chile [39]. In addition, the title and journals of publication are shown (Table 2).
Nine (75%) studies collected GCF samples with absorbent paper strips [30,31,32,33,34,35, 37,38,39] and 3 (25%) studies reported GCF collection using volumetric microcapillary pipettes [28, 29, 36]. Among the 12 included studies, 4 (33.3%) used ELISA kits without brand specification [30, 32,33,34], two (16.6%) used R&D Systems ELISA kit [36, 39], two others (16.6%) used BioSource ELISA kit [37, 38], others (8.3%) used RayBiotech [28], Diaclone [29], Bender Med Systems [31] and eBioscience [35]. Most of the studies (75%) analyzed cytokine IL-17 levels individually [28,29,30, 32, 34, 36,37,38,39], whereas, 2 others (17%) analyzed IL-23 levels [33, 35] and only one (8%) analyzed the IL-23/IL-17 axis together [31] (Table 3).
Assessment of study quality and risk of bias
The Joanna Briggs Institute (JBI) checklist was used to assess the quality of the cross-sectional studies. According to the established criteria, all articles achieved scores of 100 [28,29,30,31,32,33,34,35,36,37,38,39], resulting in a low risk of bias in all selected studies (Table 4).
Discussion
A systematic review was conducted, which evaluated IL-23/IL-17 axis levels in GCF of subjects with chronic periodontitis, gingivitis, and healthy controls, from 12 independent cross-sectional studies corresponding to seven different countries.
In the gingival sulcus, the presence of a dysbiotic microbiome, which disrupts host immune responses, constitutes the main cause of the initiation, establishment and progression of inflammation (gingivitis), and subsequent destruction of tooth-supporting tissues (periodontitis) [48]. On the one hand, innate immunity represents the initial response of the host, constituting the first line of defense against invasion by pathogens, whereas, adaptive immunity represents the response following specific exposure to a given antigen, and is mediated mainly by T cells; which participate in cellular immunity against intracellular pathogens and, B cells; which participate in humoral immunity, through the production of antibodies directed against extracellular pathogens and microbial toxins [49]. Definitely, the interaction between both systems is quite complex, but very important to regulate and maintain tissue homeostasis [50].
In relation to cellular immunity, and immediately after CD4 and CD8 (+) T naive cells have been exposed to a particular antigen and/or cytokines present in the microenvironment, these cells proliferate, and differentiate into effector cells, which include cytotoxic T lymphocytes (CTL) and T helper (Th) cells [51]. In this context, some proinflammatory cytokines such as IL-1β, IL-6 and IL-23, as well as, the transcription factors signal transducer and activator of transcription 3 (STAT3), and retinoid-related orphan receptor-γt (RORγt), participate in the differentiation of CD4 (+) T naive cells into Th17 cells [52]. Actually, what happens is that, transforming growth factor beta (TGF-β), IL-1β and IL-6 inhibit forkhead box P3 (FOXP3), which is a negative regulator of Th17 cells and simultaneously activate RORγt initiating the Th17 cell differentiation cascade [53]. When RORγt is absent, RORα gives rise to this mechanism [54]. Therefore, the presence of these transcription factors is quite important for the generation of the Th17 subset [55].
Specifically, in the IL-23/IL-17 axis, IL-23 is first secreted by antigen presenting cells such as dendritic cells, and macrophages in response to polymicrobial challenge as pathogen associated molecular patterns, and damage-associated molecular patterns (PAMPS, DAMPS respectively) as well as MAMPS. Subsequently, IL-23 interacts and binds with its receptor (IL-23R) present on the cell membrane of Th17, which on the one hand, up-regulates RORγt expression through STAT3; creating a feedback loop sustained by IL-23. And on the other it also induces the production of other proinflammatory cytokines such as some members of the tumor necrosis factor superfamily (TNF-α and RANKL), interleukin-22 (IL-22), and IL-17, contributing to the bone resorption process [56]. Importantly, IL-23 signaling alone does not induce the development of Th17 cells from CD4 (+) T naive cells, as its receptor is expressed after differentiation into Th17 cells is initiated [57] (Fig. 2).
In fact, it is presumed that these molecules could be used as complementary tools to clinical parameters to diagnose and assess the degree of progression of periodontal diseases [58, 59].
TNF-α is a proinflammatory and pleiotropic cytokine, whose main function lies in: 1)promotion of myeloid cells, 2)induction of osteoclastic activity; by RANKL-independent paracrine/autocrine signaling or by up-regulation of RANKL, 3)inhibition of osteoblastic activity; by inhibition of the wnt pathway to down-regulate osteoblast function and increase their apoptosis and 4) secretion of other cytokines/chemokines and MMPs (MMP-8, MMP-9 and MMP-13) perpetuating the proinflammatory and destructive state in the periodontium [60, 61]. Numerous studies have shown differences in TNF-α levels in saliva, serum, tissue biopsies, PICF and GCF of subjects with periodontal disease [62,63,64,65,66]. In fact, it is currently considered a potential inflammatory biomarker to distinguish between periodontal (gingivitis and chronic periodontitis) and peri-implant (mucositis and peri-implantitis) diseases [44, 67].
In relation to IL-22, there is little evidence of its role in the pathogenesis of periodontal disease, however, it is known to play an important role in mucosal immunity [68]. In this regard, it is documented that IL-22 does not act directly on immune cells, but acts on cells of tissues such as skin and mucosa. Thus, it has been shown that, in keratinocytes IL-22 induces the production of antimicrobial peptides such as human beta defensin-2 (HBD-2) which plays a key role in host defense against infection. In fact, increased levels of IL-22 and HBD-2 in GCF, as well as, a positive correlation between both proteins with periodontal clinical parameters; plaque index (PI), gingival index (GI), probing deep (PD), and clinical attachment level (CAL) have been reported in subjects with gingivitis and chronic periodontitis, however, to distinguish between both conditions, subjects with chronic periodontitis presented a significant increase of IL-22 and HBD-2 levels in GCF compared to subjects with gingivitis [69]. On the other hand, in individuals with chronic periodontitis and psoriasis, a trend of increased levels of this cytokine has also been reported compared to periodontally healthy individuals with psoriasis [70]. These findings in the literature could be explained by the characteristic polymicrobial dysbiosis in subjects with periodontal disease that stimulates the inflammatory response and thus the large repertoire of Th17 cells, with subsequent production of proinflammatory cytokines that produce osteoclastogenesis [21, 40].
The role of the IL-23/IL-17 axis has been demonstrated in other systemic diseases such as rheumatoid arthritis [71], cancer [72], psoriasis [73], kidney disease [74], inflammatory bowel disease [75] and cardiovascular diseases [76]. In addition, several studies have explored the role of the IL-23/IL-17 axis in all types of periodontal diseases. These studies have examined the levels of the IL-23/IL-17 axis in different biological samples such as saliva, serum, tissue biopsies, PICF and GCF using some immunoassay methods such as ELISA [22, 28,29,30,31,32,33,34,35,36,37,38,39,40,41] and immunohistochemistry [77], as well as other molecular biology methods such as polymerase chain reaction [78].
The new classification of periodontal and peri-implant diseases proposed by the 2017 global workshop identified three forms of periodontitis; necrotizing periodontal disease, periodontitis as a manifestation of systemic diseases and that encompassed in a single term, as “periodontitis” (referring to chronic and aggressive periodontitis) taking into account the progression and severity of the disease; represented by stages I-IV and grades A-C [79]. In the present review, most of the studies compared individuals with chronic periodontitis with periodontally healthy subjects.
Today, GCF is considered one of the most reliable sources of oral biomarkers, due to its easy availability, with a high potential to reflect health and/or disease status. As mentioned above, there are different collection techniques. In the present study, the use of absorbent paper strips was the method most commonly used by the investigators, followed by the microcapillary pipetting technique. It is important to mention that, both techniques may have some limitations that affect the quantity and quality of the previously collected fluid, and therefore may contribute to the heterogeneity of the data. These changes mainly influence the collection time. In the first technique, a short time of 30 to 60 s is normally required, whereas, in the second technique, it varies between 40 min in healthy sites and 10 min in diseased sites. In both techniques, there is also the possibility of contamination with saliva or blood, which again motivates repeat sampling [80].
Ideally, a biomarker should meet some important criteria, such as validity, should be easy to use and measure, should be affordable, cost-effective, and able to be collected noninvasively. In addition, it should show sensitivity (identification of individuals who actually have the disease) and specificity (those who actually do not have the disease). One of the advantages of using IL-23/IL-17 axis as a diagnostic marker of periodontal disease is due to its ability to be analyzed in different oral fluids and tissues compared to other cytokines/chemokines that are more plasma specific [81].
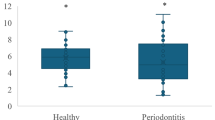
The results of our study showed an increase in the levels of IL-23/IL-17 axis in GCF of individuals with gingivitis compared to healthy controls, likewise the most important finding of this study showed that the mean level of IL-23/IL-17 axis in GCF of subjects with chronic periodontitis significantly increased compared to gingivitis and periodontally healthy individuals.
Nair et al., [28], Wankhede and Dhadse, [29], Nainee et al., [30], Kalate et al., [32], Althebeti et al., [33], Mitani et al., [34], Himani et al., [ 35], Nagireddy et al., [36], Shaker et al., [37] and Vernal et al., [39] showed in their investigations that the levels of the IL-23/IL-17 axis in GCF were increased and positively correlated with the progress and severity of periodontal disease (GI, PD, CAL). Therefore, scientific evidence suggests that this axis can potentially be considered as biomarkers of inflammation and destruction of periodontal tissues. Also, a positive evaluation was found between the levels of both molecules, which is explained by the fact that IL-23 can produce clonal expansion of Th17 and induce the expression of IL-17 [56]. It is worth mentioning that, only two studies [31, 38] found elevated levels of these cytokines in GCF from periodontally healthy individuals compared to subjects with gingivitis and chronic periodontitis. But this does not necessarily mean that there is a lower production of both cytokines in periodontal diseases. It is recommended to carry out future studies, with a better methodological design, in which individuals who present the different forms of periodontal disease are included, with a larger sample size and where the main cytokines that are involved in the axis can be evaluated. IL-23/IL-17 along with its receptors and isoforms present.
The main limitations of this review were the methodological design of the included cross-sectional studies, so researchers should be encouraged to carry out follow-up studies evaluating changes in the levels of the IL-23/IL-17 axis before and after periodontal therapy; the inclusion of a small number of articles, especially in relation to IL-23, so a meta-analysis was not possible; and a high heterogeneity of the available data, which is given by differences in the included variables, such as GCF sampling, collection time, variation of the periodontal microbiome between individuals, sex, age and systemic inflammatory condition of the body, so the results must be analyzed with great caution.
Conclusions
The levels of the IL-23/IL-17 axis are increased in GCF of subjects with chronic periodontitis and gingivitis compared to periodontally healthy individuals. Therefore, it could be used as a diagnostic tool to distinguish between periodontal diseases.
Data availability
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
Abbreviations
- IL-23:
-
Interleukin-23
- IL-17:
-
Interleukin-17
- Th17:
-
T helper cells 17
- GCF:
-
Gingival crevicular fluid
- PICF:
-
Peri-implant gingival crevicular fluid
- ELIA:
-
Enzyme-linked immunosorbent assay
- MAMPS:
-
Microbial-associated molecular patterns
- PAMPS:
-
Pathogen associated molecular patterns
- DAMPS:
-
Damage-associated molecular patterns
- IL-22:
-
Interleukin-22
- IL-6:
-
Interleukin-6
- IL-1β:
-
Interleukin-1 beta
- TGF-β:
-
Transforming growth factor beta
- TNF-α:
-
Tumor necrosis factor- alpha
- CX3CL1 Ligan 1:
-
(C-X3-C motif)
- IL-8:
-
Interleukin-8
- NETs:
-
Neutrophil extracellular traps
- MMPs:
-
Matrix metalloproteases
- RANKL:
-
Receptor activator of NF-κB ligand
- RANK:
-
Receptor activator of NF-κB
- CTL:
-
Cytotoxic T lymphocytes
- STAT3:
-
Signal transducer and activator of transcription 3
- RORγt:
-
Retinoid-related orphan receptor-γt
- FOXP3:
-
Forkhead box P3
- RORα:
-
Retinoid-related orphan receptor-α
- HBD-2:
-
Human beta defensin-2
- GI:
-
Gingival index
- PI:
-
Plaque index
- PD:
-
Probing deep
- CAL:
-
Clinical attachment level
References
Becerra-Ruiz JS, Guerrero-Velázquez C, Martínez-Esquivias F, Martínez-Pérez LA, Guzmán-Flores JM. Innate and adaptive immunity of periodontal disease. From etiology to alveolar bone loss. Oral Dis. 2022;28(6):1441–7. https://doi.org/10.1111/odi.13884. Epub 2021 Apr 28. PMID: 33884712.
Isola G, Santonocito S, Lupi SM, Polizzi A, Sclafani R, Patini R, Marchetti E. Periodontal Health and Disease in the context of systemic diseases. Mediators Inflamm. 2023;2023:9720947. https://doi.org/10.1155/2023/9720947. PMID: 37214190; PMCID: PMC10199803.
Trombelli L, Farina R, Silva CO, Tatakis DN. Plaque-induced gingivitis: Case definition and diagnostic considerations. J Periodontol. 2018;89 Suppl 1:S46-S73. https://doi.org/10.1002/JPER.17-0576. PMID: 29926936.
Cardoso EM, Reis C, Manzanares-Céspedes MC. Chronic periodontitis, inflammatory cytokines, and interrelationship with other chronic diseases. Postgrad Med. 2018;130(1):98–104. Epub 2017 Nov 8. PMID: 29065749.
Nuñez-Belmar J, Morales-Olavarria M, Vicencio E, Vernal R, Cárdenas JP, Cortez C. Contribution of -Omics technologies in the study of Porphyromonas gingivalis during Periodontitis Pathogenesis: a Minireview. Int J Mol Sci. 2022;24(1):620. https://doi.org/10.3390/ijms24010620. PMID: 36614064; PMCID: PMC9820714.
Cárdenas AM, Ardila LJ, Vernal R, Melgar-Rodríguez S, Hernández HG. Biomarkers of Periodontitis and its Differential DNA methylation and gene expression in Immune cells: a systematic review. Int J Mol Sci. 2022;23(19):12042. https://doi.org/10.3390/ijms231912042. PMID: 36233348; PMCID: PMC9570497.
Trindade D, Carvalho R, Machado V, Chambrone L, Mendes JJ, Botelho J. Prevalence of periodontitis in dentate people between 2011 and 2020: a systematic review and meta-analysis of epidemiological studies. J Clin Periodontol. 2023;50(5):604–26. https://doi.org/10.1111/jcpe.13769. Epub 2023 Jan 20. PMID: 36631982.
Genco RJ, Sanz M. Clinical and public health implications of periodontal and systemic diseases: An overview. Periodontol 2000. 2020;83(1):7–13. https://doi.org/10.1111/prd.12344. PMID: 32385880.
Kim TJ, Littlejohn CG, Richey KH, Falsafi N, Li C, Wang TJ, Lander B, Chang YC. A Modern Approach to treat Molar/Incisor Pattern Periodontitis-Review. J Clin Med. 2023;12(18):6107. https://doi.org/10.3390/jcm12186107. PMID: 37763046; PMCID: PMC10531571.
Al Shayeb KN, Turner W, Gillam DG. Periodontal probing: a review. Prim Dent J. 2014;3(3):25 – 9. https://doi.org/10.1308/205016814812736619. PMID: 25198634.
Adeoye J, Su YX. Artificial intelligence in salivary biomarker discovery and validation for oral diseases. Oral Dis. 2023 Jun 19. https://doi.org/10.1111/odi.14641. Epub ahead of print. PMID: 37335832.
Umeizudike K, Räisänen I, Gupta S, Nwhator S, Grigoriadis A, Sakellari D, Sorsa T. Active matrix metalloproteinase-8: a potential biomarker of oral systemic link. Clin Exp Dent Res. 2022;8(1):359–65. https://doi.org/10.1002/cre2.516. Epub 2021 Nov 19. PMID: 34800007; PMCID: PMC8874056.
Alarcón-Sánchez MA, Heboyan A, Fernandes GVO, Castro-Alarcón N, Romero-Castro NS. Potential impact of prosthetic biomaterials on the Periodontium: a Comprehensive Review. Molecules. 2023;28(3):1075. https://doi.org/10.3390/molecules28031075. PMID: 36770741; PMCID: PMC9921997.
Subbarao KC, Nattuthurai GS, Sundararajan SK, Sujith I, Joseph J, Syedshah YP. Gingival Crevicular Fluid: an overview. J Pharm Bioallied Sci. 2019;11(Suppl 2):135–S139. https://doi.org/10.4103/JPBS.JPBS_56_19. PMID: 31198325; PMCID: PMC6555362.
Fatima T, Khurshid Z, Rehman A, Imran E, Srivastava KC, Shrivastava D. Gingival Crevicular Fluid (GCF): a Diagnostic Tool for the Detection of Periodontal Health and diseases. Molecules. 2021;26(5):1208. https://doi.org/10.3390/molecules26051208. PMID: 33668185; PMCID: PMC7956529.
Barros SP, Williams R, Offenbacher S, Morelli T. Gingival crevicular fluid as a source of biomarkers for periodontitis. Periodontol 2000. 2016;70(1):53–64. https://doi.org/10.1111/prd.12107. PMID: 26662482; PMCID: PMC4911175.
Nazar Majeed Z, Philip K, Alabsi AM, Pushparajan S, Swaminathan D. Identification of Gingival Crevicular Fluid Sampling, Analytical methods, and oral biomarkers for the diagnosis and monitoring of Periodontal diseases: a systematic review. Dis Markers. 2016;2016:1804727. https://doi.org/10.1155/2016/1804727. Epub 2016 Dec 15. PMID: 28074077; PMCID: PMC5198140.
Prieto D, Maurer G, Sáez M, Cáceres F, Pino-Lagos K, Chaparro A. Soluble Neuropilin-1 in gingival crevicular fluid from periodontitis patients: an exploratory cross-sectional study. J Oral Biol Craniofac Res. 2021 Jan-Mar;11(1):84–7. Epub 2020 Nov 27. PMID: 33384917; PMCID: PMC7770967.
Heneberk O, Wurfelova E, Radochova V. Neopterin, the cell-mediated Immune Response Biomarker, in Inflammatory Periodontal diseases: a narrative review of a more than fifty Years Old Biomarker. Biomedicines. 2023;11(5):1294. https://doi.org/10.3390/biomedicines11051294. PMID: 37238968; PMCID: PMC10215987.
Kwon Y, Park C, Lee J, Park DH, Jeong S, Yun CH, Park OJ, Han SH. Regulation of bone cell differentiation and activation by Microbe-Associated molecular patterns. Int J Mol Sci. 2021;22(11):5805. https://doi.org/10.3390/ijms22115805. PMID: 34071605; PMCID: PMC8197933.
Bunte K, Beikler T. Th17 cells and the IL-23/IL-17 Axis in the Pathogenesis of Periodontitis and Immune-mediated inflammatory diseases. Int J Mol Sci. 2019;20(14):3394. https://doi.org/10.3390/ijms20143394. PMID: 31295952; PMCID: PMC6679067.
Medara N, Lenzo JC, Walsh KA, Reynolds EC, Darby IB, O’Brien-Simpson NM. A review of T helper 17 cell-related cytokines in serum and saliva in periodontitis. Cytokine. 2021;138:155340. https://doi.org/10.1016/j.cyto.2020.155340. Epub 2020 Nov 2. PMID: 33144024.
Ye L, Cao L, Song W, Yang C, Tang Q, Yuan Z. Interaction between apical periodontitis and systemic disease (review). Int J Mol Med. 2023;52(1):60. https://doi.org/10.3892/ijmm.2023.5263. Epub 2023 Jun 2. PMID: 37264964; PMCID: PMC10249585.
Wang J, Zhou Y, Ren B, Zou L, He B, Li M. The role of Neutrophil Extracellular traps in Periodontitis. Front Cell Infect Microbiol. 2021;11:639144. https://doi.org/10.3389/fcimb.2021.639144. PMID: 33816343; PMCID: PMC8012762.
Magán-Fernández A, Rasheed Al-Bakri SM, O’Valle F, Benavides-Reyes C, Abadía-Molina F, Mesa F. Neutrophil Extracellular traps in Periodontitis. Cells. 2020;9(6):1494. https://doi.org/10.3390/cells9061494. PMID: 32575367; PMCID: PMC7349145.
Atanasova T, Stankova T, Bivolarska A, Vlaykova T. Matrix metalloproteinases in oral health-special attention on MMP-8. Biomedicines. 2023;11(6):1514. https://doi.org/10.3390/biomedicines11061514. PMID: 37371608; PMCID: PMC10295544.
Ikeuchi T, Moutsopoulos NM. Osteoimmunology in periodontitis; a paradigm for Th17/IL-17 inflammatory bone loss. Bone. 2022;163:116500. https://doi.org/10.1016/j.bone.2022.116500. Epub 2022 Jul 20. PMID: 35870792; PMCID: PMC10448972.
Nair V, Grover V, Arora S, Das G, Ahmad I, Ohri A, Sainudeen S, Saluja P, Saha A. Comparative Evaluation of Gingival Crevicular Fluid Interleukin-17, 18 and 21 in Different Stages of Periodontal Health and Disease. Med (Kaunas). 2022;58(8):1042. https://doi.org/10.3390/medicina58081042. PMID: 36013509; PMCID: PMC9415654.
Wankhede AN, Dhadse PV. Interleukin-17 levels in gingival crevicular fluid of aggressive periodontitis and chronic periodontitis patients. J Indian Soc Periodontol. 2022 Nov-Dec;26(6):552–6. https://doi.org/10.4103/jisp.jisp_47_21. Epub 2022 Nov 14. PMID: 36582957; PMCID: PMC9793929.
Nainee N, Sanikop S, Jha A. Estimation of interleukin-17 levels in gingival crevicular fluid from healthy individuals, chronic gingivitis and chronic periodontitis patients using enzyme linked immunosorbent assay. Int J Res Med Sci. 2020;8(10):3605–10. https://doi.org/10.18203/2320-6012.ijrms20204237.
Sadeghi R, Sattari M, Dehghan F, Akbari S. Interleukin-17 and interleukin-23 levels in gingival crevicular fluid of patients with chronic and aggressive periodontitis. Cent Eur J Immunol. 2018;43(1):76–80. https://doi.org/10.5114/ceji.2018.74876. Epub 2018 Mar 30. PMID: 29736149; PMCID: PMC5927176.
Kalate FA, Gholami L, Alijani E, Hedayatipanah A, Kosari S. Level of interleukin-17 in gingival crevicular fluid of patients with chronic periodontitis. World J Dentistry. 2018;9(6):495–9.
Althebeti GR, Elfasakhany FM, Talla EA. Evaluation of Interleukin-23 in periodontal health and disease. Int J Health Sci Res. 2018;8(2):226–32.
Mitani A, Niedbala W, Fujimura T, Mogi M, Miyamae S, Higuchi N et al. Increased expression of interleukin-35 and – 17, but not – 27, in gingival tissues with chronic periodontitis. J Periodontol. https://doi.org/10.1902/jop.2014.140293.
Himani GS, Prabhuji ML, Karthikeyan BV. Gingival crevicular fluid and interleukin-23 concentration in systemically healthy subjects: their relationship in periodontal health and disease. J Periodontal Res. 2014;49(2):237–45. https://doi.org/10.1111/jre.12100. Epub 2013 May 31. PMID: 23721589.
Nagireddy RR, Chavan V, Subramanyam MB, Reddy VS, Pasupuleti MK, Avula KK. Estimation of interleukin-17 levels in gingival crevicular fluid from healthy individuals and patients with chronic periodontitis. J Dr NTR Univ Health Sci. 2013;2:191–5.
Shaker OG, Ghallab NA. IL-17 and IL-11 GCF levels in aggressive and chronic periodontitis patients: relation to PCR bacterial detection. Mediators Inflamm. 2012;2012:174764. doi: 10.1155/2012/174764. Epub 2012 Nov 26. PMID: 23226926; PMCID: PMC3513783.
Yetkin Ay Z, Sütçü R, Uskun E, Bozkurt FY, Berker E. The impact of the IL-11:IL-17 ratio on the chronic periodontitis pathogenesis: a preliminary report. Oral Dis. 2009;15(1):93–9. https://doi.org/10.1111/j.1601-0825.2008.01497.x. Epub 2008 Oct 29. PMID: 18992017.
Vernal R, Dutzan N, Chaparro A, Puente J, Antonieta Valenzuela M, Gamonal J. Levels of interleukin-17 in gingival crevicular fluid and in supernatants of cellular cultures of gingival tissue from patients with chronic periodontitis. J Clin Periodontol. 2005;32(4):383-9. https://doi.org/10.1111/j.1600-051X.2005.00684.x. PMID: 15811056.
Rodríguez-Montaño R, Ruiz-Gutiérrez AdC, Martínez-Rodríguez VMdC, Gómez-Sandoval JR, Guzmán-Flores JM, Becerra-Ruiz JS, Zamora-Perez AL, Guerrero-Velázquez C. Levels of IL-23/IL-17 Axis in plasma and Gingival Tissue of Periodontitis patients according to the New classification. Appl Sci. 2022;12:8051. https://doi.org/10.3390/app12168051.
Ozçaka O, Nalbantsoy A, Buduneli N. Interleukin-17 and interleukin-18 levels in saliva and plasma of patients with chronic periodontitis. J Periodontal Res. 2011;46(5):592-8. https://doi.org/10.1111/j.1600-0765.2011.01377.x. Epub 2011 Jun 3. PMID: 21635252.
Boronat-Catalá M, Catalá-Pizarro M, Bagán Sebastián JV. Salivary and crevicular fluid interleukins in gingivitis. J Clin Exp Dent. 2014;6(2):e175–9. https://doi.org/10.4317/jced.51403. PMID: 24790719; PMCID: PMC4002349.
Almehmadi AH, Alghamdi F. Biomarkers of alveolar bone resorption in gingival crevicular fluid: a systematic review. Arch Oral Biol. 2018;93:12–21. https://doi.org/10.1016/j.archoralbio.2018.05.004. Epub 2018 May 22. PMID: 29800801.
Madureira DF, De Abreu Lima L, Costa I, Lages GC, Martins EMB, Aparecida Da Silva CC. Tumor necrosis factor-alpha in Gingival Crevicular Fluid as a diagnostic marker for Periodontal diseases: a systematic review. J Evid Based Dent Pract. 2018;18(4):315–31. https://doi.org/10.1016/j.jebdp.2018.04.001.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;29:372n71. https://doi.org/10.1136/bmj.n71.
Higgins JP. June Cochrane Handbook for Systematic Reviews of Interventions, Version 5.0.1; The Cochrane Collaboration: London, UK,2008; Available online: http://www.cochrane-handbook.org (accessed on 20 2023).
Moola S, Munn Z, Tufanaru C, Aromataris E, Sears K, Sfetcu R, Currie M, Qureshi R, Mattis P, Lisy K, Mu P-F. Chapter 7: Systematic reviews of etiology and risk. In: Aromataris E, Munn Z, editors. JBI Manual for Evidence Synthesis. JBI, 2020. Available from https://synthesismanual.jbi.global.
Salvi GE, Roccuzzo A, Imber JC, Stähli A, Klinge B, Lang NP. Clinical periodontal diagnosis. Periodontol 2000. 2023;14. https://doi.org/10.1111/prd.12487. Epub ahead of print. PMID: 37452444.
Costalonga M, Herzberg MC. The oral microbiome and the immunobiology of periodontal disease and caries. Immunol Lett. 2014;162(2 Pt A):22–38. https://doi.org/10.1016/j.imlet.2014.08.017.
Meyle J, Dommisch H, Groeger S, Giacaman RA, Costalonga M, Herzberg M. The innate host response in caries and periodontitis. J Clin Periodontol. 2017;44(12):1215–25. https://doi.org/10.1111/jcpe.12781.
Freire MO, Van Dyke TE. Natural resolution of inflammation. Periodontol 2000. 2013;63(1):149–64. https://doi.org/10.1111/prd.12034.
Huang N, Dong H, Luo Y, Shao B. Th17 cells in Periodontitis and its regulation by A20. Front Immunol. 2021;7(12):742925. https://doi.org/10.3389/fimmu.2021.742925.
Liu J, Ouyang Y, Zhang Z, Wen S, Pi Y, Chen D, Su Z, Liang Z, Guo L, Wang Y. The role of Th17 cells: explanation of relationship between periodontitis and COPD? Inflamm Res. 2022;71(9):1011–24. https://doi.org/10.1007/s00011-022-01602-1.
Cheng WC, Hughes FJ, Taams LS. The presence, function and regulation of IL-17 and Th17 cells in periodontitis. J Clin Periodontol. 2014;41(6):541–9. https://doi.org/10.1111/jcpe.12238.
Nikolajczyk BS, Dawson DR 3rd. Origin of Th17 cells in type 2 diabetes-potentiated Periodontal Disease. Adv Exp Med Biol. 2019;1197:45–54. https://doi.org/10.1007/978-3-030-28524-1_4.
Kini V, Mohanty I, Telang G, Vyas N. Immunopathogenesis and distinct role of Th17 in periodontitis: a review. J Oral Biosci. 2022;64(2):193–201. https://doi.org/10.1016/j.job.2022.04.005.
Larsen JM. The immune response to Prevotella bacteria in chronic inflammatory disease. Immunology. 2017;151(4):363–74. https://doi.org/10.1111/imm.12760.
Duarte PM, Serrão CR, Miranda TS, Zanatta LC, Bastos MF, Faveri M, Figueiredo LC, Feres M. Could cytokine levels in the peri-implant crevicular fluid be used to distinguish between healthy implants and implants with peri-implantitis? A systematic review. J Periodontal Res. 2016;51(6):689–98. https://doi.org/10.1111/jre.12354.
Plemmenos G, Evangeliou E, Polizogopoulos N, Chalazias A, Deligianni M, Piperi C. Central Regulatory Role of Cytokines in Periodontitis and Targeting options. Curr Med Chem. 2021;28(15):3032–58. https://doi.org/10.2174/0929867327666200824112732.
Majdi Abunemer R, Saifuddin Shaheen R, Abudullah Alghamdi R. Correlation of anti-TNF-a biological therapy with periodontal conditions and osteonecrosis in autoimmune patients: a systematic review. Saudi Dent J. 2023;35(7):785–96. https://doi.org/10.1016/j.sdentj.2023.07.006.
Pan W, Wang Q, Chen Q. The cytokine network involved in the host immune response to periodontitis. Int J Oral Sci. 2019;11(3):30. https://doi.org/10.1038/s41368-019-0064-z.
Singh P, Gupta ND, Bey A, Khan S. Salivary TNF-alpha: a potential marker of periodontal destruction. J Indian Soc Periodontol. 2014;18(3):306–10. https://doi.org/10.4103/0972-124X.134566. PMID: 25024542; PMCID: PMC4095621.
Bakshi D, Kaur G, Singh D, Sahota J, Thakur A, Grover S. Estimation of plasma levels of Tumor Necrosis Factor-a, Interleukin-4 and 6 in patients with chronic periodontitis and type II diabetes Mellitus. J Contemp Dent Pract. 2018;19(2):166–9. https://doi.org/10.5005/jp-journals-10024-2231.
Pesevska S, Nakova M, Gjorgoski I, Angelov N, Ivanovski K, Nares S, Andreana S. Effect of laser on TNF-alpha expression in inflamed human gingival tissue. Lasers Med Sci. 2012;27(2):377–81. https://doi.org/10.1007/s10103-011-0898-x.
Gleiznys D, Kriauciunas A, Maminskas J, Stumbras A, Giedrimiene D, Niekrash C, Gleiznys A, Sakalauskiene J, Vitkauskiene A. Expression of Interleukin-17, Tumor Necrosis Factor-Alpha, and Matrix Metalloproteinase-8 in patients with chronic Peri-implant Mucositis. Med Sci Monit. 2021;2627:e932243. https://doi.org/10.12659/MSM.932243.
Romero-Castro NS, Vázquez-Villamar M, Muñoz-Valle JF, Reyes-Fernández S, Serna-Radilla VO, García-Arellano S, Castro-Alarcón N. Relationship between TNF-α, MMP-8, and MMP-9 levels in gingival crevicular fluid and the subgingival microbiota in periodontal disease. Odontology. 2020;108(1):25–33. https://doi.org/10.1007/s10266-019-00435-5.
Ghassib I, Chen Z, Zhu J, Wang HL. Use of IL-1 β, IL-6, TNF-α, and MMP-8 biomarkers to distinguish peri-implant diseases: a systematic review and meta-analysis. Clin Implant Dent Relat Res. 2019;21(1):190–207. https://doi.org/10.1111/cid.12694.
Isaza-Guzmán DM, Cardona-Vélez N, Gaviria-Correa DE, Martínez-Pabón MC, Castaño-Granada MC, Tobón-Arroyave SI. Association study between salivary levels of interferon (IFN)-gamma, interleukin (IL)-17, IL-21, and IL-22 with chronic periodontitis. Arch Oral Biol. 2015;60(1):91–9. https://doi.org/10.1016/j.archoralbio.2014.09.002.
Sidharthan S, Dharmarajan G, Kulloli A. Gingival crevicular fluid levels of Interleukin-22 (IL-22) and human β Defensin-2 (hBD-2) in periodontal health and disease: a correlative study. J Oral Biol Craniofac Res. 2020;10(4):498–503. https://doi.org/10.1016/j.jobcr.2020.07.021.
Jiménez C, Carvajal D, Hernández M, Valenzuela F, Astorga J, Fernández A. Levels of the interleukins 17A, 22, and 23 and the S100 protein family in the gingival crevicular fluid of psoriatic patients with or without periodontitis. Bras Dermatol. 2021;96(2):163–70. https://doi.org/10.1016/j.abd.2020.08.008.
Schinocca C, Rizzo C, Fasano S, Grasso G, La Barbera L, Ciccia F, Guggino G. Role of the IL-23/IL-17 pathway in Rheumatic diseases: an overview. Front Immunol. 2021;22(12):637829. https://doi.org/10.3389/fimmu.2021.637829.
Martin-Orozco N, Dong C. The IL-17/IL-23 axis of inflammation in cancer: friend or foe? Curr Opin Investig Drugs. 2009;10(6):543–9.
Hawkes JE, Yan BY, Chan TC, Krueger JG. Discovery of the IL-23/IL-17 signaling pathway and the treatment of Psoriasis. J Immunol. 2018;201(6):1605–13. https://doi.org/10.4049/jimmunol.1800013.
Ghali JR, Holdsworth SR, Kitching AR. Targeting IL-17 and IL-23 in Immune mediated Renal Disease. Curr Med Chem. 2015;22(38):4341–65. https://doi.org/10.2174/0929867322666151030163022.
Cătană CS, Berindan Neagoe I, Cozma V, Magdaş C, Tăbăran F, Dumitraşcu DL. Contribution of the IL-17/IL-23 axis to the pathogenesis of inflammatory bowel disease. World J Gastroenterol. 2015;21(19):5823–30. https://doi.org/10.3748/wjg.v21.i19.5823.
Mills KHG. IL-17 and IL-17-producing cells in protection versus pathology. Nat Rev Immunol. 2023;23(1):38–54. https://doi.org/10.1038/s41577-022-00746-9.
Ohyama H, Kato-Kogoe N, Kuhara A, Nishimura F, Nakasho K, Yamanegi K, Yamada N, Hata M, Yamane J, Terada N. The involvement of IL-23 and the Th17 pathway in periodontitis. J Dent Res. 2009;88(7):633–8. https://doi.org/10.1177/0022034509339889.
Ohyama H, Kato-Kogoe N, Kuhara A, Nishimura F, Nakasho K, Yamanegi K, et al. The involvement of IL-23 and the Th17 pathway in periodontitis. J Dent Res. 2009;88:633–8.
Tonetti MS, Greenwell H, Kornman KS. Staging and grading of periodontitis: Framework and proposal of a new classification and case definition. J Clin Periodontol. 2018;45(Suppl 20):S149–61. https://doi.org/10.1111/jcpe.12945. Erratum in: J Clin Periodontol. 2019;46(7):787.
Gul SS, Abdulkareem AA, Sha AM, Rawlinson A. Diagnostic accuracy of oral fluids Biomarker Profile to determine the current and future status of Periodontal and Peri-implant diseases. Diagnostics (Basel). 2020;10(10):838. https://doi.org/10.3390/diagnostics10100838.
Keskin M, Rintamarttunen J, Gülçiçek E, Räisänen IT, Gupta S, Tervahartiala T, Pätilä T, Sorsa T. A comparative analysis of treatment-related changes in the diagnostic biomarker active Metalloproteinase-8 levels in patients with Periodontitis. Diagnostics (Basel). 2023;13(5):903. https://doi.org/10.3390/diagnostics13050903.
Acknowledgements
Not applicable.
Funding
No external funding was received.
Author information
Authors and Affiliations
Contributions
Conceptualization, M.A.A.-S.; methodology, M.A.A.-S-.; software, M.A.A.-S.; validation, M.A.A.-S, R.R.-M, J.S.B.-R. and C.G.-V.; formal analysis, M.A.A.-S, R.R.-M, J.S.B.-R. and C.G.-V.; investigation, M.A.A.-S.; resources, M.A.A.-S, R.R.-M, J.S.B.-R. and C.G.-V.; data curation, M.A.A.-S.; writing—original draft preparation, M.A.A.-S, A.A., .R.-M, J.S.B.-R. and C.G.-V.; writing—review and editing, M.A.A.-S, R.R.-M, J.S.B.-R, C.G.-V, A.A. and A.H; visualization, M.A.A.-S, R.R.-M, J.S.B.-R, C.G.-V, and A.H; supervision, M.A.A.-S, R.R.-M, J.S.B.-R, C.G.-V, and A.H; project administration, M.A.A.-S. All authors have read and agreed to the published version of the manuscript.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Informed consent statement
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Alarcón-Sánchez, M.A., Guerrero-Velázquez, C., Becerra-Ruiz, J.S. et al. IL-23/IL-17 axis levels in gingival crevicular fluid of subjects with periodontal disease: a systematic review. BMC Oral Health 24, 302 (2024). https://doi.org/10.1186/s12903-024-04077-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12903-024-04077-0