Abstract
Recombinant adenovirus (rAd) regimens, including replication-competent oncolytic adenovirus (OAV) and replication-deficient adenovirus, have been identified as potential cancer therapeutics. OAV presents advantages such as selective replication, oncolytic efficacy, and tumor microenvironment (TME) remodeling. In this perspective, the principles and advancements in developing OAV toolkits are reviewed. The burgeoning rAd may dictate efficacy of conventional cancer therapies as well as cancer immunotherapies, including cancer vaccines, synergy with adoptive cell therapy (ACT), and TME reshaping. Concurrently, we explored the potential of rAd hitchhiking to adoptive immune cells or stem cells, highlighting how this approach facilitates synergistic interactions between rAd and cellular therapeutics at tumor sites. Results from preclinical and clinical trials in which immune and stem cells were infected with rAd have been used to address significant oncological challenges, such as postsurgical residual tumor tissue and metastatic tissue. Briefly, rAd can eradicate tumors through various mechanisms, resulting from tumor immunogenicity, reprogramming of the TME, enhancement of cellular immunity, and effective tumor targeting. In this context, we argue that rAd holds immense potential for enhancing cellular immunity and synergistically improving antitumor effects in combination with novel cancer immunotherapies.
Similar content being viewed by others
Introduction
Recombinant adenovirus (rAd) represents an attractive candidate for virus vaccination and cancer immunotherapy. For viral vaccination, rAd can act as a vehicle to deliver specific viral antigens into host cells, prompting the immune system to recognize and build defenses against future infections. For example, rAd-based vaccine for COVID-19, which utilizes the SARS-CoV-2 spike protein as an antigen, has demonstrated specific antibody and T-cell responses in clinical trials [1, 2]. In the realm of cancer immunotherapy, given the nuanced differences between cancer cells and their normal counterparts, the challenges are distinct. The antigens delivered by rAd usually encompass tumor-associated antigens (TAAs) or tumor-specific antigens (TSAs) such as cancer-testis antigen NY-ESO-1 and melanoma-associated cancer-testis antigen (MAGE-A3) (NCT04908111) [3], as well as personalized patient-specific or shared neoantigens (ClinicalTrials.gov: NCT03639714, NCT03953235) [4]. In another aspect, the conditionally replicating adenovirus (CRAd), clinically referred to as oncolytic adenovirus (OAV), is engineered to specifically target and annihilate tumor cells for destruction, preventing their growth and spread. Furthermore, OAV also plays a role in reshaping the tumor microenvironment (TME) and bolstering cellular immunity [5,6,7]. By harnessing the complete potential of OAV in tumors, different strategies have been used to leverage their entry, replication, and lysis capabilities to maximize antitumor immunity [8,9,10,11].
Clinically, there are basically two primary methods for rAd administration, intravenous and localized injection. Although intravenous administration of rAd is encumbered by numerous challenges, such as neutralizing antibodies (NAbs), cytokine storm syndrome, disseminated intravascular coagulation, thrombocytopenia, and hepatotoxicity, direct intravenous administration of a high titer of rAd was superior to other routes for transgene expression [12, 13]. The existing methods for delivering viruses, such as nanoparticles and PEG/lipids/calcium phosphate, have been demonstrated to allow a high dose of viruses to be administered intravenously and increase therapeutic efficacy without inducing toxicity [14, 15]. In other cases, studies of localized injection, especially intraperitoneal administration of OAV indicated benefits in treating peritoneal metastasis over systemic injections, given its ability to deliver high concentrations directly to the tumor [16,17,18]. As such, the primary focus in potentiating rAd immunotherapy lies in balancing efficacy with safety, leading to tailored modifications in the viral genome to boost cellular immunity.
Adoptive cell therapy (ACT) is a type of vibrant tumor immunotherapy that involves a broad spectrum of immune cells, including dendritic cells (DCs), chimeric antigen receptor macrophages (CAR-Ms), T-cell receptor-engineered T cells (TCR-T cells), chimeric antigen receptor T cells (CAR-T cells), cytokine-induced killer cells (CIKs), cytotoxic T lymphocytes (CTLs), tumor-infiltrating lymphocytes (TILs), and natural killer cells (NKs) [19]. Both rAd and retrovirus serve as prevailing vectors in ACT, with their selection hinging on distinct treatment objectives and disease profiles. Retrovirus, which integrates into the host genome, enables prolonged expression. Conversely, rAd can be applied in vivo with low toxicity, transient expression and effective infection. Significantly, working in combination with ACT, rAd can synergistically stimulate immunogenicity in the TME to increase ACT efficacy [20,21,22], or reach tumors through infused cells to cause oncolysis through a phenomenon known as “viral hitchhiking”, by loading on the cell surface [23] or infecting cells [24, 25]. In this regard, stem cells with excellent tumor-homing properties usually serve as a promising systemic delivery tool for rAd and demonstrate safety and efficacy against tumors [26,27,28,29,30].
To potentiate antitumor immunity, it is vital to elicit tumor immunogenicity, synergize with cellular therapies, modulate the TME and target tumor sites. This review focuses on the generating and strengthening of rAd immunotherapies and provides a comprehensive understanding of its anti-tumor distinctions and mechanisms. Notably, we highlight promising strategies in combination with ACT to improve immunotherapeutic efficacy, which are expected to provide promising approaches for successful cancer treatment.
Biological characteristics of adenovirus and the development of rAd vectors
Adenovirus is a nonenveloped DNA virus with a 36 kb genome that contains early genes (E1-E4, pIX and pIVa2) associated with viral replication and five late genes (L1-L5) involved in assembly. To date, 7 subgroups (subgroups A-G) and over 100 human adenovirus genotypes have been identified (http://hadvwg.gmu.edu/). The adenoviral capsid features a regular 20-sided structure comprising hexons, pentons, and fibers, as well as other small proteins such as pIX, pIIIa and pIVa2 (Fig. 1a). Human adenovirus type 5 (Ad5, subgroup C) infection primarily depends on the interaction between coxsackievirus-adenovirus receptor (CXADR) on the cell surface and fibers on the viral capsid protein. Ad5 endocytosis into target cells occurs through two steps: the binding of a fiber knob to CXADR, and the subsequent interaction between the Arg-Gly-Asp motif (RGD) located in the penton base and integrin subunits alpha V (αv), beta 3 (β3) and beta 5 (β5) (Fig. 1b) [31]. Other receptors, such as CD80/CD86, CD46, desmoglein 2 (DSG2) and sialic acid, are involved in infection by B or D subgroup adenoviruses (Fig. 1b) [32].
The genome structure and entry receptors of adenovirus. a Schematic representation of Ad5 genome and the major capsid proteins. b Cellular uptake and transduction mediators of various types of adenoviral vectors. The Ad5 primarily infects cells via both CXADR and integrin. The Ad5 can be engineered to increase cell entry by incorporating peptide into capsid and swapping fibers of other species of adenovirus. Surface receptors for cell binding of B, D subgroup and chimeric adenoviruses are displayed. d Summary of adenovirus entry receptors and fiber modifications.
rAd has been continuously modified to enhance gene capacity, infection efficiency, duration of gene transduction and safety. The first-generation rAd lacks E1 and E3 regions and thus is replication incompetent and innate-immunity attenuated. The Ad5 vector from the first generation is predominantly used in scientific research and clinic [33, 34]. To mitigate the robust immune response, the second-generation rAd sees further gene deletions and an expanded transgenic space. However, deletion of E2 and/or E4 genes can lead to a decreased viral titer [34]. The third-generation rAd only retains the inverted terminal repeats (ITR) and packaging signal sequences, which can accommodate up to 37kb of exogenous DNA with the aid of a helper-packaging virus [35, 36]. This high-capacity rAd minimizes residual gene expression, attenuates host immune, and achieves sustained in vivo transgenic expression. Its vast loading capability also allows it to carry prominent gene-editing systems like CRISPR/Cas9 and TALENs.
Construction and Modification Strategies for OAV
OAV is engineered to possess capabilities of direct oncolysis and immune induction. Briefly, they can cause immunogenic cell death (ICD) and trigger the production of proinflammatory cytokines, pathogen-associated molecular patterns (PAMPs), and damage-associated molecular patterns (DAMPs) from dying cancer cells, which can activate DCs and T-cells to destruct tumor. Improving the targeting and replication of OAV is an effective approach to enhance its oncolytic effects.
Entry and targeting modifications
Capsid peptide incorporation
The CXADR required for Ad5 entry is typically expressed at low levels in target cells, prompting researchers to explore alternative entry pathways [37,38,39]. The HI loop or COOH-terminus of the fiber knob, and the hypervariable region of the hexon and shaft are promising candidates for displaying foreign peptide sequences [40, 41]. The RGD motif and its extended versions, including the internalized RGD (iRGD, CRGDKGPDC), RGD4C (CDCRGDCFC) and RGDK, have been used extensively to favor adenovirus entry via the integrin-mediated pathway (Fig. 1b) [42,43,44]. Some cell-permeable peptides, such as the polylysine motif (pK7) and a stretch of 20 lysine residues (F/K20) incorporated into the fiber have also shown an improved transduction efficiency (Fig. 1b) [45,46,47,48].
Chimeric rAd engineering
Chimeric rAd can alter viral tropism and is a potent instrument for devising immune cell-based vaccination approaches. Table 1 lists the chimeric rAd in preclinical and clinical trials. The directed-evolution screened Enadenotucirev (formerly ColoAd1) comprises the Ad11 capsid structure from subgroup B and a chimeric E2B from Ad11/Ad3, which allows it to avoid the Ad5 NAbs and display robust tumor cell eradication via intravenous administration [49,50,51,52,53,54,55]. In the case of OAV ONCOS-102, integrates the Ad3 fiber knob domain into the Ad5 structure (Ad5F3). Therefore, Ad5F3 derives superior tropism via the adequately expressed Ad3 receptor (Fig. 1b) [9, 56, 57]. Derived from Ad5, Ad5F35 replaces its receptor binding site with the fiber of Ad35 and strongly increases entry into immune cells by shifting its cellular receptor from CXADR to the ubiquitously expressed complement receptor CD46 (Fig. 1b). Lokon oncolytic adenovirus (LOAd) is Ad5F35 and shows promise in cancer intervention [6, 58,59,60,61,62].
Tumor-targeted OAV modification
Another strategy for engineering tumor-targeted OAV involves incorporating a single-chain T-cell receptor (scTCR) specific for TAA into adenoviral fibers. This approach allowed CXADR-independent viral entry and selectively infected cells that presented the corresponding TAA within the context of human leukocyte antigen (HLA)-A and effectively eradicated tumors [65].
Conditional replication modifications
Adenovirus E1 is composed of E1A and E1B and initiates viral replication through interactions with cellular proteins. OAV that can conditionally activate E1 genes for replication within tumor cells is designed to selectively proliferate within and destroy tumor cells. There are generally four strategies for OAV to achieve conditional replication: a) a 24 bp constant deletion in the E1A conserved region 2 (E1A∆24), b) a conserved region 3 (CR3) deletion of the transactivator protein E1A 13S, c) an E1B55K deletion or E1B-93R mutation, d) specific promoter regulation of E1A (Fig. 2).
E1A gene modification
E1A proteins are among the first to be expressed after adenoviral infection for initiation of the viral life cycle, cell cycle modulation, and transactivation of viral and cellular genes. Normally, adenoviruses can infect and propagate in dormant cells in which the E1A CR2 interacts with the retinoblastoma protein (Rb), releasing the E2F transcription factor and advancing the cell from G1 to S-phase [66,67,68]. The strategic E1A∆24 hampers its ability to engage with functional Rb, rendering the adenovirus incapable of replicating in normal cells. However, tumor cells, characterized by excessive E2F due to the dysregulation of antioncogene Rb, support Ad5-∆24 replication [69, 70]. Both intratumoral and intravenous administration of Ad5-∆24 proved to be relatively safe and somewhat effective when injected into solid tumors [71, 72]. Table 2 summarizes the canonical OAV with E1A∆24.
OAV with CR3 deletion in E1A 13S exhibited high replication potential in Y-box binding protein 1 (YB-1)-positive cancer cells but replication defects in normal cells. Its utility is exemplified by XVir-N-31 and its derivatives for glioma and bladder cancer (Table 3) [79,80,81,82,83,84,85]. Mechanistically, YB-1 is a multifunctional protein involved in a multitude of cellular processes, including transcriptional regulation, mRNA splicing, and translation. The involvement of YB-1 in various stages of mRNA metabolism makes it an attractive collaborator for viruses, which often hijack the host cellular machinery for replication and propagation [86, 87].
E1B gene modification
The adenovirus E1B locus encodes E1B19K and E1B55K and plays a vital role in the inhibition of apoptosis, inactivation of antioncogene p53 and modulation of viral mRNA export. The viral proteins E1B55K and E4orf6 form an E3 ubiquitin ligase complex that leads to the degradation of p53, which protects infected cells from apoptosis and allows the virus to replicate effectively in normal cells. OAV lacks the ability to bind and inactivate p53 and thus can supposedly replicate efficiently only in neoplastic cells defective in p53 function [100, 101]. ONYX-015 (dl1520), a chimeric adenovirus derived from Ad2/Ad5 with a deleted E1B55K gene, earned the distinction of being the first genetically engineered therapeutic to undergo human trials (Table 3), later gaining commercial approval in China as H101 [13, 102,103,104]. A more advanced version, ZD55-IL-24, which contains the interleukin (IL-24) and retains a functional E3 region, has shown the ability to effectively combat melanoma by converting tumor cells from the nonself state as well as through the classic direct killing pathway [95].
Despite the promise of the null E1B55K OAV, clinical evaluations suggest that the selectivity mechanism is multifaceted and not solely dependent on p53 status [102, 105,106,107]. By introducing specific point mutations at the 3' splicing acceptor site of E1B-93R, the novel OAV Ixovex-1 produces the E1B-156R splice isoform and includes a functional E3B region for superior competency compared to OAVs with multiple deletions [108,109,110]. In models of lung carcinoma, Ixovex-1 significantly enhanced oncolytic efficacy, hindered tumor growth and improved mouse survival [109]. The OAVs associated with E1B modification are listed in Table 3.
Tumor-specific promoter driven E1A
To achieve specific replication within tumors, OAV typically uses tumor-specific promoters to regulate E1A gene expression. These promoters are usually inactive in normal cells but are activated in tumor cells, enabling the OAV to replicate only within tumor cells without causing damage to normal cells. The principle behind tumor-specific promoters is the exploitation of the differences in gene expression levels between tumor cells and normal cells to achieve the specific replication of OAV. The replication of OAV controlled by tumor-specific promoter is listed in Table 4. The selective E1A gene and granulocyte macrophage colony-stimulating factor (GM-CSF) encoding CG0070 (Ad5-E2F-E1A-GM-CSF) can replicate and ultimately lyse tumor cells in Rb-deficient tumor cells while releasing tumor antigens and GM-CSF, triggering a systemic antitumor immune response. It has been approved by the FDA for the treatment of nonmuscle invasive bladder cancer (NMIBC). Prior reports of intravesical CG0070 yielded an overall 74.4% complete response (CR) that was maintained for more than 6 months [111, 112]. The Ad3-hTERT-CMV-CD40L leverages human telomerase reverse transcriptase (hTERT) for selective replication, incorporates CD40L as an immune stimulant and avoids the ubiquitous Ad5 NAbs when administered intravenously [113, 114]. Administered intratumorally, OBP-301 (Ad5-hTERT-E1A-IRES-E1B) has displayed cytopathic effects on solid tumors expressing the CXADR-receptor in an hTERT-dependent manner [115, 116]. Given that hypoxia upregulates hTERT activity, compared with Ad5, OBP-301 potentially has enhanced antitumor effects, especially within hypoxic environments [117]. Furthermore, fiber-modified OBP-301 (termed OBP-405) was used to confirm its antitumor effect on non-CXADR-expressing OBP-301-resistant tumors [118].
Combination strategies for clinical OAV candidates
Combination strategies may enable the development of efficacious therapies better suited to some specific cancer. Table 1 shows the chimeric OAVs used in clinical practice. For instance, the response to ONCOS-102 (Ad5/3-Δ24-GM-CSF) is associated with increased lymphocyte infiltration and the expression of cytotoxic and costimulatory genes. A phase II study of intratumoral ONCOS-102 revealed substantial immune activation associated with 18-month survival in mesothelioma patients versus chemotherapy alone [64].
Table 2 lists the commercialized OAVs used in clinical trials, each featuring E1A∆24. DNX-2401 (Ad5-Δ24-RGD4C) is modified with E1A∆24 and endowed with the RGD4C motif to its fiber to facilitate entry independent of the CXADR receptor [131]. Intratumoral or intramural infusion of DNX-2401 for glioma intervention has been shown to change T-cell activity and reduce tumor size [74,75,76]. In a phase II trial of DNX-2401 virotherapy plus pembrolizumab, the overall survival rate was 52.7%, which was significantly greater than the prespecified control rate of 20%, albeit with no statistically significant difference in the objective response rate (ORR) [76]. An enhanced version, DNX-2440, equipped with the costimulatory molecule OX40 ligand (OX40L), can induce tumor-specific immune memory and distant effects [132, 133]. This variant has been or is being investigated in refractory cancers.
As a new favorite, BioTTT001 (Ad5-TD-nsIL-12), which carries triple gene deletions (E1A∆24, ∆E1B19K, and ∆E3gp19K) and nonsecreting (ns) IL-12, can induce tumor cell apoptosis and significantly enhance T-cell infiltration in the TME to inhibit tumor progression [134]. Intratumoral BioTTT001 is also being clinically evaluated against glioma (Table 3). Intraperitoneal delivery of BioTTT001 has been reported to cure peritoneally disseminated pancreatic cancer [98]. The products of this OAV category with E1A 13S or E1B modification are summarized in Table 3.
The OAV whose replication is further controlled by specific cellular promoters is listed in Table 4. A single intravenous administration of ICOVIR-5 (Ad5-E2F-Δ24-RGD4C) can reach tumor sites upon administration but failed to induce tumor regression in a phase I trial for melanoma patients [119]. Similarly, VCN-01 (Ad5-E2F-Δ24-RGDK-PH20) is designed to replicate selectively in tumor cells with a dysfunctional Rb pathway and produces the hyaluronidase PH20 enzyme. This enzyme degrades hyaluronic acid in the tumor extracellular matrix, facilitating drug penetration and immune cell infiltration [123, 135]. In fibrotic tumors such as pancreatic cancer, VCN-01 degrades the tumor stroma and remodels the TME, representing a new therapeutic agent for cancers with dense stroma [44, 123]. Clinical trials have shown that its combination with chemotherapy facilitates the delivery of a variety of therapeutic agents and has a 50% ORR in pancreatic cancer patients [136]. VCN-11, an evolved version of VCN-01, not only expresses the hyaluronidase PH20 but also features an albumin-binding domain (ABD) on hexon, allowing the virus to evade NAbs in the bloodstream. Preliminary studies have shown minimal side effects and enhanced tumor targeting via intravenous route [124].
rAd-based immunostimulatory therapy
Enhancing immunotherapy with rAd-induced immunostimulation
Immunostimulatory genes play a pivotal role in orchestrating immune responses against cancer. The strategy of using rAd to transduce immunostimulatory genes such as TAAs, costimulatory molecules [6, 137, 138], and cytokines such as (C-C motif) ligand 5 (CCL5), IL, interferon (IFN), GM-CSF, and tumor necrosis factor (TNF) [12, 139,140,141,142,143,144] has shown significant promise in enhancing tumor immunogenicity and 'firing up' the TME (Fig. 3). Additionally, immune checkpoint inhibitors (ICIs) against SOCS1, CTLA4, PD1, TIGIT, etc., have been incorporated into rAd for cancer immunotherapy [145, 146].
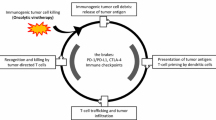
Application framework of rAd in cancer immunotherapy. a Direct oncolysis b rAd-based immune activation: DC vaccine mediated CTL priming and rAd synergy with cellular therapies c Reshaping tumor dynamics and 'firing up' the TME d Cell delivery of rAd to tumor sites. The evoked immune cells activate TME and induce the infiltration of more immune cells, thereby enhancing local and systemic anti-tumor immunity
rAd is increasingly recognized as an effective virotherapy in clinical settings, offering both systemic and local therapeutic approaches (Table 5) [12, 147, 148]. For instance, in a phase I clinical study, subcutaneous administration of the multitargeted vaccine Ad5-PSA/MUC1/Brachyury or Ad5-CEA/MUC1/Brachyury induced immunogenicity and was well-tolerated, with a median progression-free survival of 22 weeks [149, 150]. All patients mounted T-cell responses to at least one TAA, whereas 47% of patients mounted immune responses to all three TAAs [149]. In a phase III clinical trial, intravesical injection of Ad5-IFNα/Syn3 expressing IFNα and Syn3 (a polyamide surfactant used to enhance viral transduction) demonstrated efficacy in treating NMIBC. Fifty-five (53.4%) of the 103 patients achieved a complete response within 3 months, and this response was maintained in 25 (45.5%) of the 55 patients at 12 months [151].
rAd-activated DCs as vaccines
DCs orchestrate adaptive immunity by taking up and presenting antigens to T cells. Due to the potent ability of DCs to prime naive T cells, there has been longstanding interest in DC vaccines, particularly when activated by rAd infection (Fig. 3b). Notably, rAd not only efficiently promotes DC maturation and robust immunogenicity but also has a natural affinity for DCs in vitro [163, 164]. Despite lacking viral CXADR, DCs can internalize Ad5 through phagocytosis [165]. However, direct intravenous administration of rAd reduces the endocytic and cross-presentation capabilities of DCs, compromising the priming of CTLs [166].
rAd-mediated tumor antigen expression in DCs
Protective immunity can be generated with TAA-engineered DC vaccines [167, 168]. Unlike peptide-pulsed DCs, rAd-transduced DCs induce DC differentiation and maturation, expanding the range of HLA applications. rAd-induced DC vaccines can process entire TAAs into peptide pools, enabling recognition by the corresponding T-cell receptors (TCRs) and the generation of specific CTLs [169]. Furthermore, the CTL priming with viral or virally encoded epitopes gives rise to enhanced proliferation, infiltration, and distinct memory phenotypes. Preclinical studies have demonstrated the superior efficacy of DC vaccines containing rAd-encoded tyrosinase-related protein 2 (TRP2) compared to direct rAd-TRP2 injections, thus highlighting the robust immunogenicity of rAd-induced DCs [170].
rAd-induced costimulatory molecules and cytokines in DCs
To overcome immune tolerance, rAd has been engineered to deliver immunoregulatory genes and cytokines into DCs. This strategy enhances Th1 and CTL responses, which are crucial for effective antitumor immunity [171,172,173,174,175]. For instance, rAd-IL-18 transgene-engineered DC vaccines have shown potential due to the unique ability of IL-18 to induce tumor-specific CTLs [176, 177]. Similarly, compared with their externally cultured counterparts, rAd-IL-6-engineered DC vaccines enhance specific CTL responses and counter immunosuppression [178], while rAd-TNFα-transfected DCs exhibit greater maturation and T-cell activation [179]. The use of the rAd-CD137L vaccine promoted DC-induced priming of tumor-specific CD8+ T cells [7]. Additionally, Ad5F35-CD40L-IL-2 vaccines contribute to DC maturation and IL-12 production by targeting breast cancer cells overexpressing CD40 [180]. However, immature DCs transduced with rAd-CD40L can differentiate into tolerogenic DCs [181]. The complex interplay between costimulatory molecules and cytokines in DC maturation and CTL functionality has yet to be elucidated.
By collecting the lysate of tumor cells infected with replication-competent Ad3-hTERT-CMV-CD40L and culturing them with DCs, DC maturation and the production of proinflammatory cytokines can be induced, thereby augmenting the effectiveness of DC vaccination [114, 182]. Additionally, a nanovaccine derived from rAd-infected mature DCs in which specific MHC-I, anti-PD1 antibody and B7 costimulatory molecules are simultaneously anchored can self-present neoantigens to T cells and stimulate strong CTL responses in this manner [183].
Clinical trials of rAd-based DC vaccines
INGN-225 is an rAd-mediated p53-expressing DC vaccine (DC-rAd.p53). In phase II clinical trials of recurrent small cell lung cancer (Table 5), INGN-225 was demonstrated to induce significant immune responses and improve the efficacy of chemotherapy but failed to improve the ORR [155, 156, 184]. Moreover, in addition to flagellin, rAd delivers survivin and MUC1 TAAs to promote DC maturation, and a shRNA is used to suppress SOCS1, an intracellular immune checkpoint molecule (rAd-siSSF). Via inguinal injection, a phase I trial of rAd-siSSF demonstrated its safety and efficacy, with a complete remission rate of 83% in relapsed acute myeloid leukemia (AML) patients [145].
rAd as immunostimulant in cellular therapies
While rAd can effectively deliver therapeutic payloads to tumors, transient expression of these viruses necessitates repeated high-titer injections to maintain effective local concentrations [185, 186]. This strategy, combined with other therapies, is anticipated to significantly boost antitumor immunity (Fig. 3c) [142, 187, 188].
CAR-T cell and rAd synergy
Intratumorally administering rAd combined with CAR-T cells can overcome the challenges of the immunosuppressive TME and promote CAR-T cell infiltration and proliferation in vivo to eradicate local and distal tumors. Cytokine-armed OAV expressing TNFα and IL-2 (Ad5/3-E2F-Δ24-TNFa-IRES-IL-2), in conjunction with mesothelin-targeted CAR-T cells, foster T-cell infiltration and promote M1 macrophage polarization and DC maturation [189]. Moreover, the replication competent Ad5-ZD55-CCL5-IL-12, which encodes the chemokines CCL5 and IL-12, significantly increases CAR-T-cell infiltration in tumors, extending survival and restraining tumor growth. Since IL-12 enhances the phosphorylation of signal transducer and activator of transcription 4 (STAT4) in CAR-T cells and stimulates IFNγ release [22], local treatment with rAd-IL-12-PD-L1, which encodes a PD-L1-blocking antibody, and IL-12p70 was found to control both primary and metastasized head and neck squamous cell carcinoma in conjunction with HER2-specific CAR-T cells [190].
Other immunocyte therapies augmented by rAd
The rAd has documented to augment the antitumor effects of CIKs for cancer immunotherapy [191, 192]. The novel hydrogel-mediated codelivery of replication-competent rAd-IL-12-IL-15 and CIK cells could enhance the combined antitumor effects. This strategy involving an injectable and biodegradable hydrogel minimizes the dispersion of high-dose OAV and CIK cells, thus reducing nontumor exposure [193]. Another example is the intratumoral injection of ICOVIR-15K-BiTE, which expresses a bispecific T-cell adapter (BiTE) simultaneously with T-cell infusion; this method can bidirectionally recognize neoplastic cells and T cells and induce T-cell activation and tumor cell destruction [121].
rAd in tumor cell vaccination
The use of autologous unirradiated tumor cells transduced with rAd-IL-12 has shown promise in treating advanced neuroblastoma. This approach induces both a local inflammatory response and a systemic immune response, characterized by an increase in the number of circulating CD25+ and DR+CD3+ T cells and specific antitumor responses [194]. Likewise, cryoshocked tumor cells, which constitute an OAV reservoir, can eliminate viral proliferation and pathogenicity, steadily release viruses and efficiently initiate an endogenous antitumor response by increasing memory T cells and modulating the TME [195, 196]. More recently, extracellular vesicles derived from OAV-infected tumor cells have been used for delivering immunogenic OAV, inducing systemic immune responses through proinflammatory cytokines, and inhibiting primary and metastatic cancers [77, 78].
rAd reshaping tumor dynamics and the TME
rAd delivery of RNA interference (RNAi) against oncogenes
rAd has been demonstrated to enhance anti-tumor effects by expressing RNAi, targeting oncogenes or immunosuppressive genes such as PD1 and K-RAS (Fig. 3c). Notably, STAT3 is considered as a bona fide oncogene and mediates immunosuppressive functions in various immune cells including macrophages, myeloid-derived suppressor cells, and DCs [197, 198]. Given the challenges in inhibiting STAT3 through antibodies or small molecule inhibitors, siRNA serves as an ideal alternative for STAT3 inhibition. Depleting STAT3 in DCs improves their antigen-presenting activity and enhances antitumor immune responses [199]. Additionally, inhibiting STAT3 in cancer cells promotes ICD and increases IFN-responsive chemokines, facilitating immune cell infiltration [200].
rAd-rescued tumor suppressor gene expression
Tumor suppressor genes, which are crucial for inhibiting cell proliferation and tumorigenesis, often undergo mutation or inactivation in malignant tissues. rAd can target gene defects in key tumor suppressor genes, such as p53, p16/21/27, Rb, and PTEN [201,202,203]. For example, restoration of p53 in p53-deficient tumor cells has been shown to suppress tumor growth or induce apoptosis in both in vitro and in vivo models. Ad5CMV-p53, an oral infusion or intramucosal injection, has the potential to prevent oral or pharyngeal precancerous lesions, with an estimated 1-year progression-free survival rate of 92% (Table 5). In the case of the phase II trial of Ad5CMV-p53 combined with chemotherapy, the estimated 3-year survival rate was 84% [154].
rAd-mediated "suicide gene" for toxic molecule delivery
rAd can also target tumor cells by expressing enzymes that convert prodrugs into toxic compounds (Table 5). For instance, purine nucleoside phosphorylase (PNP) converts fludarabine phosphate (F-araAMP) into cytotoxic fluoroadenine. The first-in-human clinical trial found that rAd-PNP combined with intravenous F-araAMP shows potential in treating advanced glioma [204]. With herpes simplex virus thymidine kinase (HSV-TK) transforming ganciclovir into nucleotides toxic to dividing cells, rAd-TK plus ganciclovir therapy has been employed in numerous clinical trials for solid tumor treatment [158]. Additionally, cytosine deaminase (CD) converts the prodrug 5-fluorocytosine (5-FC) into toxic 5-fluorouridine (5-FU), which is metabolized into 5-fluorouracil triphosphate (5-FUTP) and 5-fluorodeoxyuridine monophosphate (5-FdUMP), causing thymidylate synthesis blockade and DNA damage. Concomitant with prodrug therapy of 5-FC and ganciclovir, clinical trials showed that replication-competent Ad5-CD/TKrep carrying CD and TK chimeric genes exerted long-term effectiveness [161, 162].
rAd in reshaping the TME
To inhibit tumor angiogenesis, vasohibin has been identified as an intrinsic and specific angiogenesis inhibitor. The therapeutic potential of the rAd-vasohibin, which encodes vasohibin, has been explored. When administered via tail vein injection, rAd-vasohibin prevented tumor cell growth in a subcutaneous tumor model by inhibiting angiogenesis without apparent side effects [205]. Additionally, targeting cancer-associated fibroblasts (CAFs) in the immunosuppressive TME is crucial. Fibroblast activation protein (FAP), a cell surface serine protease highly expressed on CAFs, was targeted by rAd-FAP. As a result, rAd-FAP enables in vivo gene delivery to stromal cells in the TME, resulting in attenuated tumor growth [206].
Synergistic interaction between rAd and cellular therapeutics for targeting tumor sites
The ability of rAd to synergistically interact with various cell types, particularly immune cells, creates a potent combination for targeting tumor sites and potentiates cellular therapies in oncology (Fig. 3d). The advantage of this approach lies in its dual functionality: while rAd can enhance the tumor-targeting ability and therapeutic payload delivery of cells, as mentioned before, the cells themselves offer a biologically compatible and dynamic method for rAd transport. This is particularly relevant in residual and metastatic tumors, where the physical barriers and immunosuppressive nature of the TME pose significant challenges for conventional therapies.
rAd hitchhiking on/in immune cells
For oncolytic virus (OV) delivery, CAR-T and TCR-T cells have been proven to be effective vehicle cells, especially for oncolytic vesicular stomatitis virus (VSV), myxoma virus (MYXV) and reovirus [23, 25]. In previous studies, OV-loaded CAR-T cells were shown to have enhanced activity and efficacy against solid tumor models and to augment expansion rate in vivo via homologous boosting [25]. OVs loaded in/on cells can be directly delivered into solid tumors in a CAR/TCR-directed fashion, avoiding recognition by the host innate defense system [23]. Analogously, Epstein-Barr virus (EBV)-specific CTLs provide an innovative approach for delivering therapeutic Ad5F35 to tumor sites, not only to locally accessible macroscopic tumors, but also to disseminated metastatic disease [24]. These CTLs, which are transgenic for the adenoviral E1 gene under the CD40L promoter, produce and release infectious Ad5F35 upon exposure to HLA-matched EBV-expressing targets but not in response to HLA-mismatched or irrelevant cells, which can avoid the risks associated with systemic administration of large doses of rAd.
Replication-competent KGHV500, carrying anti-p21Ras single-chain variable fragment antibody (scFv), has shown potential for blocking the Ras signaling pathway and inhibiting Ras-driven cancers. In several Ras-driven cancers, the CIK cell-based delivery of KGHV500 has been validated through both in vitro and in vivo studies, which confirmed the tumor-targeting efficacy and systemic safety of OAV-loaded CIK cells [129, 207,208,209]. Likewise, the oncolytic agent ZD55 introduces the CD40L promoter to regulate replication, ensuring that cell proliferation is strictly controlled by CIK cell activation. This targeted delivery by CIK cells enhances antitumor efficacy and precision in tumor targeting and minimizes infection in nontumor tissues [210].
With their innate tumor-homing ability, NK cells function as bioreactors that support OAV loading, protection, replication, amplification, and targeted release. Arming NK cells with OAV not only boosts antitumor immunity through IFN signaling but also alleviates immunosuppression in the TME, promoting DC maturation and M1 macrophage polarization. Both in vitro and in vivo data highlight the potent antitumor and antimetastatic functions of this NK cell-mediated OAV delivery system [211].
Use of stem cell as carriers of rAd
The abilities to home to tumors, shield rAd from host antiviral responses, and infiltrate tumor tissues through the TME make stem cells ideal candidates as delivery vehicles for rAd in cancer therapy [28, 212,213,214,215]. As listed in Table 6, mesenchymal stem cells (MSCs) transduced with the IFNβ expressing nonreplicating rAd (MSC-rAd.IFNβ) have shown promise in suppressing pulmonary metastasis through IFNβ production within the TME [216]. MSCs efficiently delivered rAd expressing IL-12 (MSC-rAd.IL-12) in glioma but does not completely arrest the invasive growth pattern of these lesions [217]. However, MSCs transduced with rAd carrying the secretable trimeric form of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) (MSC-rAd.stTRAIL) significantly inhibited tumor growth and prolonged survival in glioma-bearing mice [48, 218].
In terms of replication-competent rAd, the intraperitoneal administration of MSC-preloaded OAVΔ24RGD (MSC-Δ24RGD) can efficiently target ovarian tumor cells and reduce the systemic toxicity of naked virions in mice [220]. In a glioma xenograft murine model, MSC-guided delivery of MSC-Δ24RGD could migrate to tumors and exert antitumor effects via intravenous injection [213]. Furthermore, the endovascular selective intra-arterial (ESIA) infusion of MSC-Δ24RGD is a rapidly evolving strategy for treating glioma in a clinically relevant fashion [229]. In seeking to increase the viral infection and production by MSCs, a bunch of OAV chimerisms have been designed [223]. Engineered Ad5F3 expressing the TRAIL or FCU1 would enhance oncolysis by improved virus production in MSCs, thereby implementing delivery into established and primary pancreatic cancer cells [222]. In a xenograft model of glioma, systemic administration of MCSs loaded with Ad5F35 carrying IL-24 and/or endostatin and regulated by a Tet-on system (Ad5F35-Tet-on-E1B-Pro-Δ24-IL-24/endostatin) showed promise for glioma treatment while sparing normal cells [221].
In the context of hepatocellular carcinoma (HCC), MSCs loaded with OAV have demonstrated the ability to home to HCC cells and differentiate into hepatocyte-like cells within the TME [224]. They effectively package and release progeny virions that contain an adenovirus E1A gene regulated by the α-fetoprotein (AFP) promoter and microRNA-122 target sequence (AdAFPp-E1A-miR122), resulting in dramatic tumor inhibition in mouse models. Another oncolytic Ad5F35, which can replicate under the control of the AFP-positive HCC-specific Ha2bm promoter and express the WNT inhibitory (WNTi) bait receptor, was transfected into MSCs, which were then intravenously injected into HCC-bearing mice. Compared with WNTi without a cell carrier, the therapeutic effect was much more satisfactory [26]. Newer versions of OAV armed with BiTE have shown enhanced antitumor effects, reduced liver injury, and improved T-cell infiltration and activation in orthotopic transplantation model mice [225]. In conclusion, the use of rAd-mediated hitchhiking of MSCs has demonstrated safety in preclinical studies and a pleiotropic profile of tumor destruction.
Clinical trials of rAd hitchhiking
A pioneering trial of children with relapsed or refractory neuroblastoma used autologous MSCs carrying ICOVIR-5 (Ad5-E2F-Δ24-RGD4C; Table 6). The trial reported disease stabilization or remission in patients, with no toxicity or progressive disease observed [228]. This groundbreaking study paved the way for further exploration of the use of ICOVIR-5-loaded MSCs in treating late-stage solid tumors in both adults and children. Two additional ongoing trials (NCT01864759 and NCT04758533) are utilizing ICOVIR-5 in conjunction with allogeneic MSCs, to treat advanced or metastatic melanoma and gliomas. However, as of the last update, the results from these trials have not been reported. Concordantly, a separate study involving MSCs loaded with hyaluronidase-expressing ICOVIR-17 demonstrated a significant reduction in glioblastoma growth and increased survival in a clinically relevant murine model [227]. This approach is being further investigated in a phase I clinical trial (Table 6), where autologous MSCs loaded with DNX-2401 are evaluated for their therapeutic effects on glioblastoma with the support of endovascular super-selective intra-arterial infusion [226].
The commercial OAV loaded in neural stem cells has been underway [132, 230]. The safety and efficacy of the neural stem cell delivered CRAd-Survivin-pK7 (NSC-CRAd-Survivin-pK7) under the survivin promoter were evaluated in the first human clinical trial of malignant glioma, in which the median progression-free survival was 9.1 months and the median overall survival was 18.4 months (Table 6) [30].
Conclusions and future perspectives
In this review, we present an integral toolkit for rAd application according to four different antitumor mechanisms (Fig 3), i.e., a) direct oncolysis, b) immune activation: DCs priming CTL; synergies with cellular therapies, c) reshaping tumor dynamics and 'firing up' the TME, d) tumor targeting by carrier cells. With their natural tropism for epithelial cells, adenoviruses are inherently suited for targeting the majority of solid tumors. The strategic utilization of rAd in cancer therapy, particularly through replication-competent OAV, has shown significant promise. Despite challenges such as immune reactions against the virus, ongoing clinical evaluations of various rAd-equipped ACTs and combined cancer immunotherapy strategies are yielding encouraging results in terms of both safety and efficacy. In addition, the integration of immune cells and/or stem cells with rAd is pivotal in understanding cell-type-specific oncolysis within the TME and controlling tumor growth through diverse mechanisms. The development of effective rAd delivery vehicles is crucial, with criteria including high cellular infection rates, effective gene expression or replication, and the capability of carrier cells to target tumors.
Of particular interest is the role of rAd in CAR-M therapy. Macrophages, critical players in tumor progression, can be genetically modified ex vivo for adoptive transfer [231]. CAR-M, representing a groundbreaking approach in immunotherapy, exhibits functions like targeted phagocytosis, induction of a proinflammatory M1 phenotype, antigen presentation, and epitope spreading. Early studies have shown the effectiveness of CAR-M against blood tumors and solid tumors, like ovarian cancer, highlighting its potential to reshape TME [232, 233]. However, the application of CAR-M faces challenges. Macrophages' inherent defense mechanisms against viral infections, such as the production of antiviral IFNs and undergoing apoptosis, make them less susceptible to common viral vectors used in gene delivery. This challenge is addressed by the discovery of Ad5F35 as an efficient gene carrier for macrophages, leveraging their abundant CD46 expression. Ad5F35-infected macrophages not only demonstrate antigen-specific phagocytosis but also maintain a sustained proinflammatory M1 phenotype and present tumor antigens to T cells [234,235,236]. Importantly, CAR-Ms, especially those transduced with Ad5F35, have shown potential in priming T cells against neoantigens, reducing antigen escape and recurrence.
In summary, engineering rAd holds immense potential for enhancing cellular immunity and synergistically improving anti-tumor effects alongside other novel cancer immunotherapies. As research progresses, the unique advantages of rAd in targeting and reshaping the TME combined with innovative approaches will become increasingly significant in anti-tumor immunity.
Availability of data and materials
No datasets were generated or analysed during the current study.
Abbreviations
- 5-FC:
-
5-fluorocytosine
- 5-FdUMP:
-
5-fluorodeoxyuridine monophosphate
- 5-FUTP:
-
5-fluorouracil triphosphate
- 5-FU:
-
5-fluorouridine
- E1A∆24:
-
A 24bp constant deletion in the E1A CR2
- ∆24:
-
A 24bp constant region deletion
- scTCR:
-
A single-chain T-cell receptor
- F/K20:
-
A stretch of 20 lysine residues
- AML:
-
Acute myeloid leukemia
- Ad5:
-
Adenovirus type 5
- ACT:
-
Adoptive cell therapy
- ABD:
-
Albumin-binding domain
- αv:
-
Alpha V
- RGD:
-
Arg-Gly-Asp motif
- β3:
-
Beta 3
- β5:
-
Beta 5
- BiTE:
-
Bispecific T cell engager
- CAFs:
-
Cancer-associated fibroblasts
- CCL5:
-
Chemokine (C-C motif) ligand 5
- CAR-M:
-
Chimeric antigen receptor macrophages
- CAR-T:
-
Chimeric antigen receptor T cells
- CR:
-
Complete response
- CRAd:
-
Conditionally replicating adenovirus
- CR:
-
Conserved region
- CXADR:
-
Coxsackievirus-adenovirus receptor
- CIK:
-
Cytokine-induced killer cells
- CD:
-
Cytosine deaminase
- CTL:
-
Cytotoxic T lymphocytes
- DAMPs:
-
Damage-associated molecular patterns
- DC:
-
Dendritic cells
- DSG2:
-
Desmoglein 2
- E1-E4:
-
Early genes
- ESIA:
-
Endovascular selective intra-arterial
- EBV:
-
Epstein-Barr virus
- FAP:
-
Fibroblast activation protein
- F-araAMP:
-
Fludarabine phosphate
- GM-CSF:
-
Granulocyte macrophage colony-stimulating factor
- HSV-TK:
-
Herpes simplex virus thymidine kinase
- HLA:
-
Human leukocyte antigen
- hTERT:
-
Human telomerase reverse transcriptase
- ICI:
-
Immune checkpoint inhibitors
- ICD:
-
Immunogenic cell death
- IFN:
-
Interferon
- IL-24:
-
Interleukin
- iRGD:
-
Internalizing RGD
- ITR:
-
Inverted terminal repeats
- L1-L5:
-
Late genes
- LOAd:
-
Lokon oncolytic adenovirus
- MAGE:
-
Melanoma-associated cancer-testis antigen
- MSC:
-
Mesenchymal stem cell
- MYXV:
-
Myxoma virus
- NK:
-
Natural killer cells
- NAbs:
-
Neutralizing antibodies
- NMIBC:
-
Nonmuscle invasive bladder cancer
- ns:
-
Non-secreting
- ORR:
-
Objective response rate
- OAV:
-
Oncolytic adenovirus
- OVs:
-
Oncolytic viruses
- OX40L:
-
OX40 ligand
- PAMPs:
-
Pathogen-associated molecular patterns
- pK7:
-
Polylysine motif
- PNP:
-
Purine nucleoside phosphorylase
- rAd:
-
Recombinant adenovirus
- Rb:
-
Retinoblastoma protein
- RNAi:
-
RNA interference
- STAT4:
-
Signal transducer and activator of transcription 4
- scFv:
-
Single-chain variable fragment antibody
- TCR-T:
-
T cell receptor-engineered T cells
- TCRs:
-
T cell receptors
- TME:
-
Tumor microenvironment
- TNF:
-
Tumor necrosis factor
- TRAIL:
-
Tumor necrosis factor-related apoptosis-inducing ligand
- TAAs:
-
Tumor-associated antigens
- TIL:
-
Tumor-infiltrating lymphocytes
- TSAs:
-
Tumor-specific antigens
- TRP2:
-
Tyrosinase-related protein 2
- VSV:
-
Vesicular stomatitis virus
- WNTi:
-
WNT inhibitory
- AFP:
-
Α-fetoprotein
References
Zhu FC, Guan XH, Li YH, Huang JY, Jiang T, Hou LH, et al. Immunogenicity and safety of a recombinant adenovirus type-5-vectored COVID-19 vaccine in healthy adults aged 18 years or older: a randomised, double-blind, placebo-controlled, phase 2 trial. Lancet. 2020;396(10249):479–88.
Zhu FC, Li YH, Guan XH, Hou LH, Wang WJ, Li JX, et al. Safety, tolerability, and immunogenicity of a recombinant adenovirus type-5 vectored COVID-19 vaccine: a dose-escalation, open-label, non-randomised, first-in-human trial. Lancet. 2020;395(10240):1845–54.
Lin MJ, Svensson-Arvelund J, Lubitz GS, Marabelle A, Melero I, Brown BD, et al. Cancer vaccines: the next immunotherapy frontier. Nat Cancer. 2022;3(8):911–26.
Drake CG, Johnson ML, Spira AI, Manji GA, Carbone DP, Henick BS, et al. Personalized viral-based prime/boost immunotherapy targeting patient-specific or shared neoantigens: Immunogenicity, safety, and efficacy results from two ongoing phase I studies. J Clin Oncol. 2020;38(15_suppl):3137.
Liikanen I, Basnet S, Quixabeira DCA, Taipale K, Hemminki O, Oksanen M, et al. Oncolytic adenovirus decreases the proportion of TIM-3(+) subset of tumor-infiltrating CD8(+) T cells with correlation to improved survival in patients with cancer. J Immunother Cancer. 2022;10(2):e003490.
Wenthe J, Eriksson E, Hellström AC, Moreno R, Ullenhag G, Alemany R, et al. Immunostimulatory gene therapy targeting CD40, 4–1BB and IL-2R activates DCs and stimulates antigen-specific T-cell and NK-cell responses in melanoma models. J Transl Med. 2023;21(1):506.
Ding J, Jiang N, Zheng Y, Wang J, Fang L, Li H, et al. Adenovirus vaccine therapy with CD137L promotes CD8(+) DCs-mediated multifunctional CD8(+) T cell immunity and elicits potent anti-tumor activity. Pharmacol Res. 2022;175:106034.
Sallard E, Zhang W, Aydin M, Schröer K, Ehrhardt A. The Adenovirus Vector Platform: Novel Insights into Rational Vector Design and Lessons Learned from the COVID-19 Vaccine. Viruses. 2023;15(1):204.
Kuryk L, Møller AW. Chimeric oncolytic Ad5/3 virus replicates and lyses ovarian cancer cells through desmoglein-2 cell entry receptor. J Med Virol. 2020;92(8):1309–15.
Doerner J, Sallard E, Zhang W, Solanki M, Liu J, Ehrke-Schulz E, et al. Novel group C oncolytic adenoviruses carrying a miRNA inhibitor demonstrate enhanced oncolytic activity in vitro and in vivo. Mol Cancer Ther. 2022;21(3):460–70.
Lei J, Jacobus EJ, Taverner WK, Fisher KD, Hemmi S, West K, et al. Expression of human CD46 and trans-complementation by murine adenovirus 1 fails to allow productive infection by a group B oncolytic adenovirus in murine cancer cells. J Immunother Cancer. 2018;6(1):55.
Kurata H, Liu CB, Valkova J, Koch AE, Yssel H, Hirabayashi Y, et al. Recombinant adenovirus vectors for cytokine gene therapy in mice. J Allergy Clin Immunol. 1999;103(5 Pt 2):S471-84.
Zhang X, Wang Y, Lv X, Wang F, Zhou Q, Zhang F, et al. Intratumoral injection of oncolytic virus (H101) in combination with concurrent chemoradiotherapy for locally advanced cervical cancer. Int J Gynecol Cancer. 2023;33(7):1051–6.
Chen J, Gao P, Yuan S, Li R, Ni A, Chu L, et al. Oncolytic adenovirus complexes coated with lipids and calcium phosphate for cancer gene therapy. ACS Nano. 2016;10(12):11548–60.
Smith R, Wafa EI, Geary SM, Ebeid K, Alhaj-Suliman SO, Salem AK. Cationic nanoparticles enhance T cell tumor infiltration and antitumor immune responses to a melanoma vaccine. Sci Adv. 2022;8(29):eabk3150.
Koujima T, Tazawa H, Ieda T, Araki H, Fushimi T, Shoji R, et al. Oncolytic virus-mediated targeting of the ERK signaling pathway inhibits invasive propensity in human pancreatic cancer. Mol Ther Oncolytics. 2020;17:107–17.
Tanimoto T, Tazawa H, Ieda T, Nouso H, Tani M, Oyama T, et al. Elimination of MYCN-amplified neuroblastoma cells by telomerase-targeted oncolytic virus via MYCN suppression. Mol Ther Oncolytics. 2020;18:14–23.
Hattori Y, Kawakami S, Nakamura K, Yamashita F, Hashida M. Efficient gene transfer into macrophages and dendritic cells by in vivo gene delivery with mannosylated lipoplex via the intraperitoneal route. J Pharmacol Exp Ther. 2006;318(2):828–34.
Zhang P, Zhang G, Wan X. Challenges and new technologies in adoptive cell therapy. J Hematol Oncol. 2023;16(1):97.
Fang L, Yuan S, Wang M, Zhang C, Wang X, Li H, et al. Recombinant oncolytic adenovirus armed with CCL5, IL-12, and IFN-γ promotes CAR-T infiltration and proliferation in vivo to eradicate local and distal tumors. Cell Death Discov. 2023;9(1):328.
Wang G, Zhang Z, Zhong K, Wang Z, Yang N, Tang X, et al. CXCL11-armed oncolytic adenoviruses enhance CAR-T cell therapeutic efficacy and reprogram tumor microenvironment in glioblastoma. Mol Ther. 2023;31(1):134–53.
Fang L, Tian W, Zhang C, Wang X, Li W, Zhang Q, et al. Oncolytic adenovirus-mediated expression of CCL5 and IL12 facilitates CA9-targeting CAR-T therapy against renal cell carcinoma. Pharmacol Res. 2023;189:106701.
Zheng N, Fang J, Xue G, Wang Z, Li X, Zhou M, et al. Induction of tumor cell autosis by myxoma virus-infected CAR-T and TCR-T cells to overcome primary and acquired resistance. Cancer Cell. 2022;40(9):973-85.e7.
Yotnda P, Savoldo B, Charlet-Berguerand N, Rooney C, Brenner M. Targeted delivery of adenoviral vectors by cytotoxic T cells. Blood. 2004;104(8):2272–80.
Evgin L, Kottke T, Tonne J, Thompson J, Huff AL, van Vloten J, et al. Oncolytic virus-mediated expansion of dual-specific CAR T cells improves efficacy against solid tumors in mice. Sci Transl Med. 2022;14(640):eabn2231.
Yoon AR, Hong J, Li Y, Shin HC, Lee H, Kim HS, et al. Mesenchymal stem cell-mediated delivery of an oncolytic adenovirus enhances antitumor efficacy in hepatocellular carcinoma. Cancer Res. 2019;79(17):4503–14.
He X, Yao W, Zhu JD, Jin X, Liu XY, Zhang KJ, et al. Potent antitumor efficacy of human dental pulp stem cells armed with YSCH-01 oncolytic adenovirus. J Transl Med. 2023;21(1):688.
Wang P, Zhang J, Zhang Q, Liu F. Mesenchymal stem cells loaded with Ad5-Ki67/IL-15 enhance oncolytic adenovirotherapy in experimental glioblastoma. Biomed pharmacother = Biomedecine & pharmacotherapie. 2023;157:114035.
Fueyo J, Gomez-Manzano C, Lang FF, Alonso MM. Hitchhiking to brain tumours: stem cell delivery of oncolytic viruses. Lancet Oncol. 2021;22(8):1049–51.
Fares J, Ahmed AU, Ulasov IV, Sonabend AM, Miska J, Lee-Chang C, et al. Neural stem cell delivery of an oncolytic adenovirus in newly diagnosed malignant glioma: a first-in-human, phase 1, dose-escalation trial. Lancet Oncol. 2021;22(8):1103–14.
Okada N, Tsukada Y, Nakagawa S, Mizuguchi H, Mori K, Saito T, et al. Efficient gene delivery into dendritic cells by fiber-mutant adenovirus vectors. Biochem Biophys Res Commun. 2001;282(1):173–9.
Yao J, Atasheva S, Wagner N, Di Paolo NC, Stewart PL, Shayakhmetov DM. Targeted, safe, and efficient gene delivery to human hematopoietic stem and progenitor cells invivo using the engineered AVID adenovirus vector platform. Mol Ther. 2024;32(1):103–23.
Harvey BG, Hackett NR, Ely S, Crystal RG. Host responses and persistence of vector genome following intrabronchial administration of an E1(-)E3(-) adenovirus gene transfer vector to normal individuals. Mol Ther. 2001;3(2):206–15.
Danthinne X, Imperiale MJ. Production of first generation adenovirus vectors: a review. Gene Ther. 2000;7(20):1707–14.
Alba R, Bosch A, Chillon M. Gutless adenovirus: last-generation adenovirus for gene therapy. Gene Ther. 2005;12(Suppl 1):S18-27.
Biermann V, Volpers C, Hussmann S, Stock A, Kewes H, Schiedner G, et al. Targeting of high-capacity adenoviral vectors. Hum Gene Ther. 2001;12(14):1757–69.
Gaden F, Franqueville L, Magnusson MK, Hong SS, Merten MD, Lindholm L, et al. Gene transduction and cell entry pathway of fiber-modified adenovirus type 5 vectors carrying novel endocytic peptide ligands selected on human tracheal glandular cells. J Virol. 2004;78(13):7227–47.
Magnusson MK, Hong SS, Boulanger P, Lindholm L. Genetic retargeting of adenovirus: novel strategy employing “deknobbing” of the fiber. J Virol. 2001;75(16):7280–9.
Hong SS, Magnusson MK, Henning P, Lindholm L, Boulanger PA. Adenovirus stripping: a versatile method to generate adenovirus vectors with new cell target specificity. Mol Ther. 2003;7(5 Pt 1):692–9.
Kurachi S, Koizumi N, Sakurai F, Kawabata K, Sakurai H, Nakagawa S, et al. Characterization of capsid-modified adenovirus vectors containing heterologous peptides in the fiber knob, protein IX, or hexon. Gene Ther. 2007;14(3):266–74.
Robertson S, Parker AL, Clarke C, Duffy MR, Alba R, Nicklin SA, et al. Retargeting FX-binding-ablated HAdV-5 to vascular cells by inclusion of the RGD-4C peptide in hexon hypervariable region 7 and the HI loop. J Gen Virol. 2016;97(8):1911–6.
Puig-Saus C, Rojas LA, Laborda E, Figueras A, Alba R, Fillat C, et al. iRGD tumor-penetrating peptide-modified oncolytic adenovirus shows enhanced tumor transduction, intratumoral dissemination and antitumor efficacy. Gene Ther. 2014;21(8):767–74.
Tsuda H, Wada T, Ito Y, Uchida H, Dehari H, Nakamura K, et al. Efficient BMP2 gene transfer and bone formation of mesenchymal stem cells by a fiber-mutant adenoviral vector. Mol Ther. 2003;7(3):354–65.
Rojas JJ, Gimenez-Alejandre M, Gil-Hoyos R, Cascallo M, Alemany R. Improved systemic antitumor therapy with oncolytic adenoviruses by replacing the fiber shaft HSG-binding domain with RGD. Gene Ther. 2012;19(4):453–7.
Shinoura N, Yoshida Y, Tsunoda R, Ohashi M, Zhang W, Asai A, et al. Highly augmented cytopathic effect of a fiber-mutant E1B-defective adenovirus for gene therapy of gliomas. Cancer Res. 1999;59(14):3411–6.
Yoshida Y, Sadata A, Zhang W, Saito K, Shinoura N, Hamada H. Generation of fiber-mutant recombinant adenoviruses for gene therapy of malignant glioma. Hum Gene Ther. 1998;9(17):2503–15.
Kuroki LM, Jin X, Dmitriev IP, Kashentseva EA, Powell MA, Mutch DG, et al. Adenovirus platform enhances transduction efficiency of human mesenchymal stem cells: An opportunity for cellular carriers of targeted TRAIL-based TR3 biologics in ovarian cancer. PloS One. 2017;12(12):e0190125.
Youn JI, Park SH, Jin HT, Lee CG, Seo SH, Song MY, et al. Enhanced delivery efficiency of recombinant adenovirus into tumor and mesenchymal stem cells by a novel PTD. Cancer Gene Ther. 2008;15(11):703–12.
Machiels JP, Salazar R, Rottey S, Duran I, Dirix L, Geboes K, et al. A phase 1 dose escalation study of the oncolytic adenovirus enadenotucirev, administered intravenously to patients with epithelial solid tumors (EVOLVE). J Immunother Cancer. 2019;7(1):20.
Moreno V, Barretina-Ginesta MP, García-Donas J, Jayson GC, Roxburgh P, Vázquez RM, et al. Safety and efficacy of the tumor-selective adenovirus enadenotucirev with or without paclitaxel in platinum-resistant ovarian cancer: a phase 1 clinical trial. J Immunother Cancer. 2021;9(12):e003645.
Fakih M, Harb W, Mahadevan D, Babiker H, Berlin J, Lillie T, et al. Safety and efficacy of the tumor-selective adenovirus enadenotucirev, in combination with nivolumab, in patients with advanced/metastatic epithelial cancer: a phase I clinical trial (SPICE). J Immunother Cancer. 2023;11(4):e006561.
O’Cathail SM, Davis S, Holmes J, Brown R, Fisher K, Seymour L, et al. A phase 1 trial of the safety, tolerability and biological effects of intravenous Enadenotucirev, a novel oncolytic virus, in combination with chemoradiotherapy in locally advanced rectal cancer (CEDAR). Rad Oncol (London, England). 2020;15(1):151.
Sonzogni O, Zak DE, Sasso MS, Lear R, Muntzer A, Zonca M, et al. T-SIGn tumor reengineering therapy and CAR T cells synergize in combination therapy to clear human lung tumor xenografts and lung metastases in NSG mice. Oncoimmunology. 2022;11(1):2029070.
Garcia-Carbonero R, Salazar R, Duran I, Osman-Garcia I, Paz-Ares L, Bozada JM, et al. Phase 1 study of intravenous administration of the chimeric adenovirus enadenotucirev in patients undergoing primary tumor resection. J Immunother Cancer. 2017;5(1):71.
Khalil DN, Prieto González-Albo I, Rosen L, Lillie T, Stacey A, Parfitt L, et al. A tumor-selective adenoviral vector platform induces transient antiphospholipid antibodies, without increased risk of thrombosis, in phase 1 clinical studies. Investig New Drugs. 2023;41(2):317–23.
Hoffmann D, Meyer B, Wildner O. Improved glioblastoma treatment with Ad5/35 fiber chimeric conditionally replicating adenoviruses. J Gene Med. 2007;9(9):764–78.
Ulasov IV, Rivera AA, Han Y, Curiel DT, Zhu ZB, Lesniak MS. Targeting adenovirus to CD80 and CD86 receptors increases gene transfer efficiency to malignant glioma cells. J Neurosurg. 2007;107(3):617–27.
Wenthe J, Naseri S, Hellström AC, Wiklund HJ, Eriksson E, Loskog A. Immunostimulatory oncolytic virotherapy for multiple myeloma targeting 4–1BB and/or CD40. Cancer Gene Ther. 2020;27(12):948–59.
Wenthe J, Naseri S, Labani-Motlagh A, Enblad G, Wikström KI, Eriksson E, et al. Boosting CAR T-cell responses in lymphoma by simultaneous targeting of CD40/4-1BB using oncolytic viral gene therapy. Cancer Immunol Immunother. 2021;70(10):2851–65.
Labani-Motlagh A, Naseri S, Wenthe J, Eriksson E, Loskog A. Systemic immunity upon local oncolytic virotherapy armed with immunostimulatory genes may be supported by tumor-derived exosomes. Mol Ther Oncolytics. 2021;20:508–18.
Eriksson E, Moreno R, Milenova I, Liljenfeldt L, Dieterich LC, Christiansson L, et al. Activation of myeloid and endothelial cells by CD40L gene therapy supports T-cell expansion and migration into the tumor microenvironment. Gene Ther. 2017;24(2):92–103.
Eriksson E, Milenova I, Wenthe J, Ståhle M, Leja-Jarblad J, Ullenhag G, et al. Shaping the tumor stroma and sparking immune activation by CD40 and 4–1BB signaling induced by an armed oncolytic virus. Clin Cancer Res. 2017;23(19):5846–57.
Ranki T, Pesonen S, Hemminki A, Partanen K, Kairemo K, Alanko T, et al. Phase I study with ONCOS-102 for the treatment of solid tumors - an evaluation of clinical response and exploratory analyses of immune markers. J Immunother Cancer. 2016;4:17.
Ponce S, Cedrés S, Ricordel C, Isambert N, Viteri S, Herrera-Juarez M, et al. ONCOS-102 plus pemetrexed and platinum chemotherapy in malignant pleural mesothelioma: a randomized phase 2 study investigating clinical outcomes and the tumor microenvironment. J Immunother Cancer. 2023;11(9):e007552.
Sebestyen Z, de Vrij J, Magnusson M, Debets R, Willemsen R. An oncolytic adenovirus redirected with a tumor-specific T-cell receptor. Cancer Res. 2007;67(23):11309–16.
Goradel NH, Mohajel N, Malekshahi ZV, Jahangiri S, Najafi M, Farhood B, et al. Oncolytic adenovirus: a tool for cancer therapy in combination with other therapeutic approaches. J Cell Physiol. 2019;234(6):8636–46.
Fueyo J, Gomez-Manzano C, Alemany R, Lee PS, McDonnell TJ, Mitlianga P, et al. A mutant oncolytic adenovirus targeting the Rb pathway produces anti-glioma effect in vivo. Oncogene. 2000;19(1):2–12.
Berk AJ. Recent lessons in gene expression, cell cycle control, and cell biology from adenovirus. Oncogene. 2005;24(52):7673–85.
Abudoureyimu M, Lai Y, Tian C, Wang T, Wang R, Chu X. oncolytic adenovirus-a nova for gene-targeted oncolytic viral therapy in HCC. Front Oncol. 2019;9:1182.
Jakubczak JL, Ryan P, Gorziglia M, Clarke L, Hawkins LK, Hay C, et al. An oncolytic adenovirus selective for retinoblastoma tumor suppressor protein pathway-defective tumors: dependence on E1A, the E2F–1 promoter, and viral replication for selectivity and efficacy. Cancer Res. 2003;63(7):1490–9.
Wang D, Porter CE, Lim B, Rosewell Shaw A, Robertson CS, Woods ML, et al. Ultralow-dose binary oncolytic/helper-dependent adenovirus promotes antitumor activity in preclinical and clinical studies. Sci Adv. 2023;9(13):eade6790.
Heise C, Hermiston T, Johnson L, Brooks G, Sampson-Johannes A, Williams A, et al. An adenovirus E1A mutant that demonstrates potent and selective systemic antitumoral efficacy (vol 6, pg 1134, 2000). Nat Med. 2000;6(12):1412.
Cerullo V, Pesonen S, Diaconu I, Escutenaire S, Arstila PT, Ugolini M, et al. Oncolytic adenovirus coding for granulocyte macrophage colony-stimulating factor induces antitumoral immunity in cancer patients. Cancer Res. 2010;70(11):4297–309.
Gállego Pérez-Larraya J, Garcia-Moure M, Labiano S, Patiño-García A, Dobbs J, Gonzalez-Huarriz M, et al. Oncolytic DNX-2401 virus for pediatric diffuse intrinsic pontine glioma. N Engl J Med. 2022;386(26):2471–81.
Tejada S, Alonso M, Patiño A, Fueyo J, Gomez-Manzano C, Diez-Valle R. Phase I trial of DNX-2401 for diffuse intrinsic pontine glioma newly diagnosed in pediatric patients. Neurosurgery. 2018;83(5):1050–6.
Nassiri F, Patil V, Yefet LS, Singh O, Liu J, Dang RMA, et al. Oncolytic DNX-2401 virotherapy plus pembrolizumab in recurrent glioblastoma: a phase 1/2 trial. Nat Med. 2023;29(6):1370–8.
Garofalo M, Villa A, Rizzi N, Kuryk L, Rinner B, Cerullo V, et al. Extracellular vesicles enhance the targeted delivery of immunogenic oncolytic adenovirus and paclitaxel in immunocompetent mice. J Control Release. 2019;294:165–75.
Garofalo M, Saari H, Somersalo P, Crescenti D, Kuryk L, Aksela L, et al. Antitumor effect of oncolytic virus and paclitaxel encapsulated in extracellular vesicles for lung cancer treatment. J Control Release. 2018;283:223–34.
Mantwill K, Naumann U, Seznec J, Girbinger V, Lage H, Surowiak P, et al. YB-1 dependent oncolytic adenovirus efficiently inhibits tumor growth of glioma cancer stem like cells. J Transl Med. 2013;11:216.
Rognoni E, Widmaier M, Haczek C, Mantwill K, Holzmuller R, Gansbacher B, et al. Adenovirus-based virotherapy enabled by cellular YB-1 expression in vitro and in vivo. Cancer Gene Ther. 2009;16(10):753–63.
Holzmüller R, Mantwill K, Haczek C, Rognoni E, Anton M, Kasajima A, et al. YB-1 dependent virotherapy in combination with temozolomide as a multimodal therapy approach to eradicate malignant glioma. Int J Cancer. 2011;129(5):1265–76.
Lichtenegger E, Koll F, Haas H, Mantwill K, Janssen KP, Laschinger M, et al. The Oncolytic Adenovirus XVir-N-31 as a Novel Therapy in Muscle-Invasive Bladder Cancer. Hum Gene Ther. 2019;30(1):44–56.
Koch J, Schober SJ, Hindupur SV, Schöning C, Klein FG, Mantwill K, et al. Targeting the Retinoblastoma/E2F repressive complex by CDK4/6 inhibitors amplifies oncolytic potency of an oncolytic adenovirus. Nat Commun. 2022;13(1):4689.
El-Ayoubi A, Klawitter M, Rüttinger J, Wellhäusser G, Holm PS, Danielyan L, et al. Intranasal Delivery of Oncolytic Adenovirus XVir-N-31 via Optimized Shuttle Cells Significantly Extends Survival of Glioblastoma-Bearing Mice. Cancers. 2023;15(20):4912.
Klawitter M, El-Ayoubi A, Buch J, Rüttinger J, Ehrenfeld M, Lichtenegger E, et al. The Oncolytic Adenovirus XVir-N-31, in Combination with the Blockade of the PD-1/PD-L1 Axis, Conveys Abscopal Effects in a Humanized Glioblastoma Mouse Model. Int J Mol Sci. 2022;23(17):9965.
Mantwill K, Kohler-Vargas N, Bernshausen A, Bieler A, Lage H, Kaszubiak A, et al. Inhibition of the multidrug-resistant phenotype by targeting YB-1 with a conditionally oncolytic adenovirus: implications for combinatorial treatment regimen with chemotherapeutic agents. Cancer Res. 2006;66(14):7195–202.
Ehrenfeld M, Segeth F, Mantwill K, Brockhaus C, Zhao Y, Ploner C, et al. Targeting Cell Cycle Facilitates E1A-Independent Adenoviral Replication. J Virol. 2023;97(6):e0037023.
Zhang Y, Qian L, Chen K, Gu S, Wang J, Meng Z, et al. Intraperitoneal oncolytic virotherapy for patients with malignant ascites: characterization of clinical efficacy and antitumor immune response. Mol Ther Oncolytics. 2022;25:31–42.
He CB, Lao XM, Lin XJ. Transarterial chemoembolization combined with recombinant human adenovirus type 5 H101 prolongs overall survival of patients with intermediate to advanced hepatocellular carcinoma: a prognostic nomogram study. Chin J Cancer. 2017;36(1):59.
Huang L, Zhao H, Shan M, Chen H, Xu B, He Y, et al. Oncolytic adenovirus H101 ameliorate the efficacy of anti-PD-1 monotherapy in colorectal cancer. Cancer Med. 2022;11(23):4575–87.
Reid TR, Freeman S, Post L, McCormick F, Sze DY. Effects of Onyx-015 among metastatic colorectal cancer patients that have failed prior treatment with 5-FU/leucovorin. Cancer Gene Ther. 2005;12(8):673–81.
Reid T, Galanis E, Abbruzzese J, Sze D, Wein LM, Andrews J, et al. Hepatic arterial infusion of a replication-selective oncolytic adenovirus (dl1520): phase II viral, immunologic, and clinical endpoints. Cancer Res. 2002;62(21):6070–9.
Nemunaitis J, Cunningham C, Buchanan A, Blackburn A, Edelman G, Maples P, et al. Intravenous infusion of a replication-selective adenovirus (ONYX-015) in cancer patients: safety, feasibility and biological activity. Gene Ther. 2001;8(10):746–59.
Khuri FR, Nemunaitis J, Ganly I, Arseneau J, Tannock IF, Romel L, et al. a controlled trial of intratumoral ONYX-015, a selectively-replicating adenovirus, in combination with cisplatin and 5-fluorouracil in patients with recurrent head and neck cancer. Nat Med. 2000;6(8):879–85.
Hu HJ, Liang X, Li HL, Du CM, Hao JL, Wang HY, et al. The armed oncolytic adenovirus ZD55-IL-24 eradicates melanoma by turning the tumor cells from the self-state into the nonself-state besides direct killing. Cell Death Dis. 2020;11(11):1022.
Zhang ZL, Zou WG, Luo CX, Li BH, Wang JH, Sun LY, et al. An armed oncolytic adenovirus system, ZD55-gene, demonstrating potent antitumoral efficacy. Cell Res. 2003;13(6):481–9.
Barton KN, Siddiqui F, Pompa R, Freytag SO, Khan G, Dobrosotskaya I, et al. Phase I trial of oncolytic adenovirus-mediated cytotoxic and interleukin-12 gene therapy for the treatment of metastatic pancreatic cancer. Mol Ther Oncolytics. 2021;20:94–104.
Wang P, Li X, Wang J, Gao D, Li Y, Li H, et al. Re-designing Interleukin-12 to enhance its safety and potential as an anti-tumor immunotherapeutic agent. Nat Commun. 2017;8(1):1395.
Li X, Lu M, Yuan M, Ye J, Zhang W, Xu L, et al. CXCL10-armed oncolytic adenovirus promotes tumor-infiltrating T-cell chemotaxis to enhance anti-PD-1 therapy. Oncoimmunology. 2022;11(1):2118210.
Kuehnel F, Wirth TC, Zender L, Manns MP, Kubicka S. The oncolytic adenovirus Adp53dpR replicates selectively in p53 altered tumor cells and potential tumor spectrum is not limited by the p73 status. J Hepatol. 2006;44:S31–2.
Bischoff JR, Kim DH, Williams A, Heise C, Horn S, Muna M, et al. An adenovirus mutant that replicates selectively in p53-deficient human tumor cells. Science. 1996;274(5286):373–6.
McCormick F. Interactions between adenovirus proteins and the p53 pathway: the development of ONYX-015. Semin Cancer Biol. 2000;10(6):453–9.
Zhang J, Zhang Q, Liu Z, Wang J, Shi F, Su J, et al. Efficacy and safety of recombinant human adenovirus type 5 (H101) in persistent, recurrent, or metastatic gynecologic malignancies: a retrospective study. Front Oncol. 2022;12:877155.
Cheng G, Dong H, Yang C, Liu Y, Wu Y, Zhu L, et al. A review on the advances and challenges of immunotherapy for head and neck cancer. Cancer Cell Int. 2021;21(1):406.
Abbas BM, El-Mogy MA, Haj-Ahmad Y. Oncolytic E1B 55KDa-deleted adenovirus replication is independent of p53 levels in cancer cells. Cell Mol Biol (Noisy-le-Grand, France). 2017;63(7):1–11.
Sharon D, Schümann M, MacLeod S, McPherson R, Chaurasiya S, Shaw A, et al. 2-aminopurine enhances the oncolytic activity of an E1b-deleted adenovirus in hepatocellular carcinoma cells. PloS One. 2013;8(6):e65222.
Cheng PH, Wechman SL, McMasters KM, Zhou HS. Oncolytic Replication of E1b-deleted adenoviruses. Viruses. 2015;7(11):5767–79.
Sieber T, Dobner T. Adenovirus type 5 early region 1B 156R protein promotes cell transformation independently of repression of p53-stimulated transcription. J Virol. 2007;81(1):95–105.
Anwar M, Arendt ML, Ramachandran M, Carlsson A, Essand M, Akusjärvi G, et al. Ixovex-1, a novel oncolytic E1B-mutated adenovirus. Cancer Gene Ther. 2022;29(11):1628–35.
Sieber T, Scholz R, Spoerner M, Schumann F, Kalbitzer HR, Dobner T. Intrinsic disorder in the common N-terminus of human adenovirus 5 E1B–55K and its related E1BN proteins indicated by studies on E1B–93R. Virology. 2011;418(2):133–43.
Grandi P, Darilek A, Moscu A, Pradhan A, Li R. Intravesical infusion of oncolytic virus CG0070 in the treatment of bladder cancer. Methods Mol Biol (Clifton, NJ). 2023;2684:303–17.
Packiam VT, Lamm DL, Barocas DA, Trainer A, Fand B, Davis RL 3rd, et al. An open label, single-arm, phase II multicenter study of the safety and efficacy of CG0070 oncolytic vector regimen in patients with BCG-unresponsive non-muscle-invasive bladder cancer: Interim results. Urol Oncol. 2018;36(10):440–7.
Zafar S, Parviainen S, Siurala M, Hemminki O, Havunen R, Tahtinen S, et al. Intravenously usable fully serotype 3 oncolytic adenovirus coding for CD40L as an enabler of dendritic cell therapy. Oncoimmunology. 2017;6(2):e1265717.
Zafar S, Basnet S, Launonen IM, Quixabeira DCA, Santos J, Hemminki O, et al. Oncolytic adenovirus type 3 coding for CD40L facilitates dendritic cell therapy of prostate cancer in humanized mice and patient samples. Hum Gene Ther. 2021;32(3–4):192–202.
Kawashima T, Kagawa S, Kobayashi N, Shirakiya Y, Umeoka T, Teraishi F, et al. Telomerase-specific replication-selective virotherapy for human cancer. Clin Cancer Res. 2004;10(1 Pt 1):285–92.
Heo J, Liang JD, Kim CW, Woo HY, Shih IL, Su TH, et al. Safety and dose escalation of the targeted oncolytic adenovirus OBP-301 for refractory advanced liver cancer: Phase I clinical trial. Mol Ther. 2023;31(7):2077–88.
Hashimoto Y, Tazawa H, Teraishi F, Kojima T, Watanabe Y, Uno F, et al. The hTERT promoter enhances the antitumor activity of an oncolytic adenovirus under a hypoxic microenvironment. PloS One. 2012;7(6):e39292.
Sasaki T, Tazawa H, Hasei J, Kunisada T, Yoshida A, Hashimoto Y, et al. Preclinical evaluation of telomerase-specific oncolytic virotherapy for human bone and soft tissue sarcomas. Clin Cancer Res. 2011;17(7):1828–38.
García M, Moreno R, Gil-Martin M, Cascallò M, de Olza MO, Cuadra C, et al. A phase 1 trial of oncolytic adenovirus ICOVIR-5 administered intravenously to cutaneous and uveal melanoma patients. Hum Gene Ther. 2019;30(3):352–64.
Nokisalmi P, Pesonen S, Escutenaire S, Särkioja M, Raki M, Cerullo V, et al. Oncolytic adenovirus ICOVIR-7 in patients with advanced and refractory solid tumors. Clin Cancer Res. 2010;16(11):3035–43.
Fajardo CA, Guedan S, Rojas LA, Moreno R, Arias-Badia M, de Sostoa J, et al. Oncolytic adenoviral delivery of an EGFR-targeting T-cell engager improves antitumor efficacy. Cancer Res. 2017;77(8):2052–63.
Garcia-Carbonero R, Bazan-Peregrino M, Gil-Martín M, Álvarez R, Macarulla T, Riesco-Martinez MC, et al. Phase I, multicenter, open-label study of intravenous VCN-01 oncolytic adenovirus with or without nab-paclitaxel plus gemcitabine in patients with advanced solid tumors. J Immunother Cancer. 2022;10(3):e003255.
Bazan-Peregrino M, Garcia-Carbonero R, Laquente B, Álvarez R, Mato-Berciano A, Gimenez-Alejandre M, et al. VCN-01 disrupts pancreatic cancer stroma and exerts antitumor effects. J Immunother Cancer. 2021;9(11):e003254.
Mato-Berciano A, Morgado S, Maliandi MV, Farrera-Sal M, Gimenez-Alejandre M, Ginestà MM, et al. Oncolytic adenovirus with hyaluronidase activity that evades neutralizing antibodies: VCN-11. J Control Release. 2021;332:517–28.
Shirakawa Y, Tazawa H, Tanabe S, Kanaya N, Noma K, Koujima T, et al. Phase I dose-escalation study of endoscopic intratumoral injection of OBP-301 (Telomelysin) with radiotherapy in oesophageal cancer patients unfit for standard treatments. Eur J Cancer (Oxford, England : 1990). 2021;153:98–108.
Kanaya N, Kuroda S, Kakiuchi Y, Kumon K, Tsumura T, Hashimoto M, et al. Immune modulation by telomerase-specific oncolytic adenovirus synergistically enhances antitumor efficacy with Anti-PD1 antibody. Mol Ther. 2020;28(3):794–804.
Kajiwara Y, Tazawa H, Yamada M, Kanaya N, Fushimi T, Kikuchi S, et al. Oncolytic virus-mediated reducing of myeloid-derived suppressor cells enhances the efficacy of PD-L1 blockade in gemcitabine-resistant pancreatic cancer. Cancer Immunol Immunother. 2023;72(5):1285–300.
Hemminki O, Diaconu I, Cerullo V, Pesonen SK, Kanerva A, Joensuu T, et al. Ad3-hTERT-E1A, a fully serotype 3 oncolytic adenovirus, in patients with chemotherapy refractory cancer. Mol Ther. 2012;20(9):1821–30.
Dai F, Zhang PB, Feng Q, Pan XY, Song SL, Cui J, et al. Cytokine-induced killer cells carrying recombinant oncolytic adenovirus expressing p21Ras scFv inhibited liver cancer. J Cancer. 2021;12(9):2768–76.
González R, De la Rosa ÁJ, Romero-Brufau S, Barrera-Pulido L, Gallardo-Chamizo F, Pereira S, et al. Nitric oxide synthase type III overexpression by gene therapy exerts antitumoral activity in mouse hepatocellular carcinoma. Redox biology. 2015;5:420–1.
Mantwill K, Klein FG, Wang D, Hindupur SV, Ehrenfeld M, Holm PS, et al. Concepts in Oncolytic Adenovirus Therapy. Int J Mol Sci. 2021;22(19):10522.
Kazemi Shariat Panahi H, Dehhaghi M, Lam SS, Peng W, Aghbashlo M, Tabatabaei M, et al. Oncolytic viruses as a promising therapeutic strategy against the detrimental health impacts of air pollution: The case of glioblastoma multiforme. Semin Cancer Biol. 2022;86(Pt 3):1122–42.
Jiang H, Rivera-Molina Y, Gomez-Manzano C, Clise-Dwyer K, Bover L, Vence LM, et al. Oncolytic adenovirus and tumor-targeting immune modulatory therapy improve autologous cancer vaccination. Cancer Res. 2017;77(14):3894–907.
Li S, Guo Y, Ning W, Chen Y, Xu J, Zhao C, et al. Oncolytic virus Ad-TD-nsIL-12 inhibits glioma growth and reprograms the tumor immune microenvironment. Life Sci. 2024;336:122254.
Bayo-Puxan N, Gimenez-Alejandre M, Lavilla-Alonso S, Gros A, Cascallo M, Hemminki A, et al. Replacement of adenovirus type 5 fiber shaft heparan sulfate proteoglycan-binding domain with RGD for improved tumor infectivity and targeting. Hum Gene Ther. 2009;20(10):1214–21.
Garcia-Carbonero R, Bazan-Peregrino M, Gil-Martin M, Alvarez R, Macarulla T, Riesco-Martinez MC, et al. Phase I, multicenter, open-label study of intravenous VCN-01 oncolytic adenovirus with or without nab-paclitaxel plus gemcitabine in patients with advanced solid tumors. J Immunother Cancer. 2022;10(3):e003255.
Martinez-Perez AG, Perez-Trujillo JJ, Garza-Morales R, Ramirez-Avila NE, Loera-Arias MJ, Gomez-Gutierrez JG, et al. An Oncolytic Adenovirus Encoding SA-4-1BBL Adjuvant Fused to HPV-16 E7 Antigen Produces a Specific Antitumor Effect in a Cancer Mouse Model. Vaccines. 2021;9(2):149.
Ylösmäki E, Ylösmäki L, Fusciello M, Martins B, Ahokas P, Cojoc H, et al. Characterization of a novel OX40 ligand and CD40 ligand-expressing oncolytic adenovirus used in the PeptiCRAd cancer vaccine platform. Mol Ther Oncolytics. 2021;20:459–69.
Gattacceca F, Pilatte Y, Billard C, Monnet I, Moritz S, Le Carrou J, et al. Ad-IFN gamma induces antiproliferative and antitumoral responses in malignant mesothelioma. Clin Cancer Res. 2002;8(10):3298–304.
Zhao P, Zhu YH, Wu JX, Liu RY, Zhu XY, Xiao X, et al. Adenovirus-mediated delivery of human IFNgamma gene inhibits prostate cancer growth. Life Sci. 2007;81(9):695–701.
Toloza EM, Hunt K, Swisher S, McBride W, Lau R, Pang S, et al. In vivo cancer gene therapy with a recombinant interleukin-2 adenovirus vector. Cancer Gene Ther. 1996;3(1):11–7.
Xiang J, Chen Y, Moyana T. Combinational immunotherapy for established tumors with engineered tumor vaccines and adenovirus-mediated gene transfer. Cancer Gene Ther. 2000;7(7):1023–33.
He T, Hao Z, Lin M, Xin Z, Chen Y, Ouyang W, et al. Oncolytic adenovirus promotes vascular normalization and nonclassical tertiary lymphoid structure formation through STING-mediated DC activation. Oncoimmunology. 2022;11(1):2093054.
Santos JM, Cervera-Carrascon V, Havunen R, Zafar S, Siurala M, Sorsa S, et al. Adenovirus coding for interleukin-2 and tumor necrosis factor alpha replaces lymphodepleting chemotherapy in adoptive t cell therapy. Mol Ther. 2018;26(9):2243–54.
Wang D, Huang XF, Hong B, Song XT, Hu L, Jiang M, et al. Efficacy of intracellular immune checkpoint-silenced DC vaccine. JCI insight. 2018;3(3):e98368.
Hamdan F, Ylösmäki E, Chiaro J, Giannoula Y, Long M, Fusciello M, et al. Novel oncolytic adenovirus expressing enhanced cross-hybrid IgGA Fc PD-L1 inhibitor activates multiple immune effector populations leading to enhanced tumor killing in vitro, in vivo and with patient-derived tumor organoids. J Immunother Cancer. 2021;9(8):e003000.
Gambotto A, Tüting T, McVey DL, Kovesdi I, Tahara H, Lotze MT, et al. Induction of antitumor immunity by direct intratumoral injection of a recombinant adenovirus vector expressing interleukin-12. Cancer Gene Ther. 1999;6(1):45–53.
Lee CT, Wu S, Ciernik IF, Chen H, Nadaf-Rahrov S, Gabrilovich D, et al. Genetic immunotherapy of established tumors with adenovirus-murine granulocyte-macrophage colony-stimulating factor. Hum Gene Ther. 1997;8(2):187–93.
Bilusic M, McMahon S, Madan RA, Karzai F, Tsai YT, Donahue RN, et al. Phase I study of a multitargeted recombinant Ad5 PSA/MUC-1/brachyury-based immunotherapy vaccine in patients with metastatic castration-resistant prostate cancer (mCRPC). J Immunother Cancer. 2021;9(3):e002374.
Gatti-Mays ME, Redman JM, Donahue RN, Palena C, Madan RA, Karzai F, et al. A Phase I Trial Using a Multitargeted Recombinant Adenovirus 5 (CEA/MUC1/Brachyury)-Based Immunotherapy Vaccine Regimen in Patients with Advanced Cancer. Oncologist. 2020;25(6):479-e899.
Boorjian SA, Alemozaffar M, Konety BR, Shore ND, Gomella LG, Kamat AM, et al. Intravesical nadofaragene firadenovec gene therapy for BCG-unresponsive non-muscle-invasive bladder cancer: a single-arm, open-label, repeat-dose clinical trial. Lancet Oncol. 2021;22(1):107–17.
Shore ND, Boorjian SA, Canter DJ, Ogan K, Karsh LI, Downs TM, et al. Intravesical rAd-IFNα/Syn3 for patients with high-grade, bacillus calmette-guerin-refractory or relapsed non-muscle-invasive bladder cancer: a phase ii randomized study. J Clin Oncol. 2017;35(30):3410–6.
Dinney CP, Fisher MB, Navai N, O’Donnell MA, Cutler D, Abraham A, et al. Phase I trial of intravesical recombinant adenovirus mediated interferon-α2b formulated in Syn3 for Bacillus Calmette-Guérin failures in nonmuscle invasive bladder cancer. J Urol. 2013;190(3):850–6.
Cristofanilli M, Krishnamurthy S, Guerra L, Broglio K, Arun B, Booser DJ, et al. A nonreplicating adenoviral vector that contains the wild-type p53 transgene combined with chemotherapy for primary breast cancer: safety, efficacy, and biologic activity of a novel gene-therapy approach. Cancer. 2006;107(5):935–44.
Chiappori AA, Williams CC, Gray JE, Tanvetyanon T, Haura EB, Creelan BC, et al. Randomized-controlled phase II trial of salvage chemotherapy after immunization with a TP53-transfected dendritic cell-based vaccine (Ad.p53-DC) in patients with recurrent small cell lung cancer. Cancer Immunol Immunother. 2019;68(3):517-27.
Soliman H, Khambati F, Han HS, Ismail-Khan R, Bui MM, Sullivan DM, et al. A phase-1/2 study of adenovirus-p53 transduced dendritic cell vaccine in combination with indoximod in metastatic solid tumors and invasive breast cancer. Oncotarget. 2018;9(11):10110–7.
Rosenthal EL, Chung TK, Parker WB, Allan PW, Clemons L, Lowman D, et al. Phase I dose-escalating trial of Escherichia coli purine nucleoside phosphorylase and fludarabine gene therapy for advanced solid tumors. Ann Oncol. 2015;26(7):1481–7.
Umemura Y, Orringer D, Junck L, Varela ML, West MEJ, Faisal SM, et al. Combined cytotoxic and immune-stimulatory gene therapy for primary adult high-grade glioma: a phase 1, first-in-human trial. Lancet Oncol. 2023;24(9):1042–52.
Sun K, Xu Y, Zhang L, Niravath P, Darcourt J, Patel T, et al. A phase 2 trial of enhancing immune checkpoint blockade by stereotactic radiation and in situ virus gene therapy in metastatic triple-negative breast cancer. Clin Cancer Res. 2022;28(20):4392–401.
Westphal M, Ylä-Herttuala S, Martin J, Warnke P, Menei P, Eckland D, et al. Adenovirus-mediated gene therapy with sitimagene ceradenovec followed by intravenous ganciclovir for patients with operable high-grade glioma (ASPECT): a randomised, open-label, phase 3 trial. Lancet Oncol. 2013;14(9):823–33.
Freytag SO, Khil M, Stricker H, Peabody J, Menon M, DePeralta-Venturina M, et al. Phase I study of replication-competent adenovirus-mediated double suicide gene therapy for the treatment of locally recurrent prostate cancer. Cancer Res. 2002;62(17):4968–76.
Freytag SO, Stricker H, Pegg J, Paielli D, Pradhan DG, Peabody J, et al. Phase I study of replication-competent adenovirus-mediated double-suicide gene therapy in combination with conventional-dose three-dimensional conformal radiation therapy for the treatment of newly diagnosed, intermediate- to high-risk prostate cancer. Cancer Res. 2003;63(21):7497–506.
Morelli AE, Larregina AT, Ganster RW, Zahorchak AF, Plowey JM, Takayama T, et al. Recombinant adenovirus induces maturation of dendritic cells via an NF-kappaB-dependent pathway. J Virol. 2000;74(20):9617–28.
Chondronasiou D, Eisden T, Stam AGM, Matthews QL, Icyuz M, Hooijberg E, et al. Improved Induction of Anti-Melanoma T Cells by Adenovirus-5/3 Fiber Modification to Target Human DCs. Vaccines. 2018;6(3):42.
Zhong L, Granelli-Piperno A, Choi Y, Steinman RM. Recombinant adenovirus is an efficient and non-perturbing genetic vector for human dendritic cells. Eur J Immunol. 1999;29(3):964–72.
Sutherland RM, Londrigan SL, Brady JL, Azher H, Carrington EM, Zhan Y, et al. Shutdown of immunological priming and presentation after in vivo administration of adenovirus. Gene Ther. 2012;19(11):1095–100.
Pardee AD, Yano H, Weinstein AM, Ponce AA, Ethridge AD, Normolle DP, et al. Route of antigen delivery impacts the immunostimulatory activity of dendritic cell-based vaccines for hepatocellular carcinoma. J Immunother Cancer. 2015;3:32.
Liau LM, Ashkan K, Brem S, Campian JL, Trusheim JE, Iwamoto FM, et al. Association of autologous tumor lysate-loaded dendritic cell vaccination with extension of survival among patients with newly diagnosed and recurrent glioblastoma: a phase 3 prospective externally controlled cohort trial. JAMA Oncol. 2023;9(1):112–21.
Wen PY, Reardon DA, Armstrong TS, Phuphanich S, Aiken RD, Landolfi JC, et al. A randomized double-blind placebo-controlled phase II trial of dendritic cell vaccine ICT-107 in newly diagnosed patients with glioblastoma. Clin Cancer Res. 2019;25(19):5799–807.
Steitz J, Bruck J, Knop J, Tuting T. Adenovirus-transduced dendritic cells stimulate cellular immunity to melanoma via a CD4(+) T cell-dependent mechanism. Gene Ther. 2001;8(16):1255–63.
Ojima T, Iwahashi M, Nakamura M, Matsuda K, Naka T, Nakamori M, et al. The boosting effect of co-transduction with cytokine genes on cancer vaccine therapy using genetically modified dendritic cells expressing tumor-associated antigen. Int J Oncol. 2006;28(4):947–53.
Nakamura M, Iwahashi M, Nakamori M, Ueda K, Matsuura I, Noguchi K, et al. Dendritic cells genetically engineered to simultaneously express endogenous tumor antigen and granulocyte macrophage colony-stimulating factor elicit potent therapeutic antitumor immunity. Clin Cancer Res. 2002;8(8):2742–9.
Vujanovic L, Ranieri E, Gambotto A, Olson WC, Kirkwood JM, Storkus WJ. IL-12p70 and IL-18 gene-modified dendritic cells loaded with tumor antigen-derived peptides or recombinant protein effectively stimulate specific Type-1 CD4+ T-cell responses from normal donors and melanoma patients in vitro. Cancer Gene Ther. 2006;13(8):798–805.
Ye YL, Lee YL, Chuang ZJ, Lai HJ, Chen CC, Tao MH, et al. Dendritic cells modulated by cytokine-expressing adenoviruses alleviate eosinophilia and airway hyperresponsiveness in an animal model of asthma. J Allergy Clin Immunol. 2004;114(1):88–96.
Tatsumi T, Huang J, Gooding WE, Gambotto A, Robbins PD, Vujanovic NL, et al. Intratumoral delivery of dendritic cells engineered to secrete both interleukin (IL)-12 and IL-18 effectively treats local and distant disease in association with broadly reactive Tc1-type immunity. Cancer Res. 2003;63(19):6378–86.
Tanaka F, Hashimoto W, Okamura H, Robbins PD, Lotze MT, Tahara H. Rapid generation of potent and tumor-specific cytotoxic T lymphocytes by interleukin 18 using dendritic cells and natural killer cells. Cancer Res. 2000;60(17):4838–44.
Fan X, Ye M, Xue B, Ke Y, Wong CK, Xie Y. Human dendritic cells engineered to secrete interleukin-18 activate MAGE-A3-specific cytotoxic T lymphocytes in vitro. Immunol Investig. 2012;41(5):469–83.
Bhanumathy KK, Zhang B, Ahmed KA, Qureshi M, Xie Y, Tao M, et al. Transgene IL-6 enhances DC-stimulated CTL responses by counteracting CD4+25+Foxp3+ regulatory T cell suppression via IL-6-induced Foxp3 downregulation. Int J Mol Sci. 2014;15(4):5508–21.
Zhang W, Chen Z, Li F, Kamencic H, Juurlink B, Gordon JR, et al. Tumour necrosis factor-alpha (TNF-alpha) transgene-expressing dendritic cells (DCs) undergo augmented cellular maturation and induce more robust T-cell activation and anti-tumour immunity than DCs generated in recombinant TNF-alpha. Immunology. 2003;108(2):177–88.
Guo Z, Gao HY, Zhang TY, Lou JX, Yang K, Liu XD, et al. Adenovirus co-expressing CD40 ligand and interleukin (IL)-2 contributes to maturation of dendritic cells and production of IL-12. Biomed Rep. 2016;5(5):567–73.
Tuettenberg A, Fondel S, Steinbrink K, Enk AH, Jonuleit H. CD40 signalling induces IL-10-producing, tolerogenic dendritic cells. Exp Dermatol. 2010;19(1):44–53.
Xu W, Li Y, Wang X, Wang C, Zhao W, Wu J. Anti-tumor activity of gene transfer of the membrane-stable CD40L mutant into lung cancer cells. Int J Oncol. 2010;37(4):935–41.
Liu C, Liu X, Xiang X, Pang X, Chen S, Zhang Y, et al. A nanovaccine for antigen self-presentation and immunosuppression reversal as a personalized cancer immunotherapy strategy. Nat Nanotechnol. 2022;17(5):531–40.
Chiappori AA, Soliman H, Janssen WE, Antonia SJ, Gabrilovich DI. INGN-225: a dendritic cell-based p53 vaccine (Ad.p53-DC) in small cell lung cancer: observed association between immune response and enhanced chemotherapy effect. Exp Opin Biol Ther. 2010;10(6):983-91.
Liu W, Wang X, Feng X, Yu J, Liu X, Jia X, et al. Oncolytic adenovirus-mediated intratumoral expression of TRAIL and CD40L enhances immunotherapy by modulating the tumor microenvironment in immunocompetent mouse models. Cancer Lett. 2022;535:215661.
Nyati S, Stricker H, Barton KN, Li P, Elshaikh M, Ali H, et al. A phase I clinical trial of oncolytic adenovirus mediated suicide and interleukin-12 gene therapy in patients with recurrent localized prostate adenocarcinoma. PloS One. 2023;18(9):e0291315.
Emtage PC, Wan Y, Bramson JL, Graham FL, Gauldie J. A double recombinant adenovirus expressing the costimulatory molecule B7–1 (murine) and human IL-2 induces complete tumor regression in a murine breast adenocarcinoma model. J Immunol (Baltimore, Md : 1950). 1998;160(5):2531–8.
Addison CL, Bramson JL, Hitt MM, Muller WJ, Gauldie J, Graham FL. Intratumoral coinjection of adenoviral vectors expressing IL-2 and IL-12 results in enhanced frequency of regression of injected and untreated distal tumors. Gene Ther. 1998;5(10):1400–9.
Watanabe K, Luo Y, Da T, Guedan S, Ruella M, Scholler J, et al. Pancreatic cancer therapy with combined mesothelin-redirected chimeric antigen receptor T cells and cytokine-armed oncolytic adenoviruses. JCI insight. 2018;3(7).
Rosewell Shaw A, Porter CE, Watanabe N, Tanoue K, Sikora A, Gottschalk S, et al. Adenovirotherapy delivering cytokine and checkpoint inhibitor augments CAR T cells against metastatic head and neck cancer. Mol Ther. 2017;25(11):2440–51.
Ye JF, Lin YQ, Yu XH, Liu MY, Li Y. Immunotherapeutic effects of cytokine-induced killer cells combined with CCL21/IL15 armed oncolytic adenovirus in TERT-positive tumor cells. Int Immunopharmacol. 2016;38:460–7.
Yang Z, Zhang Q, Xu K, Shan J, Shen J, Liu L, et al. Combined therapy with cytokine-induced killer cells and oncolytic adenovirus expressing IL-12 induce enhanced antitumor activity in liver tumor model. PloS One. 2012;7(9):e44802.
Du YN, Wei Q, Zhao LJ, Fan CQ, Guo LR, Ye JF, et al. Hydrogel-based co-delivery of CIK cells and oncolytic adenovirus armed with IL12 and IL15 for cancer immunotherapy. Biomed Pharmacother = Biomedecine & pharmacotherapie. 2022;151:113110.
Bowman L, Grossmann M, Rill D, Brown M, Zhong WY, Alexander B, et al. IL-2 adenovector-transduced autologous tumor cells induce antitumor immune responses in patients with neuroblastoma. Blood. 1998;92(6):1941–9.
Liu X, Xu J, Yao T, Ding J, Li S, Su R, et al. Cryo-shocked cancer cells as an oncolytic adenovirus reservoir for glioblastoma immunotherapy. ACS Appl Mater Interfaces. 2023;15(1):67–76.
Wu Q, Huang H, Sun M, Zhang R, Wang J, Zheng H, et al. Inhibition of tumor metastasis by liquid-nitrogen-shocked tumor cells with oncolytic viruses infection. Adv Mater (Deerfield Beach, Fla). 2023;35(28):e2212210.
Kortylewski M, Yu H. Role of Stat3 in suppressing anti-tumor immunity. Curr Opin Immunol. 2008;20(2):228–33.
Wang Y, Shen Y, Wang S, Shen Q, Zhou X. The role of STAT3 in leading the crosstalk between human cancers and the immune system. Cancer Lett. 2018;415:117–28.
Iwata-Kajihara T, Sumimoto H, Kawamura N, Ueda R, Takahashi T, Mizuguchi H, et al. Enhanced cancer immunotherapy using STAT3-depleted dendritic cells with high Th1-inducing ability and resistance to cancer cell-derived inhibitory factors. J Immunol (Baltimore, Md : 1950). 2011;187(1):27–36.
Yang H, Yamazaki T, Pietrocola F, Zhou H, Zitvogel L, Ma Y, et al. STAT3 inhibition enhances the therapeutic efficacy of immunogenic chemotherapy by stimulating type 1 interferon production by cancer cells. Cancer Res. 2015;75(18):3812–22.
Hassin O, Oren M. Drugging p53 in cancer: one protein, many targets. Nat Rev Drug Discov. 2023;22(2):127–44.
Gohara Y, Tomonobu N, Kinoshita R, Futami J, Audebert L, Chen Y, et al. Novel extracellular role of REIC/Dkk-3 protein in PD-L1 regulation in cancer cells. J Mol Med (Berlin, Germany). 2023;101(4):431–47.
Li J, Shi L, Zhang X, Kang X, Wen Y, Qian H, et al. Recombinant adenovirus IL-24-Bax promotes apoptosis of hepatocellular carcinoma cells in vitro and in vivo. Cancer Gene Ther. 2010;17(11):771–9.
Hong JS, Waud WR, Levasseur DN, Townes TM, Wen H, McPherson SA, et al. Excellent in vivo bystander activity of fludarabine phosphate against human glioma xenografts that express the escherichia coli purine nucleoside phosphorylase gene. Cancer Res. 2004;64(18):6610–5.
Li D, Zhou K, Wang S, Shi Z, Yang Z. Recombinant adenovirus encoding vasohibin prevents tumor angiogenesis and inhibits tumor growth. Cancer Sci. 2010;101(2):448–52.
Hartmann KP, van Gogh M, Freitag PC, Kast F, Nagy-Davidescu G, Borsig L, et al. FAP-retargeted Ad5 enables in vivo gene delivery to stromal cells in the tumor microenvironment. Mol Ther. 2023;31(10):2914–28.
Wang M, Hong Y, Feng Q, Pan X, Song S, Cui J, et al. Recombinant adenovirus KGHV500 and CIK cells codeliver anti-p21-Ras scFv for the treatment of gastric cancer with wild-type ras overexpression. Mol Ther Oncolytics. 2018;11:90–101.
Lin XR, Zhou XL, Feng Q, Pan XY, Song SL, Fang H, et al. CIK cell-based delivery of recombinant adenovirus KGHV500 carrying the anti-p21Ras scFv gene enhances the anti-tumor effect and safety in lung cancer. J Cancer Res Clin Oncol. 2019;145(5):1123–32.
Qian J, Yang M, Feng Q, Pan XY, Yang LL, Yang JL. Inhibition of glioma by adenovirus KGHV500 encoding anti-p21Ras scFv and carried by cytokine-induced killer cells. Exp Biol Med (Maywood, NJ). 2021;246(10):1228–38.
Jiang R, Zhang Z, Liao X, Huang L, Liao Y, Deng W. Combination of oncolytic adenovirus ZD55 harboring TRAIL-IETD-MnSOD and cytokine-induced killer cells against lung cancer. Ann Transl Med. 2021;9(20):1527.
Ding L, Gao Q, Xu Z, Cai L, Chen S, Zhang X, et al. An Inter-Supplementary Biohybrid System Based on Natural Killer Cells for the Combinational Immunotherapy and Virotherapy of Cancer. Adv Sci (Weinheim, Baden-Wurttemberg, Germany). 2022;9(2):e2103470.
Nakashima H, Kaur B, Chiocca EA. Directing systemic oncolytic viral delivery to tumors via carrier cells. Cytokine Growth Fact Rev. 2010;21(2–3):119–26.
Shimizu Y, Gumin J, Gao F, Hossain A, Shpall EJ, Kondo A, et al. Characterization of patient-derived bone marrow human mesenchymal stem cells as oncolytic virus carriers for the treatment of glioblastoma. J Neurosurg. 2022;136(3):757–67.
McKenna MK, Englisch A, Brenner B, Smith T, Hoyos V, Suzuki M, et al. Mesenchymal stromal cell delivery of oncolytic immunotherapy improves CAR-T cell antitumor activity. Mol Ther. 2021;29(5):1808–20.
Ahmed AU, Tyler MA, Thaci B, Alexiades NG, Han Y, Ulasov IV, et al. A comparative study of neural and mesenchymal stem cell-based carriers for oncolytic adenovirus in a model of malignant glioma. Mol Pharm. 2011;8(5):1559–72.
Studeny M, Marini FC, Dembinski JL, Zompetta C, Cabreira-Hansen M, Bekele BN, et al. Mesenchymal stem cells: potential precursors for tumor stroma and targeted-delivery vehicles for anticancer agents. J Natl Cancer Inst. 2004;96(21):1593–603.
Hong X, Miller C, Savant-Bhonsale S, Kalkanis SN. Antitumor treatment using interleukin- 12-secreting marrow stromal cells in an invasive glioma model. Neurosurgery. 2009;64(6):1139–46 (discussion 46-7).
Kim SM, Lim JY, Park SI, Jeong CH, Oh JH, Jeong M, et al. Gene therapy using TRAIL-secreting human umbilical cord blood-derived mesenchymal stem cells against intracranial glioma. Cancer Res. 2008;68(23):9614–23.
Zhang Q, Yuan XF, Lu Y, Li ZZ, Bao SQ, Zhang XL, et al. Surface expression of anti-CD3scfv stimulates locoregional immunotherapy against hepatocellular carcinoma depending on the E1A-engineered human umbilical cord mesenchymal stem cells. Int J Cancer. 2017;141(7):1445–57.
Dembinski JL, Spaeth EL, Fueyo J, Gomez-Manzano C, Studeny M, Andreeff M, et al. Reduction of nontarget infection and systemic toxicity by targeted delivery of conditionally replicating viruses transported in mesenchymal stem cells. Cancer Gene Ther. 2010;17(4):289–97.
Zhang J, Chen H, Chen C, Liu H, He Y, Zhao J, et al. Systemic administration of mesenchymal stem cells loaded with a novel oncolytic adenovirus carrying IL-24/endostatin enhances glioma therapy. Cancer Lett. 2021;509:26–38.
Hammer K, Kazcorowski A, Liu L, Behr M, Schemmer P, Herr I, et al. Engineered adenoviruses combine enhanced oncolysis with improved virus production by mesenchymal stromal carrier cells. Int J Cancer. 2015;137(4):978–90.
Guo Y, Zhang Z, Xu X, Xu Z, Wang S, Huang D, et al. Menstrual blood-derived stem cells as delivery vehicles for oncolytic adenovirus virotherapy for colorectal cancer. Stem Cells Dev. 2019;28(13):882–96.
Yuan X, Zhang Q, Li Z, Zhang X, Bao S, Fan D, et al. Mesenchymal stem cells deliver and release conditionally replicative adenovirus depending on hepatic differentiation to eliminate hepatocellular carcinoma cells specifically. Cancer Lett. 2016;381(1):85–95.
Yuan X, Lu Y, Yang Y, Tian W, Fan D, Liu R, et al. Systemic administration of mesenchymal stem cells loaded with a novel oncolytic adenovirus carrying a bispecific T cell engager against hepatocellular carcinoma. Oncoimmunology. 2023;12(1):2219544.
Chen SR, Chen MM, Ene C, Lang FF, Kan P. Perfusion-guided endovascular super-selective intra-arterial infusion for treatment of malignant brain tumors. J Neurointerv Surg. 2022;14(6):533–8.
Martinez-Quintanilla J, He D, Wakimoto H, Alemany R, Shah K. Encapsulated stem cells loaded with hyaluronidase-expressing oncolytic virus for brain tumor therapy. Mol Ther. 2015;23(1):108–18.
Ruano D, López-Martín JA, Moreno L, Lassaletta Á, Bautista F, Andión M, et al. First-in-human, first-in-child trial of autologous MSCs carrying the oncolytic virus icovir-5 in patients with advanced tumors. Mol Ther. 2020;28(4):1033–42.
Kan P, Srinivasan VM, Gumin J, Garcia R, Chen SR, Johnson JN, et al. Development of a Rabbit Human Glioblastoma Model for Testing of Endovascular Selective Intra-Arterial Infusion (ESIA) of Novel Stem Cell-Based Therapeutics. Neuro-oncology. 2024;26(1):127–36.
Tobias AL, Thaci B, Auffinger B, Rincón E, Balyasnikova IV, Kim CK, et al. The timing of neural stem cell-based virotherapy is critical for optimal therapeutic efficacy when applied with radiation and chemotherapy for the treatment of glioblastoma. Stem Cells Transl Med. 2013;2(9):655–66.
Pan K, Farrukh H, Chittepu V, Xu H, Pan CX, Zhu Z. CAR race to cancer immunotherapy: from CAR T, CAR NK to CAR macrophage therapy. J Exp Clin Cancer Res. 2022;41(1):119.
Zhang L, Tian L, Dai X, Yu H, Wang J, Lei A, et al. Pluripotent stem cell-derived CAR-macrophage cells with antigen-dependent anti-cancer cell functions. J Hematol Oncol. 2020;13(1):153.
Su S, Lei A, Wang X, Lu H, Wang S, Yang Y, et al. Induced CAR-Macrophages as a Novel Therapeutic Cell Type for Cancer Immune Cell Therapies. Cells. 2022;11(10):1652.
Anderson NR, Minutolo NG, Gill S, Klichinsky M. Macrophage-based approaches for cancer immunotherapy. Cancer Res. 2021;81(5):1201–8.
Klichinsky M, Ruella M, Shestova O, Lu XM, Best A, Zeeman M, et al. Human chimeric antigen receptor macrophages for cancer immunotherapy. Nat Biotechnol. 2020;38(8):947–53.
Jayasingam SD, Citartan M, Thang TH, Mat Zin AA, Ang KC, Ch’ng ES. Evaluating the polarization of tumor-associated macrophages into M1 and M2 phenotypes in human cancer tissue: technicalities and challenges in routine clinical practice. Front Oncol. 2019;9:1512.
Acknowledgements
Not applicable.
Funding
This study was supported by Chinese National Major Project for New Drug Innovation (2019ZX09201002003). National Natural Science Foundation of China (82030076, 82070161, 81970151, 81670162 and 81870134). Shenzhen Science and Technology Foundation (JCYJ20190808163601776, JCYJ20200109113810154). Shenzhen Key Laboratory Foundation (ZDSYS20200811143757022). Sanming Project of Medicine in Shenzhen (SZSM202111004).
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
All authors critically reviewed and approved the final manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Zeng, M., Zhang, W., Li, Y. et al. Harnessing adenovirus in cancer immunotherapy: evoking cellular immunity and targeting delivery in cell-specific manner. Biomark Res 12, 36 (2024). https://doi.org/10.1186/s40364-024-00581-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s40364-024-00581-1