Abstract
Background
This study was performed to determine the therapeutic effects of diosgenin (DG) which is a steroidal saponin, administered at different doses on alveolar bone loss (ABL) in rats with experimental periodontitis using immunohistochemical and cone-beam computed tomography (CBCT).
Methods
Thirty-two male Wistar rats divided into four equal groups: control (non-ligated), periodontitis (P), DG-48, and DG-96. Sutures were placed at the gingival margin of the lower first molars to induce experimental periodontitis. Then, 48 and 96 mg/kg of DG was administered to the study groups by oral gavage for 29 days. At day 30, the animals were sacrificed and ABL was determined via CBCT. The expression patterns of osteocalcin (OCN), alkaline phosphatase (ALP), type I collagen (Col-1), B cell lymphoma 2 (Bcl 2), Bcl 2-associated X protein (Bax), bone morphogenetic protein 2 (BMP-2), and receptor activator of NF κB ligand (RANKL) were examined immunohistochemically.
Results
Histopathologic examination showed all features of the advanced lesion in the P group. DG use decreased all these pathologic changes. It was observed that periodontitis pathology decreased as the dose increased. DG treatment increased the ALP, OCN, Bcl 2, Col-1, and BMP-2 levels in a dose-dependent manner, compared with the P group (p < 0.05). DG decreased the expression of RANKL and Bax in a dose-dependent manner (p < 0.05). ABL was significantly lower in the DG-48 and DG-96 groups than in the P group (p < 0.05).
Conclusion
Collectively, our findings suggest that DG administration protects rats from periodontal tissue damage with a dose-dependent manner, provides an increase in markers of bone formation, decreases in Bax/Bcl-2 ratio and osteoclast activation.
Similar content being viewed by others
Background
Various factors cause the bone loss including systemic diseases, trauma, osteoporosis and periodontal disease [1,2,3]. Bone loss in jaws is critical because especially severe losses leads the loss of function and complicates dental treatment. Therefore, studies have been focused protective and therapeutic treatment of the bone loss [4].
Periodontitis is an inflammatory disease in which the interactions between periodontal bacteria and the host tissue response lead to tissue destruction [4]. Specific groups of oral bacteria populate in dental plaque play a precursor role in the development of periodontal disease, however that once the disease has been stimulated, other factors effect the progression of periodontitis and aggravate the treatment of disease [5]. Increases in oxidative stress, proinflammatory cytokines, and osteoclast cells have major roles in periodontal destruction [2]. Oxidative stress stimulates the transformation of precursor osteoclast cells into mature osteoclasts, leading to pathological changes, followed by the destruction of affected tissue [6,7,8]. Reactive oxygen species (ROS) are highly reactive by-products of oxygen metabolism and they have crucial role in various cellular processes as signalling molecules [9]. ROS causes apoptosis by reducing B-cell lymphoma 2 proteins (Bcl-2) and elevating the expression of Bcl-2-associated X protein (BAX) [10]. In addition, the elevation of ROS levels can damage tissue cells by stimulating proinflammatory cytokine cells and modulating the several pathways such as activation of NFκB ligand (RANKL) pathway, decreasing the protective effect of Nuclear factor red line 2 related factor 2 pathway, c-Jun N-terminal kinase signaling pathway, NOD-like receptor protein 3 [4, 11,12,13]. RANKL is known a member of the tumor necrosis factor superfamily. RANKL is an apoptosis regulator gene and it is a binding partner of osteoprotegerin. RANKL is expressed by several types of cells, including osteoblasts, osteocytes, fibroblasts, and lymphocytes [14]. RANKL induces the activation of osteoclast cells and osteoclastogenesis because it stimulates the formation of osteoclast precursor cells. RANKL-mediated osteoclastogenesis has a critical role in periodontal destruction (Fig. 1) [4, 15, 16].
Schematic representation of periodontal destruction by ROS-induced inflammation. To summarize ROS for periodontal inflammation through multiple targets and multiple pathways. MMPs: matrix metalloproteinases; Nf-kB: nuclear factor kappa B; Nrf2: Nuclear factor red line 2 related factor 2; JNK: c-Jun N-terminal kinase; NRLP3: NOD-like receptor protein 3
Growth factors organize cellular activities and improve tissue healing by binding to specific cell receptors. Several studies have used growth factors to enhance periodontal tissue and bone regeneration [17, 18]. BMP-2 belongs to the TGF-β superfamily of proteins and it is a growth factor with roles in tissue regeneration, including the transformation of undifferentiated mesenchymal cells and enhancement of osteoblast differentiation [19]. Furthermore, it stimulates the secretion of several osteoblastic-specific molecules, such as alkaline phosphatase (ALP), osteocalcin (OCN), and type I collagen (Col-1) [15, 20].
Various agents have been used to reduce the effects of ROS on periodontitis, and diosgenin (DG) is one of them. DG is a naturally occurring bioactive steroid saponin. It has been used in several steroidal drugs in the pharmaceutical industry because its chemical structure is similar to the structures of sex hormones [21, 22]. DG exhibits various therapeutic effects, including antioxidative, antidiabetic, anti-inflammatory, and antihyperlipidemic activities [21, 23, 24]. Moreover, DG modulates RANKL and OCN levels, regulates oxidative stress, stimulates signaling in the BMP pathways, and prevents apoptosis [22, 24,25,26,27,28].
To our knowledge, no study has evaluated the effects of different doses DG treatment on periodontal destruction in rats with systemically healthy. Therefore, it is unclear the influence and mechanisms of different doses DG treatment in systemically healthy rats with periodontitis. Here, we hypothesized that DG has antioxidative, anti-inflammatory and anti-resorptive properties and it could prevent periodontal tissue destruction by decreasing RANKL levels, inhibiting periodontal inflammation and cell apoptosis, and inducing bone formation. This study was performed to investigate the therapeutic effects of DG on ALP, OCN, Col-1, BAX, Bcl-2, BMP-2, and RANKL levels, as well as alveolar bone loss (ABL), in rats with experimental periodontitis to ensure basic information for potential DG application and further researches studies.
Methods
Animals
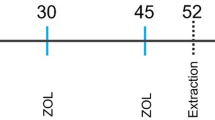
All experimental procedures in the present study were approved by the University Ethics Committee for Animal Experiments, Denizli (PAUHADYEK-2018/33). The Animal Research: Reporting of In Vivo Experiments guidelines were followed in this study. Thirty-two male Wistar albino rats (4 months old, 350–400 g), which were obtained from Pamukkale University Experimental Surgery Application and Research Center, were used in this study. Before initiation of the experimental procedures, the rats were adapted to the experimental environment for ten days; they were housed separately in cages in a room at 21 ± 2 °C and with a 12-h light:12-h dark cycle. All animals had free access to water and food. G* Power 3.1 software was used to calculating of sample size, considering the global significance level of α = 0.05, a sampling power of 95%, and f = 0.86 [24]. Rats were divided into groups by simple randomization using the coin flip method into four groups (n = 8/group): control (non-ligated), periodontitis (P; ligature only), DG-48 (ligature + DG 48 mg/kg/day), and DG-96 (ligature + DG 96 mg/kg/day). The DG (Sigma-Aldrich, Saint Louis, MO, USA) was dissolved in distilled water and administered by oral gavage for 29 days, as in previous studies [22]. Rats in the control and P groups were given 1 ml distilled water by oral gavage during the experiment. All rats were sacrificed at day 30 [15, 29]. Before sacrification, 50 mg/kg body weight of ketamine (Eczacibasi Ilac Sanayi, Istanbul, Turkey) and 5 mg/kg xylazine chloride (Virbaxil®, São Paulo, Brazil) were used for general anesthesia. Hence, all animals were unconscious. The animals were stabilized and their head were placed in the small animal guillotine opening by a specialist animal technician for sacrification. Subsequently, the rats were decapitated rapidly.
Induction of periodontitis model
The experimental procedure was performed under general anesthesia. 50 and 5 mg/kg body weight of ketamine and xylazine chloride respectively were administered intraperitoneally to provide general anesthesia. The cervical areas of the first lower right and left mandibular molars were submarginally ligatured using a 4 − 0 sterile silk suture (Dogsan Ilac Sanayi, Istanbul, Turkey) to stimulate plaque accumulation and periodontal inflammation. The ligatures were checked daily by two operators to prevent the observer bias (AK and ALA).
Three-dimensional imaging
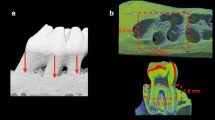
A supine-position cone-beam computed tomography (CBCT) unit (Newtom 5G-XL; QR, Verona, Italy) was used for three-dimensional imaging. The smallest field-of-view of this device (6 cm × 6 cm) was chosen; the exposure settings were 100 mm voxel, 110 kV, 11.4 mA, 9.0 s exposure time, 26.0 s scanning time, enhanced scan, boosted dose, and high-resolution (HiRes) mode. The unit’s proprietary software (NNT, version 12.1; QR) was used for image analysis. All specimens were exposed in the same position with the same exposure parameters. A dentomaxillofacial radiologist with 9 years of experience was blinded to the specimens (MO); this radiologist performed all tomographic procedures and analyzed the images. Figure 2 shows three-dimensional reconstructed and cross-sectional slice images. The distance was measured at cementoenamel junction to the alveolar bone crest and averaged across six areas (the mesial, medial, and distal parts of the buccal–lingual surfaces) of the mandibular first molar teeth for evaluating the linear bone loss (in mm).
Histopathological method
After rats had been sacrificed, mandibular samples were obtained and fixed in 10% neutral-buffered formalin for histopathological evaluation. The samples were decalcified in a solution (Osteofast 1; Biognost, Zagreb, Croatia) for 2 weeks, then routinely processed using automatic tissue processor equipment (Leica ASP300S; Leica Microsystems, Wetzlar, Germany) and immersed in paraffin. Subsequently, a rotary microtome (Leica RM 2155; Leica Microsystems) was used to obtained 5 μm sections from each sample. Each sample was cut along the long axis of the tooth in the mesiodistal direction and stained with hematoxylin and eosin. Histopathological examinations were performed by a single specialist who was blinded to the samples (ÖÖ).
Observations were conducted using a light microscope at ×40 magnification, according to a modified version of histopathological scoring criteria established by Leitao et al. [30]. To standardize the data, five areas from each rat were evaluated and their averages were taken. Neutrophil leukocyte infiltrations were specifically assessed. Inflammatory cell infiltrations, alveolar bone resorption, and degeneration and destruction of the cementum were scored according to previous study [30].
Immunohistochemical method
The streptoavidin-biotin peroxidase technique was performed to the sections selected for immunohistochemical processing. Sections were immunohistochemically stained at 1/100 dilution for all primary antibodies using anti-BMP 2 (ab59348; all from Abcam plc, Cambridge, UK), anti-RANKL (ab216484), anti-ALP (ab224335), Bax (ab53154), anti-Bcl-2 (ab59348), anti-Col-1 (ab34710) and anti-OCN (ab93876) antibody kits according to the manufacturer’s recommendations.
The sections were then embedded with hydrogen peroxide in 3% methanol for 20 min to eliminate activity of endogenous peroxidase. Sections were boiled twice for 5 min with citrate buffer solution and washed in phosphate buffered saline (PBS). The UltraVision Detection System Anti-Polyvalenti HRP Kit (Mouse and Rabbit Specific HRP/DAB Detection Kit-Micro-polymer, ab236466; Abcam plc) was used as the secondary antibody was used as the secondary antibody and 3,3’-diaminobenzidine (DAB) as the chromogen. Sections were incubated with primary antibodies for 60 min at room tempareture. Immunohistochemistry was then performed using biotinylated secondary antibody and streptavidin-alkaline phosphatase conjugate. Sections were incubated with DAB for 3–5 min.
For negative controls, an antibody dilution solution was used instead of primary antibodies. Harris haematoxylin was used for contrast staining and slides were examined under a light microscope. Immunohistochemical findings were scored on a scale of 0 to 3, where 0 = no staining, 1 = mild staining, 2 = moderate staining, and 3 = heavy staining [31]. All immunohistochemical evaluations were performed by a specialized pathologist who was blinded to the samples (ÖÖ). Immunohistochemical analyses were performed using ImageJ 1,48 version (National Institutes of Health, Bethesda MD).
After the classic microscopic analyses, we obtained histomorphometric and immunohistochemical evaluations using an automated image analysis system (Olympus CX41; Olympus Corporation, Tokyo, Japan). The lesioned area was evaluated using proprietary software (cellSens Life Science Imaging Software System; Olympus Corporation).
Statistical analysis
The Shapiro–Wilk test was used to assess whether data exhibited normal distributions. The post hoc Duncan multiple comparison test and one-way analysis of variance were used to analyze the ABL. Independent variables (ALP, BAX, Bcl-2, BMP-2, Col-1, OCN, RANKL, and histopathological scores) were evaluated using the Kruskal–Wallis test. All data are reported as means ± standard deviations for each group (p < 0.05). All analyses were conducted using SPSS software (version 23; IBM Corporation, Armonk, NY, USA).
Results
CBCT findings
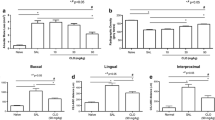
Periodontitis was induced in all ligated groups, according to the CBCT findings. The results showed that the control group had no ABL. ABL was significantly lower in the DG-48 and DG-96 groups than in the P group (p < 0.05; Figs. 2 and 3).
Histopathological findings
Histological examination showed that the control group had normal gingival tissue architecture and gingival epithelium; it showed no pathological findings. Hyperemia, ulcers in the gingival epithelial layer, inflammatory reactions in the gingival tissue and periodontal ligament, partial to severe cement destruction, and alveolar bone degradation were observed in the P group. Microscopic evaluations of the DG-48 and DG-96 groups revealed that the treatments ameliorated the pathological findings, compared with the P group. Furthermore, cellular infiltration, ABL, and cement destruction were reduced in the DG-96 group, compared with the DG-48 group (Fig. 4).
Histopathological appearance of the groups. Normal gingival histology in the control group (arrow); gingival epithelial loss and severe inflammatory reaction in the periodontitis group; moderate inflammatory reaction in the diosgenin 48 mg/kg/day (DG-48) group; and decreased inflammatory reaction and periodontal lesions and increased epithelization in the diosgenin 96 mg/kg/day (DG-96) group. T: teeth; the bars represent 100 μm
Immunohistochemical findings
The expression patterns of ALP, Bcl-2, BAX, Col-1, BMP-2, OCN, and RANKL in mesenchymal cells in all groups were observed immunohistochemically. Positive immunoexpression was indicated by a brown color. During examinations of the ALP, BAX, Bcl-2, BMP-2, Col-1, OCN, and RANKL immunostained sections, slight to negative immunoexpression findings were observed in the control group.
ALP, Bcl-2, BMP-2, Col-1, and OCN expression levels were significantly lower in the P group than in the control group (p < 0.05). Treatment significantly increased the expression levels of ALP, Bcl-2, BMP-2, Col-1, and OCN in the DG groups, compared with the P group (p < 0.05). Additionally, DG-96 was more effective than DG-48 for normalizing immunoexpression (Fig. 5). Statistical analysis results of the immunohistochemical scores are shown in Fig. 3.
Immunohistochemical expression results for all groups according to the streptavidin–biotin method. Negative-to-slight expression in the control group; marked decreases in alkaline phosphatase (ALP), Bcell lymphoma 2 (Bcl-2), bone morphogenetic protein 2 (BMP-2), Col-1, and osteocalcin (OCN) but increases in Bcl-2-associated X protein (BAX) and receptor activator of NF-κB ligand (RANKL) in the periodontitis (P) group; and amelioration by diosgenin 48 mg/kg/day (DG-48) and diosgenin 96 mg/kg/day (DG-96) in those groups. The arrows indicate cells expressing markers; the bars represent 50 μm
BAX and RANKL expression levels increased in the P group, compared with the control group (p < 0.05). Treatment significantly decreased RANKL and BAX levels in the DG groups, compared with the P group (p < 0.05). Finally, DG-96 significantly decreased the expression levels of RANKL and BAX, compared with DG-48 (p < 0.05; Figs. 3 and 5).
Discussion
In the present study, we used histomorphometry, immunohistochemistry, and CBCT to evaluate the effects of DG dose on ABL in experimental periodontitis. To our knowledge, this is the first study regarding the effects of DG in healthy rats with experimental periodontitis. The doses of DG were determined on the basis of previous findings [22, 32, 33].
There are three methods that are frequently used to induce periodontal disease, which are: ligature application, oral bacterial inoculation, and the lipopolysaccharide injection technique. In the ligature model, sterile non-absorbable sutures or orthodontic wires are widely used to induce local irritation and bacterial plaque accumulation. Secondly, mono and mixed cultures of periodontal bacteria are inoculated orally by gavage or topical application. Lastly, lipopolysaccharide extracted from pathogenic bacteria can be directly injected into the gingival sulcus to induce inflammation and stimulate osteoclastogenesis and alveolar bone loss. Among these methods, ligature application induces inflammation and alveolar bone resorption more promptly compared to other methods [34]. Hence, we preferred this method for inducing bone loss.
Micro-computerized tomography is regarded as the “gold standard” method for analyzing trabecular bone and tooth microstructure, evaluating the development of the skull bones, and assessing tissue engineering [35]. However, several studies have evaluated the efficacy of CBCT as an alternative for assessing periodontal defects because micro-computerized tomography involves ultra-high radiation doses and is not routinely used in clinical settings [36]. Thus, Tayman et al. investigated the use of CBCT to measure periodontal defects; they concluded that it provides useful linear and volumetric measurements of such defects in vitro [36]. Other studies have suggested that CBCT can be used to evaluate periodontal defects and the structures and trabecular microarchitecture of alveolar bone [37]. In an experimental study, Lektemur Alpan et al. demonstrated that CBCT measurements of ABL levels were accurate [37]. Thus, we measured ABL using CBCT in this study.
RANKL is the primary regulator of osteoclastogenesis; it has a critical role in osteoclast-associated diseases [38]. Several studies have demonstrated that ABL is associated with high RANKL levels [35, 39]. Zhang et al. performed 24 mg/kg body weight/day, 48 mg/kg body weight/day and 96 mg/kg body weight/day as dosages of DG and they reported that a high dose of DG decreased bone loss by modulating the RANKL and osteoprotegerin levels in an ovariectomized rat model [22]. In an another study, Zhang et al. evaluated protective effects of DG on ABL in ovariectomized rats and they indicated that DG inhibited osteogenesis and osteoclastogenesis by regulating the releasing of important molecules in the Wnt, RANKL or osteoclastogenic cytokine pathways [40]. In previous study, we evaluated the effects of DG on RANKL in diabetic rats with periodontitis and our results showed that 96 mg/kg DG treatment significantly decreased in RANKL levels and ABL. In the present study, DG treatments significantly downregulated the RANKL levels and inhibited RANKL-induced osteoclastogenesis in rats in a dose-dependent manner, compared with the untreated periodontitis group. Furthermore, DG significantly decreased ABL in a dose-dependent manner, compared with the untreated group. These results suggested that DG prevents ABL by inhibiting RANKL expression and RANKL-induced osteoclastogenesis, consistent with the findings of earlier reports [22, 24, 40].
Inflammation can increase oxidative stress, thus, worsening DNA damage and tissue apoptosis [41]. Moreover, periodontal disease reportedly leads to an imbalance between pro- and anti‐apoptotic processes [42]. Therefore, we evaluated apoptotic marker levels in our study. BAX is a member of the Bcl-2 family; expression levels of BAX and Bcl-2 are considered indicators of apoptosis or survival in cells [15, 43]. Wu et al. applied the 10, 50, or 100 mg/kg DG daily in ovariectomized rats and they reported that DG treatment decreases BAX and BAX/Bcl-2 levels and it has a therapeutic potential for ovariectomy-induced cardiac apoptosis [44]. In vitro study demonstrated that 2, 6, and 8 µM doses of DG alleviates the apoptosis by maintaining the Bcl-2 expression [45]. Additionally, 96 mg/kg DG treatment significantly reduced Bax and increased Bcl-2 levels in our previous experimental study [24]. In the present study, we evaluated BAX and Bcl-2 levels to identify the effects of DG on apoptosis signaling pathways. The results showed that experimental periodontitis upregulated and downregulated the expression levels of BAX and Bcl-2, respectively. In contrast, DG treatment upregulated and downregulated the expression levels of Bcl-2 and BAX, respectively, in our experimental periodontitis model. Particularly, DG significantly increased the Bcl-2 levels in higher dose group than low dose group. These results indicate that dose depending DG treatment decreases periodontitis–induced apoptosis by suppressing the expression of BAX and inducing the expression of Bcl-2; these results are also consistent with previous findings [24, 26, 44, 45]. Additionally, apoptosis is a complex process, and additional markers or assays might be needed to confirm this effect comprehensively.
Several biochemical markers have been used to evaluate bone metabolic activity, including ALP, Col-1, OCN, and BMP-2 [10, 37]. ALP is released by osteoblast cells; measurements of ALP level are used to evaluate osteoblastic activity. OCN controls mineral deposition; thus, it has critical roles in bone formation and remodeling [46]. Furthermore, BMP-2 mediates the differentiation of osteoblastic cells and induces the release of ALP, OCN, and Col-1 [15, 19, 20]. Zhao et al. performed 10 mg/kg, 30 mg/kg, and 90 mg/kg DG in retinoic acid-induced osteoporosis in rats and they indicated DG significantly reduced the ALP levels and increased OCN levels in 30 mg/kg, and 90 mg/kg DG groups and promoted bone formation and inhibits bone absorption by regulating bone metabolism and mineralization [27]. Another study applied DG via oral gavage at a dosage of 100 mg/kg body weight daily and they found that DG could enhance the bone formation process through increased Wnt and BMP signaling activity; these pathways regulate the osteogenic differentiation of mesenchymal stem cells and preosteoblasts [25]. Liao et al. found that the arginyl–DG conjugate stimulates BMP-2-induced osteoblastic differentiation with synergistic effects on ALP activity and mineralization [47]. Additionally, Zhang et al. treated the DG group rats by oral gavage with 100 mg/kg body weight DG and they showed that DG has anti-bone loss efficiacy on rat alveolar bone by alleviating the OCN levels [40]. In diabetic rats with experimental periodontitis, we used the 96 mg/kg DG treatment and previous results reported that DG treatment significantly improved the expression of ALP, OCN and BMP-2 in test group [24]. In the present study, DG significantly promoted the expression of ALP, OCN and BMP-2 in test groups than P group. Also, high dose DG treatment significantly promoted BMP-2 and ALP levels compared the low dose group. These findings suggest that DG treatment, especially high dose of DG, enhances bone formation by increasing new bone activity through enhanced expression of ALP, OCN, and BMP-2; this is also consistent with previous findings [24, 25, 27, 40, 47].
Col-1 is an important factor that stimulates osteoblast differentiation and mineral matrix deposition [48]. The increasing of Col-1 supports the ABL formation in the experimental periodontitis [43]. A few studies investigated the association between DG and Col-1 level. In our previous study, 96 mg/kg DG treatment significantly increased the Col-1 levels in diabetic rats with periodontitis [24]. The present study showed periodontitis decreased the Col-1 level and both dose of DG treatment significantly increased the Col-1 level and confirmed the previous study the association between the periodontitis and Col-1 [24, 43]. However, further studies are needed to evaluating DG on Col-1 levels.
This study have several limitation. We did not evaluate the effect of DG on the Wnt pathways or osteoprotegerin levels or other relevant markers of bone metabolism, inflammation and did not compare the CBCT findings with micro-computerized tomography; these were limitations of the present study. Therefore, further studies are needed to investigate the effects of DG on the other bone metabolic pathways and relevant markers expression in periodontal disease. Another limitation of our study is the inability to examine DG in humans by histological examination due to ethical barriers and potential side effects. Ligature induced periodontitis causes acute inflammation in rats however periodontitis is a chronic course in humans in terms of proinflammatory, anti-inflammatory cytokine activities and oxidant/antioxidant balance and that is a limitation. Additionally, we preferred DG doses according to previous studies nonetheless different doses of DG could be evaluated further studies.
Conclusion
The present study indicated that both doses of DG—particularly the higher dose—regulate bone activity, prevent RANKL-induced osteoclastogenesis and improve new bone activity and bone formation. Although limitations, our results indicate that DG administration can prevent alveolar bone damage in periodontal disease.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ROS:
-
Reactive oxygen species
- Bcl-2:
-
B-cell lymphoma 2 proteins
- BAX:
-
Bcl-2-associated X protein
- RANKL:
-
NFκB ligand
- BMP-2:
-
Bone morphogenetic protein 2
- ALP:
-
Alkaline phosphatase Osteocalcin (OCN)
- Col-1:
-
Type I collagen
- DG:
-
Diosgenin
- ABL:
-
Alveolar bone loss
- P:
-
Periodontitis
- DG-48:
-
Ligature + DG 48 mg/kg/day
- DG-96:
-
Ligature + DG 96 mg/kg/day
- CBCT:
-
Cone-beam computed tomography
References
Darcey J, Devlin H, Lai D, et al. An observational study to assess the association between osteoporosis and periodontal disease. Br Dent J. 2013;215(12):617–21.
Aldelaimi TN, Khalil AA. Surgical management of pediatric mandibular trauma. J Craniofac Surg. 2013;24(3):785–87.
Enezei HH, Khalil AA, Naif TNA. Clinical analysis of surgically managed Mandibular fractures: Epidemiology, Clinical Profile, patterns, treatments, and outcomes. Int Med J Malays. 2020;27(4):1–4.
Arabacı T, Kermen E, Özkanlar S, et al. Therapeutic effects of melatonin on alveolar bone resorption after experimental periodontitis in rats: a biochemical and immunohistochemical study. J Periodontol. 2015;86(7):874–81.
Scannapieco FA, Gershovich E. The prevention of periodontal disease—An overview. Periodontol 2000. 2020;84(1):9–13.
Domazetovic V, Marcucci G, Iantomasi T, Brandi ML, Vincenzini MT. Oxidative stress in bone remodeling: role of antioxidants. Clin Cases Min Bone Metab. 2017;14(2):209–16.
Balci YH, Toker H, Yildirim A, Tekin MB, Gevrek F, Altunbas N. The effect of luteolin in prevention of periodontal disease in Wistar rats. J Periodontol. 2019;90(12):1481–89.
Sczepanik FSC, Grossi ML, Casati M, et al. Periodontitis is an inflammatory disease of oxidative stress: we should treat it that way. Periodontol 2000. 2020;84(1):45–68.
Georgiou A, Ulloa PC, Van Kessel GMH, Crielaard W, Van der Waal SV. Reactive oxygen species can be traced locally and systemically in apical periodontitis: a systematic review. Arch Oral Biol. 2021;129:105167.
Rastogi A, Joshi P, Contreras E, Gama V, Gama. Remodeling of mitochondrial morphology and function: an emerging hallmark of cellular reprogramming. Cell Stress. 2019;3(6):181–94.
Özcan E, Saygun NI, Ilıkçı R, Karslıoğlu Y, Muşabak U, Yeşillik S. Increased visfatin expression is associated with nuclear factor-kappa B and phosphatidylinositol 3-kinase in periodontal inflammation. Clin Oral Investig. 2017;21(4):1113–21.
Thomas B, Ramesh A, Suresh S, Prasad BR. A comparative evaluation of antioxidant enzymes and selenium in the serum of periodontitis patients with diabetes mellitus type 2. Contemp Clin Dent. 2013;4(2):176–80.
Vo TTT, Chu PM, Tuan VP, Te JSL, Lee IT. The promising role of antioxidant phytochemicals in the prevention and treatment of periodontal disease via the inhibition of oxidative stress pathways: updated insights. Antioxidants. 2020;9(12):1211–29.
Kim AR, Kim JH, Choi YH, et al. The presence of neutrophils causes RANKL expression in periodontal tissue, giving rise to osteoclast formation. J Periodontal Res. 2020;55(6):868–76.
Alpan AL, Kızıldağ A, Özdede M, Karakan NC, Özmen Ö. The effects of taxifolin on alveolar bone in experimental periodontitis in rats. Arch Oral Biol. 2020;117:104823.
Kızıldağ A, Arabaci T, Albayrak M, et al. Therapeutic effects of caffeic acid phenethyl ester on alveolar bone loss in rats with endotoxin-induced periodontitis. J Dent Sci. 2019;14(4):339–45.
Wei L, Teng F, Deng L, et al. Periodontal regeneration using bone morphogenetic protein 2 incorporated biomimetic calcium phosphate in conjunction with barrier membrane: a pre-clinical study in dogs. J Clin Periodontol. 2019;46(12):1254–63.
Nakamura S, Ito T, Okamoto K, et al. Acceleration of bone regeneration of horizontal bone defect in rats using collagen-binding basic fibroblast growth factor combined with collagen scaffolds. J Periodontol. 2019;90(9):1043–52.
Kuroda Y, Kawai T, Goto K, Matsuda S, Matsuda. Clinical application of injectable growth factor for bone regeneration: a systematic review. Inflamm Regeneration. 2019;39(1):1–10.
Hashimi SM. Exogenous noggin binds the BMP-2 receptor and induces alkaline phosphatase activity in osteoblasts. J Cell Biochem. 2019;120(8):13237–42.
Pari L, Monisha P, Jalaludeen AM, Jalaludeen. Beneficial role of diosgenin on oxidative stress in aorta of streptozotocin induced diabetic rats. Eur J Pharmacol 2012(1–3); 691:143 – 50.
Zhang Z, Song C, Fu X, et al. High-dose diosgenin reduces bone loss in ovariectomized rats via attenuation of the RANKL/OPG ratio. Int J Mol Sci. 2014;15(9):17130–47.
Ma MH, Wu XH, He Y, Huang W. Anti-inflammatory and analgesic effects of saponins from D. Zingiberensis CH Wright and diosgenin derivative on mice. Sichuan Da Xue Xue Bao Yi Xue Ban. 2011;42(4):494–7.
Kızıldağ A, Alpan AL, Özdede M, Aydın T, Özmen Ö, et al. Therapeutic effects of diosgenin on alveolar bone loss and apoptosis in diabetic rats with experimental periodontitis. Iran J Basic Med Sci. 2023;26(7):785–90.
Zhang Z, Chen Y, Xiang L, Wang Z, Xiao GG, Ju D. Diosgenin protects against alveolar bone loss in ovariectomized rats via regulating long non–coding RNAs. Exp Ther Med. 2018;16(5):3939–50.
Khosravi Z, Sedaghat R, Baluchnejadmojarad T, Roghani M. Diosgenin ameliorates testicular damage in streptozotocin-diabetic rats through attenuation of apoptosis, oxidative stress, and inflammation. Int Immunopharmacol. 2019;70:37–46.
Zhao S, Niu F, Xu CY. Diosgenin prevents bone loss on retinoic acid-induced osteoporosis in rats. Ir J Med Sci. 2016;185(3):581–7.
Khateeb S, Albalawi A, Alkhedaide A. Diosgenin modulates oxidative stress and inflammation in high-fat diet-induced obesity in mice. Diabetes Metab Syndr Obes. 2022;15:1589–96.
Toker H, Balci YH, Lektemur Alpan A, Gevrek F, Elmastas M, Elmastas. Morphometric and histopathological evaluation of the effect of grape seed proanthocyanidin on alveolar bone loss in experimental diabetes and periodontitis. J Periodontal Res. 2018;53(3):478–86.
Leitão R, Ribeiro R, Chaves H, Rocha F, Lima V, Brito G. Nitric oxide synthase inhibition prevents alveolar bone resorption in experimental periodontitis in rats. J Periodontol. 2005;76(6):956–63.
Z Çiftçi ZZ, Kırzıoğlu Z, Nazıroğlu M, Özmen Ö. Effects of prenatal and postnatal exposure of Wi-Fi on development of teeth and changes in teeth element concentration in rats. Biol Trace Elem Res. 2015;163(1):193–201.
Gong G, Qin Y, Huang W, Zhou S, et al. Protective effects of diosgenin in the hyperlipidemic rat model and in human vascular endothelial cells against hydrogen peroxide-induced apoptosis. Chem Biol Interact. 2010;184(3):366–75.
Gong G, Qin Y, Huang W, Huang. Anti-thrombosis effect of diosgenin extract from Dioscorea Zingiberensis CH Wright in vitro and in vivo. Phytomedicine. 2011;18(3):458–63.
Khuda F, Baharin B, Anuar NNM, Satimin BSF, Nasruddin NS. Effective modalities of periodontitis induction in rat model. J Vet Dent. 2024;41(1):49–57.
Braz-Silva PH, Bergamini ML, Mardegan AP, De Rosa CS, Hasseus B, Jonasson P. Inflammatory profile of chronic apical periodontitis: a literature review. Acta Odontol Scand. 2019;77(3):173–80.
Tayman MA, Kamburoğlu K, Küçük Ö, Ateş FS, Günhan M. Comparison of linear and volumetric measurements obtained from periodontal defects by using cone beam-CT and micro-CT: an in vitro study. Clin Oral Investig. 2019;23(5):2235–44.
Alpan AL, Çalisir M, Kizildag A, Özdede M, Özmen Ö. Effects of a glycogen synthase kinase 3 inhibitor tideglusib on bone regeneration with calvarial defects. J Craniofac Surg. 2020;31(5):1477–82.
Tsukasaki M. RANKL and osteoimmunology in periodontitis. JBMM. 2020;82–90.
Araújo AA, Souza TO, Moura LM, et al. Effect of telmisartan on levels of IL-1, TNF‐α, down‐regulated COX‐2, MMP‐2, MMP‐9 and RANKL/RANK in an experimental periodontitis model. J Clin Periodontol. 2013;40(12):1104–11.
Zhang Z, Yue L, Wang Y. A circRNA-miRNA-mRNA network plays a role in the protective effect of diosgenin on alveolar bone loss in ovariectomized rats. BMC Complement Med Ther. 2020;20(1):1–16.
Li X, Hu L, Ma L, Chang S, et al. Severe periodontitis may influence cementum and dental pulp through inflammation, oxidative stress, and apoptosis. J Periodontol. 2019;90(11):1297–306.
Figueredo CM, Alves JC, de Souza Breves Beiler TFC, Fischer RG. Anti-apoptotic traits in gingival tissue from patients with severe generalized chronic periodontitis. JICD. 2019;10(3):12422.
Alpan AL, Bakar O, Kızıldağ A, Özdede M, Topsakal Ş, Özmen Ö. Effects of taxifolin on bone formation and apoptosis in experimental periodontitis in diabetic rats. Biotech Histochem. 2022;97(4):306–14.
Wu XB, Lai CH, Ho YJ. Anti-apoptotic effects of diosgenin on ovariectomized hearts. Steroids. 2022;179:108980.
Zhong Y, Jin C, Han J. Diosgenin Protects against Kidney Injury and mitochondrial apoptosis Induced by 3-MCPD through the regulation of ER stress, Ca2 + homeostasis, and Bcl2 expression. Mol Nutr Food Res. 2021;65(15):2001202.
Yang D, Liu R, Liu L, Liao H, Wang C, Cao Z. Involvement of CD 147 in alveolar bone remodeling and soft tissue degradation in experimental periodontitis. J Periodontal Res. 2017;52:704–12.
Liao AM, Jung H, Yu JW, et al. Synthesis and biological evaluation of arginyl–diosgenin conjugate as a potential bone tissue engineering agent. Chem Biol Drug Des. 2018;91(1):17–28.
Varela HA, Souza JC, Nascimento RM, et al. Injectable platelet rich fibrin: cell content, morphological, and protein characterization. Clin Oral Investig. 2019;23(3):1309–18.
Acknowledgements
Not applicable.
Funding
This study was self-funded by the authors.
Author information
Authors and Affiliations
Contributions
AK, ALA and TA induced periodontitis, applied experimental procedures. MÖ performed radiographic analyzed and ÖÖ applied the histological examinations. AK and ALA wrote the article. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All experimental procedures in the present study were approved by the University Ethics Committee for Animal Experiments, Denizli (PAUHADYEK-2018/33).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it.The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Kızıldağ, A., Alpan, A.L., Aydın, T.K. et al. Assessment and comparative study of diosgenin doses in alleviating experimental periodontitis. BMC Oral Health 24, 859 (2024). https://doi.org/10.1186/s12903-024-04646-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12903-024-04646-3