Abstract
Background
Obesity is a significant public health issue associated with various chronic diseases. Research has indicated that chia seeds have the potential to improve cardiometabolic health. However, due to the diversity of research and inconsistencies in study design, further investigation is needed to fully understand their clinical effects on overweight individuals. This review aims to comprehensively analyze the available evidence on the effects of chia seeds on cardiometabolic indices in overweight populations through a meta-analysis.
Methods
A comprehensive literature search was performed across PubMed, Web of Science, Scopus, and Embase databases from their inception until 01-03-2024 to identify randomized controlled trials (RCTs) evaluating the effect of chia on cardiometabolic indices in overweight subjects. The search strategy incorporated both Medical Subject Headings (MeSH). Following the screening, ten RCTs were finally included. The data, including subject characteristics, study design, and changes in serum biomarkers, were extracted and analyzed using Stata software version 18.
Results
The meta-analysis results reveal that chia supplementation no significant changes in lipid profile, including triglycerides (TG) (MD: − 5.80 mg/dL, p = 0.47), total cholesterol (TC) (MD: − 0.29 mg/dL, p = 0.95), high-density lipoprotein (HDL) (MD: 1.53 mg/dL, p = 0.33), and low-density lipoprotein (LDL) (MD: 0.63 mg/dL, p = 0.88). Similarity fasting blood glucose (FBG) (MD: − 0.03 mg/dL, p = 0.98), hemoglobin A1c (HbA1c) (MD: − 0.13%, p = 0.13), and insulin levels (MD: 0.45 µIU/mL, p = 0.78). However, chia seed supplementation was associated with a significant reduction in C-reactive protein (CRP) (MD: − 1.18 mg/L, p < 0.0001), but no significant changes were observed in interleukin-6 (IL-6) (MD: − 0.15, p = 0.70) or tumor necrosis factor-alpha (TNF-α) (MD: 0.03, p = 0.91). There was no significant effect on body mass index (BMI) (MD: 0.1 kg/m2, p = 0.91), but a significant reduction in waist circumference (WC) (MD: − 2.82 cm, p < 0.001) was noted. Additionally, chia seed supplementation resulted in a significant reduction in systolic blood pressure (BP) (MD: − 3.27 mmHg, p = 0.03), though diastolic BP changes were non-significant (MD: − 2.69 mmHg, p = 0.09). The studies showed low to moderate heterogeneity in outcome measures, with I2 < 50%.
Conclusion
Chia seed supplementation does not significantly impact most lipid profile parameters and glycemic markers. However, it shows potential benefits in reducing WC, BP, and CRP. While chia seeds can be a valuable addition to cardiometabolic health management, they should be part of a broader health strategy that includes a balanced diet, exercise, and lifestyle modifications for optimal results.
Similar content being viewed by others
Introduction
In recent decades, the rates of overweight and obesity have significantly increased, becoming major public health concerns [1]. Obesity, characterized by excessive fat accumulation that poses health risks, affects over 650 million people worldwide, according to the World Health Organization (WHO). This rising prevalence of obesity signifies a widespread pandemic with significant health implications [2]. Obesity is linked to a higher risk of developing other conditions such as type 2 diabetes mellitus (T2DM), dyslipidemia, metabolic syndrome (MetS), and cardiovascular diseases (CVD) [3]. Cardiometabolic conditions remain the leading causes of death and illness globally, according to a 2000–2019 assessment [4]. Controlling anthropometric measures, blood pressure, glycemic indices, lipid profiles, and inflammatory markers is crucial for maintaining health [5]. These markers are strongly linked to a higher risk of chronic illnesses like obesity [6]. The interest in developing effective therapies to enhance these health indicators is increasing. Dietary adjustments are highlighted as crucial in preventing metabolic diseases, supported by experimental and epidemiological research [7, 8]. There is a growing demand for nutritionally balanced herbal products, with chia seed supplementation gaining particular interest [9, 10].
Salvia hispanica, commonly known as chia seeds, is an herbaceous plant in the Lamiaceae family. It is rich in protein, omega-3 fatty acids, and both soluble and insoluble fibers. Traditionally used as a dietary and therapeutic resource, chia seeds have recently garnered interest for their unique chemical profile [11, 12]. They may aid in weight reduction and address obesity-related challenges, offering potential health benefits. Additionally, chia seeds possess anti-inflammatory and antioxidant properties, further contributing to their therapeutic potential [11,12,13].
Although research specifically targeting overweight patients is limited, chia seeds are thought to offer several cardiometabolic benefits. According to recent research, chia seeds may improve lipid profile, glycemic markers, antioxidant properties, blood pressure (BP) regulation, and weight management [14, 15]. Over the last two decades, research has indicated that chia seeds have beneficial outcomes regarding insulin resistance, lipid abnormality, glucose tolerance, and obesity [16,17,18,19,20]. According to recent research, chia seeds may improve blood lipid profiles because of their numerous beneficial components; this is due to their ability to lower blood glucose levels, fight against microbes, reduce blood pressure, and stimulate the immune system [21]. Nevertheless, these studies exhibit significant disparities in the number of participants, individual characteristics, and sample composition, with a greater emphasis on animal research rather than human studies.
Despite the increasing number of studies examining the effects of chia seed supplementation on health markers related to overweight and cardiometabolic conditions, there remains a significant gap in the consistent and comprehensive analysis of these effects in human populations. Existing research shows varied results across different sample sizes and characteristics. Therefore, this study aims to systematically review and evaluate the cardiometabolic effects of chia seeds on overweight individuals from diverse populations, specifically focusing on their impact on cardiometabolic factors.
Methods
Protocol and registration
This systematic review and meta-analysis were conducted following the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement to ensure transparency and comprehensive reporting [22]. The research protocol was registered with the International Prospective Register of Systematic Reviews (PROSPERO) under the registration number CRD42024538150 to facilitate methodological rigor and reproducibility.
Search strategy
This systematic review and meta-analysis involved a comprehensive search of several scientific databases, including PubMed, Web of Science, Scopus, Embase, and Google Scholar, from inception until March 2024. The objective was to identify RCTs evaluating the effect of Chia (Salvia hispanica L.) on cardiometabolic indices in overweight patients. The search strategy utilized a combination of relevant MeSH (Medical Subject Headings) terms and non-MeSH phrases, employing Boolean operators (AND, OR) by searching [Title/Abstract] without any time restrictions. The search was limited to articles published in English. Additionally, the reference lists of all relevant articles were manually searched to identify any potentially eligible studies, including grey literature, that may have been missed during the database searches.
Inclusion and exclusion criteria
We included original RCTs that investigated the impact of the Chia (intervention) on cardiometabolic indices (outcome) among overweight healthy subjects (population). Furthermore, our inclusion criteria embrace studies exclusively in English. Our exclusion criteria meticulously refined the focus of our research by excluding various study designs such as cross-sectional, case series, case–control, and cohort studies, as well as systematic reviews, meta-analyses, abstracts, and letters to the editor. Additionally, we excluded in vitro, in vivo, and animal studies to streamline our analysis of human subjects. Furthermore, articles not published in English were excluded to ensure consistency in data interpretation and facilitate comprehension. These stringent criteria enhanced the validity and applicability of our research findings to the target population of overweight individuals undergoing Chia supplementation. Guided by the Patient, Intervention, Comparison, and Outcome (PICO) model [23], our research question was formulated (Table 1).
Data extraction
The data from the included studies were screened and extracted independently by two reviewers (E.SH.R. and A.A.) based on the study inclusion criteria explained above, and any disagreements during data extraction were resolved through discussion between the two reviewers, if consensus could not be reached, a third reviewer (M.K.) was consulted. Extracted data including the name of the first author, year of publication, participants characteristics (mean age, gender), study design, sample sizes, country setting, and mean ± standard deviation (SD) of changes in cardiometabolic indices, including anthropometric indices (BMI, WC), blood pressure (systolic and diastolic), glycemic markers (FBG, Insulin, HbA1c,), lipid profile (LDL, HDL, TG, TC), and inflammatory markers (CRP, IL-6, TNF-α) in each group of intervention and control.
Quality assessment
Two researchers (S.P. & E.SH.R.) meticulously reviewed the methodological quality of each included study using the Revised Cochrane risk-of-bias instrument for randomized trials (RoB-2). Any disagreements were resolved through discussion between the two reviewers, and if consensus could not be reached, a third reviewer (M.K.) was consulted. RoB-2 is specifically designed to assess the likelihood of bias in randomized controlled trials (RCTs), ensuring a rigorous evaluation process. The RoB2 tool examines various domains of bias, including bias arising from the randomization process, deviations from intended interventions, missing outcome data, measurement of the outcome, and selection of the reported result [16] (see Fig. 1).
Statistical analyses
The effect size for the outcomes was evaluated using the mean difference (MD) along with 95% confidence intervals (CI) to quantify the impact of chia seed supplementation on cardiometabolic indices [24]. When studies provided only baseline and end-of-trial means without reporting standard deviations (SD) for changes, the SD for the net change was estimated using the Follmann method, assuming a correlation coefficient (R) 0.5. If the Standard Error (SE) was provided, the SD was calculated using the formula SD = SE × sqrt(n), where n represents the sample size in each group [25]. Heterogeneity among studies was assessed using the I2 statistic and Cochrane’s Q-test, with I2 values greater than 50% and a P-value of less than 0.05, indicating substantial heterogeneity [26]. A random-effects model was applied to account for potential variability across studies. For trials assessing multiple interventions, each was analyzed as a separate effect size to ensure accuracy. Sensitivity analysis was conducted to test the robustness of the findings, while subgroup analyses explored the influence of specific study characteristics and excluded studies with a high risk of bias. Begg’s and Egger’s tests were performed to assess publication bias, and funnel plots were used to visually inspect for asymmetry. All statistical analyses were conducted using Stata software version 18.
Results
Study selection
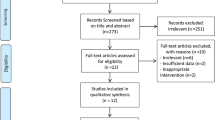
A comprehensive search of four databases, including PubMed, Web-of-Science, Scopus, Embase, and a manual search in Google Scholar, yielded a total of 2147 articles. After the removal of 784 duplicate records, 1363 unique studies were subjected to the selection process. The initial screening of titles and abstracts excluded 1346 papers from 1363 studies because of exclusion criteria (review studies, non-human studies, not being RCT, etc.), and 27 studies had inclusion criteria. This left 27 papers for full-text assessment, during which 17 more publications were excluded due to reasons such as Not being RCT (n = 5), inadequate data (n = 4), not being overweight (n = 5), or lack of relevance to the research question (n = 3). Consequently, ten studies were deemed suitable for data extraction and included all RCTs in the final quantitative meta-analysis (Fig. 2). These 10 RCTs formed the basis for the meta-analysis, providing robust data for evaluating the research hypotheses.
Study characteristics
The review included studies published from 2007 to 2023, encompassing 424 participants: 210 in intervention and 214 in control groups. Study durations ranged from 10 to 24 weeks, and participants' ages varied from 8 to 64 years.
Most studies assessed chia seed consumption in grams per day (gr/day). Chia seeds were supplied in several forms, including whole, ground, and chia seed oil. The outcome measurements were cardiometabolic indices and risk factors for metabolic and cardiovascular diseases, including anthropometric index (body weight, BMI, WC), BP, lipid profile (TG, TC, HDL, LDL), glycemic markers (FBG, Hb1AC, Insulin), and inflammatory markers (CRP, IL-6, TNF-α) (Table 2).
Effect on anthropometric indices (BMI and WC)
In this meta-analysis, five and four studies examined the effect of chia supplementation on BMI and WC, respectively. They concluded that there was no statistically significant (MD = 0.1 kg/m2, 95% CI [− 0.36, 0.43], I2 = 12.58%, p = 0.86) effect on BMI compared to the control group (Fig. 2A). However, the intervention group experienced a significant − 2.82 cm decrease in WC compared to the control group (MD = − 2.82 cm, 95% CI [− 4.32, − 1.31], I2 = 0.0%, p < 0.001) (Fig. 3B).
Effect on blood pressure (BP)
In this meta-analysis, seven studies examined the effect of chia seed supplementation on systolic BP, while five examined its impact on diastolic BP. The intervention group experienced a significant (MD: − 3.27 mmHg, 95% CI [− 6.28, − 0.27], I2 = 0.0%, p-value = 0.03) reduction of − 3.27 mmHg in systolic BP compared to the control group (Fig. 4A). On the other hand, the intervention group showed a slight non-significant (MD: − 2.69 mmHg, 95% CI [− 5.76, 0.38], I2 = 43.03%, p = 0.09) reduction in diastolic BP compared to the control group (Fig. 4B).
Effect on lipid profile
In this meta-analysis, seven studies reported triglycerides (TG), in which 5.80 mg/dL was reduced, but it was not significant (MD: − 5.80 mg/dL, 95% CI [− 21.50, 9.89], I2 = 00.01%, p = 0.47) (Fig. 5A). Similarly, eight studies reported that total cholesterol (TC) 5.80 mg/dL was reduced, but it was not significant (MD: − 0.29 mg/dL, 95% CI [− 8.49, 7.92], I2 = 18.56%, p = 0.95) (Fig. 5B). Seven studies reported mean differences in high-density lipoprotein (HDL) and low-density lipoprotein (LDL). HDL had a non-significant decrease of 1.53 mg/dL (MD: − 1.53 mg/dL, 95% CI [− 4.58, 1.53], I2 = 32.09%, p = 0.33) (Fig. 5C), and LDL exhibited a non-significant increase of 0.63 mg/dL (MD: 0.63 mg/dL, 95% CI [− 7.59, 8.86], I2 = 10.48%, p = 0.88) (Fig. 5D).
Effect on glycemic markers
The meta-analysis examining the effects of chia seed supplementation in overweight subjects reveals no significant impact on fasting blood glucose (FBG), hemoglobin A1c (HbA1c), or insulin levels. Specifically, FBG showed a negligible with a mean difference of − 0.03 mg/dL (MD: − 0.03 mg/dL, 95% CI [− 2.82, 2.75], I2 = 15.98%, p = 0.98) (Fig. 6A), insulin levels exhibited a non-significant mean difference of 0.45 µIU/mL (MD: 0.45 µIU/mL, 95% CI [− 2.76, 3.66], I2 = 73.43%, p = 0.78) (Fig. 6B), and HbA1c had a non-significant mean reduction of − 0.13% (MD: − 0.13%, 95% CI [− 0.29, 0.04], I2 = 0.0%, p = 0.13) (Fig. 6C). These results suggest that chia seed supplementation does not significantly alter these glycemic and insulin parameters in overweight individuals (Fig. 6).
Effect on Inflammatory markers
The meta-analysis on chia seed supplementation in overweight subjects shows a significant reduction in C-reactive protein (CRP) levels, with a mean difference of − 1.18 mg/L (MD: − 1.18 mg/L, 95% CI [− 2.01 to − 0.36], I2 = 87.9%, p < 0.0001) (Fig. 7A), indicating a substantial effect on reducing inflammation. However, there were no significant changes in interleukin-6 (IL-6) (MD: − 0.15 pg/ml, 95% CI [− 0.88 to 0.59], I2 = 2.16%, p = 0.7) (Fig. 7B), and tumor necrosis factor-alpha (TNF-α) levels (MD: 0.03 pg/ml, 95% CI [− 0.58 to 0.65], I2 = 9.28%, p = 0.91) (Fig. 7C), suggesting that chia seed supplementation does not significantly impact these inflammatory markers.
Heterogeneity and sensitivity analysis
The heterogeneity among studies varied in outcome measures, but it was generally low to moderate (I2 < 50%). Despite excluding studies with a high risk of bias, the overall effect sizes were equivalent, and sensitivity analyses assessed the robustness of the results. The sensitivity analysis showed that excluding any individual study did not impact the effect size of chia seed supplementation in outcomes such as FBG.
Publication bias
A funnel plot was designed to assess the parameter FBG, which was evaluated in at least ten studies. Figure 8 illustrates a moderate degree of asymmetry, indicating the presence of publication bias, possibly resulting from the difficulties in disseminating negative or inconclusive findings. Based on most outcome measures, the examination of funnel plot asymmetry and Egger's regression test implementation failed to find any significant indications of publication bias in the included papers. However, there was a notable publication bias for FBG, as indicated by a t-value of 3.13 and a P-value of 0.98.
Subgroup analysis
Subgroup analyses were performed to determine the source of high heterogeneity in the meta-analysis. The results of the subgroup analysis based on the BMI are shown in Fig. 9A. Subgroup analysis indicated that there were no statistically significant differences among different study designs, such as single-blind RCTs, double-blind RCTs, and cross-over trials (MD: 0.04, 95% CI − 0.36 to 0.43, p = 0.46, I2 = 12.58%). Similarly, subgroup analysis for TG (Fig. 9B) and systolic BP (Fig. 9C) revealed no significant differences among different studies (MD: − 0.10, 95% CI − 0.35 to 0.16, p = 0.44, I2 = 2.58%), (MD: − 0.22, 95% CI − 0.44 to 0.01, p = 0.76, I2 = 0%), respectively.
Stratifying the analysis based on study designs revealed no difference in TC (Fig. 9D) among different studies (MD: − 0.02, 95% CI − 0.25 to 0.21, p = 0.61, I2 = 8.81%). Subgroup analysis showed a negligible difference in CRP (Fig. 9E) among different study designs (MD: − 0.34, 95% CI − 0.69 to 0.01, p = 0.81). Based on the study designs, the subgroup analysis showed no significant difference in FBG (Fig. 9F) among studies (MD: − 0.0, 95% CI − 0.23 to 0.23, p = 0.26).
When analyzing the lipid profile, subgroup analysis did not reveal significant differences in LDL (Fig. 9G) or HDL (Fig. 9H) levels across various trial designs. The subgroup analysis found no statistically significant difference in different study designs (mean difference: − 0.22, 95% CI − 0.44 to 0.01, p = 0.76).
Discussion
This systematic review and meta-analysis included ten RCTs with 424 participants to assess the effects of chia seed supplementation on cardiometabolic indices in overweight subjects. The meta-analysis findings indicate that chia seed supplementation does not significantly change lipid profile parameters such as TG, TC, HDL, or LDL, nor does it change glycemic markers, including FBG, HbA1c, or insulin levels. However, chia seeds are associated with a significant reduction in CRP, an inflammatory marker, and a decrease in WC and systolic BP. Despite these positive effects, no significant changes were observed in IL-6, TNF-α, BMI, or diastolic BP. The included studies exhibited low to moderate heterogeneity in outcome measures.
Our study indicates that consuming 30 g/day of chia seeds for six months may reduce weight and WC in overweight individuals. Some previous studies support our findings, while others do not [27, 28]. For example, Guevara-Cruz et al. [29] demonstrated a significant decrease in WC in individuals with metabolic syndrome who consumed a regular diet, including chia seeds, soy, nopal, and oats. Conversely, Nieman et al. [30] found that consuming 50 g/day of chia for 12 weeks did not result in weight loss among obese or overweight individuals. Toscano et al. [31] reported a notable decrease in WC among overweight and obese individuals who consumed 35 g/day of chia for twelve weeks. However, another study found no significant changes in weight, BMI, WC, or insulin resistance after a 10-day consumption of 25 g/day of chia [32]. Lovato's research revealed significant reductions in body weight, abdominal circumference, WC, and BMI after chia intervention [33]. Similarly, Ayaz et al. [34] showed significant weight, BMI, and WC decreases after a six-month diet, including 25 g/day of chia. The differing outcomes across studies could be attributed to duration, dosage, and variations in participants’ characteristics. While chia seeds are high in dietary fiber, which can promote fullness and potentially aid in weight reduction, several factors contribute to this. Factors such as participant characteristics (like baseline metabolic rate and lifestyle habits), adherence to the supplementation regimen, and variations in measurement and reporting methods can all influence the effectiveness of chia seeds in achieving weight reduction [34,35,36,37].
Previous studies have highlighted the beneficial components of chia seeds for cardiovascular health, including their high insoluble fiber content, which more effectively lowers BP than soluble fiber [29, 38,39,40,41]. Vuksan et al. [42] found that chia seed consumption reduced systolic BP in individuals with well-managed T2DM. However, other studies reported no significant changes in systolic BP after 10 or 12 weeks of chia supplementation [30, 31].
Chia seeds have several compounds that positively impact BP and cardiovascular health. They are rich in crucial fatty acids, particularly with a high omega-3 to omega-6 ratio, linked to reduced BP and a lower risk of CVD [43,44,45]. A 100-g serving of chia provides significant amounts of magnesium, potassium, and calcium, all potent vasodilators and vascular smooth muscle contraction inhibitors, effectively reducing BP [46,47,48,49]. Additionally, chia seeds contain flavonoids such as quercetin, chlorogenic acid, and caffeic acid, which have been shown to lower BP [14, 43]. Research by Toscano et al. [39] demonstrated that chia flour intake consistently reduced BP in hypertensive individuals. The dietary fiber in chia seeds forms a gel-like substance when combined with water, aiding in satiety, reducing hunger, and potentially lowering the risk of coronary heart disease. This fiber also acts as a prebiotic, beneficial for treating hypertension and obesity [14, 43].
The chia seeds help slow down starch digestion and reduce post-meal blood glucose levels [8, 50]. Chia seeds include viscous and soluble fibers that have been shown to reduce cholesterol levels by affecting the metabolism of lipids in the liver [51]. This impact is accomplished by increased excretion of bile acids, reducing the uptake of cholesterol and other lipids in circulation, and inhibiting the production of free fatty acids in the liver [43]. Furthermore, the functional proteins and bioactive peptides found in chia can block the activity of 3 hydroxy 3 methylglutaryl coenzyme reductase, which plays a crucial role in forming cholesterol. As a result, the synthesis of cholesterol is diminished [52].
Sosa-Crespo et al. [53] demonstrated that consuming chia seeds within 30–60 min lowers plasma glucose levels in healthy individuals and those with prediabetes or T2DM. Vuksan et al. [17] also found that various doses of chia (7, 15, and 24 g) reduced postprandial blood glucose levels in healthy individuals. Additionally, a previous meta-analysis reported that higher doses of chia seeds reduced postprandial glucose levels [9]. FBG reductions were observed in several subgroups, including healthy women, overweight or obese individuals, hypertensive patients, and those with metabolic syndrome [29, 38, 39]. However, in our study, chia seed supplementation showed a slight improvement in glycemic markers among overweight individuals, but this improvement was not statistically significant. Several factors may account for this discrepancy. First, the inclusion of only 10 RCTs may have been insufficient to detect significant changes in glycemic markers. Second, differences in baseline characteristics of our study population compared to those in other studies could have influenced the outcomes. Third, variations in the amount and form of chia seed consumption and unaccounted dietary and lifestyle factors may have affected the results. Finally, individual variability in response to chia seed consumption could also explain our study's lack of significant findings.
The bioactive components in chia seeds have been linked to preventive effects against obesity and plasma oxidative stress in several studies [12, 54]. Obesity is characterized by persistent, low-level inflammation in adipose tissue, accompanied by the excessive production of pro-inflammatory adipokines [55, 56]. Elevated levels of these inflammatory mediators in the bloodstream have been linked to the activation of hepatocytes, prompting them to create CRP [54]. Elevated CRP levels are associated with a higher risk of developing coronary heart disease and increased mortality rates [57].
Although the exact anti-inflammatory mechanism of chia seeds is not fully understood, the high levels of alpha-linolenic acid (ALA) in chia seeds may suggest an anti-inflammatory effect [58]. However, the primary method of enhancing antioxidant levels in plasma is still debated, as it is hypothesized that there may be a decrease in pro-oxidative substances or an increase in antioxidant substances [59]. The antioxidant capacity of chia seeds is defined by their ability to inhibit the synthesis of reactive oxygen species (ROS) or through direct scavenging action. These antioxidants can transform into active molecules, reducing cell damage and playing a role in regulating gene expression and signal transduction. Consequently, the body initiates feedback processes to enhance cellular defense and survival [60]. A study showed that chia's anti-inflammatory effect seems to be associated with the inhibition of PPARγ and NF-κB expression (61). Furthermore, research has shown that chia seeds contain many phenolic compounds and high levels of natural antioxidants, including quercetin and kaempferol, while caffeic and chlorogenic acids are in minimal quantities [62]. These antioxidative properties make chia seeds a valuable source of antioxidants [63].
This meta-analysis used data only from RCTs, which are considered the most rigorous form of clinical scientific evidence. Our investigation did not uncover any indications of publication bias that could have affected the meta-analysis's conclusions. We also tried to minimize any potential bias in the review process by conducting an in-depth assessment of existing literature and adhering to the rigorous PRISMA requirements for conducting and reporting the review. In addition, we performed subgroup analyses to examine the impacts within distinct subgroups and identify the probable variables that may contribute to the found heterogeneity. These methodological strengths enhance the reliability of our findings, providing robust evidence to support clinical decision-making.
Despite these strengths, our study has several limitations that warrant careful consideration. The overall sample size was small, potentially affecting the statistical power of our results. Additionally, significant heterogeneity was observed, likely due to variations in study populations, dosing regimens, and treatment durations, which complicates the interpretation of the findings. Most of the included studies originated from Eastern countries, raising concerns about the generalizability of our results to Western populations. Furthermore, several outcomes were assessed by only a few studies, limiting the strength of the evidence supporting those conclusions. These factors may undermine the validity of our results and should be acknowledged when interpreting our findings.
Conclusions
The findings of this meta-analysis indicate that chia seed supplementation does not lead to significant changes in most lipid profile parameters, including TG, TC, HDL, and LDL, nor in FBG, HbA1c, or insulin levels. Despite these findings, chia seeds appear to have beneficial effects on certain health markers. Specifically, chia seed supplementation was associated with a notable reduction in CRP, suggesting a potential anti-inflammatory effect. Additionally, significant reductions in waist circumference and systolic blood pressure were observed, though changes in diastolic blood pressure were not significant. No substantial impact on BMI was found. Overall, chia seeds should be considered a valuable adjunct in managing cardiometabolic health, particularly due to their anti-inflammatory properties and effects on WC and PB. However, they should not be relied upon as a sole intervention. Chia seeds should be integrated into a comprehensive health strategy that includes a balanced diet, regular physical activity, and other lifestyle modifications for optimal health outcomes.
Future studies should aim to include larger RCTs and more diverse sample sizes with standardized dosing regimens and treatment durations to enhance statistical power, reduce heterogeneity, and improve the generalizability and validity of the findings.
Availability of data and materials
The study includes the original contributions in the article/Supplementary material. For further inquiries, please contact the corresponding authors.
Abbreviations
- CVD:
-
Cardiovascular diseases
- BMI:
-
Body Mass Index
- WC:
-
Waist circumference
- mm Hg:
-
Millimeters of mercury
- BP:
-
Blood pressure
- FBG:
-
Fasting blood glucose
- Hb1AC:
-
Hemoglobin A1c
- TG:
-
Triglycerides
- TC:
-
Total cholesterol
- HDL:
-
High-density lipoprotein
- LDL:
-
Low-density lipoprotein
- CRP:
-
C-reactive protein
- IL-6:
-
Interleukin-6
References
Skinner AC, Ravanbakht SN, Skelton JA, Perrin EM, Armstrong SC. Prevalence of obesity and severe obesity in US children, 1999–2016. Pediatrics. 2018;141(3).
Simsek HU, Albayrak MGB, Kasap M, Simsek T, Akpinar G, Guler SA, et al. Elucidation of the changes occurring at the proteome level in ovaries of high-fat diet-induced obese rats. Cell Biochem Funct. 2022;40(3):278–97.
Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1923–94.
World Health Organization cardiovascular disease risk charts. revised models to estimate risk in 21 global regions. Lancet Glob Health. 2019;7(10):e1332–45.
Jenkins DJ, Kendall CW, Augustin LS, Franceschi S, Hamidi M, Marchie A, et al. Glycemic index: overview of implications in health and disease. Am J Clin Nutr. 2002;76(1):266s-s273.
Goodpaster BH, Sparks LM. Metabolic flexibility in health and disease. Cell Metab. 2017;25(5):1027–36.
Prevalence and attributable health burden of chronic respiratory diseases, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Respir Med. 2020;8(6):585–96.
Capitani MI, Spotorno V, Nolasco SM, Tomás MC. Physicochemical and functional characterization of by-products from chia (Salvia hispanica L.) seeds of Argentina. LWT Food Sci Technol. 2012;45(1):94–102.
Teoh SL, Lai NM, Vanichkulpitak P, Vuksan V, Ho H, Chaiyakunapruk N. Clinical evidence on dietary supplementation with chia seed (Salvia hispanica L.): a systematic review and meta-analysis. Nutr Rev. 2018;76(4):219–42.
Mokgalaboni K, Phoswa WN. Corchorus olitorius extract exhibit anti-hyperglycemic and anti-inflammatory properties in rodent models of obesity and diabetes mellitus. Front Nutr. 2023;10:1099880.
Lovato AdC, Corgozinho MLMV, Alves LV, Martins SR, Duarte RCF, Cardoso CN, et al. Effect of the use of chia (Salvia hispanica L.) seeds on antioxidant status and anthropometric parameters in obese, type 2 diabetics and/or hypertensive patients. Res Soc Dev. 2022;11(4):1.
Oliveira-Alves SC, Vendramini-Costa DB, BetimCazarin CB, MarósticaJúnior MR, Borges Ferreira JP, Silva AB, et al. Characterization of phenolic compounds in chia (Salvia hispanica L.) seeds, fiber flour and oil. Food Chem. 2017;232:295–305.
Marcinek K, Krejpcio Z. Chia seeds (Salvia hispanica): health promoting properties and therapeutic applications—a review. Rocz Panstw Zakl Hig. 2017;68(2):123–9.
Knez Hrnčič M, Ivanovski M, Cör D, Knez Ž. Chia seeds (Salvia hispanica L.): an overview-phytochemical profile, isolation methods, and application. Molecules. 2019;25(1).
Agarwal A, Rizwana, Tripathi AD, Kumar T, Sharma KP, Patel SKS. Nutritional and functional new perspectives and potential health benefits of quinoa and chia seeds. Antioxidants (Basel). 2023;12(7).
Vuksan V, Jenkins AL, Brissette C, Choleva L, Jovanovski E, Gibbs AL, et al. Salba-chia (Salvia hispanica L.) in the treatment of overweight and obese patients with type 2 diabetes: a double-blind randomized controlled trial. Nutr Metab Cardiovasc Dis. 2017;27(2):138–46.
Vuksan V, Jenkins AL, Dias AG, Lee AS, Jovanovski E, Rogovik AL, et al. Reduction in postprandial glucose excursion and prolongation of satiety: possible explanation of the long-term effects of whole grain Salba (Salvia hispanica L.). Eur J Clin Nutr. 2010;64(4):436–8.
Ho H, Lee AS, Jovanovski E, Jenkins AL, Desouza R, Vuksan V. Effect of whole and ground Salba seeds (Salvia hispanica L.) on postprandial glycemia in healthy volunteers: a randomized controlled, dose-response trial. Eur J Clin Nutr. 2013;67(7):786–8.
Rui Y, Lv M, Chang J, Xu J, Qin L, Wan Z. Chia seed does not improve cognitive impairment in SAMP8 mice fed with high fat diet. Nutrients. 2018;10(8).
Chicco AG, D’Alessandro ME, Hein GJ, Oliva ME, Lombardo YB. Dietary chia seed (Salvia hispanica L) rich in alpha-linolenic acid improves adiposity and normalises hypertriacylglycerolaemia and insulin resistance in dyslipaemic rats. Br J Nutr. 2009;101(1):41–50.
Kulczyński B, Kobus-Cisowska J, Taczanowski M, Kmiecik D, Gramza-Michałowska A. The chemical composition and nutritional value of chia seeds-current state of knowledge. Nutrients. 2019;11(6).
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372: n71.
Brown D. A review of the pubmed PICO tool: using evidence-based practice in health education. Health Promot Pract. 2020;21(4):496–8.
Andrade C. Mean difference, standardized mean difference (SMD), and their use in meta-analysis: as simple as it gets. J Clin Psychiatry. 2020;81(5):11349.
Akoglu H. User’s guide to correlation coefficients. Turk J Emerg Med. 2018;18(3):91–3.
Thorlund K, Imberger G, Johnston BC, Walsh M, Awad T, Thabane L, et al. Evolution of heterogeneity (I2) estimates and their 95% confidence intervals in large meta-analyses. PLoS ONE. 2012;7(7): e39471.
Waruguru P. Systematic evaluation of the impact of chia seeds on weight loss. JMHS. 2023;2(1):87–98.
Nikpayam O, Jafari A, Safaei E, Naghshi N, Najafi M, Sohrab G. Effect of chia product supplement on anthropometric measures, blood pressure, glycemic-related parameters, lipid profile and inflammatory indicators: a systematic and meta-analysis. J Funct Foods. 2023;110: 105867.
Guevara-Cruz M, Tovar AR, Aguilar-Salinas CA, Medina-Vera I, Gil-Zenteno L, Hernández-Viveros I, et al. A dietary pattern including nopal, chia seed, soy protein, and oat reduces serum triglycerides and glucose intolerance in patients with metabolic syndrome. J Nutr. 2012;142(1):64–9.
Nieman DC, Cayea EJ, Austin MD, Henson DA, McAnulty SR, Jin F. Chia seed does not promote weight loss or alter disease risk factors in overweight adults. Nutr Res. 2009;29(6):414–8.
Tavares Toscano L, Tavares Toscano L, Leite Tavares R, da Oliveira Silva CS, Silva AS. Chia induces clinically discrete weight loss and improves lipid profile only in altered previous values. Nutr Hosp. 2014;31(3):1176–82.
Vuksan V, Choleva L, Jovanovski E, Jenkins AL, Au-Yeung F, Dias AG, et al. Comparison of flax (Linum usitatissimum) and Salba-chia (Salvia hispanica L.) seeds on postprandial glycemia and satiety in healthy individuals: a randomized, controlled, crossover study. Eur J Clin Nutr. 2017;71(2):234–8.
Lovato A, Corgozinho M, Alves L, Martins S, Duarte R, Cardoso C, et al. Effect of the use of chia (Salvia Hispanica L.) seeds on antioxidant status and anthropometric parameters in obese, type 2 diabetics and/or hypertensive patients. 2023.
Ayaz A, Akyol A, Inan-Eroglu E, Kabasakal Cetin A, Samur G, Akbiyik F. Chia seed (Salvia Hispanica L.) added yogurt reduces short-term food intake and increases satiety: randomised controlled trial. Nutr Res Pract. 2017;11(5):412–8.
Poudyal H, Panchal SK, Ward LC, Brown L. Effects of ALA, EPA and DHA in high-carbohydrate, high-fat diet-induced metabolic syndrome in rats. J Nutr Biochem. 2013;24(6):1041–52.
Tucker LA, Thomas KS. Increasing total fiber intake reduces risk of weight and fat gains in women. J Nutr. 2009;139(3):576–81.
Lattimer JM, Haub MD. Effects of dietary fiber and its components on metabolic health. Nutrients. 2010;2(12):1266–89.
Nieman DC, Gillitt N, Jin F, Henson DA, Kennerly K, Shanely RA, et al. Chia seed supplementation and disease risk factors in overweight women: a metabolomics investigation. J Altern Complement Med. 2012;18(7):700–8.
Toscano LT, da Silva CS, Toscano LT, de Almeida AE, Santos Ada C, Silva AS. Chia flour supplementation reduces blood pressure in hypertensive subjects. Plant Foods Hum Nutr. 2014;69(4):392–8.
Valenzuela BR, Barrera RC, González-Astorga M, Sanhueza CJ, Valenzuela BA. Alpha linolenic acid (ALA) from Rosa canina, sacha inchi and chia oils may increase ALA accretion and its conversion into n-3 LCPUFA in diverse tissues of the rat. Food Funct. 2014;5(7):1564–72.
Aljuraiban GS, Griep LM, Chan Q, Daviglus ML, Stamler J, Van Horn L, et al. Total, insoluble and soluble dietary fibre intake in relation to blood pressure: the INTERMAP Study. Br J Nutr. 2015;114(9):1480–6.
Vuksan V, Whitham D, Sievenpiper JL, Jenkins AL, Rogovik AL, Bazinet RP, et al. Supplementation of conventional therapy with the novel grain Salba (Salvia hispanica L.) improves major and emerging cardiovascular risk factors in type 2 diabetes: results of a randomized controlled trial. Diabetes Care. 2007;30(11):2804–10.
Ullah R, Nadeem M, Khalique A, Imran M, Mehmood S, Javid A, et al. Nutritional and therapeutic perspectives of Chia (Salvia hispanica L.): a review. J Food Sci Technol. 2016;53(4):1750–8.
Khalid W, Arshad MS, Aziz A, Rahim MA, Qaisrani TB, Afzal F, et al. Chia seeds (Salvia hispanica L.): a therapeutic weapon in metabolic disorders. Food Sci Nutr. 2023;11(1):3–16.
European Food Safety A. Opinion on the safety of ‘Chia seeds (Salvia hispanica L.) and ground whole Chia seeds’ as a food ingredient. EFSA J. 2009;7(4):996.
Suter PM, Sierro C, Vetter W. Nutritional factors in the control of blood pressure and hypertension. Nutr Clin Care. 2002;5(1):9–19.
Sontia B, Touyz RM. Role of magnesium in hypertension. Arch Biochem Biophys. 2007;458(1):33–9.
Dickinson HO, Mason JM, Nicolson DJ, Campbell F, Beyer FR, Cook JV, et al. Lifestyle interventions to reduce raised blood pressure: a systematic review of randomized controlled trials. J Hypertens. 2006;24(2):215–33.
Wang L, Manson JE, Buring JE, Lee IM, Sesso HD. Dietary intake of dairy products, calcium, and vitamin D and the risk of hypertension in middle-aged and older women. Hypertension. 2008;51(4):1073–9.
Schuchardt JP, Wonik J, Bindrich U, Heinemann M, Kohrs H, Schneider I, et al. Glycemic index and microstructure analysis of a newly developed fiber enriched cookie. Food Funct. 2016;7(1):464–74.
Riccioni G, Sblendorio V, Gemello E, Di Bello B, Scotti L, Cusenza S, et al. Dietary fibers and cardiometabolic diseases. Int J Mol Sci. 2012;13(2):1524–40.
Coelho MS, Soares-Freitas RAM, Arêas JAG, Gandra EA, Salas-Mellado MLM. Peptides from chia present antibacterial activity and inhibit cholesterol synthesis. Plant Foods Hum Nutr. 2018;73(2):101–7.
Sosa-Crespo I, Espinosa-Marrón A, Chel-Guerrero L, Lavia-da-Molina H, Betancur-Ancona D. Postprandial glycaemic effect of a peptide fraction of Salvia Hispanica in Patients with Insulin Resistance. J Biol Act Prod Nat. 2021;11(4):356–62.
Marineli RDS, Lenquiste SA, Moraes ÉA, Maróstica MR Jr. Antioxidant potential of dietary chia seed and oil (Salvia hispanica L.) in diet-induced obese rats. Food Res Int. 2015;76(Pt 3):666–74.
Karastergiou K, Mohamed-Ali V. The autocrine and paracrine roles of adipokines. Mol Cell Endocrinol. 2010;318(1–2):69–78.
Ellulu MS, Khaza’ai H, Rahmat A, Patimah I, Abed Y. Obesity can predict and promote systemic inflammation in healthy adults. Int J Cardiol. 2016;215:318–24.
Kuller LH, Tracy RP, Shaten J, Meilahn EN. Relation of C-reactive protein and coronary heart disease in the MRFIT nested case-control study. Multiple Risk Factor Intervention Trial. Am J Epidemiol. 1996;144(6):537–47.
Poudyal H, Panchal SK, Waanders J, Ward L, Brown L. Lipid redistribution by α-linolenic acid-rich chia seed inhibits stearoyl-CoA desaturase-1 and induces cardiac and hepatic protection in diet-induced obese rats. J Nutr Biochem. 2012;23(2):153–62.
Chim Y, Gallegos S, Jimenez Martinez C, Dávila Ortiz G, Guerrero L. Antioxidant capacity of Mexican chia (Salvia hispanica L.) protein hydrolyzates. J Food Meas Charact. 2018;12:1–9.
Xanthis V, Fitsiou E, Voulgaridou GP, Bogadakis A, Chlichlia K, Galanis A, et al. Antioxidant and cytoprotective potential of the essential oil Pistacia lentiscus var. chia and its major components myrcene and α-pinene. Antioxidants (Basel). 2021;10(1).
Grancieri M, Martino HSD, Gonzalez de Mejia E. Protein digests and pure peptides from chia seed prevented adipogenesis and inflammation by inhibiting PPARγ and NF-κB pathways in 3T3L-1 adipocytes. Nutrients. 2021;13(1):176.
Reyes-Caudillo E, Tecante A, Valdivia-López MA. Dietary fibre content and antioxidant activity of phenolic compounds present in Mexican chia (Salvia hispanica L.) seeds. Food Chem. 2008;107(2):656–63.
Orona-Tamayo D, Valverde M, Nieto-Rendón B, Paredes-Lopez O. Inhibitory activity of chia (Salvia hispanica L.) protein fractions against angiotensin I-converting enzyme and antioxidant capacity. Lebensmittel-Wissenschaft und-Technologie. 2015;64.
Brissette, C., The effect of Salvia hispanica L. seeds on weight loss in overweight and obese individuals with type 2 diabetes mellitus. 2013, University of Toronto Toronto.
da Silva CS, et al. Assessing the metabolic impact of ground chia seed in overweight and obese prepubescent children: a double-blind randomized clinical trial results. J Med Food. 2020;23(3):224–32.
Alwosais EZM, et al. Chia seed (Salvia hispanica L.) supplementation to the diet of adults with type 2 diabetes improved systolic blood pressure: a randomized controlled trial. Nutr Health. 2021;27(2):181–9.
Quaresma LS, et al. Chia flour (Salvia hispanica L.) intake does not affect weight loss and lipid profile but improve systolic blood pressure control in obesity. J Am Nutr Assoc. 2023;42(4):403–10.
Acknowledgements
We would like to express our gratitude to all those who supported us in this study. We also appreciate the efforts of our colleagues and collaborators, whose insights and assistance were invaluable throughout the process.
Funding
The authors confirm that they received no financial support for this article's research, authorship, and publication. Our study was carried out without any external funding or financial support from organizations or individuals that could impact the outcomes or interpretation of our findings.
Author information
Authors and Affiliations
Contributions
M.K. conceived the study idea, conducted advanced research and screening, performed data analysis, supervised the study, and revised the manuscript. E.SH.R., A.A., N.SH. and S.P. screened the studies and extracted data. M.K., N.SH., S.M.H., and SH.K. wrote the primary manuscript. S.A., K.K., and M.K. reviewed and revised the manuscript. M.K. supervised the project. All authors contributed to the manuscript writing, reviewed, and approved the final version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Competing interests
We declare that no financial conflicts of interest are associated with this research. The results presented in this study are solely based on thorough scientific analysis and are free from any financial bias or external pressure.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Karimi, M., Pirzad, S., Shirsalimi, N. et al. Effects of chia seed (Salvia hispanica L.) supplementation on cardiometabolic health in overweight subjects: a systematic review and meta-analysis of RCTs. Nutr Metab (Lond) 21, 74 (2024). https://doi.org/10.1186/s12986-024-00847-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s12986-024-00847-3