Abstract
Background
An extensive quest for genetic variables impacting the susceptibility for type 2 diabetes mellitus (T2DM) and other cardiometabolic disorders has been sparked by the global growth in the frequency of those widespread ailments. Although the association between several SNPs and T2DM has been reported in prior research, little is known about the association between SNPs FTO rs8050136, and CDKAL1 rs10946398 and T2DM in Egyptian population. This study aims to investigate these two identified SNPs in Egyptian diabetic patients to ascertain their underlying genetic influences on T2DM. This study included 50 diabetic patients and 50 healthy subjects. Each individual underwent a clinical assessment and total body examination, laboratory investigations including liver enzymes, fasting blood sugar, glycated hemoglobin (HbA1C), and lipid profile. The DNA Purification Kit was used to separate genomic DNA from the whole blood. Gene polymorphism was detected via PCR-REFLP and PCR-AS methods.
Results
There was a significant association between the presence of the C allele in the FTO gene at rs8050136 and T2DM among studied people. The patient group had a considerably higher frequency of the FTO "AC" genotype and the "C" allele (P < 0.05). Additionally, only the wild-type homozygous “GG” of the CDKAL1 rs10946398 was found in all the studied cases.
Conclusions
The FTO "CC" genotype is significantly associated with T2DM in the Egyptian population. However, no association was detected between CDKAL1 rs10946398 and T2DM. This result may be attributed to the small sample size or the rare incidence of this SNP in the Egyptian population. The study suggests verifying the findings on a larger sample and looking into the relationship between T2DM and additional gene polymorphisms.
Highlights
-
Type 2 diabetes becomes a challenge health problem owing to its growing prevalence, complex pathogenesis.
-
It is necessary to study the relationship between different SNPs and T2D to identify their underlying genetic influences on T2DM, which could help to delay the onset of the disease or prevent it or its related complications.
-
In Egypt, limited studies evaluate the association between SNPs in Fat mass and obesity-associated protein (FTO rs8050136); as well as CDKAL1 rs10946398 and T2DM.
-
Thus, it may be useful to idenfiy the association between these polymorphisms and T2D.
Similar content being viewed by others
1 Background
The most prevalent and clinically significant metabolic illness is type 2 diabetes (T2D), which has become a major global healthcare burden in recent years [1]
The International Diabetes Federation (IDF) estimates that 463 million adults between the ages of 20–80 years had diabetes in 2019, and it is anticipated to reach 700 million by the year 2045. In 2019, it caused 4.2 million deaths worldwide. At least 720 billion USD in medical expenses were related to diabetes in 2019. Additionally, since 1 in 3 diabetics—or 232 million people—had their diabetes misdiagnosed, the true disease burden of T2DM is probably underrepresented [2]. In Egypt, as in other countries, T2DM appears to be a significant public health issue among the population as there are more than 8.222 million adults (15.1%) with diabetes in 2017 [3].
T2DM is one of the most prevalent disorder caused by genetic, environmental factors, and metabolic defects [4]. Numerous single nucleotide polymorphisms (SNPs) have been implicated with the development of T2DM.
In order to prevent and cure T2DM, genetic risk factors must be investigated. Information gleaned from genome-wide association studies (GWAS) [5,6,7] indicated that several single nucleotide polymorphisms (SNPs) play a vital role in the development of T2DM. They also recognized common genetic variants associated with the discovery of diabetes susceptibility genes.
Numerous studies reported that fat-mass and obesity-associated protein (FTO) polymorphisms are firmly hazard factors for type 2 diabetes (T2D) [8, 9]. It is involved in the fatty acid metabolism, repairing DNA and post-translational modifications [10].
The CDKAL1 “cyclin-dependent kinase 5 (CDK5) regulatory subunit-associated protein 1-like 1” gene encodes a 65-kD protein which has a protein domain in common with a neuronal protein “cyclin-dependent kinase 5 (cdk5) regulatory subunit-associated protein 1 (CDK5RAP1)” that especially inhibits activation of Cyclin-dependent kinase 5 (CDK5). Serine/threonine protein kinase CDK5 is known to be a crucial molecule in the brain and has a number of extra-neuronal impacts [11]. It has been demonstrated that CDK5 inhibits the release of insulin in response to glucose, contributes to the glucotoxicity-induced reduction in insulin gene expression, and contributes to the pathophysiology of cell dysfunction and vulnerability to type 2 diabetes. One can therefore hypothesize that increased activity of CDK5 in β cells would be caused by reduced CDKAL1 expression which would lead to decreased insulin secretion [12]. Patients with diabetes were found to have a lower chance of developing diabetic nephropathy when CDKAL1 rs7756992 was present [13]. It appears that the disparity in how different people react to environmental stimuli is an important reason for ethnic variance and race-specific inheritance. Although the association between several SNPs and T2DM has been reported in prior research [14, 15], little is known about the association between SNPs FTO rs8050136, and CDKAL1 rs10946398 and T2DM in Egyptian population.
Therefore, our aim is to investigate these two identified SNPs (FTO rs8050136, and CDKAL1 rs10946398) in some Egyptian diabetic patients to identify their underlying genetic influences on T2DM in this group, which could help to delay the onset of the disease or prevent it or its related complications.
2 Subjects and methods
The present study included 100 individuals (50 with T2DM and 50 apparently healthy controls) collected from outpatients’ clinics of the Internal Medicine Department of Kasr Al Ainy Hospital Cairo, Egypt. All participants are between 22 and 60 years old.
2.1 Exclusion criteria
-
(1)
Cases and subjects with ages < 22 and > 60 years.
-
(2)
People with type 1 diabetes or any other form of diabetes.
-
(3)
People suffering from chronic endocrine or renal conditions, liver illness, thyroid issues, chromosomal abnormality or other diseases.
-
(4)
People suffering from any prior or present malignant disease.
-
(5)
Patients who are receiving corticosteroid or hormone replacement treatment.
The Medical Research Ethical Committee at the National Research Center in Cairo, Egypt, gave its approval to the study plan under number (2153042021). All participants gave their permission in writing after being fully informed.
All patients and controls underwent a thorough history taking process that included questions about smoking history, occupational chemical exposure history, drug use history, prior illnesses, etc. Body mass index (BMI) was calculated. Also, liver enzymes (ALT, AST, and ALP), serum albumin, serum creatinine, urea, lipid profile, fasting blood sugar, and HbA1C were measured in all individuals according to the manufacturer’s instructions. The chemicals were bought from Spectrum Company, Cairo, Egypt.
2.2 Samples collection and methods
10 ml venous blood was drawn from all study participants in the morning after a 12 h fast; a portion of the blood was collected on an EDTA tube for the extraction of DNA. The other portion is left to clot at room temperature. Serum was separated by centrifuging for 10 min at 3000 rpm. Sera were used immediately for other biochemical investigations including alanine aminotransferase (ALT), aspartate aminotransferase (AST), serum albumin, fasting blood glucose, cholesterol, triglycerides, HDL-C, LDL-C following the directions provided by the c. The chemicals were bought from the Cairo, Egypt-based Spectrum Company according to the manufacturer's instructions.
2.3 DNA extraction and polymorphism determination
Using GeneJET genomic DNA Purification Kit (Thermo Scientific, Lithuania, genomic DNA was extracted from blood following the manufacturer's suggestions. Genotyping was carried out via either restriction enzyme digestion of PCR product (PCR-RFLP) or using allele-specific Polymerase Chain Reaction (PCR-AS). Primers sequences [16] and the expected size of PCR products are listed in Table 1.
2.4 PCR conditions and procedure
For PCR-RFLP, each SNP’s reaction was done in a separate tube, whereas PCR-AS occurs in two reaction tubes per sample. The total volume was 20 μl for each PCR reaction including 2 µL of extracted DNA, 10 µL of PCR Master Mix (2X) (Thermo Scientific, Lithuania), 6 µL nuclease-free water, and 1µL of each forward and reverse primers (10 µM). The circumstances for the CDKAL1 gene's cycling were set up as previously advised: initial activation for 3 min at 94 °C, followed by 35 cycles of denaturation at 94 °C for 30 S, annealing at 58 °C for 30 s and extension via heated-up to 72 °C for 40 s. The plate was cooled to 4 °C after the final extension at 72 °C for 5 min. In contrast, For FTO (rs8050136) the cycling conditions were as follows: initial activation at 94 °C for 3 min, followed by thirty-five cycles of denaturation at 94 °C for 30S, annealing at 57 °C for 30 s and elongation at 72 °C for 40 s. The final extension was settled at 72 °C for 5 min and then the plate was cooled to 4 °C.
Following agarose gel electrophoresis and staining with ethidium bromide, the sizes of PCR products were observed under UV light using a 100 bp plus DNA ladder. The restriction enzyme (FastDigest Ssil) was used to digest the PCR products of the CDKAL1 gene. On 3% agarose gels, restriction fragments and 100 bp plus ladder were resolved and the outcomes were analyzed as shown in Table 1.
2.5 Statistical analysis
Utilizing the program SPSS version 21 (Chicago, USA), data were analyzed. Results are shown as mean ± standard deviation or percentages (%). The unpaired Student’s t-test or One-Way ANOVA test was used to compare quantitative clinical data of the diabetic patients and controls as appropriate. The Chi-square test was used to compare the genotype and allele frequencies. Odds ratios (OR) and their 95% confidence intervals (CI) were calculated using the test for Hardy–Weinberg equilibrium. P-values < 0.05 were considered significant.
3 Results
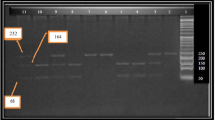
Baseline characteristics of the cases included in the present study are listed in Table 2 The diabetic group's average age was 48.4 ± 11.7 years older on average than the controls' 36.26 ± 12 years, P < 0.001). Besides, there were more females among diabetic cases (86%) compared to 62% in controls. As expected, the BMI was significantly higher in diabetic cases compared with the controls (28.4 ± 5.5 vs. 24.39 ± 3.2; P < 0.001). Also, the levels of FBS, postprandial glucose level, HbA1c, and total cholesterol (TC), were all higher in diabetic cases compared to controls (P < 0.001). Other examined parameters, however, did not vary between the two groups (P > 0.05) (Table 2). No statistically significant difference (P values > 0.05) was found between T2DM patients and controls for the genotypic and allelic frequencies for the tested SNP in the CDKAL1 gene as only the wild-type homozygous GG genotype of the CDKAL1 rs10946398 gene was found in all the samples of the present study (100%), whereas GC and CC genotypes were not found (Fig. 1 and Table 3).
Analysis of FTO gene polymorphism: the agarose gel picture showing the PCR-AS results of FTO gene polymorphism (rs8050136). The upper gel showed the samples with normal primers (normal tube) while the lower gel showed the same sample with mutant primer (mutant tube of the same sample in the upper gel. Lane M: 100 bp Plus DNA ladder, lane (1 & 1\) & (6 & 6\) & (7 & 7\) & (10 & 10\) & (13 & 13\) indicates a homozygous AA sample (246 bp in normal tube), lane (2 & 2\) & (4 & 4\) & (5 & 5\) & (9 & 9\) & (11 & 11\) & (14 & 14\) & (15 & 15\) indicates a heterozygous CA (246 bp in both tubes), lanes (3 & 3\) & (8 & 8\) indicates, a homozygous CC (246 bp in mutant tube).
However, there was a significant association between the presence of the C allele in FTO gene polymorphism (rs8050136) and T2DM among studied people. The distribution of FTO rs8050136 genotypes in diabetic cases was 10 patients with AA genotype (20%) and 25 with AC (50%) while 15 patients (30%) had homozygous mutant genotype CC. However, in the control group, 24 healthy cases had the AA genotype (48%), 15 (30%) had the AC genotype, and 11 (22%) had the CC genotype. Concerning the allele frequency, the “C” allele in the FTO rs8050136 polymorphism was more frequent in the diabetic group (55%) compared to the normal “A” allele which is more frequent in normal cases (63%), (P = 0.011) (Fig. 1 and Table 3). The present study showed that the carrier of rs8050136-C allele (CC + AC genotypes) of FTO has an increased risk for T2DM (OR = 3.69; 95% CI 1.52–8.97) (Table 4).
Table 5 illustrates the observed and expected genotype frequencies of FTO" gene' polymorphisms in the control group. It was detected that FTO genotypes deviated significantly from Hardy-Weinberg equilibrium expectations (Fig. 2).
Table 6 displays the results of the relation between FTO genotypes and the measured biochemical parameters. Statistical analyses showed that there was no significant association between different FTO genotypes and all studied investigated parameters.
4 Discussion
T2DM is considered a chronic complex disease characterized by hyperglycemia which results from reduced insulin secretion, insufficient pancreatic cell response to the ongoing onset of insulin resistance (IR) in peripheral organs, or compromised liver glucose control [17]. Identification of new genetic variants that increase susceptibility to T2DM makes it possible to translate this genetic information to the clinical practice and maybe enhance risk prediction. Additionally, it might aid in delaying or preventing the onset of disease, or any related complications. Such research seeks to resolve the intricacy that caused the illness's urgency. As T2DM is a complicated illness that is brought on by a number of variables, including several genetic and environmental factors allied to manifest the disease. Obesity, positive family history, and physical inactivity are thought to be key risk factors for this illness [18].
Numerous studies have lately been conducted to ascertain how genetic factors influence obesity and T2D susceptibility. These studies provided controversial findings, which may have been caused by the inclusion of multiple ethnic groups. Additionally, the majority of these studies used inconsistent techniques. This work aims to investigate two SNPs (FTO rs8050136, and CDKAL1 rs10946398) in Egyptian diabetic patients to examine the relationship between the presence of these SNPs and the occurrence of T2DM.
According to the current study, the diabetic group's mean age was noticeably older than that of the controls. The fact that metabolic disorders, involving T2D and cardiovascular diseases, are tightly related to the aging process may be because IR is one of the early preconditions of DM, and its consequences related to metabolic cardiovascular diseases are frequently found in the elderly. IR may occur as a result of a loss of lean body mass (LBM) and an increase in body fat, chiefly visceral adiposity, which regularly comes with aging. The mechanism related to T2DM showed that aging results in either a decline of insulin sensitivity or alteration & inadequate compensation of β-cell functional mass in the face of increasing IR [19]. Additionally, compared to controls, diabetes cases had a higher proportion of females. This may be due to diversities between men and women in predisposition clinical presentation, and development of the disease. Also, the difference in biology, socioeconomic status, lifestyle, and environmental impact may be another cause [20].
As expected, diabetic cases' mean BMIs were much higher than those of the healthy controls. The BMI is a straightforward way of determining how far an individual's body weight deviates from what is considered normal or ideal. It is estimated that 36.9% of men and 38.0% of women with BMI ≥25 kg/m2 have type 2 diabetes worldwide [7]. The present analysis of BMI showed that the majority of our enrolled control individuals (90%) have a normal weight (BMI, 19–25) while in the diabetic group, (4%) were overweight (BMI, 25.1–30) and 44% were obese (BMI, more than 30). Previous studies revealed that as weight classes increase, diabetes prevalence dose as well. Given that over half of the persons with diabetes are obese and that nearly one-four have poor glycemic control, weight loss is an important strategy in efforts to lessen the burden of diabetes on the health care system [21]. Additionally, overweight or obese patients have a higher rate of cardiac events (such as the acute coronary syndrome and heart failure) and other chronic diseases [22]. Also, the levels of FBG, postprandial glucose level, and HbA1c were all higher in diabetic cases compared to controls (all P < 0.001). The preferred test for chronic diagnosis of diabetes is currently HbA1c since it reliably measures chronic glycemia and corresponds well with the risk of long-term consequences from diabetes [23]. Additionally, it has been recommended that recuperating glycemic state regulation in T2DM patients is more important for preventing and dramatically lowering the risk of cardiovascular accidents in micro and macrovascular complications than treating dyslipidemia. It was anticipated that the fatality rate would decrease by 10% with a 0.2% decrease in HbA1c levels [24]. One of the key biochemical indicators of T2DM and cardiovascular disease risk is cholesterol. Our research revealed that, according to the lipid profile, patients' mean total cholesterol levels are significantly greater than those of controls (P < 0.001) in comparison. This result is not unexpected given that dyslipidemia is an established risk factor for cardiovascular disease and a feature of metabolic syndrome [25]. However, there were no appreciable variations in the mean triglyceride, HDL, or LDL values between patients and controls (P > 0.05).
Numerous GWAS have shown that the CDKAL1 gene increases the body's need for insulin when glucose levels are high, and pancreatic cells continue to be stimulated, which may limit CDK5 function in pancreatic cells. By decreasing the expression of insulin genes, insulin production is decreased. The related protein 1-LIAK 1 (CDKAL1) gene is regulated by CDK5, which is one of the most recurrent risk genes for T2DM, and CDK5 mutations may result in decreased insulin production, which raises the risk of T2DM. The SNP rs10946398 of CDKAL1 in particular had the highest connection with T2DM [26].
Despite the fact that CDKAL1 (rs10946398 G > C) is an important susceptibility polymorphism for T2DM in many populations, we were unable to find a relationship between the two in our cases. Only the wild-type homozygous GG genotype of CDKAL rs10946398 was identified in the current study; while GC and CC genotypes were not identified. This finding may be explained by other factors, such as the diverse genetic backgrounds of the various populations, or by the limited sample size or rare incidence of this SNP in the Egyptian population, which necessitates a larger sample size for detecting the other genotypes. Additionally, El-Lebedy et al. and Pineda-Cortel [27, 28] studies revealed no correlation between CDKAL1 rs7756992 and T2DM. However, this is in contrast with previously published study which reported that CDKAL1 RS10946398 was positively associated with T2DM [29].
FTO, a gene recently identified in genome-wide investigations for T2DM, is crucial for managing energy balance and regulating body fat mass through lipolysis [17]. Our study's objective was to examine the relationship between the genetic variant FTO rs8050136 A > C and T2DM susceptibility in the Egyptian population.
In our investigation, genotypic frequencies for the FTO (rs8050136) were statistically analyzed, and the results displayed a considerable dissimilarity between both studied groups in the FTO rs8050136 A > C genetic variant. Additionally, statistical analysis demonstrated that there is a remarkable variation between the diabetic cases and controls under the dominant model. Our results are in line with a previously published studies which suggest a significant association between T2DM and variants in FTO (rs8050136) [17, 30,31,32]. In contradiction to our findings, it has been stated that FTO (rs8050136) polymorphism does not correlate with susceptibility to T2DM [33, 34].
5 Conclusions
The present study shows for the first time that rs10946398 gene polymorphism in the CDKAL1 gene was not associated with T2DM susceptibility among the Egyptian population. This could be explained by the differences in ethnicity, the sample size, environmental risk factors, or other causes as yet undetermined. Additionally, the FTO rs8050136 gene polymorphism has shown a consistent association with the susceptibility of T2DM in the Egyptian population. This may help in understanding the biology of this complex disease. Further studies on large scale should be done to confer the association of SNPs (FTO rs8050136, and CDKAL1 rs10946398) with the susceptibility of T2DM.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- T2DM:
-
Type 2 diabetes mellitus
- FTO:
-
Fat-mass and obesity-associated protein
- CDKAL1:
-
Cyclin-dependent kinase 5 (CDK5) regulatory subunit-associated protein 1-like 1
- 2 h PPG:
-
2 Hour post-prandial blood glucose
- HbA1C:
-
Glycated hemoglobin
- SNPs:
-
Single nucleotide polymorphisms
- IDF:
-
International Diabetes Federation
- GWAS:
-
Genome-wide association studies
- CDK5:
-
Cyclin-dependent kinase 5
- BMI:
-
Body mass index
- FBG:
-
Fasting blood glucose
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- ALP:
-
Alkaline phosphatase
- ALB:
-
Albumin
- TC:
-
Total cholesterol
- TG:
-
Triglycerides
- HDL:
-
High-density lipoprotein
- LDL:
-
Low-density lipoprotein
- PCR-RFLP:
-
Restriction enzyme digestion of PCR product
- PCR-AS:
-
Allele-specific polymerase chain reaction
- OR:
-
Odds ratios
- CI:
-
Confidence intervals
- IR:
-
Insulin resistance
- LBM:
-
Lean body mass
References
Reed J, Bain S, Kanamarlapudi V (2021) A review of current trends with type 2 diabetes epidemiology, aetiology, pathogenesis, treatments and future perspectives. Diabetes Metab Syndr Obes Targets Ther 14:3567
Verma AK, Goyal Y, Bhatt D, Dev K, Alsahli MA, Rahmani AH, Almatroudi A (2021) A compendium of perspectives on diabetes: a challenge for sustainable health in the modern era. Diabetes, Metab Syndr Obes Targets Ther 14:2775
Federation I: IDF diabetes atlas eighth edition 2017. In.: International diabetes federation (2017)
Ortiz-Martínez M, González-González M, Martagón AJ, Hlavinka V, Willson RC, Rito-Palomares M (2022) Recent developments in biomarkers for diagnosis and screening of type 2 diabetes mellitus. Curr Diabetes Rep 22(3):95–115
Al-Daghri NM, Alkharfy KM, Alokail MS, Alenad AM, Al-Attas OS, Mohammed AK, Sabico S, Albagha OM (2014) Assessing the contribution of 38 genetic loci to the risk of type 2 diabetes in the Saudi Arabian Population. Clin Endocrinol 80(4):532–537
Hara K, Fujita H, Johnson TA, Yamauchi T, Yasuda K, Horikoshi M, Peng C, Hu C, Ma RC, Imamura M (2014) Genome-wide association study identifies three novel loci for type 2 diabetes. Hum Mol Genet 23(1):239–246
Ng MC, Shriner D, Chen BH, Li J, Chen W-M, Guo X, Liu J, Bielinski SJ, Yanek LR, Nalls MA (2014) Meta-analysis of genome-wide association studies in African Americans provides insights into the genetic architecture of type 2 diabetes. PLoS Genet 10(8):e1004517
Bravard A, Lefai E, Meugnier E, Pesenti S, Disse E, Vouillarmet J, Peretti N, Rabasa-Lhoret R, Laville M, Vidal H (2011) FTO is increased in muscle during type 2 diabetes, and its overexpression in myotubes alters insulin signaling, enhances lipogenesis and ROS production, and induces mitochondrial dysfunction. Diabetes 60(1):258–268
Kamura Y, Iwata M, Maeda S, Shinmura S, Koshimizu Y, Honoki H, Fukuda K, Ishiki M, Usui I, Fukushima Y (2016) FTO gene polymorphism is associated with type 2 diabetes through its effect on increasing the maximum BMI in Japanese men. PLoS One 11(11):e0165523
Gerken T, Girard CA, Tung YC, Webby CJ, Saudek V, Hewitson KS, Yeo GS, McDonough MA, Cunliffe S, McNeill LA et al (2007) The obesity-associated FTO gene encodes a 2-oxoglutarate-dependent nucleic acid demethylase. Science (New York, NY) 318(5855):1469–1472
Smith DS, Greer PL, Tsai LH (2001) Cdk5 on the brain. Cell Growth Differ. Mol. Biol. J. Am. Assoc. Cancer Res. 12(6):277–283
Dehwah MA, Wang M, Huang QY (2010) CDKAL1 and type 2 diabetes: a global meta-analysis. Genet. Mol. Res. GMR 9(2):1109–1120
Lasram K, Ben Halim N, Benrahma H, Mediene-Benchekor S, Arfa I, Hsouna S, Kefi R, Jamoussi H, Ben Ammar S, Bahri S et al (2015) Contribution of CDKAL1 rs7756992 and IGF2BP2 rs4402960 polymorphisms in type 2 diabetes, diabetic complications, obesity risk and hypertension in the Tunisian population. J Diabetes 7(1):102–113
Abosheasha MA, Zahran F, Bessa SS, El-Magd MA, Mohamed TM (2019) Association between a novel G94A single nucleotide polymorphism in ATP1A1 gene and type 2 diabetes mellitus among Egyptian patients. J Res Med Sci Off J Isfahan Univ Med Sci 24:62
Abdel-Kareim A, Ebraheem Z, ElAlfy A, Awwad M, Marei A (2023) A possible correlation between the SNP rs10830963 of the MTNR1B gene and early-onset Type 2 Diabetes mellitus (EOD) in a sample of the egyptian population. Egypt Acad J Biol Sci C, Physiol Mol Biol 15(1):393–402
Altalalgah MN, Thalathini M (2018) Genetic polymorphism and risk of having type 2 diabetes in a palestinian population: a study of 16 gene polymorphisms. Insulin 3(1):1–6
Bego T, Čaušević A, Dujić T, Malenica M, Velija-Asimi Z, Prnjavorac B, Marc J, Nekvindová J, Palička V, Semiz S (2019) Association of FTO gene variant (rs8050136) with type 2 diabetes and markers of obesity, glycaemic control and inflammation. J Med Biochem 38(2):153–163
Hu C, Wu J, Jia W (2014) Biomarkers of metabolic disorders: diagnostic and prognostic values, and insights into the pathogenesis. Biomed Res Int 2014:586272
Suastika K, Dwipayana P, Semadi MS, Kuswardhani RT (2012) Age is an important risk factor for type 2 diabetes mellitus and cardiovascular diseases. Glucose Toler 5:67–80
Kautzky-Willer A, Harreiter J, Pacini G (2016) Sex and gender differences in risk, pathophysiology and complications of type 2 diabetes mellitus. Endocr Rev 37(3):278–316
Nguyen NT, Nguyen XM, Lane J, Wang P (2011) Relationship between obesity and diabetes in a US adult population: findings from the National Health and Nutrition Examination Survey, 1999–2006. Obes Surg 21(3):351–355
Costanzo P, Cleland JG, Pellicori P, Clark AL, Hepburn D, Kilpatrick ES, Perrone-Filardi P, Zhang J, Atkin SL (2015) The obesity paradox in type 2 diabetes mellitus: relationship of body mass index to prognosis: a cohort study. Ann Intern Med 162(9):610–618
Sherwani SI, Khan HA, Ekhzaimy A, Masood A, Sakharkar MK (2016) Significance of HbA1c test in diagnosis and prognosis of diabetic patients. Biomark Insights 11:95–104
Vaag AA (2006) Glycemic control and prevention of microvascular and macrovascular disease in the Steno 2 study. Endocr Pract Off J Am Coll Endocrinol Am Assoc Clin Endocrinol 12(Suppl 1):89–92
Expert Panel on Detection E (2001) Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (adult treatment panel III). JAMA 285(19):2486–2497
Xu N, Zhang TT, Han WJ, Yin LP, Ma NZ, Shi XY (2021) Association of CDKAL1 RS10946398 gene polymorphism with susceptibility to diabetes mellitus type 2: a meta-analysis. J Diabetes Res 2021:1254968
El-Lebedy D, Ashmawy I (2016) Common variants in TCF7L2 and CDKAL1 genes and risk of type 2 diabetes mellitus in Egyptians. J Genet Eng Biotechnol 14(2):247–251
Pineda-Cortel MR, Baybayan K, Bello P, Camenforte L, Ching S, Conti K, Ignacio J, Diaz J, Villavieja A, Pandac J et al (2021) Genetic association of rs7754840 and rs7756992 polymorphisms in the <i>CDKAL1</i> gene and gestational diabetes mellitus in selected Filipino pregnant women. Asian Pac J Reprod 10(4):155–161
Verma AK, Beg MMA, Khan NA, Goyal Y, Dev K, Joshi PC (2020) CDKAL1 (rs10946398) and TCF7L2 (rs7903146) gene polymorphisms and their association with risk of type-2 diabetes mellitus in population of Uttarakhand, India. Meta Gene 26:100767
Vasan SK, Karpe F, Gu HF, Brismar K, Fall CH, Ingelsson E, Fall T (2014) FTO genetic variants and risk of obesity and type 2 diabetes: a meta-analysis of 28,394 Indians. Obesity 22(3):964–970
Almawi WY, Nemr RF, Echtay AS, Saldanha FL, Keleshian SH (2013) Association study of polymorphisms of 15 diabetes-related loci identified by genome-wide association scans with type 2 diabetes in lebanese population. Diabetes Genet Epidemiol Endocr Soc pp. MON-806-MON-806.
O’Beirne SL, Salit J, Rodriguez-Flores JL, Staudt MR, Abi Khalil C, Fakhro KA, Robay A, Ramstetter MD, Al-Azwani IK, Malek JA (2016) Correction: type 2 diabetes risk allele loci in the qatari population. PLoS One 11(8):e0161910
Al-Sinani S, Woodhouse N, Al-Mamari A, Al-Shafie O, Al-Shafaee M, Al-Yahyaee S, Hassan M, Jaju D, Al-Hashmi K, Al-Abri M (2015) Association of gene variants with susceptibility to type 2 diabetes among Omanis. World J Diabetes 6(2):358
He H, Cao WT, Zeng YH, Huang ZQ, Du WR, Guan ND, Zhao YZ, Wei BR, Liu YH, Jing CX et al (2018) Lack of associations between the FTO polymorphisms and gestational diabetes: a meta-analysis and trial sequential analysis. Gene 677:169–175
Acknowledgements
We would like to acknowledge National research Centre for technical and financially supporting the current research.
Funding
We would like to express our gratitude to Egypt's National Research Centre for financially funding the current study.
Author information
Authors and Affiliations
Contributions
HMR, and NAE: conceptualized the study; HMR,, NAE, and MAA: supervision, contributing to experiments design, reviewing, and editing and final approval of the version to be submitted for publication; MAA: samples collections, visualization and investigation, supervision and revised the manuscript; WAE: performed the experiments, analyzed the data, drafted and revised the manuscript. The final version has been read, revised, and edited by all authors to be submitted for publication.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study protocol was approved by the Medical Research Ethical Committee - National research center, Cairo, Egypt. Written informed consent was obtained from all participants.
Consent for publication
The consents of publication are available from the corresponding author on reasonable request
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ragab, H.M., El Maksoud, N.A., Amin, M.A. et al. Gene polymorphism of FTO rs8050136 and CDKAL1 rs10946398 and their association with type 2 diabetes mellitus in the Egyptian patients. Beni-Suef Univ J Basic Appl Sci 12, 64 (2023). https://doi.org/10.1186/s43088-023-00402-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43088-023-00402-8