Abstract
In this paper, we propose and study a stochastic two-species cooperation model with functional response in a polluted environment. We first perform the survival analysis and establish sufficient conditions for extinction, weak persistence, and stochastic permanence. Then we further perform the survival analysis based on the temporal average of population size and derive sufficient conditions for the strong persistence in the mean and weak persistence in the mean. Finally, we present numerical simulations to justify the theoretical results.
Similar content being viewed by others
1 Introduction
With rapid development of industries and agriculture, a mass of toxicants has been emitted into the environment, such as the industrial wastewater, domestic sewage, and other contaminants. The presence of a variety of toxicants in the environment not only seriously threatened the survival of the exposed populations but also affected the human life style. Therefore it is important to estimate the environmental toxicity, which requires quantitative estimates for the survival risk of species in a polluted environment. This motivates scholars to utilize the mathematical models to assess the effects of toxicants on various ecosystems. Hallam et al. [1–3] did pioneering works. It has been a significant topic of considerable researches, and more and more deterministic models have been proposed and analyzed (see [4–17]). They all provide a great insight into the effects of pollutants. In this paper, we mainly attempt to study a two-species cooperation model with functional response. We assume that the environment is of complete spatial homogeneity and there is no migration. Let \(x_{i}(t)\) represent the population size (population density) of the ith species at time t. We consider the following cooperation model in a polluted environment:
where the positive coefficients \(r_{1}, r_{2}\) and \(a_{1}, a_{2}\) are the intrinsic growth rates and self-inhibition rates, respectively. In the classical cooperation model the mutualism effects are described by a bilinear function, that is, \(x_{i}\) response to \(x_{j}\) is assumed to be increasingly monotonic, an inherent assumption meaning that the more \(x_{j}\) there exist in the environment, the better off the \(x_{i}\). But here the term \(c_{i}x_{i}(t)/(1+b_{i}x_{i}(t))\) represents the functional response function, and moreover, it is an increasing function with respect to \(x_{i}\) and has a saturation value for large enough \(x_{i}\). The positive coefficients \(c_{j}\) measure the mutualism effects of species \(x_{j}\) on species \(x_{i}\), and \(b_{j}\), \(i, j = 1,2\), \(i\neq j\), are positive control constants. For a relevant ecological model of system (1.1), we refer the readers to [18].
Now we are in the position to describe the dynamics of population in a polluted environment. We assume that the living organisms absorb part of toxicants into their bodies, the dynamics of the population is affected by internal toxicant, and the individuals in two species have identical concentration of organismal toxicant at time t (see [5]). To simplify the mathematical model, we assume that the capacity of the environment is so large that the change of toxicant in the environment that comes from uptake and egestion by the organisms can be neglected (see [3]). Let \(C_{0}(t)\) represent the concentration of toxicant in the organism at time t, and let \(C_{E}(t)\) represent the concentration of toxicant in the environment at time t. A coupling between species and toxicant is formulated by assuming that the intrinsic growth rate of the ith population, \(r_{i0}-r_{i1}C_{0}(t)\), is a linear function of concentration of toxicant present in the organism. Then the population dynamics can be given as follows:
where \(r_{i0}\) is the intrinsic growth rate of the ith species in the absence of toxicant, \(r_{i1}\), \(i=1,2\), is the dose-response rate of species i to the toxicant concentration, d represents the uptake rate of toxicant from environment per unit biomass, η represents the intake rate of toxicant from food per unit biomass, \(l_{1}\) represents the organismal net ingestion rates of toxicant, \(l_{2}\) represents the organismal excretion rates of toxicant, and h represents the loss rate of toxicant from the environment due to the processes such as biological transformation, chemical hydrolysis, volatilization, microbial degradation, and photosynthetic degradation. Here \(u(t)\) represents the input rate of exogenous toxin at time t, which is a nonnegative continuous function defined on \([0, \infty )\) with \(u_{1}:=\sup_{t\geq 0}u(t)>0\).
Considering that the fate of young recruits after reproduction is quite sensitive to the environmental fluctuations, so the growth rate of population is inevitably affected. May and Allen [19] have claimed that the growth rates in population systems should be fluctuating around some average values because of the environmental fluctuations. In this sense, it is necessary to incorporate the environmental fluctuations into our model. In practice, we usually estimate the growth rate \(r_{i0}\) by an average value plus an error term; by the central limit theorem the error term follows a normal distribution, and thus we can approximate the error term by \(\sigma _{i}\dot{B}_{i}(t)\), that is,
where \(\dot{B}_{i}(t)\) represents a white noise process (i.e., \(B_{i}(t)\) is a standard Brownian motion), and \(\sigma ^{2}_{i}\) is the intensity of the white noise. According to our discussion, a stochastic two-species cooperation model in polluted environment is derived as follows:
The initial conditions satisfy the conditions
In recent years, many interesting and important works about the stochastic model in polluted environment have been reported (see [20–33]). But to the best of our knowledge, there exist few published papers concerning system (1.3). Noting that the last two equations in system (1.3) can be explicitly solved, so we only need to consider the following subsystem:
Motivated by the existing results, the rest of this paper is arranged as follows. In Sect. 2, we introduce several commonly used basic lemmas and classical definitions. In Sect. 3, we perform the survival analysis and establish sufficient criteria for extinction, weak persistence, and stochastic permanence. In Sect. 4, we further discuss the survival analysis based on the temporal average of population size and derive sufficient conditions for the strong persistence in the mean and weak persistence in the mean. In Sect. 5, we present several numerical simulations to validate our theoretical results. The limitation of the model is also discussed in the last section.
2 Preliminaries
From now on, unless otherwise specified, we always work on the complete probability space \((\varOmega,\mathcal{F},P)\) with filtration \(\{\mathcal{F}_{t}\}_{t\geq 0}\) satisfying the usual conditions, that is, it is right continuous, and \(\mathcal{F}_{0}\) contains all P-null sets. Note that both \(C_{0}(t)\) and \(C_{E}(t)\) represent the concentrations of toxicant, so to be realistic, we must have \(0 \leq C_{0}(t) < 1\) and \(0 \leq C_{E}(t) <1\) for all \(t\geq 0\). In fact, we can prove that by solving the last two equations of system (1.3).
For convenience and simplicity, we introduce the following notations:
Definition 2.1
(See [34])
To state and prove our main results, we recall some classical concepts.
-
Population \(x_{i}\) is said to be extinct if \(\lim_{t\rightarrow \infty }x_{i}(t)=0, i=1,2\), a.s.
-
Population \(x_{i}\) is said to be weakly persistent if \(\limsup_{t\rightarrow \infty }x_{i}(t)>0\) a.s.
-
Population \(x_{i}\) is said to be weakly persistent in the mean if \(\limsup_{t\rightarrow \infty }\langle x_{i}(t) \rangle >0\) a.s.
-
Population \(x_{i}\) is said to be strongly persistent in the mean if \(\liminf_{t\rightarrow \infty }\langle x_{i}(t) \rangle >0\) a.s.
-
System (1.4) is said to be stochastically permanent if for every \(\varepsilon \in (0,1)\), there exists a pair of positive constants \(\alpha =\alpha (\varepsilon ),\beta =\beta (\varepsilon )\) such that for any positive initial condition \(x(0)\), the solution \(x(t)\) satisfies
$$ \liminf_{t\rightarrow \infty }P\bigl\{ x_{i}(t)\geq \alpha \bigr\} \geq 1- \varepsilon,\qquad \liminf_{t\rightarrow \infty }P\bigl\{ x_{i}(t) \leq \beta \bigr\} \geq 1- \varepsilon. $$(2.1)
3 Survival analysis
In this section, we first study the existence and uniqueness of a globally positive solution for the biological significance and then perform the survival analysis of system (1.4).
Theorem 3.1
For every initial value\(x(0)=(x_{1}(0),x_{2}(0))\in R^{2}_{+}\), system (1.4) has a unique solution\(x(t)\), and\(x(t)\)remains in\(R^{2}_{+}\)for all\(t\geq 0\)with probability one.
Proof
The proof of the theorem is standard. Firstly, let us consider the following stochastic system:
with initial condition \(y_{1}(0)=\ln x_{1}(0), y_{2}(0)=\ln x_{2}(0)\). It is easy to verify that the coefficients of system (3.1) satisfy the local Lipschitz condition, and thus system (3.1) has a unique solution \(y(t)\) on \([0, \tau _{e})\), where \(\tau _{e}\) is the explosion time (see [35]). By applying Itô’s formula and a simple calculation, it is easy to derive that \(x_{1}(t)=e^{y_{1}(t)}, x_{2}(t)=e^{y_{2}(t)}\) is the unique positive local solution to system (1.4) with initial value \(x(0)\in R^{2}_{+}\). To show that this solution is global, we only need to show that \(\tau _{e}=\infty \).
Let \(n_{0}\) be sufficiently large such that every component of \(x(0)\) remains in the interval \([\frac{1}{n_{0}}, n_{0}]\). For each integer \(n\geq n_{0}\), we define the stopping time
Here we set \(\inf \emptyset =+\infty \) (∅ denotes the empty set). Obviously, \(\tau _{n}\) is increasing as \(n\rightarrow \infty \). Let \(\tau _{\infty }=\lim_{n\rightarrow \infty }\tau _{n}\), whence \(\tau _{\infty }\leq \tau _{e}\) almost surely; if we can show that \(\tau _{\infty }=\infty \) almost surely, then \(\tau _{e}=\infty \) almost surely, and the proof is completed.
Define the \(C^{2}\)-function \(\widetilde{V}(x): R^{2}_{+}\rightarrow R_{+}\) as
The nonnegativity of this function can be seen from \(v-1-\ln v\geq 0\) for \(v>0\). Let \(T > 0\) be an arbitrary constant. Then for \(0 \leq t \leq \tau _{n} \wedge T\), by Itô’s formula we obtain
where
Clearly, \(G(x_{1},x_{2})\) is upper bounded, say by \(\mathcal{M}\). So we have
Integrating both sides from 0 to \(\tau _{n} \wedge T\), we have
Taking the expectations on both sides yields that
Let \(\varOmega _{n}=\{\tau _{n}\leq T\}\). For arbitrary \(\omega \in \varOmega _{n}\), there exists some i such that \(x_{i}(\tau _{n},\omega )\) equals either n or \(\frac{1}{n}\), and thus \(\widetilde{V}(x(\tau _{n},\omega ))\) is not less than \((n-1-\ln n)\wedge (\frac{1}{n}-1+\ln n)\triangleq \mu (n)\). It then follows from (3.6) that
where \(1_{\varOmega _{n}}\) is the indicator function of \(\varOmega _{n}\). Let \(n\rightarrow \infty \). Then \(\mu (n)\rightarrow \infty \), and (3.7) leads to \(P(\tau _{\infty }\leq T)=0\). By the arbitrariness of T we have \(P(\tau _{\infty }=\infty )=1\) almost surely. This completes the proof of Theorem 3.1. □
It is well known that the threshold is very important for assessing the risk of extinction for species exposed to toxicant from a biological point of view. In the following, we first show that the pth moment of the solution of system (1.4) is upper bounded and then establish the threshold between weak persistence and extinction for species \(x_{i}\) modeled by (1.4). To begin with, we present the fundamental assumption that \(r_{i0}>0.5\sigma ^{2}_{i}\). Unless otherwise stated, it is always assumed in this paper.
Theorem 3.2
For any\(p>1\), there exists a positive constant\(K=K(p)\)such that the solution\(x(t)\)of system (1.4) has the property that
Proof
Define \(V(x_{1},x_{2})=x^{p}_{1}+x^{p}_{2}\). By Itô’s formula, we have
where
Obviously, \(L(x_{1}, x_{2})\) is upper bounded; we denote it by \(\mathcal{L}\), that is,
Applying Itô’s formula to \(e^{t}V(x_{1},x_{2})\) yields that
Integrating from 0 to t and then taking the expectation of both sides yield that
This gives that
This completes the proof of Theorem 3.2. □
Theorem 3.3
If\(C^{*}_{0}<(r_{i0}-0.5\sigma ^{2}_{i})/r_{i1}\), then system (1.4) is stochastically permanent.
Proof
Taking arbitrary \(0<\varepsilon <1\), we first claim that there is a constant \(\alpha > 0\) such that
Since \(C^{*}_{0}<(r_{i0}-0.5\sigma ^{2}_{i})/r_{i1}\), we can choose a constant \(m>0\) such that
Define
By Itô’s formula we derive that
Let k be sufficiently small to satisfy
We define
By Itô’s formula again,
Clearly, \(J(x_{1}, x_{2})\) is upper bounded for \((x_{1}, x_{2})\in R^{2}_{+}\), that is, \(\mathcal{J}\triangleq \sup_{x\in R^{2}_{+}}J(x_{1}, x_{2})<\infty \). As a result, we have
Integrating both sides and then taking the expectations, we obtain that
that is,
On the other hand,
For arbitrary \(\varepsilon \in (0, 1)\), let \(\alpha =(\frac{\varepsilon }{\delta })^{\frac{1}{m}}\). By Chebyshev’s inequality we have
which gives that
that is,
In the following, we prove that for arbitrary \(\varepsilon \in (0,1)\), there is a constant \(\beta > 0\) such that
Let \(\beta = [\frac{K}{\varepsilon } ]^{\frac{1}{p}}\). Then by Chebyshev’s inequality and Theorem 3.2 we have
which implies that
Consequently,
This completes the proof of Theorem 3.3. □
Remark 3.1
Theorem 3.3, which directly measures the population size \(x_{i}(t)\), indicates that the population size will neither too small nor too large with large probability when the time is sufficiently large.
Theorem 3.4
If\(\langle C_{0} \rangle _{*}>(r_{i0}-0.5\sigma ^{2}_{i}+ \frac{c_{j}}{b_{j}})/r_{i1}\), \(i,j=1,2\), \(i\neq j\), then population\(x_{i}\)goes to extinction with probability one.
Proof
Applying Itô’s formula yields that
Integrating both sides yields that
Letting \(t\rightarrow \infty \) and applying the strong law of large numbers, we obtain that
In other words, \(\lim_{t\rightarrow \infty }x_{i}(t) = 0\). This completes the proof of Theorem 3.4. □
Remark 3.2
Theorem 3.4 indicates the worst case that the population will go to extinction almost surely.
Theorem 3.5
If\(\langle C_{0} \rangle _{*}<(r_{i0}-0.5\sigma ^{2}_{i}+ \frac{c_{j}}{b_{j}})/r_{i1}\), \(i,j=1,2\), \(i\neq j\), then population\(x_{i}\)is weakly persistent almost surely.
Proof
We denote \(S:=\{\omega: \limsup_{t\rightarrow \infty }x_{i}(t,\omega )=0\}\) and assume that \(P(S)>0\). Then for all \(\omega \in S\), we have \(\limsup_{t\rightarrow \infty }x_{i}(t, \omega )=0\). For arbitrary small ε satisfying \(0<\varepsilon <1\), there exists \(T(\omega )\) such that
It then follows that
By the continuity of \(x_{i}(t,\omega )\) there must be a constant K̃ such that \(x_{i}(t,\omega )\leq \tilde{K}\) for \(0\leq t\leq T(\omega )\). On the other hand,
for sufficient large t. Since ε is arbitrarily small, we obtain that
Since \(x_{i}(t)>0\), we have \(\liminf_{t\rightarrow \infty }\frac{1}{t}\int _{0}^{t}x_{i}(s, \omega )\,ds\geq 0\), and thus
Substituting (3.40) into (3.34) and using the strong law of large numbers, we deduce the contradiction
This completes the proof of Theorem 3.5. □
Remark 3.3
Theorem 3.5 admit the case that the population size is close to zero even if the time is sufficiently large. In this case the survival of species can be dangerous in reality. In addition, Theorems 3.4 and 3.5 reveal that \(\langle C_{0} \rangle _{*}=(r_{i0}-0.5\sigma ^{2}_{i}+ \frac{c_{j}}{b_{j}})/r_{i1}\) is the threshold between extinction and weak persistence.
4 The estimation of temporal averages
In this section, we further discuss the survival analysis of system (1.4) based on the temporal average of the population size \(x_{i}(t)\).
Theorem 4.1
The solution of system (1.4) has the property that
Proof
By Itô’s formula,
Integrating both sides yields that
where \(N(t)=\int _{0}^{t}\sigma _{i}e^{s}\,dB_{i}(s)\). Note that \(N(t)\) is a local martingale with quadratic variation
By the exponential martingale inequality we have
where \(\theta >1\), and \(\gamma >0\) is arbitrary. By the Borel–Cantelli lemma, for almost all \(\omega \in \varOmega \), there is a random integer \(n_{0}(\omega )\) such that for all \(n \geq n_{0}(\omega )\) and \(0\leq t \leq \gamma n\),
Substituting (4.6) into (4.3) yields that
Since \(0\leq t \leq \gamma n\) and \(x_{i}(t)>0\), there is a constant ϱ such that
that is, for \(0\leq t \leq \gamma n\), we have
Therefore, if \(\gamma (n-1)\leq t \leq \gamma n\) and \(n \geq n_{0}(\omega )\), then we obtain that
which implies that
Then letting \(\theta \rightarrow 1\) and \(\gamma \rightarrow 0\) gives the required assertion (4.1). This completes the proof of Theorem 4.1. □
Theorem 4.2
Let\(x(t)\)be a solution of system (1.4). If\(r_{i0}-0.5\sigma ^{2}_{i}-r_{i1}\langle C_{0} \rangle ^{*}>0\), then the component\(x_{i}(t)\)satisfies
Proof
We first show that \(\limsup_{t\rightarrow \infty }\frac{\ln x_{i}(t)}{t} \leq 0\). From Theorem 4.1 we have that
In the following, we show that \(\liminf_{t\rightarrow \infty }\frac{\ln x_{i}(t)}{t} \geq 0\). Since \(\lim_{t\rightarrow \infty } \frac{\int _{0}^{t}\sigma _{i}\,dB_{i}(s)}{t}=0\) a.s., for any \(\varepsilon \in (0,1)\), there exists a positive constant T such that
for \(t>s\geq T\). From equations (3.38) and (4.14) we have
This gives that
that is, \(x_{i}^{-1}(t) \leq \bar{K}e^{2\varepsilon (t+T)}\) almost surely. Therefore we obtain that
which yields that
In other words,
Then from the arbitrariness of ε it follows that
This completes the proof of Theorem 4.2. □
Theorem 4.3
Let\(x(t)\)be a solution of system (1.4). If\(r_{i0}-0.5\sigma ^{2}_{i}-r_{i1} \langle C_{0} \rangle ^{*}>0\), then the component\(x_{i}(t)\)has the following property:
Moreover,
that is, population\(x_{i}\)is strongly persistent in the mean almost surely.
Proof
Recalling (3.34), we obtain that
Then from Theorem 4.2 it follows that
On the other hand,
Similarly, we obtain that
This completes the proof of Theorem 4.3. □
Theorem 4.4
If\(\langle C_{0} \rangle _{*}<(r_{i0}-0.5\sigma ^{2}_{i})/r_{i1}\), then the component\(x_{i}(t)\)satisfies
In other words, population\(x_{i}\)is weakly persistent in the mean almost surely.
Proof
Recalling (4.25), we obtain that
For all \(\omega \in \{\omega: \limsup_{t\rightarrow \infty }\langle x_{i}(t, \omega ) \rangle =0\}\), we have \(\langle x_{i}(t,\omega ) \rangle ^{*}=0\). Then from Theorem 4.2 and (4.28) it follows that
which contradicts the assumption of Theorem 4.4. So we must have \(\limsup_{t\rightarrow \infty }\langle x_{i}(t) \rangle >0\). This completes the proof of Theorem 4.4. □
5 Numerical simulations
In this section, we present several specific examples to justify our theoretical results based on the Milstein method, which is mentioned by Higham [36]. For system (1.4), we assign \(r_{10} = 1.6, r_{11} = 0.8, a_{1} = 0.5, b_{2} = 0.4, c_{2} = 0.2, \sigma _{1}=0.6, r_{20} = 1.4, r_{21} = 0.7, a_{2} = 0.4, b_{1} = 0.5, c_{1} = 0.4,\sigma _{2}=0.7\), and the initial value \((x_{1}(0), x_{2}(0)) =(0.8, 0.6)\) and then consider the following discrete version:
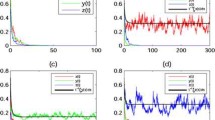
where \(\xi _{1}(k)\) and \(\xi _{2}(k)\) are independent standard Gaussian random variables. To reveal the effects of toxicant, we need to choose \(C_{0}(t)=0\) for comparison with system (5.1). By a simple calculation, \(r_{10}=1.6>0.5\sigma _{1}^{2}=0.18, r_{20}=1.4>0.5\sigma _{2}^{2}=0.245\). Then each species can live alone in a nonpolluted environment; see Fig. 1. Theorem 3.1 shows that for any positive initial value, the solution of system (5.1) remains in \(R^{2}_{+}\) almost surely. The nice property provides us a great opportunity to discuss the survival analysis of system (5.1), and so it is the fundamental conclusion in this paper.
Stochastic permanence of system (5.1) with \(C_{0}(t)=0, \Delta t=0.01\)
Example 5.1
Theorem 3.3 shows that a tiny amount of toxicant cannot disrupt the stochastic permanence; in other words, the population size will be neither too small nor too large with large probability if the time is sufficiently large. Let \(C_{0}(t)=0.4+0.1\sin (t), \Delta t=0.01\); by a simple calculation, \(C_{0}^{*}=0.5, (r_{10}-0.5\sigma _{1}^{2})/r_{11}=1.775, (r_{20}-0.5 \sigma _{2}^{2})/r_{21}=1.65\), that is, \(C_{0}^{*}<(r_{i0}-0.5\sigma _{i}^{2})/r_{i1}\). So population \(x_{i}\) is stochastically permanent by Theorem 3.3; see Fig. 2.
Stochastic permanence of system (5.1) with \(C_{0}(t)=0.4+0.1\sin (t), \Delta t=0.01\)
Example 5.2
Theorem 3.4 shows that a large amount of toxicant will force the species incline to extinction with probability one. Let \(C_{0}(t)=3.1+0.1\sin (t), \Delta t=0.01\). It is easy to compute that \(\langle C_{0} \rangle _{*}=3.1, (r_{10}-0.5\sigma _{1}^{2}+ \frac{c_{1}}{b_{1}})/r_{11}=2.4, (r_{20}-0.5\sigma _{2}^{2}+ \frac{c_{2}}{b_{2}})/r_{21}=2.79\), that is, \(\langle C_{0}\rangle _{*}>(r_{i0}-0.5\sigma _{i}^{2}+ \frac{c_{j}}{b_{j}})/r_{i1}\). So population \(x_{i}\) goes to extinction almost surely; see Fig. 3.
Extinction of system (5.1) with \(C_{0}(t)=3.1+0.1\sin (t), \Delta t=0.01\)
Example 5.3
Theorem 3.5 admits the case that the population size is closed to zero even if the time is sufficiently large. Let \(C_{0}(t)=1.78+0.1\sin (t), \Delta t=0.01\). Then by a straightforward calculation we have \(\langle C_{0} \rangle _{*}=1.78\) and \(\langle C_{0} \rangle _{*}<(r_{i0}-0.5\sigma _{i}^{2}+ \frac{c_{j}}{b_{j}})/r_{i1}\). By Theorem 3.5 population \(x_{i}\) is weakly persistent almost surely; see Fig. 4.
Weak persistence of system (5.1) with \(C_{0}(t)=1.78+0.1\sin (t), \Delta t=0.01\)
6 Discussion
In this paper, considering the fact of polluted environment, we propose and study a stochastic two-species cooperation model with functional response. However, we only consider the case that a coupling between species and toxicant is a linear function of concentration of toxicant present in the organism, that is, \(r_{i0}-r_{i1}H_{i}(C_{0})=r_{i0}-r_{i1}C_{0}(t)\), where \(H_{i}(C_{0})\) is the dose-response function of species \(x_{i}\) to the toxin. However, someone suggested that \(H_{i}(C_{0})\) should be nonlinear in many cases, such as a sigmoid dose-response curve (see [37, 38]). Liu and Wang [24] also introduced a more general case: \(H_{i}(C_{0})\) is a nondecreasing continuous function of \(C_{0}\) with \(H_{i}(0) = 0\). Clearly, our assumption is a particular case of this generalized condition. Moreover, the exogenous toxin cannot be continuous but rather of the impulse form, the growth rate of population is also inevitably affected by other environment noise such as Lévy jumps, and the regime-switching is other common random perturbation in the environment (see [39–41]). All these questions associated with the polluted environment are interesting topics to deserve further investigation, and we leave them for our future works.
References
Hallam, T.G., Clark, C.E., Lassiter, R.R.: Effects of toxicants on populations: a qualitative approach I. Equilibrium environmental exposure. Ecol. Model. 18(3–4), 291–304 (1983)
Hallam, T.G., Clark, C.E., Jordan, G.S.: Effects of toxicant on population: a qualitative approach II. First order kinetics. J. Math. Biol. 18(1), 25–37 (1983)
Hallam, T.G., de Luna, J.T.: Effects of toxicants on populations: a qualitative approach III. Environmental and food chain pathways. J. Theor. Biol. 109(3), 411–429 (1984)
Luna, J.T.D., Hallam, T.G.: Effects of toxicants on populations: a qualitative approach IV. Resource-consumer-toxicant models. Ecol. Model. 35(3), 249–273 (1987)
Liu, H.P., Ma, Z.E.: The threshold of survival for system of two species in a polluted environment. J. Math. Biol. 30(1), 49–61 (1991)
Freedman, H.I., Shukla, J.B.: Models for the effect of toxicant in single-species and predator–prey systems. J. Math. Biol. 30(1), 15–30 (1991)
Chattopadhyay, J.: Effect of toxic substances on a two-species competitive system. Ecol. Model. 84(1–3), 287–289 (1996)
Ma, Z.E., Zong, W.G., Luo, Z.X.: The thresholds of survival for an n-dimensional food chain model in a polluted environment. J. Math. Anal. Appl. 210(2), 440–458 (1997)
Pan, J.X., Jin, Z., Ma, Z.E.: Thresholds of survival for an n-dimensional Volterra mutualistic system in a polluted environment. J. Math. Anal. Appl. 252(2), 519–531 (2000)
Mukherjee, D.: Persistence and global stability of a population in a polluted environment with delay. J. Biol. Syst. 10(3), 225–232 (2002)
Liu, B., Chen, L.S., Zhang, Y.J.: The effects of impulsive toxicant input on a population in a polluted environment. J. Biol. Syst. 11(3), 265–274 (2003)
Dubey, B.: A model for the effect of time delay on the dynamics of a population living in a polluted environment. J. Biol. Syst. 12(1), 35–43 (2004)
Duan, L.X., Lu, Q.S., Yang, Z.Z., Yang, L.S.: Effects of diffusion on a stage-structured population in a polluted environment. Appl. Math. Comput. 154(2), 347–359 (2004)
Samanta, G.P., Maiti, A.: Dynamical model of a single-species system in a polluted environment. J. Appl. Math. Comput. 16(1–2), 231–242 (2004)
Dubey, B., Hussain, J.: Modelling the survival of species dependent on a resource in a polluted environment. Nonlinear Anal., Real World Appl. 7(2), 187–210 (2006)
He, J., Wang, K.: The survival analysis for a single-species population model in a polluted environment. Appl. Math. Model. 31(10), 2227–2238 (2007)
Zhao, Z., Chen, L.S., Song, X.Y.: Extinction and permanence of chemostat model with pulsed input in a polluted environment. Commun. Nonlinear Sci. Numer. Simul. 14(4), 1737–1745 (2009)
Zhang, H., Feng, F., Jing, B., Li, Y.Q.: Almost periodic solution of a multispecies discrete mutualism system with feedback controls. Discrete Dyn. Nat. Soc. 2015, Article ID 268378 (2015)
May, R.M.: Stability and Complexity in Model Ecosystems. Princeton University Press, Princeton (2001)
Liu, M., Wang, K.: Survival analysis of stochastic single-species population models in polluted environments. Ecol. Model. 220(9), 1347–1357 (2009)
Liu, M., Wang, K., Wu, Q.: Survival analysis of stochastic competitive models in a polluted environment and stochastic competitive exclusion principle. Bull. Math. Biol. 73(9), 1969–2012 (2011)
Liu, M., Wang, K.: Persistence and extinction of a stochastic single-specie model under regime switching in a polluted environment. J. Theor. Biol. 264(3), 934–944 (2010)
Geng, Z.J., Liu, M.: Analysis of stochastic Gilpin–Ayala model in polluted environments. IAENG Int. J. Appl. Math. 45(2), 128–137 (2015)
Liu, M., Wang, K.: Survival analysis of a stochastic cooperation system in a polluted environment. J. Biol. Syst. 19(2), 183–204 (2011)
Liu, M., Wang, K.: Survival analysis of a stochastic single-species population model with jumps in a polluted environment. Int. J. Biomath. 9(1), 207–221 (2016)
Liu, M., Bai, C.Z.: Persistence and extinction of a stochastic cooperative model in a polluted environment with pulse toxicant input. Filomat 29(6), 1329–1342 (2015)
Zhao, Y., Yuan, S.L., Zhang, Q.M.: The effect of Lévy noise on the survival of a stochastic competitive model in an impulsive polluted environment. Appl. Math. Model. 40(17–18), 7583–7600 (2016)
Zhao, Y., Yuan, S.L.: Optimal harvesting policy of a stochastic two-species competitive model with Lévy noise in a polluted environment. Phys. A, Stat. Mech. Appl. 477, 20–33 (2017)
Han, Q.I., Jiang, D.Q., Ji, C.Y.: Analysis of a delayed stochastic predator–prey model in a polluted environment. Appl. Math. Model. 38(13), 3067–3080 (2014)
Liu, Q., Chen, Q.M.: Analysis of a stochastic delay predator–prey system with jumps in a polluted environment. Appl. Math. Comput. 242, 90–100 (2014)
Deng, M.L., Liu, M., Bai, C.Z.: Dynamics of a stochastic delayed competitive model with impulsive toxicant input in polluted environments. Abstr. Appl. Anal. 2014, Article ID 634871 (2014)
Zhao, Y., Yuan, S.L., Ma, J.L.: Survival and stationary distribution analysis of a stochastic competitive model of three species in a polluted environment. Bull. Math. Biol. 77(7), 1285–1326 (2015)
Liu, M.: Survival analysis of a cooperation system with random perturbations in a polluted environment. Nonlinear Anal. Hybrid Syst. 18, 100–116 (2015)
Liu, M., Fan, M.: Permanence of stochastic Lotka–Volterra systems. J. Nonlinear Sci. 27(2), 425–452 (2017)
Mao, X.R.: Stochastic Differential Equations and Applications. Horwood, Chichester (1997)
Higham, D.J.: An algorithmic introduction to numerical simulation of stochastic differential equations. Soc. Ind. Appl. Math. Rev. 43(3), 525–546 (2001)
Butler, G.C.: Principles of Ecotoxicology. Wiley, New York (1979)
Filov, V.A., Golubev, A.A., Liublina, E.I., Tolokontsev, N.A.: Quantitative Toxicology. Wiley, New York (1979)
Liu, M., Bai, C.Z.: Optimal harvesting of a stochastic mutualism model with regime-switching. Appl. Math. Comput. 373, 125040 (2020)
Wang, H., Liu, M.: Stationary distribution of a stochastic hybrid phytoplankton–zooplankton model with toxin-producing phytoplankton. Appl. Math. Lett. 101, 106077 (2020)
Deng, Y., Liu, M.: Analysis of a stochastic tumor-immune model with regime switching and impulsive perturbations. Appl. Math. Model. 78, 482–504 (2020)
Acknowledgements
The author thanks the anonymous reviewers for their careful reading of the manuscript and valuable comments and suggestions.
Availability of data and materials
Data sharing is not applicable to this paper as no datasets were generated or analyzed during the current study.
Funding
The work is supported by the Scientific Research Foundation of Chongqing Technology and Business University (No. 1856048).
Author information
Authors and Affiliations
Contributions
The single author is responsible for the complete manuscript. The author read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The author declares that he has no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guo, S. Survival analysis of a stochastic cooperation system with functional response in a polluted environment. Adv Differ Equ 2020, 354 (2020). https://doi.org/10.1186/s13662-020-02816-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13662-020-02816-5