Abstract
Background
Entomopathogenic nematodes (EPNs) have a great potential as a biocontrol agent for a wide range of insect pests. EPNs have attracted much interest around the world to study their distribution, virulence, and usage in the IPM programs. This research was conducted to identify the steinernematids M.22 and M.313, which were isolated during the survey of EPNs in different agricultural and ecological systems, in the Syrian coastal region between 2017 and 2020. Additionally, to evaluate the virulence of these isolates against the last instar larvae of the greater wax moth, Galleria mellonella (Linnaeus 1758) (Lepidoptera: Pyralidae), three bioassays were carried out at 18 °C. The exposure time assay was performed by applying different times (15, 30, 45, and 60 min) using a specified concentration (400 IJs/Larva). The concentration–response and one-on-one assays were conducted by using different concentrations of the infective juveniles (IJs) (5, 15, 25, and 40 IJs/L) and an individual juvenile, respectively, for 72 h of exposure. LC50 and LT50 values were calculated.
Results
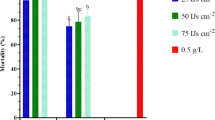
The steinernematids M.22 and M.313 were isolated from 2 of 821 soil samples (0.24%). The positive isolates were identified as Steinernema affine (Bovien, 1937), based on morphological, morphometric, and molecular characterization. Steinernema affine was recovered in the walnut orchard and grassland in AL-Kadmous area of Tartus governorate, Syria. The soil type was silty loamy and silty, PH was slightly acidic, and organic matter content was 6 and 4.1%. In the exposure time assay, the mortality ratio increased gradually by increasing time, M.22 and M.313 achieved the highest mortality ratios at 60 min, which were 90.16 and 91.49%, respectively. In the concentration–response assay, complete mortality was achieved at the concentration of 40 IJs/L for both isolates. Low mortality ratios were observed in the assay of one-on-one, 20 and 23.6%, respectively. The LC50 and LT50 values were calculated and resulted in (11.7 IJs/L, 33.47 min) and (11.5 IJs/L, 32.43 min), respectively. Nonsignificant difference between both isolates was recorded.
Conclusions
The results showed that the isolates were identical and identified as Steinernema affine. This is the first report of S. affine in Syria. It could be an effective biological control agent.
Similar content being viewed by others
Background
The entomopathogenic nematodes (EPNs) from Steinernematidae Travassos, 1927, and Heterorhabditidae Poinar, 1976, families have proven to be effective biological control agents against different agricultural insect pests (Gozel and Gozel 2016). The specialized symbiotic relationship between nematodes and bacteria, Heterorhabditis–Photorhabdus, Steinernema–Xenorhabdus, causes the rapid death of the insect host (Boemare 2002). Infective juveniles (IJs) of EPNs penetrate their hosts through their natural openings such as the mouth, anus, and spiracles. Also, Heterorhabditis sp. can penetrate through the cuticle of the host's body (Poinar and Grewal 2012). Once EPNs reach the insect hemocoel, they will release the symbiotic bacteria, which begin to grow and release cytotoxic, exoenzymes and antibiotics, causing septicemia and often killing the insect within 24–48 h (Koppenhöfer 2007). EPNs have two to three generations inside the host depending on the size of the insect as a food source. When the insect cadaver begins to decompose, the IJs emerge out, as dauer-juvenile to search for a new host in the soil. In IPM programs, the success of using EPNs is determined by the virulence of the EPNs species as well as the adaptation to the environment of the target insect (Shapiro et al. 2006). For this reason, several surveys have been carried out around the world to isolate locally adapted EPNs species (Laznik and Trdan 2012). These studies have found 102 Steinernema species, 22 Heterorhabditis species, and one Neosteinernema species, (Hazir et al. 2022). In Syria, eight species of valid EPNs include: Heterorhabditis bacteriophora Poinar, 1976, H. zealandica Poinar, 1990, H. indica Poinar, Karunakar & David, 1992, H. megidis Poinar, Jackson & Klein, 1987, Heterorhabditidodes chonmingensis (Zahang 2008), Steinernema cubanum Mráˇcek, Hernández & Boemare, 1994, S. feltiae (Filipjev, 1934), and S. carpocapsae (Weiser 1955) were recorded (Canhilal et al. 2006; Mosalam, 2009; Jawish 2016; Zeini et al. 2019). A survey in Syria’s coastal area (Latakia and Tartus governorates) was conducted, which have not been sufficiently investigated for EPNs species.
Therefore, the aim of this study was: (1) to identify Steinernema isolates based on morphological, morphometric, and molecular characterization and (2) to evaluate the virulence of both Steinernema isolates against the last instar of G. mellonella larvae.
Methods
Soil sampling and isolation of EPNs
The survey was conducted in the Syrian coastal region (4200 km2), which extends from the coastal line to 1000 m above the sea line level, during the spring and autumn months between 2017 and 2020. A total of 821 soil samples were collected randomly from different agricultural and ecological systems at the studied sites (Fig. 1). Each soil sample (approximate 2 kg) was composited of five random subsamples taken with a hand shovel to a depth of 30 cm over an area of 10 m2. The mixed samples were placed in separate polyethylene bags to prevent water loss and kept in coolers, as recommended by Kaya and Stock (1997), to be transported to the research laboratory, Department of Plant Protection, Tishreen University. EPNs were isolated from mixed soil samples using the “Galleria-bait method” (Bedding and Akhurst 1975). Ten larvae of G. mellonella were placed in each container, covered, turned upside down, and incubated in the dark at the temperature of a laboratory for 14 days. The larvae were checked every 48 h (Kaya and Stock 1997). Dead larvae, which exhibited marks of infection with EPNs, were collected and placed in a modified White trap after washing twice in distilled water (White 1929). The emerged IJs were cleaned and used to confirm Koch’s postulate of pathogenicity, then they were stored in tissue culture flasks at 10 °C. The soil characteristics (type, organic matter, PH) and habitats of the positive sampling sites are indicated in (Table 1).
Morphological and Morphometric characterization
Ten G. mellonella larvae were exposed to ca 2000 IJs in a Petri dish (9 cm diameter) lined with two moist filter papers (Whatman No. 1) (Nguyen 2007a). G. mellonella cadavers were dissected in Ringer’s solution (Sodium chloride 9.0 g, Potassium chloride 0.4 g, Calcium chloride 0.4 g, Sodium carbonate 0.2 g, Sterile distilled water to 1000 ml), to obtain the males and females of the first and second generations after 5 and 7 days post-infection, respectively. The infective juveniles (IJs) were collected during the first two days after emerging from the cadavers. For the light microscope observation and assessments, 20 specimens were examined from each life stage (males, females, and infective juveniles) alive. Additionally, specimens were killed and fixed in TAF (7 ml formalin, 2 ml Triethanolamine, 91 ml water) (Kaya and Stock 1997). Morphological traits and morphometric measurements were observed under a light microscope equipped with software (OPTIKA PROVIEW, Italy) using 4X, 10X, and 40X objectives.
The following morphometric characteristics were studied and measured, as mentioned by Nguyen (2007b). Total body length (L), maximum body diameter (MBD), distance from anterior end to excretory pore (EP), distance from anterior end to nerve ring (NR), distance from anterior end to the base of the esophagus (ES), tail length (T), anal body diameter (ABD), ratios: a = L/MBD, b = L/ES, c = L/T, D% = EP/ES × 100, E% = EP/T × 100. Additionally, for males the testis reflexion (TR), the length of the spicules (SL), the spicule width, the length of the gubernaculum (GL), the gubernaculum width, SW% = SL/ABD × 100, and GS% = GL/SL × 100 were measured. The number and arrangement of genital papillae of males were studied.
Molecular characterization
In the Laboratory of Molecular Biology, total genomic DNA was extracted from a single female of first generation using a DNeasy® Blood and Tissues Kit (Qiagen.Inc), following the manufacturer protocol. The entire internal transcribed spacer region (ITS) of rDNA was amplified using the primer TW81 (5´-GTTTCCGTAGGTGAACCTGC-´3) forward and AB28 (5´- ATATGCTTAAGTTCAGCGGGT-´3) reverse (Hominick et al. 1997). Protocol thermo cycler for the PCR was as: one cycle at 94 °C for 2 min, 35 cycles at 94 °C for 1 min, follow 55 °C for 45 s and 72 °C for 2 min then 72 °C for 10 min. Ten μl of the PCR product was loaded on a 1.2% agarose gel stained with 0.5 μg/ml ethidium bromide, with 1X TBE buffer running solation and under voltage 80 v for two hrs. 100-bp DNA ladder was used for approximant quantification of the DNA product. The gel was visualized under ultraviolet light and photographed using a gel documentation system (Bio. Doc. Analyze). The target bands were purified using the gel purification kit NucleoSpin®Extract II Kit. The purified PCR products were sequenced by Macrogen Inc. (Netherland).
Phylogenetic analysis
Evolutionary analyses were studied and the phylogenetic tree was conducted in MEGA X. for the 27 sequences of Steinernema spp. which were obtained from the NCBI database. Multiple sequence alignments of ITS gene among the extracted isolates and the existing sequences in the library were aligned using Clustal W. Caenorhabditis elegans (Maupas 1900) (accession number JN636099) was used as an out-group sequence. Ancestral states were inferred using the maximum likelihood method and Hasegawa–Kishino–Yano model (1000 bootstrap replicates) (Kumar et al. 2018).
Bioassays
Three bioassay tests were carried out to evaluate the virulence of both isolates M.22 and M.313 using the last instar larvae of G. mellonella (Glazer and Lewis 2000). All bioassays were performed using 24-well plates (1.5-cm diameter) as test arena, lined with filter paper discs (Whatman No. 1) at the bottom of each well. One-week-old IJs suspensions were used in the experiments. After washing the suspensions with distilled water several times, the concentrations of the IJs were adjusted using the formula:
where Va = the amount of water (ml) to be added (if positive) or to be removed (if negative) from the suspension. V = volume of the suspension (ml). i = initial concentration / 50 μl. c = final concentration/50 μl (Glazer and Lewis 2000). The last instar larvae of G. mellonella were reared on artificial diets (wheat flour 350 g, corn flour 200 g, milk powder 130 g,backing yeast powder 70 g, honey 100 ml, and glycerin 150 ml) (Metwally et al. 2012). The larvae were used with an average weight of (0.2–0.3) g. Each concentration was separately applied to the wells; there were 24 replicates for each treatment. Four-well plates were used. Each experiment was repeated twice. The controls were identical to the treatment except that no IJs.
Exposure time assay
The nematode suspension was adjusted at 8000 IJs/ml. After shaking the suspension, 50 µl (had around 400 IJs) was transferred to each of the filter paper-lined wells using a micropipette. Single G. mellonella larva was added to each well and the plates were sealed with their lids. All plates were kept at 18 °C. After 15, 30, 45, and 60 min, six insect larvae were removed from each plate and rinsed in water to remove nematodes from the surface. Then, they were placed in a Petri dish (5-cm diameter) lined with the moist filter paper and incubated at 22 °C. The insect mortality was recorded after 48 h.
Concentration–response assay
Different concentrations of IJs (5, 15, 25 and 45) /Larva were used. Each one was applied separately, to the wells. Single G. mellonella larva was added to each well and the plates were sealed. All plates were kept at 18 ºC. The insect mortality was recorded after 72 h of exposure.
One-on-One assay
The nematode suspension was adjusted at 100 IJs/ml. An individual infective juvenile was transferred to each of the wells in 25 µl water. It was added another 25 µl of distilled water to wash the contents of the pipette tip into each well. Single G. mellonella larva was added to each well and the plates were sealed properly. The plates were kept at 18 °C. The insect mortality was recorded after 72 h. Two plates were used in this experiment (n. 48). Half lethal concentration (LC50) and half lethal time (LT50) were estimated.
Statistical analysis
Data of experiments were analyzed in a completely randomized design. Statistical analysis was done using a statistical package for SPSS software (SPSS 16.0; IBM Corp., USA) to study the significant differences among both EPNs isolates, the means were compared using LSD test at (P < 0.01). LC50 and LT50 values were calculated using the Probit analysis. Means were compared by analysis of variance (ANOVA) and Duncan’s test of multiple comparisons at a 1% level of significance.
Results
Nematode recovery from soil samples
The steinernematids were isolated from 2 of 821 soil samples (0.24%), from the walnut orchard and grassland in Amodeyha and Fneitiq village, respectively, in Al-Kadmous area belonging to Tartus governorate (Fig. 1). Soil characteristics of the positive sites were classified as silty loam and silty, respectively, more details are mentioned in (Table 1). ITS sequences analysis supported the morphometric and morphological studies for both isolates M.22 and M.313. The isolates were identical. They were identified as Steinernema affine.
Morphometric measurements
All morphometric examination results of the males and females of the first and second generations, and the infective juveniles for both isolates are shown in (Tables 2 and 3). The morphometric characteristics of the Syrian isolates were matched with the original description of the species by Poinar (1988) and Nguyen et al. (2007b). The Syrian isolates showed a variety of measurements. In the case of the first and second generations, males in all their measurements were similar to the original description, except some values, like E% and NR values in the first generation and NR, EP, and ES values in the second generation which were higher than the original description. For females, the measurements of the first generation were higher than the reported description. While, in the second generation most of the measurements were within the reference range, except the values of ES, MBD, and ABD, which were higher than the original description. The Infective juvenile’s measurements were within the range of the reported description.
Morphological characterization of male
Cuticle smooth, head rounded, not offset from rest of body. Six lips united at the base but distinct at the tips. Each lip bears a single labial papilla forming an inner circle of six. The outer circle of four cephalic papillae is located at the base of the dorsolateral and ventrolateral papillae. Stoma partially collapsed, a posterior portion surrounded by pharyngeal tissue. Cheilorhabdions are represented by a thick ring of sclerotized material lining the fused area of lips. Lateral fields and phasmids are inconspicuous. The pharynx is muscular with a cylindrical procorpus followed by a slightly swollen, non-valvate, metacorpus. The isthmus joins a nearly spherical basal bulb containing a reduced valve with refractive ridges. The excretory pore is anterior to the nerve ring, which is located at the anterior portion of the basal bulb. The genital system monorchic, reflexed. The spicules are variable in shape, tend to be strongly curved and sometimes moderately, tips narrowed but blunt. The Capitulum (manubrium) is broadly rounded and with a small rostrum at its base. The velum is extending obliquely from the rostrum almost to the tip of the lamina. The gubernaculum was forked with the proximal portions curved upwards and overlapping the spicule lamine. The distal portion is as hook-shaped. There is the basic complement of twenty-three genital papillae consisting of eleven pairs and a single adanal ventral one (S). A row of five subventral pairs is located anterior to the cloacal aperture. Three pairs are in the region posterior to the gubernaculum. And three pairs occurred near the tail tip surrounding the tail mucron.Tip of tail bearing minute mucron. Males of the second generation are similar to the first generation but smaller in size and mucron is absent (Fig. 2).
Steinernema affine male. A: first generation entire body. B: the anterior portion, right arrow refers to nerve ring position while left arrow refers to excretory pore. C: arrows refer to velum of spicule. D: lateral view of the five pairs of papillae anterior to cloaca opening, addition to the two pairs of papillae (1st, 2nd) posterior to the gubernaculum. E: lateral view of adanal ventral one papillae (S), and two pairs of papillae posterior to the gubernaculum (2nd, 3rd). F: two pairs of papillae (1st, 2nd) posterior to the gubernaculum, the arrows refer to three pairs of papillae near the tail tip, G: arrows refer to mucro of tail which surrounded by three pairs of papillae. H: lateral view of spicules and gubernaculum. I: ventral view of gubernaculum. J: moderately curved spicules. K: second generation tail male without a mucro. A: 1481 µm. B: EP = 119 µm, NR = 126 µm. H,I: SL = 65 µm. GL = 50 µm. J = SL = 68 µm, GL = 51 µm. K = 41 µm.
Females
Cephalic extremity and cuticular structures similar to the male. The nerve ring is located more toward the center of the isthmus; in some females, it is also located toward the anterior portion of the basal bulb, as we observed. The excretory duct is cuticularized; the excretory pore is more anteriorly located than in male. The vulval aperture is located posterior to the mid-body; lips protrude by 20 μm (11–22 μm). The postanal swelling is present. The tail ends with one or two papilla-like mucrons. Females of the second generation are similar to the first generation, but smaller in size. The vulva lips protrude by 3 μm (0–4) μm. The postanal swelling slightly developed. The tail is straight, conoid, and ends in a fine mucron (Fig. 3).
Steinernema affine female. A: first-generation entire body B: second-generation entire body. C: anterior portion, right arrow refer to nerve ring position and left arrow refer to excretory pore of first generation. D: right arrow refers to nerve ring which located toward the anterior portion of basal bulb. E: vulva of first generation female. F: vulva of second-generation female. G, H: tail region with one and two papilla structure of first generation, respectively. I: tail region with papilla structure of second generation. Scale bar: A = 4245 µm. B = 1460 µm, C: EP = 122 µm, NR = 160 µm. D: NR = 167 µm, EP = 118 µm. F = 0.9 µm. G = 1 µm. H = 1 = 2.1 µm, 2 = 1.2. I = 4.3 µm. E = 19 µm. F = 2 µm. G:1 = 2.1 µm, 2 = 1.3 µm. H: 4.3 µm. I = 0.9 µm
Infective juvenile
Cephalic extremity fine rounded, offset from rest of body. Oral aperture and anus closed, the excretory pore located anterior to nerve ring in mid-pharynx region. The intestine collapsed. Tail sharply pointed. The existence of a tail terminal spine is the most significant trait of the infective juvenile (Fig. 4).
Molecular characterization
Using the ITS region of the two isolates (M.313 and M.22) resulted in a 900 bp amplicon which was sequenced and submitted to GenBank (Accession numbers: OM350119.1 and OL437016). Homology search results for Ma.313 isolate using BLAST search showed similarity to Steinernema affine strain isolate STP43/2 (Accession: MN861028.1) with a query coverage of 97 and 97.66% identity, but M.22 isolate homology search results showed similarity to the same isolate (STP43/2) with a query coverage of 99 and 97.47% identity. The unrelated species, Caenorhabditis elegans (JN_636099.1) formed an out-group during sequence alignment and phylogenetic tree construction (Fig. 5). Maximum likelihood phylogenetic (MP) tree was constructed for the two studied isolates, and 27 sequences and one C. elegans sequence were aligned to study phylogenetic relationships among them. MP analyses showed two main clades, viz., Clade I: ‘affine-intermedium’ group; Clade II: other sequences. The studied isolates positions revealed that it is distantly related to Steinernema affine species.
Maximum likelihood phylogenetic tree of Steinernema nematodes based on ITS sequences. The evolutionary relationships were computed using the Hasegawa–Kishino–Yano model. The tree with the highest log likelihood (− 8485.52) is shown. Numbers at nodes represent bootstrap values higher than 70% based on 100 replications. A discrete gamma distribution was used to model evolutionary rate differences among sites (5 categories (+ G, parameter = 2.1835)). The tree is drawn to scale, with branch lengths measured in the number of substitutions per site. This analysis involved 30 nucleotide sequences. Codon positions included were 1st + 2nd + 3rd + Noncoding. There were a total of 1184 positions in the final dataset. Evolutionary analyses were conducted in MEGA X. The obtained isolates in this study are marked with star
Bioassays
In the exposure time assay (Table 4), the mortality rate increased gradually as the time exposure lengthened. Despite that, one hour of exposure was insufficient to cause 100% mortality, during this test period, M.22 and M.313 achieved 90.16 and 91.49% insect mortality, respectively, with nonsignificant difference (P < 0.01). A similar pattern was observed in the Concentration–response assay (Table 5), but the mortality rate was 100% for both isolates, at the concentration of 40 IJs\L after 72 h of exposure. In the one-on-one assay, a low mortality rate was recorded, for both M.22 and M.313; the rate was 20.0 and 23.6%, respectively (Table 5), with nonsignificant difference (P < 0.01). The LT50 values indicated that M.22 and M.313 could kill half of G. mellonella larvae in 33.47 and 32.43 min, respectively, while the LC50 values were 11.7 and 11.5 IJs\L for M.22 and M.313, respectively, (Table 6).
Discussion
The present study documented S. affine in Syria, as a first report. It was isolated from Al-Kadmous area, which has a moderate to cold climate. Simultaneously, this research showed that this genus was not common or widely distributed in Syria's studied areas. Although this study included different agricultural and ecological systems, Steinernema was recovered at a low ratio (0.24%). The reasons could be (1) Excessive use of conventional and novel chemical pesticides in several agricultural systems, which may have an impact on the viability of IJs, as mentioned by Laznik and Trdan (2013). (2) The climate changes that have occurred in Syria in the last years were accompanied by rising temperature and decreased precipitation drop, had a negative impact on the vegetative cover. As a result, the insect hosts may have become limited. Furthermore, the temperatures (Tarasco et al. 2015), as well as the soil moisture (Grant and Villani, 2003), had a direct effect on the infectivity of EPNs and their survival. (3) Using only laboratory temperature during isolation could affect the success of the isolation process of some EPNs species (Hazir et al. 2003) (4) Using G. mellonella as a trap insect could be not a susceptible host for all EPNs species as reported by Spiridonov & Moens (1999). In general, the prevalence of the EPNs was low in the neighboring countries as Türkiye (Hazir et al. 2003), Jordan (Stock et al. 2008), and Lebanon (Noujeim et al. 2011).
Globally, S. affine was isolated from Denmark (Bovien 1937), the Alpin region in Switzerland (Steiner 1996), Spain (Pino and Palomo 1996), Italy (Tarasco et al. 2015) Belgium (Miduturi et al. 1997), Slovak (Sturhan and Lišková 1999), Czech Republic (Mráček et al. 1999), Türkiye (Hazir et al. 2003), British Colombia Canada (Mráček et al. 2005), China (Wang et al. 2014), and Pakistan (Tabassum et al. 2017). So, this species' geographic distribution might be considered to span three continents.
In this research, S. affine was recovered from walnut orchard and grassland. The global studies referred that S. affine was isolated from different habitats such as apple orchard (Mráček and Bečvář 2000), woodland (Midituri et al. 1997), riverbanks (Sturhan and Lišková 1999), a roadside verge (Midituri et al. 1997) cultivated areas (Sturhan and Lišková 1999), a fallow fields (Hazir et al. 2003), fruit crop (Wang et al. 2014), and cherry orchard (Tabassum et al. 2017), but the most frequently of them was the grassland (Mráček et al. 2005; Hazir et al. 2003), this is similar to our finding. Originally, Bovien described this species from diseased bibionid fly larvae in Denmark. The larvae of this fly feed on roots and decaying organic matter, in our research, we were unable to determine the insect's host. But we can expect the presence of insect larvae such as bibionid fly larvae in grassland and walnut orchard.
The chemical and physical characteristics of the soil affect the longevity and persistence of dauer juveniles of EPNs (Shapiro et al. 2000). So our soil samples were analyzed, and the results showed that the positive soil samples were silty loam and silty, and this is similar to the study of Mráček et al. (2003). As well as the organic matter content was 4.1 and 6%, which were within the range mentioned by Miduturi et al (1997). PH of both samples was slightly acidic, this result agrees with other studies which found the PH range of EPNs positive soil samples was 5.6–7.9 (Hazir et al. 2003). Recently, Khathwayo et al (2021) found the highest infective juvenile survival was obtained from PH 3 to 10.
The morphometric measurements and the morphological characteristics for first and second generations’ males, consider helpful and very important to identifying the EPNs species. In this research, some variations in measurements of males compared to the reported description were observed. Even so, the values of E and SW% in the second-generation male were still higher than in the first generation. This is a distinguishing feature of S. affine males, as reported by Nguyen et al. (2007b). The morphological traits were similar to the original characteristics, except for the location of the nerve ring in some females, which is located toward the anterior basal bulb. This characteristic was not mentioned in the reported description, which only indicated that the nerve ring is located toward the center of the isthmus. The differences of morphometric and morphological traits are thought to result from both intraspecific genetic variations and environmental factors.
Traditionally, species definition in steinernematids has been based on morphology and cross-breeding experiments (Hominick et al. 1997). Stock et al. (2001) noted the absence of autapomorphies for four species during the analysis of the LSU domain for Steinernema. Our ITS rDNA analysis showed sequence divergence might be considered an indication of independent evolutionary history in steinernematids, which revealed that S. intermedium and S. affine were in the same clade. The phylogenetic analyses in this study showed that the molecular relationship between the tested isolates and the reported ones in NCBI which were isolated in Belgium, Netherlands, and UK were 95% query coverage and 96% identity (Spiridonov et al. 2004), as well as S. affine isolates in Georgia mentioned by Gorgadze et al. (2015) showed 92% query coverage and 97% identity with the Syrian studied isolates. There was non-alignment between our isolates and the Pakistani ones which were reported by Tabassum et al. (2017).
In Syria to date, the use of EPNs as biological control agents is unknown among farmers, who are wholly dependent on chemical pesticides. So in the present research, some bioassays to get an initial thought about the pathogenicity and virulence of Syrian S. affine isolates, were conducted, and had given good results and achieved high mortality, this result is considered to be equal to the effect of some chemical pesticides.
Conclusion
Distribution of Steinernema spp. in the Syrian coastal region is low. For the first time in Syria, S. affine was recorded in grassland and walnut orchards in Tartus governorate. S affine isolates achieved a high mortality against G. mellonella larvae at 18 °C. The promising results obtained from bioassays encourage extending the assays to other important insect pests in different conditions; in order to use them in the Syria’s IPM programs in the future.
Availability of data and materials
All data generated or analyzed during this study are included in the text.
Abbreviations
- EPNs:
-
Entomopathogenic nematodes
- One-On-One:
-
Individual infective juvenile of nematodes against insect larva
- ITS:
-
Internal transcribed spacer
- LSD:
-
Least significant difference
- LT50 :
-
Median lethal time.
- LC50 :
-
Lethal concentration 50%
- SPSS:
-
Statistical package for the social sciences
References
Bedding RA, Akhurst RJ (1975) Simple technique for the detection of insect parasitic rhabditid nematodes in soil. Nematologica 21:109–110
Boemare N (2002) Biology, taxonomy and systematics of Photorhabdus and Xenorhabdus. In: Gaugler R (ed) Entomopathogenic nematology. CABI Publishing, UK, pp 35–56. https://doi.org/10.1079/9780851995670.0035
Bovien P (1937) Some types of association between nematodes and insects. Vidensk Medd fra Dansk Naturhistorisk Foren 101:1–14
Canhilal R, Reid W, Kutuk H, El-Bouhssini M (2006) Natural occurrence of entomopathogenic nematodes (Rhabditida: Steinernematidae and Heterorhabditidae) in Syrian soils. Res J Agric Biol Sci 2:493–497
Glazer I, Lewis EE (2000) Bioassays for entomopathogenic nematodes. In: Navon A, Ascher KRS (eds) Bioassays of entomopathogenic microbes and nematodes. CABI, Wallingford, pp 229–247
Gorgadze O, Lortkhipanidze M, Ogier JC, Tailliez P, Burjanadze M (2015) Steinernema tbilisiensis sp. n. (Nematoda: Steinernematidae)—A new species of entomopathogenic nematode from Georgia. J Agricultural Sci Technol A. https://doi.org/10.17265/2161-6256/2015.04.005
Gozel U, Gozel C (2016) Entomopathogenic nematodes in pest management. In: Gill H, Goyl G (eds) Integrated pest management (IPM): environmentally sound pest management. Intechopen, London, pp 55–69. https://doi.org/10.5772/63894
Hazir S, Keskin NS, Stock P, Kaya H, Ozcan S (2003) Diversity and distribution of entomopathogenic nematodes (Rhabditida: Steinernematidae and Heterorhabditidae) in Turkey. Biodivers Conserv 12:375–386. https://doi.org/10.1023/A:1021915903822
Hazir S, Kaya H, Touray M, Çimen H, Shapiro Ilan D (2022) Basic laboratory and field manual for conducting research with the entomopathogenic nematodes, Steinernema and the entomopathogenic nematodes, Steinernema and Heterorhabditis, and their bacterial symbionts Heterorhabditis, and their bacterial symbionts. Turkish J Zool 46:305–350. https://doi.org/10.55730/1300-0179.3085
Hominick WM, Briscoe BR, Pino GD, Jian Heng J, Hunt DJ, Kozodoy E, Mracek Z, Nguyen KB, Reid AR, Spiridonov S, Stock P, Sturhan D, Waturu C, Yoshida M (1997) Biosystematics of entomopathogenic nematodes: current status, protocols and definitions. J Helminthol 71:271–298. https://doi.org/10.1017/s0022149x00016096
Jawish A (2016). Biological and morphological characterization of entomopathogenic nematode isolates and their symbiosis bacteria and evaluation their efficiency in the control of soil insects. PhD Thesis, Damascus University Faculty of Agriculture Syria
Kaya HK, Stock P (1997) Techniques in insect nematology. In: Lacey LA (ed) Manual of techniques in insect pathology. Academic Press, Cambridge, pp 281–324. https://doi.org/10.1016/B978-012432555-5/50016-6
Khathwayo Z, Ramakuwela T, Hatting J, Shapiro DI, Cochrane N (2021) Quantification of pH tolerance levels among entomopathogenic nematodes. J Nematol 53:1–12. https://doi.org/10.21307/jofnem-2021-062
Koppenhöfer HS (2007) Bacterial Symbionts of Steinernema and Heterorhabditis. In: Nguyen K, Hunt D (eds) Entomopathogenic nematodes: systematics, phylogeny and bacterial symbionts. Brill, Leiden. https://doi.org/10.1163/ej.9789004152939.i-816.43
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35:1547–1549. https://doi.org/10.1093/molbev/msy096
Laznik Z, Trdan S (2012) Entomopathogenic nematodes (Nematoda: Rhabditida) in slovenia: from tabula rasa to implementation into crop production systems. In: Perveen Farzana (ed) Insecticides - advances in integrated pest management. InTech, London. https://doi.org/10.5772/29540
Laznik Z, Trdan S (2013) The influence of insecticides on the viability of entomopathogenic nematodes (Rhabditida: Steinernematidae and Heterorhabditidae) under laboratory conditions. Pest Manag Sci 70:784–789. https://doi.org/10.1002/ps.3614
Metwally HM, Hafez GA, Hussein MA, Hussein MA, Salem HA, Saleh MM (2012) Low cost artificial diet for rearing the greater wax moth, Galleria mellonella L. (Lepidoptera: Pyralidae) as a host for entomopathogenic nematodes. Egypt J Biol Pest Control 22:15–17
Miduturi JS, Waeyenberge L, Moens M (1997) Natural distribution of entomopathogenic nematodes (Heterorhabditidae and Steinernematidae) in Belgian soils. Russ J Nematol 5:55–65
Mosalam Z (2009) Efficacy of entomopathogenic nematodes against Capnodis spp. In stone-fruit orchards. PhD Thesis, Teshreen University, Faculty of Agriculture Syria
Mráček Z, Bečvář S, Kindlmann P (1999) Survey of entomopathogenic nematodes from the families Steinernematidae and Heterorhabditidae (Nematoda: Rhabditida) in the Czech Republic. Folia Parasitol 46:145–148
Mráček Z, Kindlmann P, Webster J (2005) Steinernema affine (Nematoda: Steinernematidae), a new record for North America and its distribution relative to other entomopathogenic nematodes in British Columbia. Nematology 7:495–507
Nguyen KB (2007) Methodology, morphology and identification. Nematology monographs & perspectives. In: Nguyen K, Hunt D (eds) Entomopathogenic nematodes: systematics, phylogeny and bacterial symbionts. Brill, Leiden. https://doi.org/10.1163/ej.9789004152939.i-816.25
Nguyen KB, Hunt DJ, Mráček Z (2007b) Steinernematidae: species descriptions. In: Nguyen K, Hunt D (eds) Entomopathogenic nematodes: systematics, phylogeny and bacterial symbionts. Brill, Leiden, pp 121–609
Noujeim E, Khater C, Pages S, Ogier JC, Tailliez P, Hamze M, Thaler O (2011) The first record of entomopathogenic nematodes (Rhabiditiae: Steinernematidae and Heterorhabditidae) in natural ecosystems in Lebanon: a biogeographic approach in the Mediterranean region. J Invertebr Pathol 107:82–85. https://doi.org/10.1016/j.jip.2011.01.004
Pino GD, Palomo A (1996) Natural occurrence of entomopathogenic nematodes (Rhabditida: Steinernematidae and Heterorhabditidae) in Spanish soils. J Invertebr Pathol 68:84–90. https://doi.org/10.1006/jipa.1996.0062
Poinar GO (1988) Redescription of Neoaplectana affinis Bovien (Rhabditida: Steinernematidae). Revue Nématol. 11:143–147
Poinar GO, Grewal PS (2012) History of entomopathogenic nematology. J Nematol 44:153–161
Shapiro DI, Mccoy CW, Fares A, Obreza T, Dou H (2000) Effects of soil type on virulence and persistence of entomopathogenic nematodes in relation to control of Diaprepes abbreviates (Coleoptera:Curculionidae). Environ Entomol 29:1083–1087. https://doi.org/10.1603/0046-225X-29.5.1083
Shapiro DI, Gouge DH, Piggott SJ, Fife JP (2006) Application technology and environmental considerations for use of entomopathogenic nematodes in biological control. Biol Control 38:124–133. https://doi.org/10.1016/j.biocontrol.2005.09.005
Spiridonov SE, Moens M (1999) Two previously unreported species of steinernematids from woodlands in Belgium. Russ J Nematol 7:39–42
Spiridonov SE, Reid AP, Podrucka K, Subbotin SA, Moens M (2004) Phylogenetic relationships within the genus Steinernema (Nematoda:Rhabditida) as inferred from analyses of sequences of the ITS1-5.8 S-ITS2 region of rDNA and morphological features. Nematology 6:547–566
Steiner W (1996) Distribution of entomopathogenic nematodes in the Swiss Alp. Revue Suisse De Zool 103:439–452
Stock SP, Campbell JF, Nadler SA (2001) Phylogeny of Steinernema Travassos, 1927 (Cephalobina:Steinernematidae) inferred from ribosomal DNA sequences and morphological characters. J Parasitol 87:877–889. https://doi.org/10.1645/0022-3395(2001)087[0877:postcs]2.0.co;2
Stock P, Luma B, Darwish R, Katbehc A (2008) Diversity and distribution of entomopathogenic nematodes (Nematoda: Steinernematidae, Heterorhabditidae) and their bacterial symbionts (γ-Proteobacteria:Enterobacteriaceae) in Jordan. J Invertebr Pathol 98:228–234. https://doi.org/10.1016/j.jip.2008.01.003
Sturhan D, Lišková M (1999) Occurrence and distribution of entomopathogenic nematodes in the Slovak republic. Nematology 1:273–277
Tabassum KA, Salma J, Sagir H (2017) Two new records of entomopathogenic nematodes (Nematoda: Steinernematidae) from Gilgit-Baltistan, Pakistan. Pak J Nematol 35:157–173. https://doi.org/10.18681/pjn.v35.i02.p157-173
Tarasco E, Oreste M, Li X, Qai-zhi L (2015) Infectivity of mediterranean native entomopathogenic nematodes (Steinerne-matidae and Heterorhabditidae) from natural habitats in relation to temperature. J Zool 98:109114
Wang H, Luana L, Dong H, Qian H, Cong B (2014) Natural occurrence of entomopathogenic nematodes in Liaoning (Northeast China). J Asia-Pacific Entomol. https://doi.org/10.1016/j.aspen.2014.03.005
White GF (1929) A method for obtaining infective nematode larvae from cultures. Science 66:302–303
Zeini GH, Abokaf N, Al-body M, Mofleh M (2019) Molecular characterization of several isolates of entomopathogenic nematode Heterorhabditis bacteriophora Poinar (Rhabditida, Heterorhabditidae) from soil of citrus orchards in Lattakia, Syria. SSRG Int J Agric Environ Sci 7:70–74
Acknowledgements
The authors thank Prof. Dr. SH from Aydin Adnan Menderes University Türkiye, for the help in confirming the identity of the nematodes species. We also express our appreciation to Prof. Dr. WS director of the Biotechnology Center, Tishreen University Syria, DYM, DHM, Technical University of Dresden, Institute of botany Germany, and DRK (Indian agriculture research institute) for the support and help to perform this study.
Funding
This work was supported by the Research Laboratory, Department of Plant Protection Faculty of agriculture at Tishreen University Syria.
Author information
Authors and Affiliations
Contributions
Mai A conceived the work, designed and performed the experiments, analyzed the data, wrote the paper. NA was a major contributor in writing the manuscript, review and editing the paper. MA review and edit the paper. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ali, M., Allouf, N. & Ahmad, M. First report of entomopathogenic nematode Steinernema affine (Nematoda: Steinernematidae) in Syria and its virulence against Galleria mellonella L. (Lepidoptera: Pyralidae). Egypt J Biol Pest Control 32, 101 (2022). https://doi.org/10.1186/s41938-022-00602-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s41938-022-00602-x