Abstract
Peritoneal dialysis is an important part of end-stage kidney disease replacement therapy. However, prolonged peritoneal dialysis can result in peritoneal fibrosis and ultrafiltration failure, forcing patients to withdraw from peritoneal dialysis treatment. Therefore, there is an urgent need for some effective measures to alleviate the occurrence and progression of peritoneal fibrosis. Mesenchymal stem cells play a crucial role in immunomodulation and antifibrosis. Numerous studies have investigated the fact that mesenchymal stem cells can ameliorate peritoneal fibrosis mainly through the paracrine pathway. It has been discovered that mesenchymal stem cells participate in the improvement of peritoneal fibrosis involving the following signaling pathways: TGF-β/Smad signaling pathway, AKT/FOXO signaling pathway, Wnt/β-catenin signaling pathway, TLR/NF-κB signaling pathway. Additionally, in vitro experiments, mesenchymal stem cells have been shown to decrease mesothelial cell death and promote proliferation. In animal models, mesenchymal stem cells can enhance peritoneal function by reducing inflammation, neovascularization, and peritoneal thickness. Mesenchymal stem cell therapy has been demonstrated in clinical trials to improve peritoneal function and reduce peritoneal fibrosis, thus improving the life quality of peritoneal dialysis patients.
Similar content being viewed by others
Introduction
Peritoneal dialysis (PD) is an essential renal replacement treatment for patients with end-stage renal disease (ESRD). Around the world, more than 196,000 individuals with ESRD are presently undergoing PD treatment [1]. In contrast to conventional hemodialysis, PD provides several benefits, including low medical resources and cost savings due to its straightforward operation, effective removal of mesomolecular molecules, minor loss of remaining renal functions, and steady hemodynamics [2]. However, PD patients are persistently exposed to biologically incompatible dialysis solutions with high concentrations of glucose, which may contribute to losing normal morphology and function in peritoneal mesothelial cells, resulting in ultrafiltration failure (UFF) and peritoneal fibrosis (PF) [3]. PF is a severe consequence of the PD procedure. It is characterized by the loss of peritoneal mesothelial cells, the aberrant proliferation of α-SMA-positive myofibroblasts, a noticeable buildup of collagen, and a gradual increase in the thickness of the submesothelial compact zone [4, 5]. Peritoneal function can be improved by controlling the occurrence of PF [6,7,8]. Epithelial-mesenchymal transition (EMT) was an initiating and reversible step of PF [9]. EMT is a cellular transdifferentiation process that involves the transformation of an epithelial phenotype into a mesenchymal phenotype. During this process, the cells lose their polarity, adherence, and tight junctions, resulting in a fibroblast-like appearance and mesenchymal cell phenotype, leading to PF and UF [2, 10]. Therefore, exploring the mechanism of EMT is crucial for alleviating PF in PD patients (Fig. 1).
Epithelial-mesenchymal transition (EMT) of peritoneal mesothelial cells. Biocompatible factors in dialysate may contribute to mesothelial cell injury, leading to EMT. EMT is an important phase in the development of peritoneal fibrosis, involving the transformation of an epithelial phenotype into a mesenchymal phenotype. During this process, cells lose their polarity, adherence, and tight junctions, resulting in a fibroblast-like appearance and mesenchymal cell phenotypic expression (including upregulation of α-SMA, vimentin, N-cadherin, fibronectin, and downregulation of E-cadherin). Mesenchymal stem cells (MSCs) could secrete HGF (hepatocyte growth factor) to promote the proliferation and restoration of mesothelial cells, which could express a large number of membrane complement regulators (Crry, CD55), further decreasing the complement activation product C3b, C5b-9 to inhibit inflammation. Created by BioRender.com
Mesenchymal stem cells (MSCs) are multipotent adult stem cells that can be isolated from multiple kinds of tissues, including bone marrow, adipose tissue, umbilical cord blood, and the placenta [11,12,13]. MSCs have regenerative, immunomodulatory, and antifibrotic capabilities [14]. As for antifibrotic ability, various studies have investigated the fact that MSCs can ameliorate fibrosis in the liver, heart, lung, and kidney among different animal models [15,16,17,18]. Additionally, MSCs can repair tissue damage, but their mechanism is controversial. Initially, it was thought that MSCs repair tissues by engrafting and differentiating to replace injured cells [19,20,21,22,23]. Nowadays, many studies have proved that MSCs exert their beneficial effects by secreting cytokines and growth factors through the paracrine pathway or cell-to-cell contacts. The cytokines, such as TNF-α-stimulated gene 6 protein (TSG-6) [6, 24], VEGF_ENREF_38 [25], and PDGF [26], can alter the tissue microenvironment to repair tissue injury. It is worth noting that the cytokines released by MSCs have the potential to be amplified through crosstalk with injured cells, resulting in the expression of even more therapeutic factors [23, 27]. Exosomes are tiny vesicles secreted by MSCs via the paracrine pathway that contain a variety of biomolecules, such as proteins, non-coding RNA, DNA, and mRNA, and act as important carriers in cell communication [28]. Recent research has shown that exosomes derived from hUMSCs (human umbilical cord-derived mesenchymal stem cells) can reduce the fibrosis of organs, such as the liver, lung, and kidney [29,30,31].
This review summarises the latest studies on MSCs in the in three parts: (1) the mechanism of MSCs in treating PF, (2) in vitro experiments, (3) animal experiments, and (4) clinical trials. In addition, we discuss the different sources of MSC, which may lead to different results. Different injection methods may have different effects on peritoneal adhesion, possible problems in applying results of animal experiments to clinical studies, and the current research progress in combining biotechniques with stem cell therapy. This review will help advance the development of regenerative medicine and provide a new direction for cell therapy for human disease. The basic characteristics of the included literature are presented in Tables 1, 2 and 3.
Mechanism
Promote macrophage polarisation
Macrophage polarization is the activation of macrophages in response to stimulation by pathogenic microbes, inflammatory reactions, cytokines, or certain physicochemical conditions, resulting in differentiation into various phenotypes [32]. Macrophage polarization is involved in the disease progression process, and when specific signals activate macrophages, they differentiate into distinct phenotypes that exert regulatory effects through a multitude of signaling pathways [33]. There are two main types of macrophage polarization: classically activated M1 and alternatively activated M2 [34]. M1 macrophages are known for their pro-inflammatory properties and secrete cytokines like IL-1β, iNOS, and TNF-α. On the other hand, M2 macrophages have anti-inflammatory properties and produce cytokines like IL-10, TGF-β, and Arg-1 [35]. Both M1 and M2 macrophages are critical in the pathogenesis of fibrosis. Therefore, macrophage polarization plays an important part in the progression of fibrotic diseases [36,37,38,39,40]. The researchers carried out cellular and animal experiments to test the hypothesis that promoting macrophage polarisation could alleviate PF. According to the study from Nagasaki et al. [41], SF-MSCs significantly reduced inflammation by stimulating the transition of proinflammatory M1 macrophages into immunosuppressive M2 macrophages. Yang et al. [7] constructed MGO-induced PF rat and cultured cells in vitro and found that TGF-β1 can stimulate ADSCs to secrete more IL-6 and increase the expression of the macrophage gene Arg-1, thereby promoting the polarization of M2 macrophages to decrease PF in rats. Research has indicated that inhibiting inflammation by inducing macrophage polarisation may help to alleviate PF, providing a new direction for the treatment of PF, but more supportive evidence is needed in this area, which requires further exploration of the involved mechanisms (Fig. 2).
The possible mechanism of MSCs (Mesenchymal stem cells) to ameliorate peritoneal fibrosis. MSCs could reduce peritoneal fibrosis by two main mechanisms, including inhibiting EMT and inflammation. a: pMSCs could improve viability and reduce apoptosis in mesothelial cells, which can be more resistant to the toxicity of uremic toxin in rat models. b: hUMSCs could secrete exosomes through the paracrine pathway, and the gene lnc-CDHR and lnc-GAS5 in the exosomes can competitively bind to mi3149 to regulate PTEN through the AKT/FOXO signaling pathway and Wnt/β-catenin signaling pathway to inhibit EMT, thus reducing peritoneal fibrosis. c: BM-MSCs could secrete HGF through the TGF-β/Smad pathway to inhibit EMT and reduce peritoneal fibrosis. Simultaneously, BM-MSCs have the potential to release TSG-6, which interacts with local macrophage CD44 receptors to suppress the TLR2/NF-kB signaling pathway. This would decrease the release of pro-inflammatory cytokines and improve peritoneal fibrosis. d: ADSCs could secrete HGF to promote the proliferation and restoration of mesothelial cells, which could express abundant membrane complement regulators (Crry, CD55), further decreasing the complement activation products C3b, and C5b-9 to inhibit inflammation. ADSCs can also secrete IL-6, promoting the polarisation of M1 macrophages to M2 macrophages to inhibit inflammation and reduce peritoneal fibrosis. e: MSCs cultured in the serum-free medium can promote M2 macrophage polarisation to suppress inflammation. Created by BioRender.com
Secrete cytokines and growth factors
Secrete TSG-6, inhibit NF-κB signaling pathway
TSG-6 is a 30 kD glycoprotein with anti-inflammatory effects in various animal models [42, 43]. Studies have shown that the absence of the gene in transgenic mice increases inflammatory reactions, while overexpression reduces inflammation reactions [44, 45]. The protein’s anti-inflammatory activities are attributed to several effects, including its ability to bind to pro-inflammatory hyaluronan fragments, inhibiting the inflammatory cascade of proteases, and preventing the neutrophil inflow into inflammatory areas [46]. Nagasaki et al. [41] found that the STK2 serum-free medium enhanced the proliferation of MSCs and the secretion of TSG-6 in MSCs to suppress infiltration of inflammatory cells to suppress PF.
NF-κB is a key transcriptional regulator of the inflammatory response [47,48,49]. NF-κB can be activated by various stimuli that regulate inflammatory and immune responses and cell survival. The most powerful activators of NF-κB are inflammatory cytokines like tumor necrosis factor (TNF) or interleukin-1 (IL-1), as well as pathogen-derived molecules that trigger Toll-like receptors (TLRs) such as lipopolysaccharide (LPS), viral, and bacterial DNA and RNA [47, 50]. NF-κB is crucial for developing liver fibrosis [51,52,53]. Liao et al. [54] developed inflammatory and fibrotic models and discovered that isoliquiritigenin protects the kidney by inhibiting the Mincle/Syk/NF-κB signaling pathway. Choi et al. [6] constructed zymosan-induced mouse peritonitis to demonstrate that MSCs activated by inflammatory signals secrete the anti-inflammatory protein TSG-6 through TLR2/NF-κB signaling to attenuate zymosan-induced mouse peritonitis in resident macrophages. It has been discovered that TSG-6 interacts with macrophages through the CD 44 receptor to inhibit zymosan/TLR2-mediated nuclear translocation of the NF-κB, lowering pro-inflammatory factor secretion by macrophages and mitigating the inflammatory cascade that is released by resident macrophages and heightened by mesothelial cells or other potential peritoneal cells. In conclusion, secretion of TSG-6 by MSCs may play an important role in alleviating PF, and TSG-6 may act by inhibiting the infiltration of inflammatory cells through the NF-κB signaling pathway. And the NF-κB signaling pathway may provide a new direction for investigating the mechanism of PF (Fig. 2).
Secrete hepatocyte growth factor (HGF), inhibit TGF-β/Smad signaling pathway
HGF is a potent antifibrotic cytokine that blocks tubular epithelial-to-mesenchymal transition (EMT) induced by TGF-β [55]. HGF can also prevent bleomycin-induced pulmonary fibrosis in mice and inhibit the TGF-β/smad signaling pathway in lung cells [56]. TGF-β1 induces EMT in peritoneal mesothelial cells and contributes to the advancement of several kinds of fibrosis, including liver, lung, heart, and kidney [2, 57,58,59]. Ueno et al. [60]. have found through in vitro and animal experiments that MSCs can inhibit TGF-β1 production by secreting HGF through the paracrine pathway and improve TGF-β1-stimulated EMT in HPMCs through the TGF-β1/smad2 signaling pathway, thereby alleviating PF and reducing peritoneal functional impairment. Therefore, HGF may play an important role in alleviating PF, and the TGF-β signaling pathway may provide a new direction for MSC treatment of PF (Fig. 2).
Secrete HGF, inhibit complement deposition
Disorders of the complement activation system may be a contributing factor to peritoneal injury [61,62,63]. Kim et al. [64] constructed the Zy/scraping peritonitis model to investigate the relationship between complement activation and initial inflammation. They found that complement activation enhanced peritoneal inflammation in this peritoneal injury model. Rat ASCs were injected into the Zy/scraping peritonitis model, and it was found that HGF secreted by rat ASCs through the paracrine pathway may contribute to the repair of peritoneal mesothelial cell injury as well as peritoneal mesothelial cell proliferation and that the peritoneal mesothelial cells expressed abundant CReg (Crry, CD55, CD59), which could potentially prevent complement activation and the deposition of complement activation products like C3b and C5b-9 (Fig. 1).
Secrete exosomes
Exosomes are tiny vesicles secreted by MSCs via the paracrine pathway that contain a variety of biomolecules, such as proteins, non-coding RNA, DNA, and mRNA, and act as important carriers in cell communication [28]. According to recent research, exosomes can alleviate fibrosis in the liver, lung, and kidney [29,30,31]. lnc RNA is widely found in fibrotic tissues such as the heart, liver, kidney, and lungs and plays an important role in the fibrotic process [65]. lnc RNA is present in the exosomes of MSCs [66]. Jiao et al. [67] discovered that hUMSCs’ exosomal lnc-CDHR binds competitively to miR-3149, regulating the target PTEN genes via AKT/FOXO signaling pathway to reduce EMT in HMrSV5. Huang et al. [68] suggested that exosomal lncRNA GAS5 competitively binds to miR21 and regulates PTEN via the Wnt/β-catenin pathway to inhibit EMT. Thus, Exosomes can alleviate PF, which might be attributed to the function of lncRNA. As an important carrier, lncRNA can play a vital role in alleviating PF. Due to the limited study, it is necessary for us to further explore the function of lncRNA in PF (Fig. 2).
In vitro experiment
MSCs can reduce peritoneal mesothelial cell death, increase their activity and migration capacity, and promote their proliferation and injury repair, thereby inhibiting peritoneal mesothelial cell EMT and alleviating peritoneal injury (Table 1).
MSCs can reduce mesothelial cell death and increase cell activity in vitro
MSCs can reduce the death of peritoneal mesothelial cells. Fan et al. [69] cocultured hUMSCs with PD-induced cell death in HPMCs and found that the hUMSCs could prevent morphological disturbances and apoptosis-like cell debris in HPMCs, improve cell viability in HPMCs and reduce the percentage of HPMCs death, which indicates a protective role of hUMSCs in peritoneal dialysis solution-induced HPMCs death. Du et al. [70] found that the secretome from pMSCs can significantly reduce peritoneal mesothelial cell death when exposed to PDS. Zhou et al. [71] incubated pMSCs-CM with H2O2-induced apoptosis and PDS-induced cell apoptosis in HPMCs and found that pMSC-CM could prevent cell death of cultured HPMCs (Table 1).
MSCs can improve the migration capacity of mesothelial cells and promote injury repair in vitro
MSCs can enhance mesothelial cell migration. Wang et al. [72] constructed a mechanical injury model in vitro and discovered that MSCs could increase the migratory capacity and the proliferation of RPMCs in the early phase of injury. By co-culturing rat ASCs supernatant and peritoneal mesothelial cells, Kim et al. [64] found that the supernatant of rat ASCs could promote the proliferation of rat peritoneal mesothelial cells as well as the repair of injuries (Table 1).
MSCs and their secretions inhibit EMT in mesothelial cells in vitro
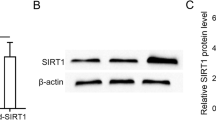
MSCs inhibit EMT in peritoneal mesothelial cells. Guo et al. [73] co-cultured hUMSCs and SIRT1-modified hUMSCs with TGF-β1-stimulated Met-5 A cells respectively, and found that SIRT1-hUMSCs markedly inhibit EMT of Met-5 A cells compared to the hUMSCs. Specifically speaking, SIRT1-hUMSCs can significantly decrease the expression of mesenchymal and fibrotic markers such as Fibronectin, α-SMA, and Snail. Meanwhile, SIRT1-modified hUMSCs can restore the downregulate of expression of E-cadherin during EMT. Huang et al. [68] found that the conditional medium of hUMSCs could inhibit the EMT of HPMCs. Jiao et al. [67] discovered that hUMSCs exosomal lnc-CDHR binds to miR-3149 competitively and regulates the target PTEN genes’ repression to lessen EMT in HMrSV5 (Table 1).
In Vivo experiment (animal experiment)
Studies have shown that long-term exposure of the peritoneal membrane to standard PD fluid with high glucose concentration results in morphological changes such as increased numbers of inflammatory cells, neovascularization, and submesothelial thickening, leading to UFF. MSCs, as a potential treatment, can reverse this change and protect peritoneal function [74] (Table 2).
MSCs can inhibit EMT in animal models
MSCs significantly suppressed the accumulation of myofibroblasts and macrophages, as well as the expression of mesenchymal markers (α-SMA and FSP-1) and extracellular matrix (ECM) proteins (collagens I and III), leading to amelioration of PF [60]. Guo et al. [73] and Li et al. [75] constructed an MGO-PD-induced rat model and investigated that hUMSCs alleviate EMT in the peritoneal injury rat model. Wakabayashi et al. [76] developed a CG-induced PF model and discovered that ASCs could inhibit the EMT to reduce experimental PF (Table 2).
MSCs have anti-fibrotic and anti-inflammatory properties in animal models
Several studies have demonstrated that MSCs have antifibrotic and anti-inflammatory effects. Yang et al. [7] and Nagasaki et al. [41] induced a PF model and found that ADSCs and BM-MSCs both can suppress PF. Yu et al. [77] constructed peritoneal injury mouse models induced by 2.5%PDF + LPS and found that BM-MSC-Exos alleviates PF related to PD and relieves peritoneal inflammation and angiogenesis in mice. Costalonga et al. [78] constructed PF models developed in uremic rats and found that ASCs have antifibrotic and anti-inflammatory effects. Specifically speaking, ASC therapy dramatically decreased macrophage and T-cell infiltration and improved the development of PF in the PF rat model. ASC infusion prevented PF by lowering the number of peritoneal myofibroblasts and changing the expression of genes involved in ECM synthesis (Table 2).
MSCs can ameliorate peritoneal function in animal models
Several studies have demonstrated that MSCs can improve peritoneal function in animal models by inhibiting peritoneal thickness, improving ultrafiltration [75, 79,80,81], and the absorption rate of glucose from the dialysate (D/D0 of glucose) [41, 77, 80]. Ueno et al. [60] discovered that MSCs can improve the D/D0 of glucose and the transport rate of blood urea nitrogen from the plasma (D/P of BUN) to inhibit functional impairment. Guo et al. [73] discovered that SIRT1-hUMSCs markedly improved ultrafiltration volume, D/P of Cr, and D/D0 of glucose in PD-treated rats.
Clinical application of MSCs
Due to the scarcity of clinical trials on the use of stem cells to treat PF, the following are three studies in which stem cell therapy was utilized to improve peritoneal function in PD patients. hUMSCs have the advantages of strong proliferation and differentiation ability, easy access to materials, and low immunogenicity, and have better application prospects than other stem cells [82]. Jiang et al. [83] found that hUMSC treatment partially improved clinical indicators of continuous ambulatory peritoneal dialysis (CAPD) patients. Within three months after hUMSCs transplantation, experimental results showed a significant increase in hemoglobin, erythropoietin, and albumin levels, decreased C-reactive protein levels, and marked improvement in cystatin C and urine volume (Table 3).
Adipose tissue is a prospective source for autologous cell-based treatment since it is more accessible than bone marrow [84]. A study conducted by Ahmadi et al. [8] suggests that injecting Autologous ADSCs may lead to a slight improvement in UF capacity and mild enhancement in peritoneal membrane function. The study found a slight increase in both systemic and peritoneal levels of CA125 and a minor decrease in gene expression levels of TGF-β, α-SMA, and FSP-1. Alatab et al. [85] conducted a study on PD patients suffering from UFF who were on CAPD for at least two years and discovered that systemic delivery of ADSCs to PD patients was feasible and well tolerated, with no severe adverse events or catheter-related problems noted. Therefore, from the above results, it is clear that applying MSCs in clinical trials can improve peritoneal function indices to a certain extent and is safe and reliable. Due to insufficient clinical trials, the exploration of MSCs is inadequate. More clinical trials are needed in the future, including the following aspects: identifying the most suitable types of MSCs, exploring how to maximize the effect of MSCs, and improving the convenience and efficiency of MSC preparation technology (Table 3).
Discussion
Prolonged PD can lead to the development of PF, which can force patients to withdraw from PD [3]. It is crucial to slow down the process of PF. MSCs have been studied in clinical trials and basic experiments due to their ability to self-regenerate, modulate the immune system, and repair tissue damage [14]. MSCs can repair tissue damage, but their mechanism is controversial. Initially, it was thought that MSCs repair tissues by engrafting and differentiating to replace injured cells [19,20,21,22,23]. Nowadays, MSCs have been proven to exert their beneficial effects by secreting cytokines and growth factors through the paracrine pathway or cell-to-cell contacts. Exosomes, which are important secretions produced by MSCs, have been found to significantly reduce fibrosis in different tissues such as the liver, lung, kidney, and peritoneum [29,30,31, 67, 68]. However, a recent study found that exosomes derived from PD effluents could result in peritoneal damage by transmitting molecules such as proteins [86]. Therefore, it is important to identify the specific components of exosomes that are beneficial or harmful and to understand their role in regulating peritoneal function.
Different sources of MSCs may have varying effects on improving peritoneal function. Yang et al. [7] argued that ADSC has a more significant antifibrotic effect than BM-MSC in reducing peritoneal membrane thickness because the researchers found that ADSC can release more IL-6 than BM-MSC and IL-6 as an important component during the process of M2 macrophage polarization, which can significantly reduce the PF. Du et al. [70] found that pMSCs were more effective than hUMSCs in protecting the peritoneal membrane and remnant kidneys in 5/6Nx rats, which was mainly explained by the fact that pMSCs exhibited greater resistance to the toxicity of uremic toxins present in uremic rats and were more protective of peritoneal mesothelial cells from death. The effect of different injection modalities of MSCs on relieving peritoneal adhesions may be different. Wang et al. [87] investigated that MSCs given intraperitoneally did not reduce peritoneal adhesion, while those injected intravenously significantly improved adhesion. The reasons may be that intravenously injected MSCs, which accumulated mainly in the lung, can survive for 7 days, are rarely phagocytosed by macrophages, and secrete TSG-6 within 12 h. In contrast, intraperitoneally injected MSCs, accumulating mainly in the spleen and can survive for only 4 h, are subsequently phagocytosed by macrophages and do not secrete TSG-6, thus failing to exert a therapeutic effect.
The therapeutic effects of MSCs in rat models and clinical trials cannot be fully equated. This may be related to the immune compatibility between the donor and recipient, MSC dosing, and the fitness of culture-adapted MSCs [88]. In animal models, MSCs are usually administered intravenously at 50 million MSC/kg/dose, but in most clinical trials, MSCs are usually administered intravenously at 1–2 million cells/kg/dose and no more than 12 million cells/kg. Therefore, there is a disproportionate relationship between dose and body weight. Thus, the disproportionate relationship between dose and body weight suggests a possible reason for the discrepancy between the efficacy of animal studies and clinical trials [89]. In addition, there may be some challenges in using MSCs in clinical trials: (1) different sources of MSCs have different extraction methods, and there are no standardized guidelines on this; (2) there are some technical difficulties in culturing and expanding MSCs; (3) different injection modality and different dosage may cause different effects, so exploring the most appropriate injection modality and dosage is important for the future; (4) The transportation and storage conditions of MSCs can impact the therapeutic effectiveness of MSCs in clinical trials, and it is necessary to conduct further study in the future. Although there are many unresolved problems in using MSCs, current clinical studies have shown that MSCs are safe and feasible, with no serious adverse effects and no catheter-related complications reported [85, 90, 91].
Since the effects of MSCs in animal experiments are not the same as in actual clinical trials, it is necessary to use technical skills to improve the therapeutic effects of MSCs. Combining stem cell therapy and biotechnology is one of the potential fields for tissue damage and repair. Huang et al. [92] found that SIRT1 significantly alleviates renal fibrosis in rat models of chronic kidney disease and murine mesangial cells. SIRT1 knockdown increases renal fibrosis and destroys renal function, whereas SIRT1 overexpression decreases TGF-β-induced extracellular matrix production and expression [92]. Guo et al. [73] constructed MGO-PD-induced rat models and found that SIRT1-modified hUMSCs can markedly reduce the PF in rat models. In addition, Huerta et al. [93] found that E-selectin gene-modified MSCs could accelerate wound healing compared to MSC and phosphate-buffered saline treatment group. Studies have indicated that a hypoxic environment is beneficial to stem cell survival rate [94]. Trisnadi et al. [95] found that hypoxic conditions-induced MSCs might significantly reduce the TGF-β level in peritoneal adhesion rat models compared to the normal MSCs. MSCs cultured in serum medium may contribute to inflection and increase the risk of transmitting viral disease [96, 97]. So, the serum-free medium is important to clinical application [98, 99]. Nagasaki et al. [41] used the serum-free conditional medium to culture MSCs, and the results indicate that SF-MSC was more effective in inhibiting PF than using 10% MSC (10% serum conditional medium). Furthermore, serum-free MSC culture has many benefits, including shortened culture cycles, reduced risk of infection from serum components, no longer having to check for variations in serum batches, and enhanced cell proliferation stability and efficiency. This provides valuable insight for the in vitro expansion of MSCs in clinical settings [96, 97].
This review provides an overview of all studies in vitro and in vivo on MSCs in treating dialysis-induced PF. Due to the lack of in-depth research in this field, there is a paucity of research study on the mechanism. Therefore, a large number of studies in vitro and in vivo are needed to demonstrate the efficacy of MSCs. Furthermore, Tables 1, 2 and 3 summarises the different sources of MSCs, different treatments of MSCs, and different animal models of fibrosis induction, MSC doses, and different indicators of peritoneal function, as well as the signaling pathways involved in MSCs. These can provide data support for subsequent researchers to get a quick overview of the current state of research, save time in collecting literature, and conduct further advance study.
Conclusion
This review summarises the latest research progress on the alleviation of PF by MSCs, including the mechanism of MSCs in alleviating PF, cellular experiments, animal experiments, and the clinical application of MSCs. Numerous studies have demonstrated that MSCs alleviate PF mainly through the paracrine pathway. The following signaling pathways were found to be involved in the alleviation of PF by MSCs: TGF-β/smad signaling pathway, AKT/FOXO signaling pathway, Wnt/β-catenin signaling pathway, TLR/NF-κB signaling pathway. MSCs can alleviate PF by secreting exosomes, which contain genes that regulate miRNA action targeting PTEN to inhibit EMT and alleviate PF. MSCs can also alleviate PF by inhibiting the infiltration of inflammatory cells, inducing macrophage polarisation, stimulating TSG-6 secretion by MSCs, and secreting HGF to inhibit complement deposition. In addition, serum-free cultured MSCs may help to suppress inflammation. The effects produced by different sources of MSCs are different, and the effects of different injection modalities in relieving peritoneal adhesions may also vary. Therefore, the field of MSCs to alleviate PF is currently understudied, and more mechanisms still need to be explored. MSCs provide a new direction for progress in the treatment of PF in dialysis patients.
Data availability
Data sharing is not applicable to this article, as no datasets were generated or analyzed during the current study.
Abbreviations
- ADSCs:
-
Adipose-derived mesenchymal stem cells
- Alb:
-
Albumin
- Arg-1:
-
Arginine-1
- BM-MSCs:
-
Bone marrow mesenchymal stem cells
- BUN:
-
Blood urea nitrogen
- BMP-7:
-
Bone morphogenic protein 7
- BMI:
-
Body mass index
- CA125:
-
Cancer antigen 125
- Ccr:
-
Creatinine clearance rate
- CM:
-
Conditional medium
- CG:
-
Chlorhexidine gluconate
- CBUN :
-
Clearance of blood urea nitrogen
- D/P of BUN:
-
Dialysate-to-plasma ratio of blood urea nitrogen
- D/Pcr:
-
The ratio of dialysate and plasma creatinine concentration
- EMT:
-
Epithelial-to-mesenchymal transition
- ECM:
-
Extracellular matrix
- EPO:
-
Erythropoietin
- Exo:
-
Exosome
- FSP-1:
-
Fibroblast-specific protein-1
- FWT:
-
Free water transport
- GLU D/P:
-
The glucose dialysate-to-plasma ratio
- Hb:
-
Hemoglobin
- HG:
-
High concentration of glucose
- HIF-1α:
-
Hypoxia-inducible factor-1α
- HPMCs:
-
Human peritoneal mesothelial cells
- Hs-CRP:
-
High-sensitivity C-reactive protein
- HGF:
-
Hepatocyte growth factor
- HMrSV5:
-
Human peritoneal mesothelial cell line
- hUMSCs:
-
Human umbilical cord-derived mesenchymal stem cells
- iNOS:
-
Inducible nitric oxide synthase
- IP.inject:
-
Intraperitoneal injection
- IV.inject:
-
Intravenous injection
- IL-1β:
-
Interleukin-1β
- IL-6:
-
Interleukin 6
- LPS:
-
Lipopolysaccharides
- MSCs:
-
Mesenchymal stem cells
- 10%MSCs:
-
MSCs cultured in a medium containing 10% fetal bovine serum
- MPO:
-
Myeloperoxidase
- MCP-1:
-
Monocyte chemoattractant protein-1
- MGO:
-
Methylglyoxal
- Met-5A:
-
An immortalized human pleural mesothelial line
- NC:
-
Negative control
- OCG:
-
Osmotic conductance to glucose
- PMN:
-
Polymorphonuclear cells
- PD:
-
Peritoneal dialysis
- PMNs:
-
PolyMorphonuclear neutrophil
- PF:
-
Peritoneal fibrosis
- PDGF:
-
Platelet-derived growth factor
- PM:
-
Peritoneal membrane
- pMSCs:
-
PD effluent-derived mesenchymal stromal cells
- PDS:
-
PD solution
- RPMC:
-
Rat peritoneal mesothelial cells
- Rat-MC-CM:
-
Rat mesothelial-CM
- SD-Rat:
-
Sprague-Dawley rat
- α-SMA:
-
α-Smooth muscle actin
- SMC:
-
Submesothelial compact
- Scr:
-
Serum creatinine
- SF:
-
Serum-free medium
- SF-MSCs:
-
MSCs in serum-free medium
- TSG-6:
-
TNF-α–stimulated gene 6 protein
- TGF-β1:
-
Transforming growth factor-β1
- THP-1 cells:
-
A human monocytic cell line
- UFF:
-
Ultrafiltration failure
- UFSP:
-
Ultrafiltration- small pore
- UF:
-
Ultrafiltration
- UFT:
-
Ultrafiltration total
- VEGF:
-
Vascular endothelial-derived growth factor
- ZO-1:
-
Zonula occludens-1
References
Jain AK, Blake P, Cordy P, Garg AX. Global trends in rates of peritoneal dialysis. J Am Soc Nephrol. 2012;23(3):533–44.
Yáñez-Mó M, Lara-Pezzi E, Selgas R, Ramírez-Huesca M, Domínguez-Jiménez C, Jiménez-Heffernan JA, et al. Peritoneal dialysis and epithelial-to-mesenchymal transition of mesothelial cells. N Engl J Med. 2003;348(5):403–13.
Zhang K, Zhang H, Zhou X, Tang WB, Xiao L, Liu YH, et al. miRNA589 regulates epithelial-mesenchymal transition in human peritoneal mesothelial cells. J Biomed Biotechnol. 2012;2012:673096.
Williams JD, Craig KJ, Topley N, Von Ruhland C, Fallon M, Newman GR, et al. Morphologic changes in the peritoneal membrane of patients with renal disease. J Am Soc Nephrol. 2002;13(2):470–9.
Mateijsen MA, van der Wal AC, Hendriks PM, Zweers MM, Mulder J, Struijk DG, et al. Vascular and interstitial changes in the peritoneum of CAPD patients with peritoneal sclerosis. Perit Dial Int. 1999;19(6):517–25.
Choi H, Lee RH, Bazhanov N, Oh JY, Prockop DJ. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-κB signaling in resident macrophages. Blood. 2011;118(2):330–8.
Yang CY, Chang PY, Chen JY, Wu BS, Yang AH, Lee OK. Adipose-derived mesenchymal stem cells attenuate dialysis-induced peritoneal fibrosis by modulating macrophage polarization via interleukin-6. Stem Cell Res Ther. 2021;12(1):193.
Ahmadi A, Moghadasali R, Najafi I, Shekarchian S, Alatab S. Potential of autologous adipose-derived mesenchymal stem cells in peritoneal fibrosis: a pilot study. Arch Iran Med. 2023;26(2):100–9.
de Graaff M, Zegwaard AH, Zweers MM, Vlijm A, de Waart DR, Vandemaele F, et al. The effects of a dialysis solution with a combination of glycerol/amino acids/dextrose on the peritoneal membrane in chronic renal failure. Perit Dial Int. 2010;30(2):192–200.
Acloque H, Adams MS, Fishwick K, Bronner-Fraser M, Nieto MA. Epithelial-mesenchymal transitions: the importance of changing cell state in development and disease. J Clin Invest. 2009;119(6):1438–49.
Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;276(5309):71–4.
Lee RH, Kim B, Choi I, Kim H, Choi HS, Suh K, et al. Characterization and expression analysis of mesenchymal stem cells from human bone marrow and adipose tissue. Cell Physiol Biochem. 2004;14(4–6):311–24.
Wang HS, Hung SC, Peng ST, Huang CC, Wei HM, Guo YJ, et al. Mesenchymal stem cells in the Wharton’s jelly of the human umbilical cord. Stem Cells. 2004;22(7):1330–7.
Parekkadan B, Milwid JM. Mesenchymal stem cells as therapeutics. Annu Rev Biomed Eng. 2010;12:87–117.
Nagaya N, Kangawa K, Itoh T, Iwase T, Murakami S, Miyahara Y, et al. Transplantation of mesenchymal stem cells improves cardiac function in a rat model of dilated cardiomyopathy. Circulation. 2005;112(8):1128–35.
Ortiz LA, Gambelli F, McBride C, Gaupp D, Baddoo M, Kaminski N, et al. Mesenchymal stem cell engraftment in lung is enhanced in response to bleomycin exposure and ameliorates its fibrotic effects. Proc Natl Acad Sci U S A. 2003;100(14):8407–11.
Oyagi S, Hirose M, Kojima M, Okuyama M, Kawase M, Nakamura T, et al. Therapeutic effect of transplanting HGF-treated bone marrow mesenchymal cells into CCl4-injured rats. J Hepatol. 2006;44(4):742–8.
Ninichuk V, Gross O, Segerer S, Hoffmann R, Radomska E, Buchstaller A, et al. Multipotent mesenchymal stem cells reduce interstitial fibrosis but do not delay progression of chronic kidney disease in collagen4A3-deficient mice. Kidney Int. 2006;70(1):121–9.
Prockop DJ. Repair of tissues by adult stem/progenitor cells (MSCs): controversies, myths, and changing paradigms. Mol Ther. 2009;17(6):939–46.
Nauta AJ, Fibbe WE. Immunomodulatory properties of mesenchymal stromal cells. Blood. 2007;110(10):3499–506.
Uccelli A, Moretta L, Pistoia V. Mesenchymal stem cells in health and disease. Nat Rev Immunol. 2008;8(9):726–36.
Caplan AI. Why are MSCs therapeutic? New data: new insight. J Pathol. 2009;217(2):318–24.
Ohtaki H, Ylostalo JH, Foraker JE, Robinson AP, Reger RL, Shioda S, et al. Stem/progenitor cells from bone marrow decrease neuronal death in global ischemia by modulation of inflammatory/immune responses. Proc Natl Acad Sci U S A. 2008;105(38):14638–43.
Lee RH, Pulin AA, Seo MJ, Kota DJ, Ylostalo J, Larson BL, et al. Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell. 2009;5(1):54–63.
Meirelles Lda S, Fontes AM, Covas DT, Caplan AI. Mechanisms involved in the therapeutic properties of mesenchymal stem cells. Cytokine Growth Factor Rev. 2009;20(5–6):419–27.
Gnecchi M, Zhang Z, Ni A, Dzau VJ. Paracrine mechanisms in adult stem cell signaling and therapy. Circ Res. 2008;103(11):1204–19.
Gunn WG, Conley A, Deininger L, Olson SD, Prockop DJ, Gregory CA. A crosstalk between myeloma cells and marrow stromal cells stimulates production of DKK1 and interleukin-6: a potential role in the development of lytic bone disease and tumor progression in multiple myeloma. Stem Cells. 2006;24(4):986–91.
Tavasolian F, Hosseini AZ, Soudi S, Naderi M. miRNA-146a improves Immunomodulatory effects of MSC-derived exosomes in Rheumatoid Arthritis. Curr Gene Ther. 2020;20(4):297–312.
Li T, Yan Y, Wang B, Qian H, Zhang X, Shen L, et al. Exosomes derived from human umbilical cord mesenchymal stem cells alleviate liver fibrosis. Stem Cells Dev. 2013;22(6):845–54.
Xu C, Zhao J, Li Q, Hou L, Wang Y, Li S, et al. Exosomes derived from three-dimensional cultured human umbilical cord mesenchymal stem cells ameliorate pulmonary fibrosis in a mouse silicosis model. Stem Cell Res Ther. 2020;11(1):503.
Ji C, Zhang J, Zhu Y, Shi H, Yin S, Sun F, et al. Exosomes derived from hucMSC attenuate renal fibrosis through CK1δ/β-TRCP-mediated YAP degradation. Cell Death Dis. 2020;11(5):327.
Atri C, Guerfali FZ, Laouini D. Role of human macrophage polarization in inflammation during infectious diseases. Int J Mol Sci. 2018;19(6).
Sica A, Erreni M, Allavena P, Porta C. Macrophage polarization in pathology. Cell Mol Life Sci. 2015;72(21):4111–26.
Yunna C, Mengru H, Lei W, Weidong C. Macrophage M1/M2 polarization. Eur J Pharmacol. 2020;877:173090.
Shapouri-Moghaddam A, Mohammadian S, Vazini H, Taghadosi M, Esmaeili SA, Mardani F, et al. Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol. 2018;233(9):6425–40.
Murray LA, Rosada R, Moreira AP, Joshi A, Kramer MS, Hesson DP, et al. Serum amyloid P therapeutically attenuates murine bleomycin-induced pulmonary fibrosis via its effects on macrophages. PLoS ONE. 2010;5(3):e9683.
Chu D, Du M, Hu X, Wu Q, Shen J. Paeoniflorin attenuates schistosomiasis japonica-associated liver fibrosis through inhibiting alternative activation of macrophages. Parasitology. 2011;138(10):1259–71.
Murphy BS, Bush HM, Sundareshan V, Davis C, Hagadone J, Cory TJ, et al. Characterization of macrophage activation states in patients with cystic fibrosis. J Cyst Fibros. 2010;9(5):314–22.
Duffield JS. Macrophages and immunologic inflammation of the kidney. Semin Nephrol. 2010;30(3):234–54.
Wynn TA, Barron L. Macrophages: master regulators of inflammation and fibrosis. Semin Liver Dis. 2010;30(3):245–57.
Nagasaki K, Nakashima A, Tamura R, Ishiuchi N, Honda K, Ueno T, et al. Mesenchymal stem cells cultured in serum-free medium ameliorate experimental peritoneal fibrosis. Stem Cell Res Ther. 2021;12(1):203.
Milner CM, Higman VA, Day AJ. TSG-6: a pluripotent inflammatory mediator? Biochem Soc Trans. 2006;34(Pt 3):446–50.
Wisniewski HG, Vilcek J. Cytokine-induced gene expression at the crossroads of innate immunity, inflammation and fertility: TSG-6 and PTX3/TSG-14. Cytokine Growth Factor Rev. 2004;15(2–3):129–46.
Szántó S, Bárdos T, Gál I, Glant TT, Mikecz K. Enhanced neutrophil extravasation and rapid progression of proteoglycan-induced arthritis in TSG-6-knockout mice. Arthritis Rheum. 2004;50(9):3012–22.
Mindrescu C, Dias AA, Olszewski RJ, Klein MJ, Reis LF, Wisniewski HG. Reduced susceptibility to collagen-induced arthritis in DBA/1J mice expressing the TSG-6 transgene. Arthritis Rheum. 2002;46(9):2453–64.
Getting SJ, Mahoney DJ, Cao T, Rugg MS, Fries E, Milner CM, et al. The link module from human TSG-6 inhibits neutrophil migration in a hyaluronan- and inter-alpha -inhibitor-independent manner. J Biol Chem. 2002;277(52):51068–76.
Karin M, Ben-Neriah Y. Phosphorylation meets ubiquitination: the control of NF-[kappa]B activity. Annu Rev Immunol. 2000;18:621–63.
Ghosh S, Karin M. Missing pieces in the NF-kappaB puzzle. Cell. 2002;109 Suppl:S81-96.
Xiao C, Ghosh S. NF-kappaB, an evolutionarily conserved mediator of immune and inflammatory responses. Adv Exp Med Biol. 2005;560:41–5.
West AP, Koblansky AA, Ghosh S. Recognition and signaling by toll-like receptors. Annu Rev Cell Dev Biol. 2006;22:409–37.
Luedde T, Beraza N, Kotsikoris V, van Loo G, Nenci A, De Vos R, et al. Deletion of NEMO/IKKgamma in liver parenchymal cells causes steatohepatitis and hepatocellular carcinoma. Cancer Cell. 2007;11(2):119–32.
Bettermann K, Vucur M, Haybaeck J, Koppe C, Janssen J, Heymann F, et al. TAK1 suppresses a NEMO-dependent but NF-kappaB-independent pathway to liver cancer. Cancer Cell. 2010;17(5):481–96.
Inokuchi S, Aoyama T, Miura K, Osterreicher CH, Kodama Y, Miyai K, et al. Disruption of TAK1 in hepatocytes causes hepatic injury, inflammation, fibrosis, and carcinogenesis. Proc Natl Acad Sci U S A. 2010;107(2):844–9.
Liao Y, Tan RZ, Li JC, Liu TT, Zhong X, Yan Y, et al. Isoliquiritigenin attenuates UUO-Induced renal inflammation and fibrosis by inhibiting Mincle/Syk/NF-Kappa B Signaling Pathway. Drug Des Devel Ther. 2020;14:1455–68.
Yang J, Dai C, Liu Y. A novel mechanism by which hepatocyte growth factor blocks tubular epithelial to mesenchymal transition. J Am Soc Nephrol. 2005;16(1):68–78.
Shukla MN, Rose JL, Ray R, Lathrop KL, Ray A, Ray P. Hepatocyte growth factor inhibits epithelial to myofibroblast transition in lung cells via Smad7. Am J Respir Cell Mol Biol. 2009;40(6):643–53.
Yang AH, Chen JY, Lin JK. Myofibroblastic conversion of mesothelial cells. Kidney Int. 2003;63(4):1530–9.
Margetts PJ, Bonniaud P, Liu L, Hoff CM, Holmes CJ, West-Mays JA, et al. Transient overexpression of TGF-{beta}1 induces epithelial mesenchymal transition in the rodent peritoneum. J Am Soc Nephrol. 2005;16(2):425–36.
Loureiro J, Aguilera A, Selgas R, Sandoval P, Albar-Vizcaíno P, Pérez-Lozano ML, et al. Blocking TGF-β1 protects the peritoneal membrane from dialysate-induced damage. J Am Soc Nephrol. 2011;22(9):1682–95.
Ueno T, Nakashima A, Doi S, Kawamoto T, Honda K, Yokoyama Y, et al. Mesenchymal stem cells ameliorate experimental peritoneal fibrosis by suppressing inflammation and inhibiting TGF-β1 signaling. Kidney Int. 2013;84(2):297–307.
Mizuno M, Ito Y, Hepburn N, Mizuno T, Noda Y, Yuzawa Y, et al. Zymosan, but not lipopolysaccharide, triggers severe and progressive peritoneal injury accompanied by complement activation in a rat peritonitis model. J Immunol. 2009;183(2):1403–12.
Mizuno T, Mizuno M, Morgan BP, Noda Y, Yamada K, Okada N, et al. Specific collaboration between rat membrane complement regulators Crry and CD59 protects peritoneum from damage by autologous complement activation. Nephrol Dial Transpl. 2011;26(6):1821–30.
Mizuno M, Ito Y, Mizuno T, Harris CL, Suzuki Y, Okada N, et al. Membrane complement regulators protect against fibrin exudation increases in a severe peritoneal inflammation model in rats. Am J Physiol Ren Physiol. 2012;302(10):F1245–51.
Kim H, Mizuno M, Furuhashi K, Katsuno T, Ozaki T, Yasuda K, et al. Rat adipose tissue-derived stem cells attenuate peritoneal injuries in rat zymosan-induced peritonitis accompanied by complement activation. Cytotherapy. 2014;16(3):357–68.
Cao G, Zhang J, Wang M, Song X, Liu W, Mao C, et al. Differential expression of long non-coding RNAs in bleomycin-induced lung fibrosis. Int J Mol Med. 2013;32(2):355–64.
Huang X, Yuan T, Tschannen M, Sun Z, Jacob H, Du M, et al. Characterization of human plasma-derived exosomal RNAs by deep sequencing. BMC Genomics. 2013;14:319.
Jiao T, Huang Y, Sun H, Yang L. Exosomal lnc-CDHR derived from human umbilical cord mesenchymal stem cells attenuates peritoneal epithelial-mesenchymal transition through AKT/FOXO pathway. Aging. 2023;15(14):6921–32.
Huang Y, Ma J, Fan Y, Yang L. Mechanisms of human umbilical cord mesenchymal stem cells-derived exosomal lncRNA GAS5 in alleviating EMT of HPMCs via Wnt/β-catenin signaling pathway. Aging. 2023;15(10):4144–58.
Fan YP, Hsia CC, Tseng KW, Liao CK, Fu TW, Ko TL, et al. The therapeutic potential of human umbilical mesenchymal stem cells from Wharton’s Jelly in the treatment of Rat Peritoneal Dialysis-Induced Fibrosis. Stem Cells Transl Med. 2016;5(2):235–47.
Du Y, Zong M, Guan Q, Huang Z, Zhou L, Cai J, et al. Comparison of mesenchymal stromal cells from peritoneal dialysis effluent with those from umbilical cords: characteristics and therapeutic effects on chronic peritoneal dialysis in uremic rats. Stem Cell Res Ther. 2021;12(1):398.
Zhou L, Zong M, Guan Q, da Roza G, Wang H, Qi H, et al. Protection of the peritoneal membrane by peritoneal Dialysis effluent-derived mesenchymal stromal cells in a rat model of chronic peritoneal Dialysis. Stem Cells Int. 2019;2019:8793640.
Wang N, Li Q, Zhang L, Lin H, Hu J, Li D, et al. Mesenchymal stem cells attenuate peritoneal injury through secretion of TSG-6. PLoS ONE. 2012;7(8):e43768.
Guo Y, Wang L, Gou R, Wang Y, Shi X, Pang X, et al. SIRT1-modified human umbilical cord mesenchymal stem cells ameliorate experimental peritoneal fibrosis by inhibiting the TGF-β/Smad3 pathway. Stem Cell Res Ther. 2020;11(1):362.
Tülpar S, Poyrazoğlu MH, Özbilge H, Baştuğ F, Gündüz Z, Torun YA, et al. Modulation of inflammation by mesenchymal stem cell transplantation in peritoneal dialysis in rats. Ren Fail. 2012;34(10):1317–23.
Li D, Lu Z, Li X, Xu Z, Jiang J, Zheng Z, et al. Human umbilical cord mesenchymal stem cells facilitate the up-regulation of miR-153-3p, whereby attenuating MGO-induced peritoneal fibrosis in rats. J Cell Mol Med. 2018;22(7):3452–63.
Wakabayashi K, Hamada C, Kanda R, Nakano T, Io H, Horikoshi S, et al. Adipose-derived mesenchymal stem cells transplantation facilitate experimental peritoneal fibrosis repair by suppressing epithelial-mesenchymal transition. J Nephrol. 2014;27(5):507–14.
Yu F, Yang J, Chen J, Wang X, Cai Q, He Y, et al. Bone marrow mesenchymal stem cell-derived Exosomes Alleviate Peritoneal Dialysis-Associated Peritoneal Injury. Stem Cells Dev. 2023;32(7–8):197–211.
Costalonga EC, Fanelli C, Garnica MR, Noronha IL. Adipose-derived mesenchymal stem cells modulate fibrosis and inflammation in the Peritoneal Fibrosis Model developed in uremic rats. Stem Cells Int. 2020;2020:3768718.
Baştuğ F, Gündüz Z, Tülpar S, Torun YA, Akgün H, Dörterler E, et al. Compare the effects of intravenous and intraperitoneal mesenchymal stem cell transplantation on ultrafiltration failure in a rat model of chronic peritoneal dialysis. Ren Fail. 2014;36(9):1428–35.
Zhao JL, Zhao L, Zhan QN, Liu M, Zhang T, Chu WW. BMSC-derived exosomes ameliorate peritoneal Dialysis-associated peritoneal fibrosis via the Mir-27a-3p/TP53 pathway. Curr Med Sci. 2024;44(2):333–45.
Bastug F, Gündüz Z, Tülpar S, Torun YA, Akgün H, Dörterler E, et al. Mesenchymal stem cell transplantation may provide a new therapy for ultrafiltration failure in chronic peritoneal dialysis. Nephrol Dialysis Transplantation. 2013;28(10):2493–501.
Wang Y, Ying Y, Cui X. Effects on Proliferation and differentiation of human umbilical cord-derived mesenchymal stem cells Engineered to Express neurotrophic factors. Stem Cells Int. 2016;2016:1801340.
Jiang HY, Wang JP, Bai YH, Yang M, Zeng Y, Liao YJ, et al. Clinical observation of umbilical cord mesenchymal stem cell transplantation for treating patients receiving peritoneal dialysis. Minerva Urol Nefrol. 2018;70(1):95–101.
Dykstra JA, Facile T, Patrick RJ, Francis KR, Milanovich S, Weimer JM, et al. Concise Review: Fat and Furious: harnessing the full potential of adipose-derived stromal vascular fraction. Stem Cells Transl Med. 2017;6(4):1096–108.
Alatab S, Shekarchian S, Najafi I, Moghadasali R, Ahmadbeigi N, Pourmand MR, et al. Systemic infusion of autologous adipose tissue-derived mesenchymal stem cells in peritoneal Dialysis patients: feasibility and safety. Cell J. 2019;20(4):483–95.
Corciulo S, Nicoletti MC, Mastrofrancesco L, Milano S, Mastrodonato M, Carmosino M et al. AQP1-Containing exosomes in Peritoneal Dialysis Effluent as Biomarker of Dialysis Efficiency. Cells. 2019;8(4).
Wang N, Shao Y, Mei Y, Zhang L, Li Q, Li D, et al. Novel mechanism for mesenchymal stem cells in attenuating peritoneal adhesion: accumulating in the lung and secreting tumor necrosis factor α-stimulating gene-6. Stem Cell Res Ther. 2012;3(6):51.
Galipeau J, Sensébé L. Mesenchymal stromal cells: Clinical challenges and Therapeutic opportunities. Cell Stem Cell. 2018;22(6):824–33.
Shanks N, Greek R, Greek J. Are animal models predictive for humans? Philos Ethics Humanit Med. 2009;4:2.
Lalu MM, McIntyre L, Pugliese C, Fergusson D, Winston BW, Marshall JC, et al. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic review and meta-analysis of clinical trials. PLoS ONE. 2012;7(10):e47559.
Thewes B, McCaffery K, Davis E, Garvey G. Insufficient evidence on health literacy amongst indigenous people with cancer: a systematic literature review. Health Promot Int. 2018;33(2):195–218.
Huang XZ, Wen D, Zhang M, Xie Q, Ma L, Guan Y, et al. Sirt1 activation ameliorates renal fibrosis by inhibiting the TGF-β/Smad3 pathway. J Cell Biochem. 2014;115(5):996–1005.
Huerta CT, Ortiz YY, Li Y, Ribieras AJ, Voza F, Le N, et al. Novel gene-modified mesenchymal stem cell therapy reverses impaired Wound Healing in ischemic limbs. Ann Surg. 2023;278(3):383–95.
Hawkins KE, Sharp TV, McKay TR. The role of hypoxia in stem cell potency and differentiation. Regen Med. 2013;8(6):771–82.
Trisnadi S, Muhar AM, Putra A, Kustiyah AR. Hypoxia-preconditioned mesenchymal stem cells attenuate peritoneal adhesion through TGF-β inhibition. Universa Med. 2020;39(2):97–104.
Pérez-Ilzarbe M, Díez-Campelo M, Aranda P, Tabera S, Lopez T, del Cañizo C, et al. Comparison of ex vivo expansion culture conditions of mesenchymal stem cells for human cell therapy. Transfusion. 2009;49(9):1901–10.
Nimura A, Muneta T, Koga H, Mochizuki T, Suzuki K, Makino H, et al. Increased proliferation of human synovial mesenchymal stem cells with autologous human serum: comparisons with bone marrow mesenchymal stem cells and with fetal bovine serum. Arthritis Rheum. 2008;58(2):501–10.
Chase LG, Yang S, Zachar V, Yang Z, Lakshmipathy U, Bradford J, et al. Development and characterization of a clinically compliant xeno-free culture medium in good manufacturing practice for human multipotent mesenchymal stem cells. Stem Cells Transl Med. 2012;1(10):750–8.
Gottipamula S, Ashwin KM, Muttigi MS, Kannan S, Kolkundkar U, Seetharam RN. Isolation, expansion and characterization of bone marrow-derived mesenchymal stromal cells in serum-free conditions. Cell Tissue Res. 2014;356(1):123–35.
Acknowledgements
Figures are created with BioRender.com.
Funding
This study was supported by Provincial science and technology innovation strategy special project funding program (Shanfuke [2021]88-28: 210714086900312).
Author information
Authors and Affiliations
Contributions
The authors confirm contribution to the paper as follows: review conception and design: Tianbiao Zhou; data collection, and interpretation of results, draft manuscript preparation: Lingqian Zheng. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zheng, L., Chen, W., Yao, K. et al. Clinical and preclinical studies of mesenchymal stem cells to alleviate peritoneal fibrosis. Stem Cell Res Ther 15, 237 (2024). https://doi.org/10.1186/s13287-024-03849-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13287-024-03849-3