Abstract
Background
Acute kidney injury (AKI) is common and associated with adverse outcomes as well as important healthcare costs. However, evidence examining the epidemiology of acute kidney disease (AKD)—recently defined as AKI persisting between 7 and 90 days—remains limited. The aims of this study were to establish the rates of early AKI recovery, progression to AKD and non-recovery; examine risk factors associated with non-recovery and investigate the association between recovery timing and adverse outcomes, in a population-based cohort.
Methods
All adult residents of Tayside & Fife, Scotland, UK, with at least one episode of community or hospital-managed AKI using KDIGO creatinine-based definition during the period 1 January 2010 to 31 December 2018 were identified. Logistic regression was used to examine factors associated with non-recovery, and Cox modelling was used to establish associations between AKI recovery timing and risks of mortality and development of de novo CKD.
Results
Over 9 years, 56,906 patients with at least one AKI episode were identified with 18,773 (33%) of these progressing to AKD. Of those progressing to AKD, 5059 (27%) had still not recovered at day 90 post AKI diagnosis. Risk factors for AKD included: increasing AKI severity, pre-existing cancer or chronic heart failure and recent use of loop diuretics. Compared with early AKI recovery, progression to AKD was associated with increased hazard of 1-year mortality and de novo CKD (HR = 1.20, 95% CI 1.13 to 1.26 and HR = 2.21, 95% CI 1.91 to 2.57 respectively).
Conclusions
These findings highlight the importance of early AKI recognition and management to avoid progression to AKD and long-term adverse outcomes.
Similar content being viewed by others
Background
Globally, 13 million people worldwide are thought to be affected by acute kidney injury (AKI) every year [1]. The incidence is estimated between 7 and 18% amongst hospital in-patients with rates ranging between 30 and 70% in the critically ill [2], making it one of the most common complications following hospital admission. AKI also affects about 400 per 100,000 persons per year in community-based populations with an increasing incidence [3]. It is well established that acute kidney injury (AKI) is associated with adverse outcomes including development or worsening of CKD, [4, 5] kidney failure, cardiovascular events [6, 7], and reduced survival [8]. There is however limited evidence examining post-AKI renal recovery and how short-term recovery affects longer term outcomes. Even though community-acquired may be the most common form of AKI [9], evidence regarding community-acquired/community-managed AKI is sparse. Compared to those managed in-hospital, AKI cases managed in the community could represent a different sample of patients with fewer risk factors, milder cases and better outcomes [10] or conversely a palliative care population. Including these patients allows for a comprehensive depiction of real-word AKI burden and therefore generalizable findings. Over the past 15 years, definitions of both AKI and CKD have been agreed in formal consensus studies, and these definitions are currently applied widely in both research and clinical practice. However, no official definition for AKI recovery currently exists with a lack of consensus on how recovery should be defined [11]. Recently, the term acute kidney disease (AKD) has been proposed by Acute Disease Quality Initiative (ADQI) Workgroup to define an “acute or subacute damage and/or loss of kidney function for a duration of between 7 and 90 days after exposure to an AKI initiating event” [12]. This bridges the gap between AKI and CKD, reflecting increasing recognition that AKI and CKD are interconnected and likely represent a continuum, with patients who have sustained an episode of AKI having an increased risk of either developing de novo CKD or experiencing worsening of underlying CKD [13, 14]. However, important knowledge gaps on the epidemiology including the clinical course of AKD need to be addressed before this terminology can be meaningfully used in clinical practice or research to differentiate early (in the first 7 days) and delayed (between 8 to 90 days) renal recovery after AKI. Furthermore, with the exception of some general key recommendations proposed by KDIGO [15], there are a lack of guidelines targeting AKI and AKD follow-up care.
The aim of this study is to (i) establish the rates of early recovery, progression to AKD and non-recovery following AKI using population based routinely collected healthcare data, (ii) understand which factors are associated with progression to AKD and non-recovery and (iii) explore the relationship between recovery timing and survival as well as development of de novo CKD.
Methods
Study population
This was a population-based cohort formed of all adults (aged 18 or above) in Tayside & Fife, Scotland, UK, who had at least two serum creatinine measurements on different days and presented an AKI episode between 1 January 2010 and 31 December 2018. Cohort entry (index date) was defined as the first day of the first AKI diagnosis during the study period.
Data sources
Data were provided by the Health Informatics Centre (HIC) [16] at the University of Dundee which enables anonymised linkage of health records of all residents of Tayside and Fife, Scotland (population of approximately 800,000 individuals), using the unique Community Health Index (CHI) number, which is used across the whole National Health Service (NHS) healthcare system. The following datasets were linked: creatinine laboratory results (community and hospital), Scottish Morbidity Record of hospital admissions (SMR01), medicines dispensed by community pharmacies, the Scottish Care Initiative-Diabetes Collaboration, National Records of Scotland (NRS) death records and the Scottish Renal Registry.
Linkage to SMR01 data provided information on all hospital admission and discharge dates as well as reasons for admission. Deprivation category was derived from the Scottish Index of Multiple Deprivation [17]. Information on diabetes type and date of diagnosis was obtained from the Scottish Care Information-Diabetes Collaboration [18]. Patients receiving chronic dialysis or with a kidney transplant were identified using the Scottish Renal Registry [19]. Comorbidities were identified at the index date and computed based on past ICD-10 hospitalisation codes using the Quan adaptation [20] of the Deyo Charlson mapping algorithm [21].
Outcomes
The primary outcomes were AKI recovery/non-recovery, death and progression to chronic KRT (Kidney Replacement Therapy). These were assessed at day 7 and day 90 post AKI diagnosis.
Secondary outcomes included progression to de novo chronic kidney disease and recovery timing—in terms of “days to recovery from the first day of AKI diagnosis”. Secondary outcomes were only assessed in a subset of the cohort: amongst patients without pre-existing chronic kidney disease and amongst patients with hospital-managed AKI respectively. The association between recovery timing and 1 year-mortality as well as 1-year de novo CKD were also explored.
Definitions
Detailed descriptions of all the concepts defined below are also available in Table 1 and illustrated in Fig. 1.
Acute kidney injury (AKI)
AKI definition was based on the Kidney Disease: Improving Global Outcomes (KDIGO) creatinine-based criteria [22], using the NHS England AKI e-alert algorithm [23]. The mean creatinine was calculated if there was more than one serum creatinine (SCr) measurement taken on the same day. By linking creatinine measurements to hospital admission data, AKI was further classified into 3 categories: community-acquired/community-managed (CA-CM), community-acquired/hospital-managed (CA-HM) and hospital-acquired (HA). An AKI episode diagnosed in the community was categorised as CA-CM AKI if there was no hospital admission within 7 days post AKI diagnosis. CA-HM AKI was defined as either an AKI episode diagnosed in the community with hospital admission within 7 days post AKI diagnosis or an AKI episode diagnosed on the day (J0) or the next day (J1) following an hospital admission. Finally, the definition of HA AKI was met for patients developing an AKI episode after 2 days in hospital (J2) or later.
Acute kidney disease (AKD)
AKD was defined as a loss of kidney function for a duration between 7 and 90 days after exposure to an AKI initiating event, as per the ADQI Workgroup definition [12]. By default, patients tested within the first 7 days post AKI diagnosis who did not meet criteria for recovery at day 7 entered the AKD phase at that point (provided they did not die or initiate chronic KRT before day 7). Their loss of kidney function was described as AKD until either criterion for recovery was met or day 90 after the AKI initiating event, whichever came first.
Chronic kidney disease (CKD)
CKD was defined according to the KDIGO definition [24] where eGFR was calculated using the CKD-EPI Creatinine Equation [25] using standardised SCr level.
Therefore, the presence of 2 eGFR records below 60 mL/min/1.73 m2 separated by more than 90 days was used to define CKD. Pre-existing CKD was determined using all SCr measurements strictly prior to the index date (first day of AKI diagnosis) whilst de novo CKD was determined using all SCr measurements sampled strictly after the 90th day following the index date.
Progression to CKD was only investigated in patients who had no pre-existing CKD identified prior to the index date.
AKI recovery
Creatinine-based recovery was defined as having a creatinine measurement within 90 days post AKI diagnosis that was either < 1.2 times higher than reference value 1 (RV1) (for AKI identified by creatinine ratio) or < 1.2 times higher than RV1 and < 26.5 μmol/L higher than reference value 2 (RV2) (for AKI identified by creatinine increment) [26]. All SCr measurements within the 90 days post AKI diagnosis were used to search for creatinine-based recovery. The earliest date with a SCr measurement meeting the recovery criteria described above was defined as the date of recovery. In order to avoid misclassification, two additional criteria had to be met to fulfil the definition of creatinine-based recovery: (1) absence of chronic KRT initiation in the 30 days following the date of creatinine recovery and (2) recovery status sustained for at least 3 days (day of creatinine recovery + the two following days—although this could only be applied if tests were available over 3 consecutive days, thereby only avoiding misclassification of detected early relapses as recoveries). Recovery timing was then defined as early if the patient recovered within the first 7 days (day 7 included) or as delayed if criteria for recovery were not met in the first 7 days but were further met during the AKD phase (day 8 to day 90 following AKI diagnosis).
At day 7 and day 90 post AKI diagnosis, patient status was classified into one of the states described in Table 1. Patients who either recovered, died or started chronic KRT during a time period were excluded from the sub-cohort for the next time period (or censored on the date of recovery, death or chronic KRT initiation in survival analyses).
Patients untested within the first 7 days, who did not die or commence chronic KRT during that period, were described but excluded from all statistical analyses as no assumption can be made regarding their recovery status.
Chronic KRT
Chronic KRT was defined as either dialysis initiation (haemodialysis or peritoneal dialysis) or kidney transplantation. The date of chronic dialysis initiation or kidney transplantation is recorded in the Scottish Renal Registry for all patients starting chronic KRT in Scotland with 100% coverage.
Statistical analysis
Characteristics of the study population were summarised by medians and interquartile ranges for continuous measurements (due to non-Normal distributions) and as percentages for categorical factors. Scottish Index of Multiple Deprivation (SIMD) quintiles were summarised as a categorical factor. Age was converted to a categorical variable with approximately similar numbers within each category (less than 65, 65 to 74, 75 to 84, 85+ years old) and youngest patients (< 65 years old) taken as the reference level. Multivariable logistic regression models were implemented to identify risk factors associated with progression to AKD, taking patients with early recovery as the reference level. We then considered patients who entered the AKD phase and determined risk factors associated with non-recovery at day 90 post-AKI diagnosis, using another multivariable logistic model. For both models, we excluded patients who died or initiated chronic KRT, between day 1 and day 7, and between day 8 and day 90 respectively. In a sensitivity analysis, the models were rerun keeping patients who died or initiated chronic KRT during the period considered in the non-recovery group, and risk factors for non-recovery were re-identified. The same candidate risk factors were included in both models: demographic characteristics (age at AKI diagnosis, sex and social deprivation); baseline comorbidities (decreased baseline eGFR, cancer, coronary artery disease, congestive heart failure, diabetes and hypertension) and medications (ACE inhibitors or ARBs, loop diuretics, metformin, NSAIDs, statins) received in the 90 days prior to the index date. Those variables were checked for multicollinearity using a correlation matrix and the variance inflation factor. Frequency of creatinine measurements can provide additional important information that other variables cannot capture and was therefore included in the models as a continuous variable.
Associations between recovery timing and 1-year mortality or de novo CKD were evaluated in the recovery cohort (patients with proven recovery within the 90 days following AKI diagnosis) amongst those who had been tested within the first 7 days, using multivariable Cox proportional hazards (PH) models. People in the recovery cohort were followed up from the recovery date (time 0) until either occurrence of one of the study outcomes (death or de novo CKD) or censored at the last date of data availability (29-05-2019). Development of de novo CKD was assessed using a cause-specific Cox proportional hazard model with all-cause mortality as a competing endpoint. All AKI categories were included when exploring the association between delayed versus early recovery and adverse outcomes. However, the association between days to recovery and adverse outcomes was only investigated in patients with hospital-managed AKI, since the testing frequency (number of SCr measurements divided by number of days from AKI to recovery) was too low in those with community-managed AKI to allow for a precise determination of recovery timing, hence the exclusion of this AKI subgroup from this specific analysis. Days to recovery was included as a continuous variable using P-splines [27] to allow for non-linear effects on the hazard of study outcomes, with reference set as the median recovery time (4 days, HR = 1). Previous work has demonstrated good accuracy of penalised spline smoothing methods to account for nonlinear effects of covariates in Cox models [28]. Selection of the optimal smoothing parameter controlling the penalty applied to the curve was determined on the basis of the Akaike Information Criteria (AIC) [29]. For each individual Cox model, the proportional hazards (PH) assumption was checked using graphical diagnosis based on the scaled Schoenfeld residuals and testing of independence between residuals and time.
All data were analysed using the R statistical programming language (Version 3.6.2, Vienna, Austria) using the following packages: dplyr, data.table, survival, survminer, networkD3, graphics and sjPlot.
Results
Description of the cohort
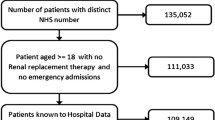
The study cohort consisted of 56,906 patients who had at least one AKI episode during the period 1 January 2010 to 31 December 2018 (Fig. 2). They were followed-up for a median time of 2.1 years (IQR: 0.4 to 4.7 years). Of those 56,906 patients, 13,443 (24%) had AKI diagnosed and managed in the community (community-acquired/community-managed), 22,637 (40%) had AKI diagnosed in the community but managed in hospital (community-acquired/hospital-managed), and for 20,826 (36%), AKI was acquired and managed in-hospital (hospital-acquired). Out of all first AKI episodes during the study period, 45,361 (80%) were stage 1 at diagnosis, 7599 (13%) were stage 2, and 3946 (7%) were stage 3. The median age of the cohort was 75 years old (IQR: 63 to 83) with an evenly distributed men/women ratio. Patients with community-acquired/community-managed AKI were younger (median: 69 years old, IQR: 52 to 80), with a larger proportion of women (65%) and fewer comorbidities, compared to those managed in hospital. Patients’ characteristics at baseline stratified by AKI category are summarised in Table 2. Table 3 summarises outcomes at 90 days and 1 year following the AKI episode, stratified by AKI category and AKI severity. At 1-year post-AKI, 18,381 patients (32.3%) had died, with the lowest crude mortality observed amongst those with community-acquired/community-managed AKI (15.8%) followed by community-acquired/hospital-managed AKI (36.4%) and hospital-acquired AKI (38.5%). Additional file 1: Fig. S1 depicts the overall survival following the AKI episode, stratified by AKI category. Mortality was associated with AKI severity, with 1-year survival of 69.8% and 57.3% for those with AKI stage 1 and 3 respectively.
From the 56,906 patients in the cohort, only 535 (0.94%) commenced chronic KRT after the AKI episode. This was strongly associated with AKI severity, with 6.9% of patients with AKI stage 3 further initiating chronic KRT.
Additional file 1: Table S1 summarises recovery status at 7 and 90 days post AKI, stratified by AKI category and AKI stage at diagnosis. During the first 7 days post AKI diagnosis, 20,041 (35.2%) out of 56,906 recovered, 18,773 (33.0%) were tested but had not recovered, 13,154 (23.1%) were not tested, 4892 (8.6%) died and 46 (0.08%) initiated chronic KRT. Proven recovery rate was highest in people with community-acquired/hospital-managed AKI (47.0%), followed by hospital-acquired (38.4%), and was only 10.4% in people with community-acquired/community-managed AKI. However, a large proportion (66.7%) of patients with community-acquired/community-managed AKI were not tested in the first 7 days, which was not the case amongst those with community-acquired/hospital-managed AKI (8.7% untested) or hospital-acquired AKI (10.7% untested). In a sensitivity analysis, we compared the characteristics of patients with community-acquired/community-managed AKI who were tested versus untested within the first 7 days post-AKI. This analysis, which only included those who had survived and not initiated chronic KRT at day 7, showed that untested patients tended to be younger (median age: 68 vs 72 years old), with fewer comorbidities, a higher baseline eGFR (eGFR> 90 in 47% vs 29%) and milder AKI (stage 1: 93% vs 83%) compared with tested patients (Additional file 1: Table S2). Compared with community-managed AKI, those with hospital-managed AKI (community- and hospital-acquired) were also more often tested within 90 days post-AKI (median number of SCr tests: 6 versus 2).
At day 8, 18,773 (33%) patients from the initial cohort entered the AKD cohort. Of these, 7698 (41%) had a delayed recovery, with a similar proportion in the different AKI categories, whilst 5059 (27%) had still not recovered at day 90. A total of 3695 (19.7%) patients with AKD died between day 8 and day 90, with a higher proportion amongst those who were managed in hospital (21.8% for community-acquired/hospital-managed AKI and 21.4% for hospital-acquired AKI versus 8.7% for community-acquired/community-managed AKI). Of note, 11.7% of those who had been tested but had not recovered at day 7 were not retested between day 8 and day 90 whilst 36.2% of those who had not been tested within the first 7 days had still not been tested at day 90 (Additional file 1: Table S3).
Day 7 status for the whole cohort, as well as day 90 status for those who entered the AKD phase, can be visualised in the Sankey diagrams, with and without stratification by AKI categories (Figs. 3 and 4a,b,c respectively).
Factors associated with progression to AKD and non-recovery
Risk factors associated with progression to AKD amongst tested individuals are summarised in Table 4 and Additional file 1: Figure S2 for the main analysis, in Additional file 1: Table S4 and Figure S3 for the sensitivity analysis (in which patients who died or initiated chronic KRT within the first 7 days were not excluded but rather considered as having not recovered during that period). More severe AKI at diagnosis (stages 2 and 3), a history of cancer diagnosis, a history of congestive heart failure and recent exposure to loop diuretics or metformin were significantly associated with progression to AKD. Conversely, prior exposure to ACE/ARB was associated with early AKI recovery (adjusted OR = 0.85, 95% CI 0.81 to 0.89, p < 0.001). The adjusted odds of progressing to AKD were 2.3 times higher (95% CI 2.2 to 2.5) in those with community-acquired/community-managed AKI than in those with community-acquired/hospital-managed AKI. An older age was negatively associated with progression to AKD, however this trend disappeared in the sensitivity analysis.
The results also showed that a higher number of SCr tests performed over the first 7 days was associated with early AKI recovery (OR = 0.89 for one supplementary test, 95% CI 0.88 to 0.90, p < 0.001). Risk factors associated with non-recovery at day 90 are summarised in Table 5 and Additional file 1: Figure S4 for the main analysis, in Additional file 1: Table S5 and Figure S5 for the sensitivity analysis (in which patients who died or initiated chronic KRT between day 8 and day 90 were not excluded but rather considered as having not recovered during that period). Later AKI stages, hospital-acquired/hospital-managed AKI, community-acquired/community-managed AKI, a history of cancer or chronic heart failure increased the odds for non-recovery in the main and sensitivity analyses. Prior recent exposure to ACE/ARB was also consistently associated with proven recovery at day 90 (aOR = 0.84, 95% CI 0.77 to 0.92 in main analysis, aOR = 0.76, 95% CI 0.70 to 0.81 in the sensitivity analysis). Lower baseline eGFR values were associated with recovery during the AKD phase.
Age was linearly associated with increased odds of non-recovery at day 90 in the sensitivity analysis only. However, community-acquired/community-managed AKI was no longer a risk factor for non-recovery at day 90 in the sensitivity analysis (aOR = 0.99, 95% CI 0.90 to 1.09).
No multicollinearity was detected between the different predictors investigated, with all correlation coefficients below 60% (Additional file 1: Figure S6).
Timing of recovery and long-term outcomes
Tested people with either early or delayed proven recovery formed a recovery cohort (n = 29,330) with 14,486 individuals free of pre-existing CKD. Of those, 2805 (19.4%) subsequently developed de novo CKD with similar proportions across the different AKI categories (19.3%, 257/1334 of those with community-acquired/community-managed AKI, 19.4% 1534/7890 of those with community-acquired/hospital-managed AKI, and 19.3% 1014/5262 of those with hospital-acquired AKI).
Compared to early recovery, delayed recovery was significantly associated with higher risk of death (HR = 1.20, 95% CI 1.13 to 1.26) and de novo CKD (HR = 2.21, 95% CI 1.91 to 2.57) in the subsequent year following the AKI episode (Additional file 1: Table S6, and Fig. 5). This trend was observed in all AKI categories, but the risk was highest in those with community-acquired/community-managed AKI (HR = 1.55, 95 % CI 1.23 to 1.95 for 1-year mortality and HR = 3.25, 95% CI 1.99 to 5.31 for 1-year risk of de novo CKD). Cox analyses showed that the association between delayed recovery and adverse outcomes was time-varying, with the strongest risks observed over the year following the AKI episode and subsequent wearing off, and no significant association after 2 years.
Figure 6 shows the association between all values of recovery timing comprised within 1 and 90 days and relative rates of 1-year mortality (a) as well as development of de novo CKD (b) in patients with hospital-managed AKI (including community-acquired/hospital-managed and hospital-acquired AKI) tested within the first 7 days. Since we would not be able to derive an accurate recovery timing for patients with community-acquired/community-managed AKI (due to the lack of repeat testing), they were excluded from this analysis as well as patients from any AKI category that were untested within the first 7 days post AKI diagnosis. The relative hazard for 1-year mortality increased with recovery timing in a nonlinear fashion, with a sharp initial rise over the first 14 days followed by a plateau. The risk of developing de novo CKD increased more progressively and linearly with recovery timing over the first month following the AKI episode. Beyond this period the risk then stabilised or may even decline.
Discussion
In this large comprehensive population-based cohort study, there were 56,906 patients with community or hospital-acquired AKI, with a median follow-up of 2.1 years. Overall, 35% of the initial cohort had proven creatinine-recovery at day 7 and 49% at day 90 post AKI diagnosis. Risk factors for progression to AKD included AKI severity, pre-existing cancer or chronic heart failure, recent use of loop diuretics, community-managed AKI as well as hospital-acquired AKI. Of note, being exposed to ACE/ARB was consistently associated with AKI recovery at both day 7 and day 90 (adjusted OR: 0.85, 95% CI 0.81-0.89 and 0.86, 95% CI 0.78–0.95 respectively). Compared with early AKI recovery, progression to AKD was associated with increased risks of 1-year mortality and de novo CKD (HR = 1.20, 95% CI 1.13 to 1.26 and HR = 2.21, 95% CI 1.91 to 2.57 respectively). The first 14 days following an AKI episode were identified as a critical window where each additional day was associated with a rapid increase in risk for adverse outcomes.
It is concerning that in our cohort, a remarkably high proportion of patients with community-acquired/community-managed AKI (67%) were untested at day 7. Amongst those, 36% remained untested at day 90. Patients untested within the first 7 days appeared to be younger, with a higher baseline eGFR and milder AKI which may explain the lack of repeat testing in this fitter population. Furthermore, it is worth noting that in this area of Scotland, repeat testing within 7 days post AKI diagnosis only became more common after the introduction of the National Health Service England Acute Kidney Injury electronic alert algorithm in 2015 [30]. Therefore, a major part of our data captures the practices in place prior to the introduction of this system. Although testing is not always appropriate, for example in palliative care settings or in the context of particularly frail patients, these findings raise questions regarding the management of AKI in the community setting. Moreover, we showed that AKI severity, history of cancer, chronic heart failure and receiving loop diuretics were consistent risk factors for progression to AKD. In line with our result, a previous study aiming to predict recovery following dialysis-requiring AKI showed that patients who recovered were less likely to have a history of heart failure [31]. The same study also identified younger age as a predictor for recovery. Our sensitivity analysis showed that an older age was associated with an increased risk of early death following the AKI episode but not with non-recovery. In both unadjusted and adjusted analyses, we consistently found that prior use of ACE/ARB was significantly associated with AKI recovery at both day 7 and day 90. The use of ACE/ARB in the context of AKI is widely debated. The KDIGO recommendation is to stop potentially nephrotoxic drug (including ACE/ARB in this category) during AKI. However, emerging evidence suggests that ACE/ARB should not be considered as nephrotoxic [32] and could even be associated with improved AKI recovery and reduced subsequent mortality [33,34,35]. The association between NSAIDs exposure and non-recovery was either non-significant or protective at both day 7 and day 90. NSAIDs-related AKI remain rare events and this observed association may be related to some residual confounding by indication where physicians avoid prescribing NSAIDs to frailer patients they perceive to be at higher risk of NSAIDs-related adverse outcomes such as AKI [36]. Another hypothesis for this finding is the usually rapid renal recovery of NSAIDs-induced AKI (typically within 72 to 96 h provided diagnosis is made early and NSAIDs are promptly discontinued) [37]. Surprisingly, our models suggested an association between lower baseline eGFR values and AKI recovery. This may be because those with lower baseline eGFR values will have greater fluctuations in serum creatinine related to volume status. Recent metformin use appeared as a risk factor for AKD but then as a strong protective factor in our sensitivity analysis when considering non-recovery at day 90. The latter is consistent with previous work reporting improved short-term survival following incident AKI in those exposed to metformin [38, 39].
The association between AKI, AKD and CKD is complex and mortality as well as progression to CKD after an AKI episode have been documented in many studies [8]. Our results showed that compared with early AKI recovery, progression to AKD was associated with both 1-year mortality and development of de novo CKD. This is consistent with previously published data conducted amongst patients admitted for cardiovascular reasons, which demonstrated that AKD was associated with both short- (90 days) [40] and long-term (5 years) [41] risk of death and adverse renal events. However, in a cohort of patient admitted for sepsis-associated AKI, individuals with early AKI reversal had similar mortality rates as those developing AKD [42].
Similarly, we found that risk of death and de novo CKD increased progressively with recovery timing. This is in line with previous work conducted in a cohort of adult US veterans, suggesting that recovery timing may act as an independent predictor for future loss of kidney function [43]. Bhatraju et al. found that recovering within the first 72 h immediately following the AKI episode may be crucial to avoid major adverse kidney events [44]. Compared to a rapid reversal (within 48 h), persistent AKI was also significantly associated with a higher 1-year mortality rate [45]. Recovery timing therefore appears to be a major factor in the context of AKI recovery, which adds important prognostic information regarding adverse long-term outcomes following an AKI episode. In this study, modelling of precise recovery timing showed that the first 2 to 3 weeks following an AKI episode represent a critical window where risk for adverse outcomes increase most rapidly and where interventions are therefore most likely to reduce risk of progression to CKD or early mortality. Mechanisms potentially explaining the association between longer recovery timing and worse outcomes include persistent inflammation, prolonged renin-angiotensin system activation with long-term hypertension even after recovery and repeated cellular injury due to local ischemia leading to kidney damage such as tubular or glomerular injury [46].
Our study has several strengths. These include the comprehensive nature of the unselected population-based cohort covering a large geographical population of Scotland (about 790,000 individuals), the large number of AKI episodes recorded over a 9-year period and the robust methodology accounting for major confounders, with sensitivity analyses ensuring the consistency of findings. The inclusion of community-managed AKI brings new insights regarding level of care and risks associated with treatment outside hospitals, for which data are currently lacking. Finally, our strict definition of sustained recovery reduces misclassification of relapse as recovery. This work helps fill important knowledge gaps in the current understanding of renal recovery after AKI but also comes with a number of limitations. Firstly, this study was conducted in a specific geographic area of Scotland and may not be generalizable to other AKI cohorts worldwide. However, it remains an unselected population-based cohort whose characteristics are similar to that of previous studies and therefore generalizable to other high-income countries. It should be noted that a large proportion (67%) of patients with community-managed AKI were untested during the first 7 days post AKI diagnosis; hence, all conclusions made on this subgroup were based on the subset of patients who had available follow-up SCr data, with subsequent risks of ascertainment and selection bias. However, our sensitivity analysis showed that patients with community-acquired/community-managed AKI tested within the first 7 days have very similar characteristics to that of patients with hospital-acquired AKI, making comparisons relevant. It should be noted that in the absence of any accepted definition [11], we chose the definition of AKI recovery (< 1.2 times higher than baseline SCr) as per previous work [26] but other studies in the field may have used different thresholds, making between-study comparisons less straightforward. Furthermore, we chose to focus on AKD occurring after an AKI event and do not examine AKD occurring without a preceding AKI episode [47]. Another limitation of this study is the lack of data availability regarding the use of temporary dialysis for AKI management, with subsequent risk of AKI recovery misclassification in a small proportion of hospital-managed AKI episodes. Finally, due to the observational nature of this study, risk of residual confounding remains, despite our efforts to control for all important variables.
Conclusions
Our data demonstrates that AKD is common in patients with AKI and associated with deleterious outcomes such as early mortality or de novo CKD, especially when AKI management takes place outside hospitals. Patients with community-managed AKI should be more widely tested within the first 7 days post AKI diagnosis to ensure optimal management. As risks for adverse outcomes increase sharply during the immediate period (2 to 3 weeks) following AKI diagnosis, this work stresses the importance of early AKI recovery to avoid long-term consequences. Patients with cancer, chronic heart failure and those exposed to diuretics may be at particularly high risk of progression to AKD and non-recovery, therefore deserving extra attention. Although more evidence is needed to guide clinical practice, our results suggested that ACE/ARB may have a protective effect in a context of AKI, with improved recovery amongst recently exposed individuals. Increased awareness and strategies for the management of patients with AKD are needed to maximise early recovery and minimise AKI-related harms.
Availability of data and materials
The data controller of the data analysed is NHS Tayside. Patient level data are available subject to standard information governance requirements for use of anonymised, unconsented NHS data https://www.dundee.ac.uk/hic/.
Abbreviations
- ACE/ARB:
-
Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers
- ADQI:
-
Acute Disease Quality Initiative
- AKD:
-
Acute kidney disease
- AKI:
-
Acute kidney injury
- CA-CM AKI:
-
Community-acquired/community-managed AKI
- CA-HM AKI:
-
Community-acquired/hospital-managed AKI
- CKD:
-
Chronic kidney disease
- eGFR:
-
Estimates glomerular filtration rate
- HA AKI:
-
Hospital-acquired/hospital-managed AKI
- HIC:
-
Health Informatics Centre
- HR:
-
Hazard ratio
- ICD-10:
-
International Classification of Diseases – version 10
- IQR:
-
Interquartile range
- KDIGO:
-
Kidney Disease Improving Global Outcomes
- KRT:
-
Kidney replacement therapy
- KTR:
-
Kidney transplant recipients
- NHS:
-
National Health Services
- NRS:
-
National Record of Scotland
- NSAIDs:
-
Nonsteroidal anti-inflammatory drugs
- OR:
-
Odds ratio
- RV:
-
Reference value
- SCr:
-
Serum creatinine
- SIMD:
-
Scottish Index of Multiple Deprivation
- SMR01:
-
Scottish Morbidity Record of hospital admissions
- UK:
-
United Kingdom
- US:
-
United States
References
Mehta RL, Cerda J, Burdmann EA, Tonelli M, Garcia-Garcia G, Jha V, et al. International Society of Nephrology’s 0by25 initiative for acute kidney injury (zero preventable deaths by 2025): a human rights case for nephrology. Lancet. 2015;385(9987):2616–43.
Lewington AJ, Cerda J, Mehta RL. Raising awareness of acute kidney injury: a global perspective of a silent killer. Kidney Int. 2013;84(3):457–67.
Hsu CY, McCulloch CE, Fan D, Ordonez JD, Chertow GM, Go AS. Community-based incidence of acute renal failure. Kidney Int. 2007;72(2):208–12.
Rubin S, Orieux A, Clouzeau B, Rigothier C, Combe C, Gruson D, et al. The incidence of chronic kidney disease three years after non-severe acute kidney injury in critically ill patients: a single-center cohort study. J Clin Med. 2019;8(12):2215. https://doi.org/10.3390/jcm8122215.
Coca SG, Singanamala S, Parikh CR. Chronic kidney disease after acute kidney injury: a systematic review and meta-analysis. Kidney Int. 2012;81(5):442–8.
Chawla LS, Amdur RL, Shaw AD, Faselis C, Palant CE, Kimmel PL. Association between AKI and long-term renal and cardiovascular outcomes in United States veterans. Clin J Am Soc Nephrol. 2014;9(3):448–56.
Omotoso BA, Abdel-Rahman EM, Xin W, Ma JZ, Scully KW, Arogundade FA, et al. Acute kidney injury (AKI) outcome, a predictor of long-term major adverse cardiovascular events (MACE). Clin Nephrol. 2016;85(1):1–11.
Coca SG, Yusuf B, Shlipak MG, Garg AX, Parikh CR. Long-term risk of mortality and other adverse outcomes after acute kidney injury: a systematic review and meta-analysis. Am J Kidney Dis. 2009;53(6):961–73.
Mesropian PD, Othersen J, Mason D, Wang J, Asif A, Mathew RO. Community-acquired acute kidney injury: a challenge and opportunity for primary care in kidney health. Nephrology (Carlton). 2016;21(9):729–35.
Huang L, Xue C, Kuai J, Ruan M, Yang B, Chen X, et al. Clinical characteristics and outcomes of community-acquired versus hospital-acquired acute kidney injury: a meta-analysis. Kidney Blood Press Res. 2019;44(5):879–96.
Guthrie G, Guthrie B, Walker H, James MT, Selby NM, Tonelli M, et al. Developing an AKI consensus definition for database research: findings from a scoping review and expert opinion using a Delphi process. Am J Kidney Dis. 2022;79(4):488–96.e1. https://doi.org/10.1053/j.ajkd.2021.05.019.
Chawla LS, Bellomo R, Bihorac A, Goldstein SL, Siew ED, Bagshaw SM, et al. Acute kidney disease and renal recovery: consensus report of the Acute Disease Quality Initiative (ADQI) 16 Workgroup. Nat Rev Nephrol. 2017;13(4):241–57.
Takaori K, Yanagita M. Insights into the mechanisms of the acute kidney injury-to-chronic kidney disease continuum. Nephron. 2016;134(3):172–6.
Chou YH, Huang TM, Chu TS. Novel insights into acute kidney injury-chronic kidney disease continuum and the role of renin-angiotensin system. J Formos Med Assoc. 2017;116(9):652–9.
Lameire NH, Levin A, Kellum JA, Cheung M, Jadoul M, Winkelmayer WC, et al. Harmonizing acute and chronic kidney disease definition and classification: report of a Kidney Disease: Improving Global Outcomes (KDIGO) Consensus Conference. Kidney Int. 2021;100(3):516–26.
University of Dundee, Health Informatics Centre Services. Available from: www.dundee.ac.uk/hic/hicservices/. Accessed 9 June 2022.
Scottish Government, Scottish Index of Multiple Deprivation 2020. Available from: www.gov.scot/collections/scottish-index-of-multiple-deprivation-2020/. Accessed 9 June 2022.
NHS Scotland, The Scottish Care Information – Diabetes Collaboration (SCI-DC), 2015. Available from: www.sci-diabetes.scot.nhs.uk/. Accessed 9 June 2022.
NHS Scotland, The Scottish Renal Registry (SRR), 2022. Available from: www.srr.scot.nhs.uk/. Accessed 9 June 2022.
Quan H, Sundararajan V, Halfon P, Fong A, Burnand B, Luthi JC, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. 2005;43(11):1130–9.
Wasey J.O. Package ‘icd’, 2017. Available from: cran.nexr.com/web/packages/icd/icd.pdf. Accessed 9 June 2022.
KDIGO, Acute kidney injury (AKI), 2012. Available from: kdigo.org/guidelines/acute-kidney-injury/. Accessed 9 June 2022.
England N. Algorithm for detecting acute repeat kidney injury (AKI) based on serum creatinine changes with time. London: Renal Association, UK Renal Registry; 2014. Available from: https://www.england.nhs.uk/wp-content/uploads/2014/06/psa-aki-alg.pdf
Levey AS, Eckardt KU, Tsukamoto Y, Levin A, Coresh J, Rossert J, et al. Definition and classification of chronic kidney disease: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int. 2005;67(6):2089–100.
Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–12.
Sawhney S, Marks A, Fluck N, Levin A, Prescott G, Black C. Intermediate and long-term outcomes of survivors of acute kidney injury episodes: a large population-based cohort study. Am J Kidney Dis. 2017;69(1):18–28.
Eilers PH, Marx BD. Flexible smoothing with B-splines and penalties. Stat Sci. 1996;11(2):89–121.
Roshani D, Ghaderi E. Comparing smoothing techniques for fitting the nonlinear effect of covariate in Cox models. Acta Inform Med. 2016;24(1):38–41.
Malloy EJ, Spiegelman D, Eisen EA. Comparing measures of model selection for penalized splines in Cox models. Comput Stat Data Anal. 2009;53(7):2605–16.
Aiyegbusi O, Witham MD, Lim M, Gauld G, Bell S. Impact of introducing electronic acute kidney injury alerts in primary care. Clin Kidney J. 2019;12(2):253–7.
Lee BJ, Hsu CY, Parikh R, McCulloch CE, Tan TC, Liu KD, et al. Predicting renal recovery after dialysis-requiring acute kidney injury. Kidney Int Rep. 2019;4(4):571–81.
Jones M, Tomson C. Acute kidney injury and ‘nephrotoxins’: mind your language. Clin Med (Lond). 2018;18(5):384–6.
Tomson C, Tomlinson LA. Stopping RAS inhibitors to minimize AKI: more harm than good? Clin J Am Soc Nephrol. 2019;14(4):617–9.
Palevsky PM, Zhang JH, Seliger SL, Emanuele N, Fried LF, Study VN-D. Incidence, severity, and outcomes of AKI associated with dual renin-angiotensin system blockade. Clin J Am Soc Nephrol. 2016;11(11):1944–53.
Gayat E, Hollinger A, Cariou A, Deye N, Vieillard-Baron A, Jaber S, et al. Impact of angiotensin-converting enzyme inhibitors or receptor blockers on post-ICU discharge outcome in patients with acute kidney injury. Intensive Care Med. 2018;44(5):598–605.
Zhang X, Donnan PT, Bell S, Guthrie B. Non-steroidal anti-inflammatory drug induced acute kidney injury in the community dwelling general population and people with chronic kidney disease: systematic review and meta-analysis. BMC Nephrol. 2017;18(1):256.
Whelton A. Nephrotoxicity of nonsteroidal anti-inflammatory drugs: physiologic foundations and clinical implications. Am J Med. 1999;106(5B):13S–24S.
Bell S, Farran B, McGurnaghan S, McCrimmon RJ, Leese GP, Petrie JR, et al. Risk of acute kidney injury and survival in patients treated with Metformin: an observational cohort study. BMC Nephrol. 2017;18(1):163.
Yang Q, Zheng J, Wen D, Chen X, Chen W, Chen W, et al. Association between metformin use on admission and outcomes in intensive care unit patients with acute kidney injury and type 2 diabetes: a retrospective cohort study. J Crit Care. 2021;62:206–11.
Matsuura R, Iwagami M, Moriya H, Ohtake T, Hamasaki Y, Nangaku M, et al. The clinical course of acute kidney disease after cardiac surgery: a retrospective observational study. Sci Rep. 2020;10(1):6490.
Chen JJ, Lee TH, Kuo G, Yen CL, Chen SW, Chu PH, et al. Acute kidney disease after acute decompensated heart failure. Kidney Int Rep. 2022;7(3):526–36.
Peerapornratana S, Priyanka P, Wang S, Smith A, Singbartl K, Palevsky PM, et al. Sepsis-associated acute kidney disease. Kidney Int Rep. 2020;5(6):839–50.
Siew ED, Abdel-Kader K, Perkins AM, Greevy RA Jr, Parr SK, Horner J, et al. Timing of recovery from moderate to severe AKI and the risk for future loss of kidney function. Am J Kidney Dis. 2020;75(2):204–13.
Bhatraju PK, Zelnick LR, Chinchilli VM, Moledina DG, Coca SG, Parikh CR, et al. Association between early recovery of kidney function after acute kidney injury and long-term clinical outcomes. JAMA Netw Open. 2020;3(4):e202682.
Ozrazgat-Baslanti T, Loftus TJ, Ren Y, Adiyeke E, Miao S, Hashemighouchani H, et al. Association of persistent acute kidney injury and renal recovery with mortality in hospitalised patients. BMJ Health Care Inform. 2021;28(1):e100458. https://doi.org/10.1136/bmjhci-2021-100458.
Tan HL, Yap JQ, Qian Q. Acute kidney injury: tubular markers and risk for chronic kidney disease and end-stage kidney failure. Blood Purif. 2016;41(1-3):144–50.
James MT, Levey AS, Tonelli M, Tan Z, Barry R, Pannu N, et al. Incidence and prognosis of acute kidney diseases and disorders using an integrated approach to laboratory measurements in a universal health care system. JAMA Network Open. 2019;2(4):e191795.
Acknowledgements
We wish to thank the Health Informatics Centre (HIC), University of Dundee, for providing the data.
Funding
This work was funded by Tenovus Tayside (Grant No. T18-26).
Author information
Authors and Affiliations
Contributions
HW and SB conceived the study. HW, SB and EL designed the study. HW and EL analysed the data and performed the statistical analyses. EL, HW and SB wrote the manuscript. All authors edited the manuscript and approved the final version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Data linkage and anonymisation was carried out under HIC Standard Operating Procedures which have been approved by the NHS Research Ethics Service, and all analyses were conducted on anonymised data in the HIC secure Safe Haven. The study was approved by the NHS Tayside Caldicott Guardian, and individual study approval by the NHS Research Ethics Service was therefore not required.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Additional file 1: Figure S1
: Survival curve stratified by AKI category. Table S1: Recovery status at 7- and 90-days post-AKI diagnosis. Table S2: Characteristics of tested versus untested patients with CA-CM AKI. Table S3: 90-day status of patients untested within the first 7 days post AKI diagnosis. Figure S2: Association between individual predictors and progression to AKD (main analysis). Table S4 and Figure S3: Association between individual predictors and non-recovery at day 7 (sensitivity analysis). Figure S4: Association between individual predictors and non-recovery at day 90 in patients who entered the AKD phase (main analysis). Table S5 and Figure S5: Association between individual predictors and non-recovery at day 90 in patients who entered the AKD phase (sensitivity analysis). Figure S6: Correlation matrix for pairs of candidate risk factors. Table S6: Association between progression to AKD and subsequent risk of death or development of de novo CKD.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Wang, H., Lambourg, E., Guthrie, B. et al. Patient outcomes following AKI and AKD: a population-based cohort study. BMC Med 20, 229 (2022). https://doi.org/10.1186/s12916-022-02428-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12916-022-02428-8