Abstract
Background
In recent years, there has been a noteworthy increase in research exploring the relationship between gut microbiota and bariatric surgery. This increase can be attributed to the growing recognition of the importance of gut microbiota in diverse health issues, such as obesity, and its alterations following bariatric surgery. Consequently, this study seeks to employ bibliometric analysis to show the current research landscape and identify key areas of focus for future investigations on the link between bariatric surgery and gut microbiota.
Methods
We conducted a comprehensive search of the Scopus database to gather literature concerning bariatric surgery and its impact on the gut microbiota. The search encompassed documents published between 2009 and 2023. To analyze the bibliometric data, we employed diverse metrics, including publication count, citation count, and evaluation of the performance of countries, institutions, and journals. Additionally, we utilized VOSviewer to visually present the findings.
Results
A total of 555 articles focused on the correlation between bariatric surgery and the gut microbiota. Original research articles constituted 55.32% of all publications on this topic, followed by reviews at 37.30%. The leading countries in terms of publication volume were the USA (n = 156; 28.11%), China (n = 73; 13.15%), and the UK (n = 52; 9.37%). Co-occurrence analysis highlighted three main clusters of research topics: (1) the impact of bariatric surgery on the gut microbiota composition in relation to obesity remission, (2) the relationship between glucose metabolism, circulating bile acids, gut hormones, and gut microbiome remodeling, and (3) the connections between alterations in the gut microbiota and insulin resistance.
Conclusions
Ongoing research has investigated the connection between bariatric surgery and the gut microbiota, providing new perspectives on metabolic improvements after surgery. Further studies are necessary to comprehensively grasp how bariatric procedures influence the gut microbiota and to formulate precise interventions aimed at optimizing the health advantages of surgery.
Similar content being viewed by others
Introduction
Overweight and obesity are medical terms for chronic diseases and are among the public health issues that have reached epidemic proportions in the developed world. The World Health Organization (WHO) screening criteria are based on body mass index (BMI), expressed in units of kg/m² as the body weight in kilograms divided by the square of the body height in meters. In adults, overweight or preobesity is defined as a BMI of 25–29.9 kg/m², while a BMI ≥ 30 kg/m² indicates obesity. Globally, 39% of adults aged 18 years and over were overweight or obese in 2016. Abdominal obesity with a waist circumference > 102 cm for men and > 88 cm for women is one of the diagnostic criteria for metabolic syndrome and can be linked directly to medical conditions such as diabetes, cancer, depression and cardiovascular disease, with a higher rate of mortality worldwide [1]. Thus, various treatment options can be used effectively, including lifestyle modifications, yet the noninvasive treatment and remission of morbid obesity have largely yielded limited results compared with surgical intervention. Therefore, several studies have reported that bariatric surgery is the most effective treatment modality for severe obesity-related comorbidities [2, 3] and that improved metabolic changes and sustained weight loss are achieved after bariatric surgery through malabsorption, accelerated gastric emptying, hormonal changes, and alterations in bile acid metabolism. Nevertheless, gastric outlet obstruction, internal herniation, nutritional deficiencies, mesh erosion, and gastroesophageal reflux are the most frequent complications that may occur [4, 5].
Several studies have shown that surgery results in profound changes in the microbiome, resulting in weight loss acceleration, particularly during the first year after surgery, when increases in Bacteroides and Proteobacteria and decreases in Firmicutes were observed in the majority of studies [6,7,8]. Bariatric surgery is considered the most effective treatment for obesity and associated disorders, such as diabetes mellitus, as 80% of type 2 diabetes mellitus cases (T2DM) are controlled after bariatric surgery compared to conventional therapy [9].
Bariatric surgery, such as gastric bypass and sleeve gastrectomy, has positive-negative effects on the patient’s gut microbiota [10, 11], varies from individual to individual, and is greatly influenced by diet, age, and the specific bariatric surgery performed. Further research is needed to fully understand the impact of bariatric surgery on the gut microbiota and how it affects patient health. Thus, a comprehensive bibliometric analysis of research related to bariatric surgery and the gut microbiota is still lacking. In this study, a comprehensive bibliometric analysis was conducted in the field of bariatric surgery and gut microbiota.
Bibliometric studies encompass quantitative assessments of scientific literature, wherein research performance, productivity, effects, and trends within a particular topic are evaluated [11, 12]. Hence, the present study is significant for the scientific community, healthcare professionals, and policymakers. This study provides a quantitative assessment of the research conducted on bariatric surgery and the gut microbiota and highlights significant areas of emphasis. As a result, this analysis aids in making well-informed decisions and directing future research endeavors to enhance the comprehension and application of bariatric surgery within the context of the gut microbiota.
Materials and methods
Data source
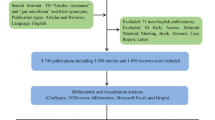
The present bibliometric investigation was conducted using the Scopus database, widely acknowledged as the most appropriate database for bibliometric analyses. The data collection tasks were fully completed on March 1, 2024. We included all the documents on gut microbiota and bariatric surgery within the Scopus database from all previous years up to December 31, 2023. Scopus distinguishes itself from other databases in bibliometric analysis through several distinctive features, as highlighted by key points in the literature: (1) Scopus stands out as the world’s largest curated abstract and citation database for research literature, encompassing research journals, conference proceedings, and scholarly books [13, 14]. (2) It boasts extensive coverage across various subject areas, publication years, and document types, drawing from a vast array of sources from more than 5,000 publishers globally, which includes patents and funding data [13]. (3) Scopus maintains rigorous content selection standards through processes overseen by an independent board of scientists, ensuring the indexing of high-quality content [13, 14]. (4) Additionally, Scopus provides in-depth author and institution profiles derived from advanced profiling algorithms and manual curation, guaranteeing high precision and recall [13, 14]. (5) Scopus is widely used for research assessments, research landscape studies, science policy evaluations, and university rankings due to its trustworthiness and high-quality data [14]. (6) It provides enriched metadata records of scientific articles, and the data have been utilized for large-scale analyses in the academic research community. The academic research community has published numerous publications after analyzing the data, and they include papers focusing on researcher mobility and network visualizations [14]. (7) A comparative study of Scopus and Web of Science revealed that the databases serve similar functions to those of the academic community, but the owners’ business philosophy contributes to the differences in the content and coverage [15,16,17]. (8) In June 2019, the International Center for the Study of Research initiated its operations, with the primary objective of collaborating with the scientometric research community. It aims to provide a virtual laboratory facilitating researchers’ access to Scopus data. Scopus undergoes ongoing monitoring and enhancement via diverse quality assurance protocols. Moreover, it provides complimentary data access to specific academic research endeavors, made feasible through application programming interfaces such as VOSviewer [14].
Search strategy
We began our research by extracting relevant terms related to the gut microbiota and bariatric surgery from the Medical Subject Headings (MeSH) in PubMed and previous studies [18,19,20]. We subsequently used these terms to search Scopus, and the detailed research strategy is available in the supplementary materials (Additional file 1). To enhance the precision of our data retrieval, we limited our search to the titles and abstracts of publications within the Scopus database, covering all years up to December 31, 2023. This restriction was applied to avoid obtaining numerous irrelevant publications, often referred to as false positive data, which would have occurred if we had extended our search to include all fields such as keywords or the full text of publications. While this approach significantly improves specificity, it may slightly reduce sensitivity [21, 22]. Additionally, we excluded any publications that had been published as errata.
Bibliometric analysis
Bibliometric data, encompassing the annual volume and types of publications, originating countries and institutions, funding sources, journal impact factors, and citation counts, were compiled into a Microsoft Excel spreadsheet for subsequent analysis and tabulation (Additional file 2). Parameters for analysis included publication date, type, international collaboration patterns, affiliations, journals, impact factors (IF), h-index, and citation frequency. International collaborations were identified based on affiliations spanning multiple countries. To assess the impact and productivity of research on bariatric surgery and the gut microbiota, bibliometric indicators such as the h-index and IF were utilized. The h-index, conceived by Hirsch in 2005, offers a more comprehensive evaluation of academic achievement, with a higher value indicating greater influence [23]. Journal impact factors (IFs) were sourced from the 2023 Journal Citation Report (JCR; Clarivate Analytics) [24]. As of the data collection cutoff date (March 1, 2024), only the top ten publications in terms of each of the parameters are represented in the Tables, which are ranked in standard competition rankings and which places the most prolific publication in position 1. Furthermore, with such a long period of time since the publication of some articles, it is possible that the frequency of citations for some of them was high. Therefore, an adjusted citation index value was calculated for each article, which reflects the frequency of references taking into account the publication time [25]. Thus, by grasping the meaning behind these metrics, readers can further develop their critical thinking skills as consumers of science: they can better comprehend and interpret the scientific literature, assess the reliability and applicability of results, and gain more insight into the links between bariatric surgery and the gut microbiota.
Visualization analysis
The data visualization in this study was performed using VOSviewer version 1.6.20 software. VOSviewer is a software tool that allows the visualization and analysis of bibliometric data, coauthorship networks, and term co-occurrence in scientific papers [26, 27]. It is utilized by researchers to find patterns and relationships in data, thus enabling a deeper understanding of the hierarchical structure in the field. Since version 1.6.20 gained a coauthorship network across countries, term co-occurrence in the titles and/or abstracts of scientific papers can be established. As a result, one can determine the leading subjects in a specific scientific discipline and the correlation among these subjects.
VOSviewer was used here to construct coauthorship networks between different countries and term co-occurrences in the title and/or abstract. In VOSviewer, different clusters within the network map can be represented by different colors to make identifying different groups of nodes easier. For example, the size of the circles in the network can represent the number of documents associated with a particular node. In contrast, the thickness of the connection lines between nodes can indicate the strength of the link, such as the number of collaborations between two nodes. While VOSviewer remains favored by experts and scholars in scientometrics and domain analysis [28], it is crucial to acknowledge its limitations and potential biases in bibliometric analysis, such as limited visibility [29] and the possibility of subjective interpretation [30].
Statistical and validation analysis
The data are presented as averages or numbers, with percentages in parentheses. An exponential regression was performed on the publishing trend to evaluate the growth pattern of the research output. The reliability of our method was evaluated using a pilot sample (n = 100 documents) to check the document type and compare it with the sources. Two separate researchers (i.e., journals) carried out this assessment. SZ and SA each conducted their evaluations of the information included within the 100 documents that were chosen. Cohen’s kappa test revealed a correlation coefficient of 0.933 between the two researchers. Therefore, it has been claimed that more than 90% reliability should be achieved [31,32,33]. The fact that the two observers and two methodologies achieved such a high level of concordance demonstrates that our method was reliable. These methodologies have been utilized in prior bibliometric studies [34,35,36,37]. Microsoft Excel 2013 and the Statistical Package for Social Sciences version 21 (IBM-SPSS) were used throughout the statistical analyses.
Results
An overview of publications
Between 2009 and 2023, a total of 555 articles were published on bariatric surgery and the gut microbiota. The original articles comprised 55.32% of all publications on bariatric surgery and the gut microbiota, followed by reviews at 37.30%. Figure 1 depicts the annual number of bariatric surgery and gut microbiota-related publications. In general, despite fluctuations over the past 15 years, the number of annual papers increased from 2 in 2009 to 71 in 2023, with a peak in 2022 with 85 publications. There is substantial evidence supporting this trend, indicated by a statistically significant moderate positive correlation (R2 = 0.8683, p < 0.001) between the publication year and the number of publications pertaining to this subject.
Analysis of countries
Seventy-one countries contributed to research related to bariatric surgery and the gut microbiota, of which the top 10 combined countries accounted for 76.22% (n = 423) of all published articles. The top 10 countries in terms of bariatric surgery and gut microbiota are listed in Table 1. The three countries with the most publications were the USA (n = 156; 28.11%), China (n = 73; 13.15%), and the UK (n = 52; 9.37%). Figure 2 illustrates a network visualization map representing collaborative relationships among countries, wherein each country has made a minimum contribution of 10 documents. The map encompasses 21 countries. The centrality of the collaboration map showed that the top countries were the USA, followed by the UK.
Country coauthorship network of publications related to bariatric surgery and the gut microbiota. There were 21 participating countries, each of which contributed a minimum of 10 publications. Publications with international authors are most prevalent in nations with large node sizes. The thickness of the connecting line indicates the strength of the research collaboration
Analysis of institutions
Within the scope of research in this particular field, a total of 2217 institutions participated, and the top 10 deciles among them collectively produced 18.01% (n = 100) of the entire body of published articles. Noteworthy contributors include INSERM, leading with 27 papers, followed by Imperial College London (n = 24 papers), Sorbonne Université (n = 22 papers), and AP-HP Assistance Publique - Hopitaux de Paris (n = 21 papers) (see Table 2). It is pertinent to highlight France’s substantial presence, encompassing four institutions on the list, while Spain exhibited two, and the United Kingdom, Sweden, Denmark, and Netherlands each contributed one institution to the distinguished cohort of top-ranking entities.
Analysis of funding agencies
Table 3 shows the top 10 most distributed funding agencies in research related to bariatric surgery and the gut microbiota. The top 10 funding sources contributed approximately one-third of the funding (n = 164). The top three funding agencies with the most publications were the National Institutes of Health (n = 46; 8.29%), National Institute of Diabetes and Digestive and Kidney Diseases (n = 37; 6.67%), and National Natural Science Foundation of China (n = 32; 5.77%).
Analysis of journals
The findings are presented in 287 journals, with Table 4 showing the top ten journals contributing to research on bariatric surgery and the gut microbiota. Approximately 27.75% of the articles were featured in these prominent publications. Most of the articles were published on Obesity Surgery (n = 38, 6.85%), followed by Surgery for Obesity and Related Diseases (n = 33, 5.95%), and Nutrients (n = 25, 4.50%).
Analysis of citations
The retrieved documents had a total of 23,494 citations, each earning an average of 42.33. The h-index for the retrieved studies was 71. Among these documents, 84 had no citations, whereas 51 garnered 100 citations or more. The combined citation count of the top ten articles, ordered by their number of citations, reached 6746 citations [38,39,40,41,42,43,44,45,46,47]. The total number of citations for these publications ranged from 305 to 1,480 (Table 5). In the list of the top 10 most-cited articles, the adjusted citation index varied between 30.5 and 151.67. When factoring in the adjusted citation index, the paper that stood out as the most influential identified gut microbial species associated with obesity, which in turn were linked to alterations in circulating metabolites [40].
Term co-occurrence cluster analysis of research hotspots
The co-occurrence analysis term provided a comprehensive overview of the predominant themes explored in research concerning bariatric surgery and the gut microbiota. Using VOSviewer, 178 terms occurring at least 20 times were identified through an examination of the titles and abstracts of the included manuscripts and were presented as a bubble map. This analysis aimed to identify the most commonly used terms. The VOSviewer term co-occurrence visualization map categorizes all terms into clusters and employs different colors to highlight distinctions between these clusters (see Fig. 3). The analysis identified three distinct clusters: first, the green cluster, which focused on the effects of bariatric surgery on the gut microbiota composition in relation to obesity remission; second, the blue cluster, which investigated the correlation between glucose metabolism, circulating bile acids, gut hormones, and the reshaping of the gut microbiome; and third, the red cluster, which explored the connections between alterations in the gut microbiota and insulin resistance. A color bar located in the lower right-hand corner of the map facilitates overlay visualization. Keywords are shaded differently based on their average publication year (see Fig. 4). For instance, the term shaded yellow‒green suggests that research on the effects of bariatric surgery on the gut microbiota composition in relation to obesity remission has gained increasing attention in recent years and may soon become a focal point of study.
Discussion
In this investigation, we searched the Scopus database for bibliographic information on published research on topics concerning bariatric surgery and the gut microbiota to explore the emerging trends in this field. In total, 474 different documents published between 2009 and 2023 were found. Even if there were only a few publications initially, the annual number of publications has exhibited a trend that indicates a quickly rising pace, particularly in recent years.
It is evident that they are interested in understanding the importance of analyzing the impact of bariatric surgery on the gut microbiota across various publications, countries, institutions, funding sources, journals, impact factors, and citations, as well as the key findings, differences across funding sources, journals, and impact factors, implications for readers and researchers, and associated challenges and limitations.
Bariatric surgery has been found to induce significant modifications to the gut microbiota, resulting in changes in both the timing and distribution of intestinal bacteria after surgery [48]. The gut microbiota is recognized as a key contributor to the metabolic enhancements observed after bariatric surgery, and alterations in its composition are linked to the prognosis of individuals undergoing such procedures [48]. The composition of the gut microbiota prior to surgery may serve as an indicator of the patient’s response to bariatric surgery, and shortly after surgery, patients who achieve successful weight loss exhibit an increase in microorganisms associated with positive effects on host metabolism [49]. Bariatric surgery leads to enduring weight loss and the resolution of obesity-related complications, with the microbiome potentially playing a role in this process [6, 50]. Various studies have investigated the impact of bariatric surgery on the composition and function of the gut microbiota, with some focusing on functional aspects rather than changes in composition [51]. The correlation between the gut microbiota and outcomes of bariatric surgery has prompted the exploration of personalized medicine, as specific patterns in the gut microbiota could predict remission of type 2 diabetes or failure to lose weight postsurgery [51]. Thus, examining the effects of bariatric surgery on the gut microbiota is crucial for comprehending its role in surgical outcomes and for potentially predicting responses to surgery and devising personalized treatment strategies [49, 51].
All of the top 3 contributors to the research on bariatric surgery and gut microbiota were from the United States, China, and the United Kingdom. Indeed, similar trends were observed in other obesity treatment-focused studies [52,53,54,55,56,57,58]. This finding is likely related to the greater number of bariatric surgeries being conducted in the US than in other countries [59]. A variety of factors are likely to contribute to the high publication rates. First, the gut microbiota is a particularly quickly developing field within obesity treatment research. Indeed, the US, China, and the UK are three leading publishers in the identified area, with the United States being first [60]. Second, developed countries have high international publication collaboration, which likely drove the high number of publications [60]. Third, the United States has also demonstrated consistent leadership in almost every measure of productivity, from active institutions, even journals, and authors to citations and collaboration across active institutions abroad [55]. Significant correlations were found between the number of publications in bariatric surgery and factors such as gross domestic product (GDP), indicating that research infrastructure and funding play a crucial role in publication rates [55]. Furthermore, it can be inferred that the high publication rates in these countries may be influenced by collaborative efforts and partnerships with other countries, especially given the emphasis on international collaboration [60].
There have been several incidents of obesity in the Western Hemisphere, primarily in the USA and the United Kingdom, over the years, the vast majority of which have been neglected. An illustration of this can be found in 2020, when the rate of obesity in the USA (based on data from 2017 to 2018) reached 42% among adults, with severe obesity (BMI ≥ 40 kg/m2) coming close to reaching double digits at 9% [61]. It is expected that the prevalence of morbid obesity (BMI ≥ 40 kg/m2) will increase to 5%, 8%, and 11% in Scotland, England, and Wales, respectively, by 2035 [62]. The equivalent statistics in China are currently seeing a highly concerning rise in obesity, most notably abdominal obesity [63]. The prevalence of overweight and obesity in China was 28.1% and 5.2%, respectively, with considerable variation across provinces [64]. A study spanning from 2013 to 2018 reported an average annual increase in the prevalence of adult obesity in China of 3.2%, with higher rates among men and specific age and education subgroups [65].
Research on the gut microbiota has grown rapidly in recent years, and China has made significant contributions to this field. In recent years, Chinese researchers have published numerous studies on the gut microbiota and its role in various health conditions, including obesity, metabolic disorders, digestive diseases, and even mental health [66,67,68,69]. For example, some studies have explored the differences in the gut microbiota between healthy individuals and those with obesity, metabolic disorders, and other related diseases and how these differences could contribute to the development and progression of these conditions. Other studies have investigated the potential of probiotics and prebiotics in modulating the gut microbiota composition and improving health outcomes [70, 71].
On the other hand, as was found by earlier bibliometric research [72,73,74,75,76], the United States of America is currently in first place for international collaborative papers. Not only was the relevance of international collaboration centered on the advancement of knowledge and the enhancement of research capacity [77], but it also had the potential to increase citation rates and improve the quality of research [78].
The topic of “effects of bariatric surgery on gut microbiota composition related to obesity remission” was one of the main hot topics in the current study. In reviewing the literature, we found that bariatric surgery produced long-lasting changes in the gut microbiota composition and the fecal metabolome that may be related to the remission of obesity [79]. Energy extraction from dietary nutrients is one of the primary contributions of the gut microbiota to the emergence of obesity. Short-chain fatty acids derived from plant polysaccharides are among the energetic metabolites that the microbiota of obese people is thought to produce in greater quantities [80]. Additionally, the microbial metabolism of dietary nutrients may produce potentially toxic chemicals that contribute to establishing an obesity-related low-grade inflammatory state [81]. In addition to increasing the abundance of favorable Verrucomicrobia species and reducing the Firmicutes/Bacteroidetes ratio, which are strongly linked to obesity, weight loss through dietary treatments also promotes functional changes in the microbiota. It modifies its derived metabolites [82, 83]. In line with the positive effects of this treatment, bariatric surgery may also cause a shift in the metabolic capacity of the gut microbiota toward a lean-like phenotype, both in terms of composition and functionality [79].
Another subject that has received much attention is the association between glucose metabolism and circulating bile acid and gut hormones and remodeling of the gut microbiome. The anatomical changes resulting from bariatric surgery, especially when excluding the long part of the small intestine from food passages, will lead to an increase in the concentration of pure bile acid (without being mixed with food) that reaches the intestine and subsequently affects glucose metabolism and body mass loss [84,85,86,87]. Changes in the gut microbiota can impact bile acid levels, as the microbiota plays a vital role in converting primary bile acids to secondary bile acids. This process may affect human glucose metabolism and enhance insulin sensitivity by interacting with membrane or nuclear receptors in various organs, including the liver, intestines, adipose tissue, skeletal muscle, and pancreas [88, 89]. Bariatric surgery is linked to an increased release of gut hormones postmeal, particularly GLP-1, which boosts insulin secretion and glucose clearance [90]. Elevated levels of circulating gut hormones following bariatric surgery have been shown to induce temporary remission of type 2 diabetes even before weight loss occurs [91]. These hormone levels increase shortly after bariatric procedures such as Roux-en-Y gastric bypass and sleeve gastrectomy, potentially serving as crucial regulators of postoperative changes in eating patterns and glucose balance [92].
Another hot topic is “links between gut microbiota alterations and insulin resistance.” Moreover, alterations in the content of the gut microbiota after bariatric surgery affect and reduce the amount of adipose tissue. This long-term effect on reducing adipose tissue leads to reduced inflammation and improved insulin sensitivity. Research has confirmed the link between bariatric surgery and the management of diabetes in people who have both diabetes and significant obesity. Furthermore, these surgical procedures have been shown to be effective at preventing both microvascular and macrovascular complications among this specific group of individuals. Bariatric surgery may prevent diabetes in those who suffer from insulin resistance. These findings seem to exert this beneficial effect by modulating insulin sensitivity and the gut microbiota in surgical patients, possibly via a decreased inflammatory response and improved signaling of gut hormones [93,94,95]. Evidence further indicates that bariatric surgeons can manipulate gut bacteria, which can transform how sugar is processed in the body and enable energy homeostasis [96,97,98]. Unfortunately, the connection between a change in the gut microbiome of an individual and increased insulin sensitivity after surgery remains unknown [98, 99]. Thus, learning more about how to manipulate gut bacteria may help improve the metabolic health of obese patients. After undergoing bariatric surgery, alterations in the gut microbiota may contribute to improved insulin resistance. Therefore, further research is necessary to comprehend the mechanisms underlying this relationship.
Strengths and limitations
The Scopus database was used for this study, the first bibliometric evaluation of the literature on bariatric surgery and the gut microbiota. This study visualizes the current state, hot spots, and growing trends of bariatric surgery and the gut microbiota from 2009 to 2023 to serve as a rapid and unbiased reference for interested scholars. However, there will always be certain limitations. First, the Scopus database is dynamically and regularly updated. Even if all database searches were completed in a single day, some fresh data might still be missing. Second, the dispersion of counts and clusters is caused by the several ways that terms can be expressed. Although these issues were addressed by the software’s merging and normalization features, they cannot be completely avoided. Third, another limitation of this study may be that the publications are ranked based on total citations rather than annual citation averages. Furthermore, the total number of citations fluctuates and increases with time. Other recently released high-quality publications did not have enough citations to be included in the top 10 most referenced papers. Publications from the most recent years could not be included due to space constraints; nevertheless, this does not make the articles published in those years any less significant. Fourth, as a result, readers should continue to take into account the likelihood of false-positive and negative results. It is anticipated that the use of title-abstract searches will reduce any research errors.
Conclusions
We found that the annual output of publications related to bariatric surgery and the gut microbiome has grown dramatically over the past few years. Therefore, for better clinical care of obese people seeking bariatric surgery, physicians and dietitians should have substantial and complete professional knowledge and abilities. Future scholars and practitioners in the microbiome or bariatric surgery may find this study useful due to its potential to provide a thorough review and reference. In addition, this research may immediately aid newcomers in understanding the current popular research areas in the discipline and the leading research countries/institutions. Simultaneously, this research will aid scholars and the general public in better understanding the field’s dynamic evolution, promoting collaboration across academic fields.
Data availability
All the data generated or analyzed during this study are included in this published article. In addition, other data sets used during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- WHO:
-
World Health Organization
- BMI:
-
Body mass index
- T2DM:
-
Type 2 diabetes mellitus
- MeSH:
-
Medical subject headings
- IBS:
-
Irritable bowel syndrome
- IBD:
-
Inflammatory bowel disease
- FMT:
-
Fecal microbiota transplantation
References
The Lancet Gastroenterology H. Obesity: another ongoing pandemic. Lancet Gastroenterol Hepatol. 2021;6(6):411.
Carmona MN, Santos-Sousa H, Lindeza L, Sousa-Pinto B, Nogueiro J, Pereira A, Carneiro S, Costa-Pinho A, Lima-da-Costa E, Preto J, et al. Comparative effectiveness of Bariatric Surgeries in patients with type 2 diabetes Mellitus and BMI >/= 25 kg/m(2): a systematic review and network Meta-analysis. Obes Surg. 2021;31(12):5312–21.
Jimenez A, Ibarzabal A, Moize V, Pane A, Andreu A, Molero J, de Hollanda A, Flores L, Ortega E, Lacy A, et al. Ten-year outcomes after Roux-en-Y gastric bypass and sleeve gastrectomy: an observational nonrandomized cohort study. Surg Obes Relat Dis. 2019;15(3):382–8.
Miras AD, le Roux CW. Mechanisms underlying weight loss after bariatric surgery. Nat Rev Gastroenterol Hepatol. 2013;10(10):575–84.
Ma IT, Madura JA. 2nd: gastrointestinal complications after bariatric surgery. Gastroenterol Hepatol (N Y). 2015;11(8):526–35.
Luijten J, Vugts G, Nieuwenhuijzen GAP, Luyer MDP. The importance of the Microbiome in bariatric surgery: a systematic review. Obes Surg. 2019;29(7):2338–49.
Crommen S, Mattes A, Simon MC. Microbial Adaptation due to gastric bypass surgery: the nutritional impact. Nutrients 2020, 12(4).
Akagbosu CO, Nadler EP, Levy S, Hourigan SK. The role of the gut Microbiome in Pediatric obesity and bariatric surgery. Int J Mol Sci 2022, 23(23).
Kaska L, Sledzinski T, Chomiczewska A, Dettlaff-Pokora A, Swierczynski J. Improved glucose metabolism following bariatric surgery is associated with increased circulating bile acid concentrations and remodeling of the gut microbiome. World J Gastroenterol. 2016;22(39):8698–719.
Ulker I, Yildiran H. The effects of bariatric surgery on gut microbiota in patients with obesity: a review of the literature. Biosci Microbiota Food Health. 2019;38(1):3–9.
Stefura T, Zapala B, Gosiewski T, Skomarovska O, Pedziwiatr M, Major P. Changes in the composition of oral and intestinal Microbiota after Sleeve Gastrectomy and Roux-En-Y gastric bypass and their impact on outcomes of bariatric surgery. Obes Surg. 2022;32(5):1439–50.
Ellegaard O, Wallin JA. The bibliometric analysis of scholarly production: how great is the impact? Scientometrics 2015, 105(3):1809–31.
Schotten M, El Aisati M, Meester WJN, Steiginga S, Ross CA. A brief history of Scopus: The world’s largest abstract and citation database of scientific literature. In: Research Analytics: Boosting University Productivity and Competitiveness through Scientometrics edn.; 2017: 31–58.
Baas J, Schotten M, Plume A, Côté G, Karimi R. Scopus as a curated, high-quality bibliometric data source for academic research in quantitative science studies. Quant Sci Stud. 2020;1(1):377–86.
Stahlschmidt S, Stephen D. Varying resonance chambers: A comparison of citation-based valuations of duplicated publications in Web of Science and Scopus. In: 17th International Conference on Scientometrics and Informetrics, ISSI 2019 - Proceedings: 2019; 2019: 1698–1709.
Falagas ME, Pitsouni EI, Malietzis GA, Pappas G. Comparison of PubMed, Scopus, web of Science, and Google Scholar: strengths and weaknesses. FASEB J. 2008;22(2):338–42.
Kulkarni AV, Aziz B, Shams I, Busse JW. Comparisons of citations in web of Science, Scopus, and Google Scholar for articles published in general medical journals. JAMA. 2009;302(10):1092–6.
Yang J, Deng Y, Cai Y, Liu Y, Peng L, Luo Z, Li D. Mapping trends and hotspot regarding gastrointestinal microbiome and neuroscience: a bibliometric analysis of global research (2002–2022). Front Neurosci. 2022;16:1048565.
van Veldhuisen SL, Gorter TM, van Woerden G, de Boer RA, Rienstra M, Hazebroek EJ, van Veldhuisen DJ. Bariatric surgery and cardiovascular disease: a systematic review and meta-analysis. Eur Heart J. 2022;43(20):1955–69.
Cӑtoi AF, Vodnar DC, Corina A, Nikolic D, Citarrella R, Perez-Martinez P, Rizzo M. Gut microbiota, obesity and bariatric surgery: current knowledge and future perspectives. Curr Pharm Des. 2019;25(18):2038–50.
Sweileh WM. A bibliometric analysis of global research output on health and human rights (1900–2017). Glob Health Res Policy. 2018;3(1):30.
Sweileh WM, Al-Jabi SW, Zyoud SH, Shraim NY, Anayah FMA, Sawalha AF, AbuTaha AS. Bibliometric analysis of global publications in medication adherence (1900–2017). Int J Pharm Pract. 2019;27(2):112–20.
Hirsch JE. An index to quantify an individual’s scientific research output. Proc Natl Acad Sci U S A. 2005;102(46):16569–72.
Clarivate. Journal Citation Reports. 2023. https://jcr.clarivate.com/jcr/home (accessed March 5 2024).
Zyoud SH, Waring WS, Al-Jabi SW, Sweileh WM, Awang R. The 100 most influential publications in Paracetamol poisoning treatment: a bibliometric analysis of human studies. Springerplus. 2016;5(1):1534.
van Eck NJ, Waltman L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics. 2010;84(2):523–38.
Van Eck NJ, Waltman L. Text mining and visualization using VOSviewer. ISSI Newsl. 2011;7(3):50–4.
Li J, Wei R. VOSviewer Application Status and its knowledge base. J Libr Inf Sci Agric. 2022;34(6):61–71.
Tomaszewski R. Visibility, impact, and applications of bibliometric software tools through citation analysis. Scientometrics. 2023;128(7):4007–28.
Nobanee H, Alhajjar M, Alkaabi MA, Almemari MM, Alhassani MA, Alkaabi NK, Alshamsi SA. AlBlooshi HH: a bibliometric analysis of objective and subjective risk. Risks 2021, 9(7).
Zyoud SH, Al-Jabi SW, Sweileh WM, Awang R, Waring WS. Bibliometric profile of the global scientific research on methanol poisoning (1902–2012). J Occup Med Toxicol. 2015;10(1):17.
Allison JJ, Wall TC, Spettell CM, Calhoun J, Fargason CA Jr., Kobylinski RW, Farmer R, Kiefe C. The art and science of chart review. Jt Comm J Qual Improv. 2000;26(3):115–36.
Banks NJ. Designing medical record abstraction forms. Int J Qual Health Care. 1998;10(2):163–7.
Zyoud SH. Global landscape of COVID-19 research: a visualization analysis of randomized clinical trials. Clin Exp Med. 2024;24(1):14.
Zyoud SH. Mapping the landscape of research on insulin resistance: a visualization analysis of randomized clinical trials. J Health Popul Nutr. 2024;43(1):6.
Sweileh WM. Analysis and mapping of Scientific Literature on Detention and Deportation of International migrants (1990–2022). J Immigr Minor Health. 2023;25(5):1065–76.
Sweileh WM. Technology-based interventions for tobacco smoking prevention and treatment: a 20-year bibliometric analysis (2003–2022). Subst Abuse Treat Prev Policy. 2024;19(1):13.
Liou AP, Paziuk M, Luevano JM Jr., Machineni S, Turnbaugh PJ, Kaplan LM. Conserved shifts in the gut microbiota due to gastric bypass reduce host weight and adiposity. Sci Transl Med. 2013;5(178):178ra141.
Furet JP, Kong LC, Tap J, Poitou C, Basdevant A, Bouillot JL, Mariat D, Corthier G, Dore J, Henegar C, et al. Differential adaptation of human gut microbiota to bariatric surgery-induced weight loss: links with metabolic and low-grade inflammation markers. Diabetes. 2010;59(12):3049–57.
Liu R, Hong J, Xu X, Feng Q, Zhang D, Gu Y, Shi J, Zhao S, Liu W, Wang X, et al. Gut microbiome and serum metabolome alterations in obesity and after weight-loss intervention. Nat Med. 2017;23(7):859–68.
Castaner O, Goday A, Park YM, Lee SH, Magkos F, Shiow STE, Schroder H. The gut Microbiome Profile in obesity: a systematic review. Int J Endocrinol. 2018;2018:4095789.
Kong LC, Tap J, Aron-Wisnewsky J, Pelloux V, Basdevant A, Bouillot JL, Zucker JD, Dore J, Clement K. Gut microbiota after gastric bypass in human obesity: increased richness and associations of bacterial genera with adipose tissue genes. Am J Clin Nutr. 2013;98(1):16–24.
Festi D, Schiumerini R, Eusebi LH, Marasco G, Taddia M, Colecchia A. Gut microbiota and metabolic syndrome. World J Gastroenterol. 2014;20(43):16079–94.
Zhang H, DiBaise JK, Zuccolo A, Kudrna D, Braidotti M, Yu Y, Parameswaran P, Crowell MD, Wing R, Rittmann BE, et al. Human gut microbiota in obesity and after gastric bypass. Proc Natl Acad Sci U S A. 2009;106(7):2365–70.
Cani PD, Osto M, Geurts L, Everard A. Involvement of gut microbiota in the development of low-grade inflammation and type 2 diabetes associated with obesity. Gut Microbes. 2012;3(4):279–88.
Graessler J, Qin Y, Zhong H, Zhang J, Licinio J, Wong ML, Xu A, Chavakis T, Bornstein AB, Ehrhart-Bornstein M, et al. Metagenomic sequencing of the human gut microbiome before and after bariatric surgery in obese patients with type 2 diabetes: correlation with inflammatory and metabolic parameters. Pharmacogenomics J. 2013;13(6):514–22.
Tremaroli V, Karlsson F, Werling M, Stahlman M, Kovatcheva-Datchary P, Olbers T, Fandriks L, le Roux CW, Nielsen J, Backhed F. Roux-en-Y gastric bypass and Vertical Banded Gastroplasty Induce Long-Term changes on the human gut Microbiome contributing to Fat Mass Regulation. Cell Metab. 2015;22(2):228–38.
Cao C, Shao YK, Yao QY. [Role and change of the gut microbiota after bariatric surgery]. Zhonghua Wei Chang Wai Ke Za Zhi. 2022;25(7):648–53.
Gutierrez-Repiso C, Garrido-Sanchez L, Alcaide-Torres J, Cornejo-Pareja I, Ocana-Wilhelmi L, Garcia-Fuentes E, Moreno-Indias I, Tinahones FJ. Predictive role of gut microbiota in weight loss achievement after bariatric surgery. J Am Coll Surg. 2022;234(5):861–71.
Mika A, Janczy A, Waleron K, Szymanski M, Kaska L, Sledzinski T. The impact of the interplay of the intestinal microbiome and diet on the metabolomic and health outcomes of bariatric surgery. Obes Rev. 2022;23(8):e13455.
Gutierrez-Repiso C, Moreno-Indias I, Tinahones FJ. Shifts in gut microbiota and their metabolites induced by bariatric surgery. Impact of factors shaping gut microbiota on bariatric surgery outcomes. Rev Endocr Metab Disord. 2021;22(4):1137–56.
Choi HS, Chun HJ. Recent trends in endoscopic bariatric therapies. Clin Endosc. 2017;50(1):11–6.
Dabi Y, Darrigues L, Katsahian S, Azoulay D, De Antonio M, Lazzati A. Publication trends in bariatric surgery: a bibliometric study. Obes Surg. 2016;26(11):2691–9.
Ozsoy Z, Demir E. Which Bariatric Procedure is the most popular in the World? A bibliometric comparison. Obes Surg. 2018;28(8):2339–52.
Ozsoy Z, Demir E. The evolution of Bariatric Surgery Publications and Global Productivity: a bibliometric analysis. Obes Surg. 2018;28(4):1117–29.
Paolino L, Pravettoni R, Epaud S, Ortala M, Lazzati A. Comparison of Surgical Activity and Scientific publications in bariatric surgery: an epidemiological and Bibliometric Analysis. Obes Surg. 2020;30(10):3822–30.
Toro-Huamanchumo CJ, Moran-Marinos C, Salazar-Alarcon JL, Barros-Sevillano S, Huamanchumo-Suyon ME, Salinas-Sedo G. Latin American research on bariatric surgery: a bibliometric study. Obes Surg. 2021;31(4):1869–76.
Zhao N, Tao K, Wang G, Xia Z. Global obesity research trends during 1999 to 2017: a bibliometric analysis. Med (Baltim). 2019;98(4):e14132.
Angrisani L, Santonicola A, Iovino P, Formisano G, Buchwald H, Scopinaro N. Bariatric surgery Worldwide 2013. Obes Surg. 2015;25(10):1822–32.
Yao H, Wan JY, Wang CZ, Li L, Wang J, Li Y, Huang WH, Zeng J, Wang Q, Yuan CS. Bibliometric analysis of research on the role of intestinal microbiota in obesity. PeerJ. 2018;6(6):e5091.
Hales CM, Carroll MD, Fryar CD, Ogden CL. Prevalence of obesity and severe obesity among adults: United States, 2017–2018. NCHS Data Brief 2020(360):1–8.
Keaver L, Xu B, Jaccard A, Webber L. Morbid obesity in the UK: a modelling projection study to 2035. Scand J Public Health. 2020;48(4):422–7.
Hemmingsson E. The unparalleled rise of obesity in China: a call to action. Int J Obes (Lond). 2021;45(5):921–2.
Zhang L, Wang Z, Wang X, Chen Z, Shao L, Tian Y, Zheng C, Li S, Zhu M, Gao R, et al. Prevalence of overweight and obesity in China: results from a cross-sectional study of 441 thousand adults, 2012–2015. Obes Res Clin Pract. 2020;14(2):119–26.
Zhao ZP, Zhang M, Li C, Yu MT, Zhang X, Wang LM, Zhou MG. [Growth rate of adult obesity prevalence in China and target population for prevention and control from 2013 to 2018]. Zhonghua Xin xue guan bing za zhi. 2024;52(1):34–41.
Xu Z, Jiang W, Huang W, Lin Y, Chan FKL, Ng SC. Gut microbiota in patients with obesity and metabolic disorders - a systematic review. Genes Nutr. 2022;17(1):2.
Wang Y, Li D, Jia Z, Hui J, Xin Q, Zhou Q, Cong W, Xu F. A Bibliometric Analysis of Research on the Links between Gut Microbiota and Atherosclerosis. Front Cardiovasc Med. 2022;9:941607.
Cabanillas-Lazo M, Quispe-Vicuna C, Barja-Ore J, Fernandez-Giusti A, Munive-Degregori A, Retamozo-Siancas Y, Guerrero ME, Mayta-Tovalino F. A 10-Year Bibliometric Analysis of Global Research on Gut Microbiota and Parkinson’s Disease: Characteristics, Impact, and Trends. Biomed Res Int 2022, 2022:4144781.
Wu W, Ouyang Y, Zheng P, Xu X, He C, Xie C, Hong J, Lu N, Zhu Y, Li N. Research trends on the relationship between gut microbiota and colorectal cancer: a bibliometric analysis. Front Cell Infect Microbiol. 2022;12:1027448.
Liu Y, Wang J, Wu C. Modulation of Gut Microbiota and Immune System by Probiotics, Pre-biotics, and post-biotics. Front Nutr. 2021;8:634897.
Xu L, Yang CS, Liu Y, Zhang X. Effective regulation of Gut Microbiota with Probiotics and Prebiotics May prevent or alleviate COVID-19 through the gut-lung Axis. Front Pharmacol. 2022;13:895193.
Liu F, Lin A, Wang H, Peng Y, Hong S. Global research trends of geographical information system from 1961 to 2010: a bibliometric analysis. Scientometrics. 2015;106(2):751–68.
Cheng T, Zhang G. Worldwide research productivity in the field of rheumatology from 1996 to 2010: a bibliometric analysis. Rheumatology (Oxford). 2013;52(9):1630–4.
Zyoud SH, Al-Jabi SW. Mapping the situation of research on coronavirus disease-19 (COVID-19): a preliminary bibliometric analysis during the early stage of the outbreak. BMC Infect Dis. 2020;20(1):561.
Zyoud SH. The arab region’s contribution to global COVID-19 research: bibliometric and visualization analysis. Global Health. 2021;17(1):31.
Sweileh WM. Bibliometric analysis of peer-reviewed literature on antimicrobial stewardship from 1990 to 2019. Global Health. 2021;17(1):1.
Soreide K, Alderson D, Bergenfelz A, Beynon J, Connor S, Deckelbaum DL, Dejong CH, Earnshaw JJ, Kyamanywa P, Perez RO, et al. Strategies to improve clinical research in surgery through international collaboration. Lancet. 2013;382(9898):1140–51.
Stek PE, van Geenhuizen MS. The influence of international research interaction on national innovation performance: a bibliometric approach. Technol Forecast Soc Change. 2016;110:61–70.
Juarez-Fernandez M, Roman-Saguillo S, Porras D, Garcia-Mediavilla MV, Linares P, Ballesteros-Pomar MD, Urioste-Fondo A, Alvarez-Cuenllas B, Gonzalez-Gallego J, Sanchez-Campos S et al. Long-Term Effects of Bariatric Surgery on Gut Microbiota Composition and Faecal Metabolome Related to Obesity Remission. Nutrients 2021, 13(8).
de la Cuesta-Zuluaga J, Mueller NT, Alvarez-Quintero R, Velasquez-Mejia EP, Sierra JA, Corrales-Agudelo V, Carmona JA, Abad JM, Escobar JS. Higher Fecal Short-Chain Fatty Acid Levels Are Associated with Gut Microbiome Dysbiosis, Obesity, Hypertension and Cardiometabolic Disease Risk Factors. Nutrients 2018, 11(1).
Tilg H, Zmora N, Adolph TE, Elinav E. The intestinal microbiota fuelling metabolic inflammation. Nat Rev Immunol. 2020;20(1):40–54.
Rinninella E, Cintoni M, Raoul P, Ianiro G, Laterza L, Lopetuso LR, Ponziani FR, Gasbarrini A, Mele MC. Gut microbiota during Dietary restrictions: New insights in Non-communicable diseases. Microorganisms 2020, 8(8).
Heianza Y, Sun D, Smith SR, Bray GA, Sacks FM, Qi L. Changes in gut microbiota-related metabolites and long-term successful weight loss in response to weight-loss diets: the POUNDS Lost Trial. Diabetes Care. 2018;41(3):413–9.
Quercia I, Dutia R, Kotler DP, Belsley S, Laferrere B. Gastrointestinal changes after bariatric surgery. Diabetes Metab. 2014;40(2):87–94.
Sweeney TE, Morton JM. Metabolic surgery: action via hormonal milieu changes, changes in bile acids or gut microbiota? A summary of the literature. Best Pract Res Clin Gastroenterol. 2014;28(4):727–40.
Schmidt JB, Pedersen SD, Gregersen NT, Vestergaard L, Nielsen MS, Ritz C, Madsbad S, Worm D, Hansen DL, Clausen TR, et al. Effects of RYGB on energy expenditure, appetite and glycaemic control: a randomized controlled clinical trial. Int J Obes (Lond). 2016;40(2):281–90.
Penney NC, Kinross J, Newton RC, Purkayastha S. The role of bile acids in reducing the metabolic complications of obesity after bariatric surgery: a systematic review. Int J Obes (Lond). 2015;39(11):1565–74.
Fiorucci S, Distrutti E. Bile acid-activated receptors, intestinal microbiota, and the treatment of metabolic disorders. Trends Mol Med. 2015;21(11):702–14.
Dutia R, Embrey M, O’Brien CS, Haeusler RA, Agenor KK, Homel P, McGinty J, Vincent RP, Alaghband-Zadeh J, Staels B, et al. Temporal changes in bile acid levels and 12alpha-hydroxylation after Roux-en-Y gastric bypass surgery in type 2 diabetes. Int J Obes (Lond). 2015;39(5):806–13.
Finelli C, Padula MC, Martelli G, Tarantino G. Could the improvement of obesity-related co-morbidities depend on modified gut hormones secretion? World J Gastroenterol. 2014;20(44):16649–64.
Will S, Hornigold DC, Baker DJ, Coghlan MP, Mesquita M, Trevaskis JL, Naylor J. Gut check on diabesity: leveraging gut mechanisms for the treatment of type 2 diabetes and obesity. Curr Opin Pharmacol. 2017;37:10–5.
Papamargaritis D, le Roux CW. Do gut hormones contribute to weight loss and glycaemic outcomes after bariatric surgery? Nutrients 2021, 13(3).
Rao RS, Yanagisawa R, Kini S. Insulin resistance and bariatric surgery. Obes Rev. 2012;13(4):316–28.
Kashyap SR, Gatmaitan P, Brethauer S, Schauer P. Bariatric surgery for type 2 diabetes: weighing the impact for obese patients. Cleve Clin J Med. 2010;77(7):468–76.
Lee PC, Tan HC, Pasupathy S, Ganguly S, Eng AKH, Nadkarni N, Tham KW. Effectiveness of bariatric surgery in diabetes prevention in high-risk Asian individuals. Singap Med J. 2018;59(9):472–5.
Dang JT, Mocanu V, Park H, Laffin M, Hotte N, Karmali S, Birch DW, Madsen KL. Roux-en-Y gastric bypass and sleeve gastrectomy induce substantial and persistent changes in microbial communities and metabolic pathways. Gut Microbes. 2022;14(1):2050636.
Munzker J, Haase N, Till A, Sucher R, Haange SB, Nemetschke L, Gnad T, Jager E, Chen J, Riede SJ, et al. Functional changes of the gastric bypass microbiota reactivate thermogenic adipose tissue and systemic glucose control via intestinal FXR-TGR5 crosstalk in diet-induced obesity. Microbiome. 2022;10(1):96.
Yadav J, Liang T, Qin T, Nathan N, Schwenger KJP, Pickel L, Xie L, Lei H, Winer DA, Maughan H, et al. Gut microbiome modified by bariatric surgery improves insulin sensitivity and correlates with increased brown fat activity and energy expenditure. Cell Rep Med. 2023;4(5):101051.
Ciobarca D, Catoi AF, Copaescu C, Miere D, Crisan G. Bariatric surgery in obesity: effects on Gut Microbiota and Micronutrient Status. Nutrients 2020, 12(1).
Acknowledgements
The author thanks An-Najah National University for all administrative assistance during the implementation of the project.
Funding
No support was received for conducting this study.
Author information
Authors and Affiliations
Contributions
Zyoud SH designed the study, collected the data, analyzed the data, made major contributions to the literature search and interpretation of the manuscript and drafted the manuscript; Barqawi A contributed to the conceptualization and methodology of the study, participated in the interpretation of the data, contributed to the manuscript writing and made revisions to the initial draft; Shakhshir M, Abushanab AS, Koni A, Khilfeh S, Shahwan M, Jairoun A, Abu Taha A, Abushamma F and Al-Jabi SW participated in the interpretation of the data, contributed to the manuscript writing and made revisions to the initial draft; all authors provided a critical review and approved the final manuscript before submission.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
Because the current study did not include any human interactions, it did not require the permission of the Ethics Committee.
Consent for publication
Not applicable.
Competing interests
The author declares that he has no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
41231_2024_173_MOESM1_ESM.xlsx
Additional file 1: Keywords and Phrases Utilized: Methodological Approach for Document Retrieval and Search Strategy in Scopus
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zyoud, S.H., Shakhshir, M., Barqawi, A. et al. Comprehensive visualization of bariatric surgery and gut microbiota research: a global analysis. transl med commun 9, 13 (2024). https://doi.org/10.1186/s41231-024-00173-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41231-024-00173-z