Abstract
Giardiasis, which is caused by Giardia duodenalis, has clinical symptoms such as steatorrhea and can be very dangerous in children. In addition, some documents reported that this parasite is present inside the tissue of patients with cancer. In this study, we analyzed the gene expression profiles of some main genes important to apoptosis and anti-apoptosis in humans.
Expression profile arrays of Genomic Spatial Event (GSE) 113666, GSE113667, and GSE113679 obtained from Gene Expression Omnibus were used for meta-analysis using R commands. Cytoscape and STRING databases used the protein–protein Interaction network. Then, the Kyoto Encyclopedia of Genes and Genomes and Gene Ontology analysis was performed. Similar genes in Homo sapiens were identified using Basic Local Alignment Search Tool analysis. The validation was performed on eight people using real-time Polymerase chain reaction. In addition to the candidate genes, the gene expression of some other genes, including Serine/Threonine Kinase 1 (AKT1), Cyclin Dependent Kinase Inhibitor 2A (CDKN2A), Kirsten Rat Sarcoma (KRAS), and Phosphatidylinositol-4,5-Bisphosphate 3-Kinase Catalytic Subunit Alpha (PIK3CA) were also examined. Analysis of the expression of serum amyloid A1 (SAA1), Regenerating Islet-Derived 3 Gamma (REG3G), and REG3A genes did not show any difference between the two groups of healthy and diseased people. Examining the mean expression of the four genes AKT1, CDKN2A, KRAS, and PIK3CA showed that three genes of AKT1, CDKN2A, and KRAS had increased expression in people with a history of giardiasis compared to healthy people. We showed that the gene expression pattern differs in apoptosis and anti-apoptosis signaling in people with a history of giardiasis. Giardia duodenalis seems to induce post-non-infectious symptoms with stimulation of human gene expression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Giardiasis is one of the most common parasitic infectious diseases of the digestive system worldwide caused by Giardia duodenalis (Vivancos et al. 2018). G. duodenalis is a flagellate unicellular eukaryotic parasite (Barwick et al. 2000) transmitted through contaminated water and food, even though direct transmission has also been reported (Adam 2021). Two forms of G. duodenalis are present in the life cycle, including trophozoite and cyst (Bernander et al. 2001). The prevalence of giardiasis in developed countries is 2–7%, and in developing countries, it is 20–30% (Dixon 2011). From 2000 to 2020, giardiasis was reported in around 5.2% of fields (Teimouri et al. 2021). While asymptomatic giardiasis is common, symptomatic infections occur more often in young children than in adults (Allain and Buret 2020), including diarrhea, steatorrhea, malabsorption, anorexia, nausea, weight loss, vomiting, severe abdominal pain, and flatulence (Mørch and Hanevik 2020). Despite significant measures done toward understanding the pathogenesis of giardiasis in the past decades, the pathophysiology of this disease is still under investigation. Epithelial dysfunction during acute and chronic infections has also been reported (Allain & Buret 2020; Teimouri et al. 2021), but the pathophysiology of gastrointestinal manifestations associated with asymptomatic and symptomatic infections remains unclear. G. duodenalis is a non-invasive protozoan that infects the small intestine and colonizes the epithelial surface and lumen (Certad et al. 2017). However, there are some documents regarding the presence of G. duodenalis trophozoites inside the tissues of the pancreas (Furukawa et al. 2011), stomach (Carroccio et al. 1997), caecum (Carter et al. 2007) or distal small intestine (Halliez and Buret 2013). Correspondingly, some scholars have reported this parasite inside the tissue of cancer patients, such as Terra et al. (Terra et al. 2020) that isolated G. duodenalis from the anal mass of a 57-year-old woman with neoplasia; Garg (Garg et al. 2018) who reported the isolation of G. duodenalis from the lymph node of a 64-year-old man with cholangiocarcinoma; and Mitchell et al. (Mitchell et al. 2011) isolating the parasite from pancreatic cancer mass. Giardiasis has been recognized as a risk for the development of post-infectious functional gastrointestinal disorders (Dormond et al. 2016; Hanevik et al. 2009; Litleskare et al. 2018; Nakao et al. 2017).
To better understand the complex system of molecular processes and identify pathways and mechanisms involved in cell response to diseases, it is indispensable to use statistical and computational approaches. High-throughput technologies such as microarray, which are being used for gene expression analyses, have made it feasible to study a large number of genes simultaneously in different conditions. This study’s microarray and sample annotation data have been deposited in the NCBI Gene Expression Omnibus (GEO) repository under accession number SuperSeries.
In the present study, we analyzed the expression of some candidate genes in apoptosis signaling in persons with a history of giardiasis compared to healthy people with SYBR Green Real-Time PCR using delta Ct to perform relative quantification of gene expression. First, the Gene Expression Omnibus (GEO) datasets were analyzed to select the candidate genes. This dataset in NCBI has comparable samples processed using the same Platform and includes the experimental variables. Then, more analysis was performed to find the upregulated genes after being infected by G. duodenalis. For an accurate evaluation, a meta-analysis was carried out. Meta-analysis is a standard statistical procedure for combining datasets from multiple studies to systematically assess previously published data to derive more comprehensive conclusions about that research field. This technique provides a broad perspective on specific biological questions and more reliable results than individual studies.
Then, the important genes related to the liver that had overexpression after giardiasis infection were selected to evaluate in an experimental study with two groups of healthy people to compare the gene expression of selected genes from datasets in individuals with no history of giardiasis and digestive disorders with the ones in people with the history of giardiasis in the past with no specific symptoms of giardiasis.
Materials and methods
Microarray databases
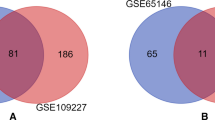
We obtained microarray data from the Gene Expression Omnibus (GEO) database (http://www.ncbi.nlm.nih.gov/geo). Since there were no datasets, including the gene expression profile of the liver from Homo sapiens infected with G. duodenalis, we used the gene expression profile in the liver from Mus musculus after the Giardia infection. We received three datasets with accession numbers GSE113679, GSE113666, and GSE113667 regarding Mus musculus, which had 32 samples in total, involving 16 samples as control and 16 samples as treatment; the GSE113697 series on the GPL24228 platform, the GSE113666 series on the GPL10333 platform, and the GSE113667 series on the GPL19795 platform.
Meta-analysis of the gene expression in datasets
We checked the quality control of each data set using boxplot diagrams and density histograms. We also removed the noises in the meta-analysis of microarray data to use datasets with different platforms by effect Batch method and by ComBat and SVA packages in R software (version 4.2.1). The comparison of genes was carried out with other expression genes (DEGs) using the limma package in R software. The DEGs were selected with commonly used equal change ∣log2 (FC)∣ > 1 threshold and p-value < 0.05.
Functional analysis
We performed GO, KEGG pathway, and REACTOME pathway using the Database for Annotation, Visualization, and Integrated Discovery (DAVID; https://david.ncifcrf.gov/) https://david.ncifcrf.gov/for DEGs. The significance criterion was set for screening enriched GO terms, KEGG, and REACTOME pathways at p < 0.05.
PPI network construction and hub gene selection
We predicted the PPI network of target genes using an online STRING database (http://string-db.org) and subsequently visualized it in Cytoscape (version 3.9.0). Then, we used the cytoHubba plug-in in Cytoscape and identified ten hub genes.
The three genes with significant expression, including Saa3, Reg3g, and Reg3b, were detected in Mus musculus. Subsequently, due to our goal to assess the most important hub genes in Homo sapiens, we detected the Homo sapiens genes, which were equivalent to Saa3, Reg3g, and Reg3b in Mus musculus. The mentioned genes in humans were SAA1, REG3G, and REG3A. Additionally, the expression of the above genes was analyzed using SYBR Green real-time PCR.
AKT1, KRAS, PIK3CA, and CDKN2A expression profile
According to microarray studies, our samples did not express three genes, SAA1, REG3A, and REG3G. Consequently, we added and assessed AKT1, KRAS, PIK3CA, and CDKN2A genes implicated in cancer development pathways by referring to various articles. The AKT gene inhibits cell apoptosis and stimulates cell proliferation following activation of protein kinase B, serine/threonine kinase (Abeyrathna and Su 2015). The PIK3CA gene encodes the catalytic subunit of phosphatidylinositol 3-kinase (PI3K) and is one of the most common genes in tumor malignancies (Cai et al. 2020). KRAS gene, a member of the RAS superfamily, encodes a small GTPase that regulates diverse cellular processes, including cell proliferation, differentiation, survival, and migration (Downward 2003). CDKN2A is an important tumor suppressor gene (Foulkes et al. 1997).
Primer design
Finally, we designed specific primer pairs for key genes using Primer3 Software (Table 1).
Patients
In this study, two groups with a history of giardiasis and one without parasite exposure were examined (Table 2). Individuals with a history of giardiasis were coded as A22, A4, and A8. Only those patients with the code of A2 had digestive symptoms. The healthy people with no history of giardiasis were coded as A1, A3, A5, A6, and A7. None of them had digestive symptoms, but sample A7 has rheumatism.
Sampling
In this study, blood sampling was done from eight people, including five healthy ones and three with giardiasis who had a history of giardiasis for three months to 15 years before selection. Blood collection was done and immediately used for RNA extraction.
RNA extraction and cDNA synthesis
The total RNA was extracted from each blood sample using a total RNA extraction kit (Parstous, Mashhad, Iran) according to the manufacturer’s instructions. We used DNase I (CinnaGen, Tehran, Iran) to treat the extracted RNA to avoid genomic contamination. The RNA quantity was done using Nanodrop (Thermo Fisher Scientific, USA). The complementary DNA (cDNA) was synthesized from total RNA (1 μg) using the RevertAid First-Strand cDNA Synthesis Kit (Thermo Fisher Scientific, USA) based on the manufacturer’s instructions.
Gene expression
We assessed the gene expression of the genes (Table 3) using SYBR Green Real-Time PCR. The housekeeping gene of GAPDH was considered an internal or endogenous control for normalization purposes. Amplification was done in a total volume of 10 μl containing 1 μl of cDNA, 5 μl of SYBR Green Real-Time Master Mix (2x), and 900 nM of each primer using a StepOne thermocycler (ABI, USA). The thermal condition of each reaction was 95 °C for 10 min to the first denaturation, followed by 40 cycles of 95 °C for 15 s and 60 °C for 1 min. Then, the melt curve analysis was set up for each run. All tests were done in duplicate. We determined the relative amount of amplification by each primer pair based on the threshold cycle value (Ct) of the gene of interest, normalized to the reference gene GAPDH. The gene expression analysis was performed using delta delta Ct.
Statistical analysis
The collected data were computerized with SPSS Software version 18, and the results were presented as tables, graphs, and statistical indices. The normality of the data was quantified by the Kolmogorov–Smirnov statistic. T-test was used for comparison of the gene epression. The p-value less than 0.05 was considered significant.
Results
Meta-analysis of microarray data
Following the data quality control assessment, the data with a low-quality sample were excluded from further examination. The results of data analysis showed that DEGs contained 34,847 common genes, including 27 genes with increased expression or up (FDR ≤ 0.05, Log FC ≥ 0.7) and 18 genes with decreased expression or down (FDR ≤ 0.05, Log FC ≤ -0.7).
PPI network construction and hub gene selection
The Cytoscape database and STRING analysis used to construct a Protein–Protein Interaction network showed that the PPI network consisted of 38 nodes and 10 edges. The PPI network revealed that Reg3g and Reg3b genes had the most interactions. Then, we identified the top 10 hub genes in Cytoscape using cytoHubba.
Functional analysis
The results related to the functional role of up and down genes (DEGs) displayed that these genes were significantly enriched in the extracellular space and extracellular region (Fig. 1). Also, for the Molecular Functions category, the genes were enriched in oligosaccharide binding (Fig. 2). In the category of Biological Processes, the genes were enriched in the acute-phase response (Fig. 3). According to KEGG pathway enrichment analysis, the DEGs were significantly enriched in Phagosome pathway, and in REACTOME pathway analysis, the DEGs were significantly enriched in Innate Immune System pathway (Fig. 4).
Genes enriched in the range of cellular compartments. Count: indicates the number of genes in each path, the larger the circle, the more genes. p. adjusted: the same p. Value that includes FDRs smaller than 0.05. GeneRatio: It is the ratio of the number of genes of each GO to the total number of DEGs
Consequently, among the genes with increased expression, three genes of Saa3, Reg3b, and Reg3g had the highest expression compared to others, which we chose as key genes. Since our samples were human, we examined three genes, SAA1, REG3G, and REG3A, which correspond to human genes as they do in mice.
qPCR analysis
The relative expression level of the AKT1 gene in samples with a history of giardiasis disease A4 and A8 increased by 59.63 and 13.57 times, respectively, compared to the average expression of the AKT1 gene in healthy subjects. No expression was observed in sample A2 (Fig. 5; p > 0.05). The relative expression of the CDKN2A gene in samples with a history of giardiasis A2 and A4 increased by 4.56 and 2.99 times, respectively, compared to the average expression of the CDKN2A gene in healthy individuals, and the expression of the gene in sample A8 was almost equal to the standard (p > 0.05). The relative expression of the KRAS gene in samples with a history of giardiasis A2 and A8 decreased by 0.75 and 0.65 times, respectively, compared to the average expression of the said gene in healthy individuals. Gene expression in sample A4 was 4.86 compared to the average expression of the said gene in healthy people, showing an increase (p > 0.05). The relative expression level of the PIK3CA gene in samples with a history of giardiasis disease A4 and A8 decreased by 0.25 and 0.21 times, respectively, compared to the average expression of the said gene in healthy individuals, and no expression was observed in sample A2 (p > 0.05). The fold changes of the AKT1, KRAS, CDKN2A, and PIK3CA genes in people with giardiasis are shown in Fig. 5.
Relative gene expression (RQ) of a KRAS, b PIK3CA, c AKT1, d CDKN2A in people with giardiasis by Real-time PCR method. Standard: Non-infected group. A2, A4, A8: Sample number of people with giardiasis. a The expression of gene KRAS was observed to decrease in samples A2 and A8 and increase in sample A4. b The expression of gene PIK3CA was decreased in samples A4 and A8, and there was no expression in sample A2. c The expression of gene AKT1 was increased in samples A4 and A8, and it was absent in sample A2. d The expression of gene CDKN2A in samples A2 and A4 had increased expression, and in sample A8, gene expression was almost equal to the standard sample
Discussion
Based on knowledge, a Giardia infection may result in cancers of the colorectal area and liver (Mahdavi et al. 2022). Also, there are some case reports regarding associations of Giardia with pancreatic cancer (Hurník et al. 2019) and gall bladder cancer (Nagasaki et al. 2011). Therefore, it is necessary to investigate the possible long-term effects of this parasite on other areas of the digestive tract, especially in the liver, pancreas, gall bladder, etc. In this study, we evaluated the persons with a history of giardiasis. These people, like non-infected people, were both females and males, and the age range of both groups was almost equal, from 21 to 33. The non-infected group, unlike the infected group, has no symptoms. In the infected group, only sample A2 showed digestive symptoms, including diarrhea and neurological symptoms such as excessive stress.
In the first step, the expression pattern of liver genes of mice infected with giardiasis was compared to healthy mice using bioinformatics methods to select the candidate genes. Then, to verify the gene expression pattern in people infected with parasites, it was compared with healthy people using laboratory methods. This study’s bioinformatics section found that three genes, Saa3, Reg3b, and Reg3g, had the highest expression in mice, similar to SAA1, REG3A, and REG3G genes in humans. We showed that none of these genes were expressed in the studied samples. It may be due to some different pathways in mice and humans. Mice strains are often used to model human disease states, therapeutic principles, and drug efficacy testing. However, a direct translation of mouse experimental data to human pathological events often fails due to substantial differences in the immune systems of both species (Zschaler et al. 2014). Since it is quite obvious that humans and mice differ in size, behavior, lifespan, living conditions, ecological situation, and other characteristics (Russell 1985), modern genetic approaches allow detailed analysis of genomes in humans and mice. Due to the common progenitor of both species, significant amounts of orthologous genes are found along with a sufficient number of individual genes associated with the species (Waterston et al. 2002). There are 15.187 genes from both species that are functionally associated, which means 75% of mouse genes and 80% of human genes are orthologous. These one-to-one orthologs have nucleotide and amino acid identities of 85.3% and 88.2%, respectively (Church et al. 2009). In defense against invading microorganisms, resistance mechanisms dominate humans, while tolerance mechanisms dominate mice (Schneider & Ayres 2008). Species differences exist in the activation and function of effector molecules released by immune cells to control pathogens (Warren et al. 2010). In addition, several pathogens found predominantly in humans but rarely or not at all in mice include Mycobacterium tuberculosis, Mycobacterium leprae, Shigella flexneri, Plasmodium falciparum, and viruses such as measles and dengue virus (Murray and Wynn 2011). Since the main immune mechanisms in mice are entirely different from those in humans, and the results of the present study also confirmed this, it seems that generalizing the results of experiments in mice to humans is not a suitable method.
The present study found that the mean relative expression level of the AKT gene in people with a history of giardiasis is increased compared to healthy people. However, among this study’s samples, one sample had lower AKT gene expression related to a woman. AKT signaling pathway is involved in inhibiting cell apoptosis and stimulating cell proliferation following activation of protein kinase B and serine/threonine kinase (Abeyrathna and Su 2015). Based on these studies, it has been determined that Giardia plays a role in triggering the apoptotic pathway in humans. Liu et al. (2020) showed that G. duodenalis induces Caco-2 cell apoptosis through a caspase-dependent pathway mediated by ROS and mitochondria (Liu et al. 2020). Therefore, it may be a natural reaction of the immune cell against the apoptosis effect of Giardia at the site of the disease; however, it is suggested that the AKT expression be investigated in patients with acute symptoms. Yang et al. (2022) also observed that AKT overexpression in p38/ERK/AKT/NF-kB signaling could inhibit anti-intestinal epithelial cell (IEC) apoptosis during giardiasis mediated by COX-2-mediated and ROS/NO production (Yang et al. 2022). The high AKT gene expression is a way to control the destruction caused by this parasite. Our results indicated that A2 showed low expression of the AKT gene. Zhao (2021) observed that G. duodenalis extracellular vesicles (GEV) could increase the levels of parasite-induced inflammatory response in mouse macrophages through activation of p38, ERK, and NF-kB signaling pathways, and activation reduces the AKT signaling pathway (Zhao et al. 2021). Li et al. (2017) presented that macrophages play a role in the immune response to giardiasis by activating AKT/MAPK signaling (Li et al. 2017). A document suggests exposure of the human’s biofilms to the gut microbiota of Giardia spp. induces the release of buoyant planktonic bacteria, leading to the induction of epithelial apoptosis, promoting bacterial transmigration, and a pro-inflammatory reaction. It suggests that it may increase the production of CXCL8 (interleukin-8) (Beatty et al. 2017). On the other hand, the PI3K/AKT signaling pathway is one of the major pathways that regulates IL-8 expression and enhances tumor cell migration or invasion. However, in our study, PI3K had reduced expression in all individuals with a history of giardiasis. The PIK3CA gene encodes the catalytic subunit of phosphatidylinositol 3-kinase (PI3K), one of the most common genes in tumor malignancies. Activation of the PI3K pathway leads to the induction of the cyclooxygenase-2 (COX-2) enzyme and the production of immunosuppressive prostaglandin E2 (PGE2) (Cai et al. 2020). PIK3CA gene is confirmed in various cancers, including colorectal, breast, head, and neck (Cai et al. 2020), ovarian cancer (Shayesteh et al. 1999), and cervical cancers (Ma et al. 2000). Since this gene is the primary gene in upstream of the AKT gene, it seems that some other mechanisms are involved in the over-expression of AKT in two samples of A4 and A8.
The mean of KRAS gene expression in people with a history of giardiasis was higher than in healthy people, although the high expression was reported just in the A4 sample. The KRAS gene, a member of the RAS superfamily, encodes a small GTPase that regulates diverse cellular processes, including cell proliferation, differentiation, survival, and migration (Downward 2003). KRAS gene is involved in several cancers, namely lung cancer, colorectal cancer (Wang and Fakih 2021), pancreatic cancer (Grant et al. 2016), and ovarian cancer (Jumaa 2022). As with AKT, the high expression of the KRAS gene may be related to the prevention of apoptosis by Giardia. On the other hand, two samples with a history of giardiasis showed low expression of KRAS.
In this study, we demonstrated that the mean relative expression of the CDKN2A gene in people with a history of giardiasis is more than the expression of this gene in healthy people. However, it should be noted that sample A8 displayed low expression of this gene. CDKN2A is an important tumor suppressor gene (Foulkes et al. 1997). The CDKN2A is a potent cyclin-dependent kinase inhibitor and a critical G1-specific negative regulator that arrests cell cycle progression at the G1-S phase boundary. Loss of its function can lead to uncontrolled cell proliferation (Serrano et al. 1993). Giardia may stimulate the CDKN2A gene expression for apoptosis (Cánepa et al. 2007). Since sample A8 had a history of giardiasis for more than 10 years, the gene may reduce gene expression after these years.
In conclusion, Genes AKT1, CDKN2A, KRAS, and PIK3CA were expressed in our samples (human). We showed that the gene expression of some main genes in apoptosis and anti-apoptosis signaling in people with a history of giardiasis differs. Giardia duodenalis seems to induce post-non-infectious symptoms with stimulation of human gene expression. Gene expression patterns can be used as a biomarker. Due to the few samples in this study, we suggest evaluating the studied gene expression in more cases.
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- GEO:
-
Gene expression omnibus
- DEGs:
-
Differential expression genes
- PPI:
-
Protein–protein interaction
- KEGG:
-
Kyoto encyclopedia of genes and genomes
- GO:
-
Gene ontology
References
Abeyrathna P, Su Y (2015) The critical role of Akt in cardiovascular function. Vascul Pharmacol 74:38–48. https://doi.org/10.1016/j.vph.2015.05.008
Adam RD (2021) Giardia duodenalis: Biology and Pathogenesis. Clin Microbiol Rev 34(4):e0002419. https://doi.org/10.1128/cmr.00024-19
Allain T, Buret AG (2020) Pathogenesis and post-infectious complications in giardiasis. Adv Parasitol 107:173–199. https://doi.org/10.1016/bs.apar.2019.12.001
Barwick RS, Levy DA, Craun GF, Beach MJ, Calderon RL (2000) Surveillance for waterborne-disease outbreaks–United States, 1997–1998. MMWR CDC Surveill Summ 49(4):1–21
Beatty JK, Akierman SV, Motta JP, Muise S, Workentine ML, Harrison JJ, Bhargava A, Beck PL, Rioux KP, McKnight GW, Wallace JL, Buret AG (2017) Giardia duodenalis induces pathogenic dysbiosis of human intestinal microbiota biofilms. Int J Parasitol 47(6):311–326. https://doi.org/10.1016/j.ijpara.2016.11.010
Bernander R, Palm JE, Svärd SG (2001) Genome ploidy in different stages of the Giardia lamblia life cycle. Cell Microbiol 3(1):55–62. https://doi.org/10.1046/j.1462-5822.2001.00094.x
Cai Y, Yousef A, Grandis JR, Johnson DE (2020) NSAID therapy for PIK3CA-altered colorectal, breast, and head and neck cancer. Adv Biol Regul 75:100653. https://doi.org/10.1016/j.jbior.2019.100653
Cánepa ET, Scassa ME, Ceruti JM, Marazita MC, Carcagno AL, Sirkin PF, Ogara MF (2007) INK4 proteins, a family of mammalian CDK inhibitors with novel biological functions. IUBMB Life 59(7):419–426. https://doi.org/10.1080/15216540701488358
Carroccio A, Montalto G, Iacono G, Ippolito S, Soresi M, Notarbartolo A (1997) Secondary impairment of pancreatic function as a cause of severe malabsorption in intestinal giardiasis: a case report. Am J Trop Med Hyg 56(6):599–602. https://doi.org/10.4269/ajtmh.1997.56.599
Carter JE, Nelson JJ, Eves M, Boudreaux C (2007) Giardia lamblia infection diagnosed by endoscopic ultrasound-guided fine-needle aspiration. Diagn Cytopathol 35(6):363–365. https://doi.org/10.1002/dc.20636
Certad G, Viscogliosi E, Chabé M, Cacciò SM (2017) Pathogenic mechanisms of Cryptosporidium and Giardia. Trends Parasitol 33(7):561–576. https://doi.org/10.1016/j.pt.2017.02.006
Church DM, Goodstadt L, Hillier LW, Zody MC, Goldstein S, She X, Bult CJ, Agarwala R, Cherry JL, DiCuccio M, Hlavina W, Kapustin Y, Meric P, Maglott D, Birtle Z, Marques AC, Graves T, Zhou S, Teague B, Ponting CP (2009) Lineage-specific biology revealed by a finished genome assembly of the mouse. PLoS Biol 7(5):e1000112. https://doi.org/10.1371/journal.pbio.1000112
Dixon BR (2011) Protozoan parasites: Cryptosporidium, Giardia, Cyclospora, and Toxoplasma. In: Hoorfar J (ed) Rapid detection, characterization and enumeration of food-borne pathogens. ASM Press, Washington, USA, pp 349–370
Dormond M, Gutierrez RL, Porter CK (2016) Giardia lamblia infection increases risk of chronic gastrointestinal disorders. Trop Dis Travel Med Vaccines 2:17. https://doi.org/10.1186/s40794-016-0030-0
Downward J (2003) Targeting RAS signalling pathways in cancer therapy. Nat Rev Cancer 3(1):11–22. https://doi.org/10.1038/nrc969
Foulkes WD, Flanders TY, Pollock PM, Hayward NK (1997) The CDKN2A (p16) gene and human cancer. Mol Med 3(1):5–20
Furukawa M, Lee L, Ikegami T, Maeda T, Nishiyama K, Itaba S, Funakoshi A (2011) Giardiasis in the pancreas accompanied by pancreatic cancer. Pancreas 40(1):168–169. https://doi.org/10.1097/MPA.0b013e3181f6a1b9
Garg R, Lehrke HD, AbuDayyeh BK, Rustagi T (2018) Incidental giardiasis diagnosed during transduodenal EUS-FNA. Gastrointest Endosc 87(1):308–309
Grant TJ, Hua K, Singh A (2016) Molecular pathogenesis of pancreatic cancer. Prog Mol Biol Transl Sci 144:241–275. https://doi.org/10.1016/bs.pmbts.2016.09.008
Halliez MC, Buret AG (2013) Extra-intestinal and long term consequences of Giardia duodenalis infections. World J Gastroenterol 19(47):8974–8985. https://doi.org/10.3748/wjg.v19.i47.8974
Hanevik K, Dizdar V, Langeland N, Hausken T (2009) Development of functional gastrointestinal disorders after Giardia lamblia infection. BMC Gastroenterol 9:27. https://doi.org/10.1186/1471-230x-9-27
Hurník P, Žiak D, Dluhošová J, Židlík V, Šustíková J, Uvírová M, Urban O, Dvořáčková J, Nohýnková E (2019) Another case of coincidental Giardia infection and pancreatic cancer. Parasitol Int 71:160–162. https://doi.org/10.1016/j.parint.2019.04.013
Jumaa MG (2022) Pattern of kras gene expression in Iraqi women ovarian carcinoma. Wiad Lek 75(1):765–769. https://doi.org/10.36740/WLek202204103
Li X, Zhang X, Gong P, Xia F, Li L, Yang Z, Li J (2017) TLR2(-/-) Mice display decreased severity of giardiasis via enhanced pro-inflammatory cytokines production dependent on AKT signal pathway. Front Immunol 8:1186. https://doi.org/10.3389/fimmu.2017.01186
Litleskare S, Rortveit G, Eide GE, Hanevik K, Langeland N, Wensaas KA (2018) Prevalence of irritable bowel syndrome and chronic fatigue 10 years after Giardia infection. Clin Gastroenterol Hepatol 16(7):1064-1072.e1064. https://doi.org/10.1016/j.cgh.2018.01.022
Liu L, Fang R, Wei Z, Wu J, Li X, Li W (2020) Giardia duodenalis induces apoptosis in intestinal epithelial cells via reactive oxygen species-mediated mitochondrial pathway in vitro. Pathogens. https://doi.org/10.3390/pathogens9090693
Ma YY, Wei SJ, Lin YC, Lung JC, Chang TC, Whang-Peng J, Liu JM, Yang DM, Yang WK, Shen CY (2000) PIK3CA as an oncogene in cervical cancer. Oncogene 19(23):2739–2744. https://doi.org/10.1038/sj.onc.1203597
Mahdavi F, Sadrebazzaz A, Chahardehi AM, Badali R, Omidian M, Hassanipour S, Asghari A (2022) Global epidemiology of Giardia duodenalis infection in cancer patients: a systematic review and meta-analysis. Int Health 14(1):5–17. https://doi.org/10.1093/inthealth/ihab026
Mitchell CM, Bradford CM, Kapur U (2011) Giardia lamblia trophozoites in an ultrasound-guided fine-needle aspiration of a pancreatic mucinous neoplasm. Diagn Cytopathol 39(5):352–353
Mørch K, Hanevik K (2020) Giardiasis treatment: an update with a focus on refractory disease. Curr Opin Infect Dis 33(5):355–364. https://doi.org/10.1097/qco.0000000000000668
Murray PJ, Wynn TA (2011) Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol 11(11):723–737. https://doi.org/10.1038/nri3073
Nagasaki T, Komatsu H, Shibata Y, Yamaguchi H, Nakashima M (2011) A rare case of gallbladder cancer with giardiasis. Nihon Shokakibyo Gakkai Zasshi 108(2):275–279
Nakao JH, Collier SA, Gargano JW (2017) Giardiasis and subsequent irritable bowel syndrome: a longitudinal cohort study using health insurance data. J Infect Dis 215(5):798–805. https://doi.org/10.1093/infdis/jiw621
Russell ES (1985) A history of mouse genetics. Annu Rev Genet 19:1–28. https://doi.org/10.1146/annurev.ge.19.120185.000245
Schneider DS, Ayres JS (2008) Two ways to survive infection: what resistance and tolerance can teach us about treating infectious diseases. Nat Rev Immunol 8(11):889–895. https://doi.org/10.1038/nri2432
Serrano M, Hannon GJ, Beach D (1993) A new regulatory motif in cell-cycle control causing specific inhibition of cyclin D/CDK4. Nature 366(6456):704–707
Shayesteh L, Lu Y, Kuo WL, Baldocchi R, Godfrey T, Collins C, Pinkel D, Powell B, Mills GB, Gray JW (1999) PIK3CA is implicated as an oncogene in ovarian cancer. Nat Genet 21(1):99–102. https://doi.org/10.1038/5042
Teimouri A, Keshavarz H, Mohtasebi S, Goudarzi F, Mikaeili F, Borjian A, Allahmoradi M, Yimam Y, Abbaszadeh Afshar MJ (2021) Intestinal parasites among food handlers in Iran: a systematic review and meta-analysis. Food Microbiol 95:103703. https://doi.org/10.1016/j.fm.2020.103703
Terra SB, Henry MR, Pritt BS, Hartley CP, Zanish JM, Chua HK, Sturgis CD (2020) Incidental Giardia duodenalis cysts in exfoliative anal cytology: an immunocompetent adult female with prior squamous dysplasia. Diagn Cytopathol 48(11):1141–1143
Vivancos V, González-Alvarez I, Bermejo M, Gonzalez-Alvarez M (2018) Giardiasis: characteristics, pathogenesis and new insights about treatment. Curr Top Med Chem 18(15):1287–1303. https://doi.org/10.2174/1568026618666181002095314
Wang C, Fakih M (2021) Targeting KRAS in colorectal cancer. Curr Oncol Rep 23(3):28. https://doi.org/10.1007/s11912-021-01022-0
Warren HS, Fitting C, Hoff E, Adib-Conquy M, Beasley-Topliffe L, Tesini B, Liang X, Valentine C, Hellman J, Hayden D, Cavaillon JM (2010) Resilience to bacterial infection: difference between species could be due to proteins in serum. J Infect Dis 201(2):223–232. https://doi.org/10.1086/649557
Waterston RH, Lindblad-Toh K, Birney E, Rogers J, Abril JF, Agarwal P, Agarwala R, Ainscough R, Alexandersson M, An P, Antonarakis SE, Attwood J, Baertsch R, Bailey J, Barlow K, Beck S, Berry E, Birren B, Bloom T, Lander ES (2002) Initial sequencing and comparative analysis of the mouse genome. Nature 420(6915):520–562. https://doi.org/10.1038/nature01262
Yang Y, Zhao Y, Liu L, Zhu W, Jia S, Li X, Li W (2022) The anti-apoptotic role of COX-2 during in vitro infection of human intestinal cell line by Giardia duodenalis and the potential regulators. Infect Immun 90(3):e00672-e621
Zhao P, Cao L, Wang X, Li J, Dong J, Zhang N, Li X, Li S, Sun M, Zhang X, Liang M, Pu X, Gong P (2021) Giardia duodenalis extracellular vesicles regulate the pro-inflammatory immune response in mouse macrophages in vitro via the MAPK, AKT and NF-Κb Pathways. Parasit Vectors 14(1):358. https://doi.org/10.1186/s13071-021-04865-5
Zschaler J, Schlorke D, Arnhold J (2014) Differences in innate immune response between man and mouse. Crit Rev Immunol 34(5):433–454
Acknowledgements
The authors express their gratitude to Shahid Sadoughi University of Medical Sciences for the financial support provided. Special thanks are extended to Saeedeh Sadat Hosseini for her valuable contributions to the methodology.
Funding
We would like to thank Shahid Sadoughi University of Medical Sciences, Yazd, Iran, with Grant Number 8986.
Author information
Authors and Affiliations
Contributions
PS wrote the manuscript draft, applied the method, and collected the data; GE revised the manuscript, designed the protocol, and supervised the project; MT revised the manuscript and designed the methodology; AAJ supervised the methodology and revised the manuscript; MV analyzed the data statistically. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The ethical statement of this study was approved by the Ethical Committee of Shahid Sadoughi University of Medical Sciences with the code of IR.SSU.MEDICINE.REC.1400.399 This project was approved by Shahid Sadoughi University of Medical Sciences, Yazd, Iran, by the code 8986.
Consent for publication
Consent for publication was obtained from patients.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Saeedi, P., Eslami, G., Tohidfar, M. et al. Differential gene expression (DGE) analysis in persons with a history of giardiasis. AMB Expr 14, 1 (2024). https://doi.org/10.1186/s13568-023-01657-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13568-023-01657-1