Abstract
Background
Endometrial hyperplasia is one of the common causes of bleeding in perimenopausal women. Variable treatment options aim to induce regression. The current study evaluated the regression rate of endometrial hyperplasia after treatment with levonorgestrel intrauterine system (LNG- IUS) versus Metformin.
Methods
This randomized clinical trial was conducted at the obstetrics and gynecology department of Suez Canal University hospital. We recruited women diagnosed with endometrial hyperplasia without atypia. Patients were allocated into two groups. Group one included patients treated with levonorgestrel-releasing intrauterine system and group two treated with Metformin. The rate of regression of hyperplasia in both groups after six months of intervention was the main outcome measure.
Results
Significant regression of hyperplasia was noted in the LNG-IUS group (96% versus 64%, p-value 0.009). There was a significant decrease in the endometrial thickness after treatment in both groups (17.65 ± 4.62 and 5.3 ± 2.01 in the LNG- IUS with a p-value < 0.001) (19.57 ± 6.84 and 11.22 ± 7.51 in the metformin group with a p-value < 0.001). Factors that correlated with the Δ endometrial thickness included parity in the LNG- IUS group (p-value 0.019) and age and BMI in the metformin group (p-value 0.043 and 0.004 respectively).
Conclusion
Metformin had a regressive effect on endometrial hyperplasia; however, it was not significant as that achieved with the levonorgestrel intrauterine system.
Trial registrations
PACTR201908498370196.
Date of registration: 21/8/2019.
Date of first patient enrollment: 25/8/2019.
URL: https://pactr.samrc.ac.za/Researcher/TrialRegister.aspx?TrialID=9335
Similar content being viewed by others
Background
Simple endometrial hyperplasia is a common pathological finding in women with abnormal uterine bleeding reaching up to 31% necessitating treatment to control patient’s symptoms [1]. It is characterized by excessive proliferation of endometrial cells [2]. During the proliferative phase, estrogen induces proliferative changes to the glands and stroma. Then, in the secretory phase, progesterone induces secretory changes on the endometrium as well as inhibiting the proliferative effect of estrogen. When ovulation is delayed or absent, the effect of estrogen becomes unopposed leading to continued proliferation [3].
Progestogens play a significant role in the management of endometrial hyperplasia. Different forms and dosing regimens exist to treat endometrial hyperplasia when surgery is declined [4, 5]. These included megestrol acetate, medroxyprogesterone acetate, norethindrone acetate, and levonorgestrel [6]. The levonorgestrel intrauterine system has been used for contraception as well as management of menorrhagia. It provides higher concentrations of progestogens than that provided after oral therapy, making it useful in managing endometrial hyperplasia [7]. However, it is associated with irregular bleeding in about 82% of women, and many women do not accept it [8].
Metformin is a potent inhibitor of cellular proliferation and capable of inducing apoptosis when used in higher concentrations [9, 10]. The use of Metformin in the management of endometrial hyperplasia has not been studied extensively. Available researches showed it has been used in combination with progestogens [11,12,13]. It has been used in previous trials, either alone or combined with progestogens, but its effectiveness and safety remained uncertain [8]. It has been shown to augment the inhibitory effect of progesterone on the proliferating endometrium [14].
The current trial was conducted to evaluate the effect of metformin versus the levonorgestrel intrauterine system for the management of endometrial hyperplasia. We hypothesized that metformin might have a regressive effect on the endometrium which would be beneficial in low resource countries.
Methods
This was a randomized clinical trial conducted in the obstetrics and gynecology department of a tertiary hospitals. The trial was conducted from August 2019 to July 2020 after approval of the research ethics committee. We recruited women according to inclusion and exclusion criteria.
-
Inclusion criteria included a) women aged 30- 75 years, b) any woman in the childbearing period presenting with abnormal uterine bleeding that needed endometrial sampling, c) postmenopausal women presenting with postmenopausal bleeding and thickened endometrium (> 5mm) that needed endometrial sampling, and d) histological evidence of endometrial hyperplasia without atypia.
-
Exclusion criteria included a) suspected pregnancy, b) contraindication to Metformin (Impaired renal functions, Cirrhosis of the liver, Hepatitis, and Alcoholism), c) contraindications to Mirena (e.g., acute genital tract inflammatory disease, genital bleeding of unknown etiology, hypersensitivity to any component of this product, congenital or acquired uterine anomaly, known or suspected breast cancer, known or suspected uterine and cervical neoplasia or acute liver disease or liver tumor), d) Women with concurrent endometrial cancer or atypical hyperplasia, e) women with secretory or disordered proliferative endometrium at biopsy, f) Women with a history of a hormone-dependent malignancy (e.g., breast cancer), g) Women taking tamoxifen, and h) diabetes taking hypoglycemic agents.
Randomization was factorial and balanced in a 1:1 manner using a computer-generated randomization list, allocating patients into two groups. Group one included patients diagnosed with endometrial hyperplasia without atypia treated with the levonorgestrel-releasing intrauterine system. Group two included patients with endometrial hyperplasia without atypia treated with Metformin. Randomization was done after evaluating the participants for eligibility. The allocation sequence was concealed from the researcher enrolling and assessing participants using opaque sealed envelopes. Patients and researchers were aware of group allocation, but outcome assessors and data analysts were kept blinded.
Study procedures
Complete history taking (age, occupation, education, complaint, chronic illness) was done. Proper examination (weight, height, BMI, gynecological examination to exclude local causes of abnormal uterine bleeding) was done.
Transvaginal ultrasound for evaluation of the uterus and ovaries was done. The woman was put in the lithotomy position, with the bladder empty. The evaluation was done using Mindray DC- 60 machine with a transvaginal probe V 11-3B (7 MHz). The endometrium was measured in a sagittal plane, with the entire endometrium through to the endocervical canal in the view. Examination was done on day 3 of the menstrual cycle for menstruating women. A thickness of > 11 mm and > 5 mm was considered pathological in premenopausal and postmenopausal women, respectively [15].
Dilatation and curettage were done under anesthesia in the operating theatre. The same researcher operated all cases before and after the intervention (the standard practice in our institute). Specimens were impregnated with 10% formaldehyde. Paraffin sections were prepared. Samples were evaluated microscopically by the same pathologist (Pathology department, Suez Canal University), who was blinded for patients' allocation.
Patients allocated for group one had the LNG- IUS, Mirena (Bayer, Leverkusen, Germany) fitted in the outpatient clinic. They were instructed that another evaluation was deemed to be done six months later. Patients allocated for group two received metformin (Cidoghage, CID pharmaceutical) treatment 850 mg tablets once daily for two months then twice daily for four months. They were instructed that another evaluation would be performed after six months of treatment. A final pill count determined cumulative exposure and treatment compliance.
Both groups were instructed to report any side effects of either treatment. After six months, both groups had a transvaginal ultrasound for evaluation of the endometrial thickness. Dilatation and curettage were performed once again after completion of treatment (the day after the end of the six- month period). The LNG- IUS was removed before performing the transvaginal ultrasound.
The primary outcome measure was rates of regression of hyperplasia in both groups after six months of intervention. Regression was defined as the return of endometrial hyperplasia to normal with associated secretory changes and atrophy [4]. Secondary outcome measures included endometrial thickness after treatment in both groups and side effects reported by the participants.
The sample size was calculated at a significance level of 95%and an error level of 20% with a proportion of hyperplasia regression among Mirena group as 89% [16] and a proportion of hyperplasia regression among Metformin group as 56% [17]. A drop-out proportion of 10% was added to the raw result giving a final count of 25 women per group.
Statistical analysis
Data were statistically described in terms of mean and standard deviation, frequencies (number of cases), and percentages when appropriate. P values of less than 0.05 were considered statistically significant. All statistical calculations were done using computer program SPSS (Statistical Package for the Social Science; SPSS Inc., Chicago, IL, USA) release 22 for Microsoft Windows. Chi-square test was used for categorical variables and (t) test for continuous variables with normally distributed data. Non-normally distributed data were tested using Fisher’s exact for categorical variables and Mann–Whitney U tests for continuous variables. Spearman correlation was used to test the correlation between patients' characteristics and the delta change in endometrial thickness.
Results
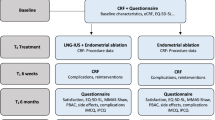
The patients’ flowchart is presented in Fig. 1. Seventy- one women were screened for eligibility and 65 patients were eligible for the study. Seven patients declined to participate in the study, leaving 58 participants ready for allocation. Each group included 29 patients. In the LNG-IUS group, four patients were excluded for inadequate samples after treatment; leaving 25 patients completed the study. In the metformin group, three patients refused to continue the study, and one patient was excluded because of inadequate sampling after treatment; leaving a total of 25 patients completed the study. No patient discontinued metformin treatment or requested LNG- IUS removal.
There was no difference between both groups in their primary demographic characters (Table 1).
The recruited cases showed simple endometrial hyperplasia before treatment. Significant regression of hyperplasia was noted in the LNG-IUS group compared to the metformin group (96% versus 64%, p-value 0.009). Nine cases had persistent hyperplasia in the metformin group versus one patient in the Mirena group (p-value 0.009) (Table 2).
There was a significant decrease in the endometrial thickness after treatment in both groups (17.65 ± 4.62 and 5.3 ± 2.01 in the LNG- IUS with a p value < 0.001) (19.57 ± 6.84 and 11.22 ± 7.51 in the metformin group with a p value < 0.001) (Table 3).
Both modalities of treatment were associated with some tolerable side effects. The most commonly reported side effect in the metformin group was gastrointestinal upset (9, 36%) (Table 4).
Factors that correlated with the Δ endometrial thickness included parity in the LNG- IUS group (r = -0.486, p value 0.019) and age and BMI in the metformin group (r = 0.425, p value 0.043 and r = 0.581, p value 0.004 respectively) (Table 5).
Discussion
In the study conducted by Ørbo et al., endometrial hyperplasia was more evident in women aged 45- 51. Half of their participants were overweight and multiparous (2 or more children), which agreed with our findings. The majority of their participants were premenopausal [7] in contradiction to the current study. This differed from another study that recruited women with atypical hyperplasia/endometrial carcinoma and desired fertility-sparing (median age was 35, and BMI was 37.7) [18].
There was a significant decrease in the endometrial thickness after treatment in both groups but was more prominent in the LNG- IUS group. Significant regression of hyperplasia was noted in the LNG-IUS group (96%). A previous study reports an overall regression rate of 95%, with 100% regression among women with endometrial hyperplasia without atypia [19]. Another one reported an overall regression rate of 90% with 92% regression among endometrial hyperplasia without atypia, although they adopted a longer follow-up duration [20]. All progestogens were found to induce regression of endometrial hyperplasia with the LNG- IUS reporting more significant improvement [7]. Progestogens modulate secretory differentiation of the endometrial glands, inhibit estrogen receptor function, and endometrial cell division. Progestogens decrease the expression and activity of insulin-like growth factor- 1, which is a potent proliferative factor, with increased expression in endometrial hyperplasia [21].
We reported a regression rate of 64% in the metformin group. There was no difference in complete response rates among women treated with progestogen alone and those treated with progestogen and Metformin (69% versus 68%, p-value 0.90) in women diagnosed with atypical endometrial hyperplasia/ endometrial carcinoma [18]. Besides, Metformin was not associated with a complete response in univariate and multivariate analysis (P = 0.066 and 0.123, respectively) [18]. This would be rendered to the different treatment regimens between both studies as we provided Metformin alone as a treatment option. Also, the target population was different between both studies.
Although Metformin has an anti-proliferative effect, up-regulates progesterone receptors, and blocks epidermal growth factor signaling [12], it has been reported that its effect on regression rates of endometrial hyperplasia (with or without atypia) lacked evidence [22, 23]. Trial results showed no evidence to support or decline Metformin as a treatment option for endometrial hyperplasia alone or combined with progestogens [8]. However, another recommended its use in combination with progestogens for the treatment of endometrial hyperplasia and early-stage endometrial carcinoma [24].
Factors correlated with the change in endometrial thickness included parity in the LNG- IUS group and age and BMI in the metformin group. This differed from the reported results in a previous study that declared parity, BMI, and menopausal status did not correlate to treatment response [7]. This might be attributed to the use of progestogens only in their study (LNG- IUS, cyclic oral MPA, and continuous oral MPA).
Both modalities of treatment were associated with some tolerable side effects. The most commonly reported side effect in the metformin group was gastrointestinal upset (9, 36%). It has been reported that Metformin was associated with gastrointestinal adverse effects in 20- 30% of patients [25] (abdominal pain, diarrhea, nausea, vomiting, and change of taste), limiting its use [8]. Irregular bleeding at first use was reported by 8% of the recruited women in the LNG- IUS group. This agreed with previous results about the LNG- IUS [7].
Strengths and limitations
This was the first trial to compare LNG- IUS, and Metformin alone as a treatment option for endometrial hyperplasia. The design of the study and the balanced randomization are considered as main strengths. Low-quality specimens with fragmentation and scanty endometrial tissue were excluded. All specimens were examined by the same pathologist who was blinded to group allocation. This study was conducted in a single tertiary hospital, which ensured the unique evaluation and management of all participants. The small sample recruited is an evident limitation for the study. Only the pathologist was blinded to group allocation. Treatment time not less than six months was adopted as recommended by previous studies [26]. A longer duration of follow up would be more conclusive. The recruitment of women with endometrial hyperplasia without atypia limited the generalizability of the results.
Conclusion
Eventually we report that Metformin had a regressive effect on the endometrium in patients with endometrial hyperplasia. The regressive effect was not as significant as that achieved after LNG- IUS treatment.
Availability of data and materials
Not applicable.
Abbreviations
- LNG-IUS:
-
Levonorgestrel- intrauterine system
- BMI:
-
Body mass index
- MPA:
-
Medroxyprogesterone acetate
References
Rezk M, Masood A, Dawood R (2015) Perimenopausal bleeding: Patterns, pathology, response to progestins and clinical outcome. J Obstet Gynaecol 35(5):517–521
Wise MR, Farrant C, Coop C (2017) Levonorgestrel‐releasing intrauterine system for endometrial hyperplasia. Cochrane Database Syst Rev 2017(5):CD012658. https://doi.org/10.1002/14651858.CD012658.
Anderson MC, Robboy SJ (1997) Aetiology and histopathology of endometrial hyperplasia and carcinoma. Curr Obstet Gynaecol 7(1):2–7
Horn LC, Schnurrbusch U, Bilek K, Hentschel B, Einenkel J (2004) Risk of progression in complex and atypical endometrial hyperplasia: clinicopathologic analysis in cases with and without progestogen treatment. Int J Gynecol Cancer 14:348–353
Wheeler DT, Bristow RE, Kurman RJ (2007) Histologic alterations in endometrial hyperplasia and well-differentiated carcinoma treated with progestins. Am J Surg Pathol 31:98898
Yuk JS, Song JY, Lee JH, Park WI, Ahn HS, Kim HJ (2017) Levonorgestrel-releasing intrauterine systems versus oral cyclic medroxyprogesterone acetate in endometrial hyperplasia therapy: a meta-analysis. Ann Surg Oncol 24:1322–1329
Ørbo A, Vereide AB, Arnes M, Pettersen I, Straume B (2014) Levonorgestrel-impregnated intrauterine device as treatment for endometrial hyperplasia: a national multicentre randomised trial. BJOG 121:477–486
Clement NS, Oliver TRW, Shiwani H, Sanner JRF, Mulvaney CA, Atiomo W. Metformin for endometrial hyperplasia. Cochrane Database of Systematic Reviews. 2017;(10)Art. No.: CD012214. https://doi.org/10.1002/14651858.CD012214.pub2
Cantrell LA, Zhou C, Mendivil A, Malloy KM, Gehrig PA, Bae-Jump VL (2010) Metformin is a potent inhibitor of endometrial cancer cell proliferation—implications for a novel treatment strategy. Gynecol Oncol 116(1):92–98
Xie Y, Wang YL, Yu L, Hu Q, Ji L, Zhang Y et al (2011) Metformin promotes progesterone receptor expression via inhibition of mammalian target of rapamycin (mTOR) in endometrial cancer cells. J Steroid Biochem Mol Biol 126(3–5):113–120
Mitsuhashi A, Sato Y, Kiyokawa T, Koshizaka M, Hanaoka H, Shozu M (2016) Phase II study of medroxyprogesterone acetate plus Metformin as a fertility-sparing treatment for atypical endometrial hyperplasia and endometrial cancer. Ann Oncol 27(2):262–266
Mitsuhashi A, Habu Y, Kobayashi T, Kawarai Y, Ishikawa H, Usui H et al (2019) Long term outcomes of progestin plus Metformin as a fertility-sparing treatment for atypical endometrial hyperplasia and endometrial cancer patients. J. Gynecol. Oncol. 30(6):e90-e
Shan W, Wang C, Zhang Z, Gu C, Ning C, Luo X et al (2014) Conservative therapy with Metformin plus megestrol acetate for endometrial atypical hyperplasia. J Gynecol Oncol 25(3):214–220
Cao C, Zhou JY, Xie SW, Guo XJ, Li GT, Gong YJ, Zhu Y (2019) Metformin enhances nomegestrol acetate suppressing growth of endometrial cancer cells and may correlate to downregulating mTOR activity in vitro and in vivo. Int J Mol Sci. 20(13):3308
Gupta A, Desai A, Bhatt S (2017) Imaging of the Endometrium: physiologic changes and diseases: women’s imaging. Radiographics. 37(7):2206–2207. a review publication of the Radiological Society of North America, Inc
Behnamfar F, Ghahiri A, Tavakoli M (2014) Levonorgestrel-releasing intrauterine system (Mirena) in compare to medroxyprogesterone acetate as a therapy for endometrial hyperplasia. J Res Med Sci 19(8):686–690
Ko EM, Sullivan S, Rambally B, O’Connor S, Everett R, Thakker D, Moore DT, Byron J, Baejump VL (2016) Metformin for the treatment of endometrial hyperplasia. J Clin Oncol. 34(15):5592–5592
Acosta-Torres S, Murdock T, Matsuno R et al (2020) The addition of Metformin to progestin therapy in the fertility-sparing treatment of women with atypical hyperplasia/endometrial intraepithelial neoplasia or endometrial cancer: Little impact on response and low live-birth rates. Gynecol Oncol 157(2):348–356. https://doi.org/10.1016/j.ygyno.2020.02.008
Wildemeersch D, Janssens D, Pylyser K et al (2007) management of patients with non-atypical and atypical endometrial hyperplasia with a levonorgestrel- releasing intrauterine system: long-term follow-up. Maturitas 57:210–213
Ra V, Soneja H, Bhatia K, Ganesan R, Rollason T, Justin Clark T, Gupta JK (2008) The effectiveness of a levonorgestrel-releasing intrauterine system (LNG-IUS) in the treatment of endometrial hyperplasia—A long-term follow-up study. European Journal of Obstetrics & Gynecology and Reproductive Biology 139:169–175
Kim ML, Seong SJ (2013) Clinical applications of levonorgestrel-releasing intrauterine system to gynecologic diseases. Obstet Gynecol Sci 56:67–75
Sharifzadeh F, Aminimoghaddam S, Kashanian M, Fazaeli M, Sheikhansari N. A comparison between the effects of Metformin and megestrol on simple endometrial hyperplasia. Gynecological Endocrinology: 2016:152–5. https://doi.org/10.1080/09513590.2016.1223285
Tabrizi AD, Melli MS, Foroughi M, Ghojazadeh M, Bidadi S (2014) Antiproliferative effect of Metformin on the endometrium - a clinical trial. Asian Pac J Cancer Prev 15(23):10067–10070
Nwanodi O (2017) Progestin intrauterine devices and metformin: endometrial hyperplasia and early stage endometrial cancer medical management. Healthcare. 5(3):30. https://doi.org/10.3390/healthcare5030030
Fatima M, Sadeeqa S, Nazir SUR (2018) Metformin and its gastrointestinal problems: A review. Biomed Res 29(11):2285–2289
Gunderson CC, Fader AN, Carson KA, Bristow RE (2012) Oncologic and reproductive outcomes with progestin therapy in women with endometrial hyperplasia and grade 1 adenocarcinoma: a systematic review. Gynecol Oncol 125:477–482
Acknowledgements
Not applicable.
Funding
Self-funded.
Author information
Authors and Affiliations
Contributions
M Shaaban: Protocol/project development, manuscript writing/editing. MM Abd-Elgelil: Data collection and analysis. EA Kishk: Data collection and management, Manuscript writing/editing. OT Taha: Data collection and management, Data analysis, Manuscript writing/editing. RE Khamees: Data collection and management, Manuscript writing/editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in in the study were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. The study was conducted after the approval of our research ethics committee. Informed consent was obtained from all participants before recruitment.
Consent for publication
Not applicable.
Competing interests
None.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Taha, O.T., Abd-Elgelil, M.M., Kishk, E.A. et al. Metformin versus levonorgestrel-releasing intrauterine system in the management of endometrial hyperplasia: a randomized clinical trial. Middle East Fertil Soc J 28, 20 (2023). https://doi.org/10.1186/s43043-023-00145-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43043-023-00145-7