Abstract
Obstructive sleep apnea (OSA) is accompanied by different metabolic disorders and is linked with a heightened incidence of cardiovascular diseases and death. The information actively engaged in relating obstructive sleep apnea and dyslipidemia is quite uncertain.
This research intended to determine the correlation between obstructive sleep apnea severeness and lipid profile abnormalities. Our study involved 120 cases diagnosed with OSA, who came to the chest department: at Benha University Hospital from September 2021 to December 2022. Included patients indicated for polysomnography because of suspected sleep-breathing disorders. Full history and clinical examination, and full polysomnography and blood samples for lipid profile, were done for all the cases.
Results A total of 120 patients were involved in the study. Most cases were severe OSA (70%). The mean age in severe obstructive sleep apnea was 44.04 years with SD 9.49, Males were 92.9% in severe OSA, and the mean BMI in severe OSA was 36.63 with SD 7.79. LDL was positively correlated with AHI and DI (significantly); triglycerides were positively correlated with AHI, DI, and min oxygen saturation (significantly); and HDL was negatively correlated with AHI and DI significantly. Among lipid profile variables, LDL was the most predictor for AHI severity.
Conclusion Lipid abnormalities are associated with OSA severity. Patients diagnosed with obstructive sleep apnea should be screened for lipid profile to correct any abnormality, hoping to prevent their harmful effects.
Similar content being viewed by others
Background
Obstructive sleep apnea (OSA) is known as recurrent obstruction of the upper airway causing intermittent hypoxemia and fragmentation of sleep caused by arousals [1]. About 13% of males and 6% of females have moderate to severe OSA, more often in old age and individuals with high body mass index [2]. Most manifestations comprise daytime sleepiness, snoring, and sleep interruption. OSA is found in 26% of adults between 30 and 70 years old [3].
The severity of OSA can be defined by the number of apneas or hypopneas and the number of oxygen desaturation episodes recorded per hour of sleep [4].
Intermittent hypoxia in patients with obstructive sleep apnea results in increased oxidative stress and free radical production, which could explain endothelial dysfunction, increased sympathetic activity, systemic inflammatory response, and impaired glucose and lipid metabolism [5].
Methods
This retrospective research comprised 120 patients diagnosed with OSA who came to the Chest Department at Benha University Hospital from September 2021 to December 2022. Patients were ≥ 18 years old and came to the sleep laboratory due to questionable sleep-breathing abnormalities. Exclusion criteria included the presence of infection, the wound on the skin in contact with electrodes, contact allergy, significant neurological disease, and medications influencing sleep.
Full history taking and physical examination including body mass index were done for all included patients, full sleep polysomnography, and blood samples for CBC, ESR, and lipid profile.
Determination of obstructive sleep apnea and its severity was done by polysomnography, performed with the Alice Four Sleep System (Respironics Inc., Murrysville, PA, USA). Sleep parameters were evaluated per the 2007 American Academy of Sleep Medicine (AASM) protocol (Berry et al., 2012). Apnea–hypopnea index (AHI — the number of apneas or hypopneas recorded per hour of sleep) was derived from level 1 polysomnography (PSG).
Discontinuation of airflow or decrease > 90% lasting 10 or more seconds was used for defining apnea, and a decrease of > 30% in airflow associated with 3% or 4% oxygen desaturation was used for defining hypopnea. Mild sleep apnea was described by AHI between 5 and 14, moderate sleep apnea was delineated by AHI between 15 and 29, while the patients with AHI more than 30 had severe sleep apnea.
Oxygen desaturation index (ODI): The number of dips in So2 > 3 or 4% from baseline is divided by total sleep time. The pulse oximeter was attached to the index finger of the nondominant hand.
Statistical analysis
The program used was SPSS version 20. Quantitative data were analyzed using mean and standard deviation, while frequency and percentage were used with qualitative data. The chi-square test to compare frequencies and analysis of variance (ANOVA) was used to compare means. Pearson correlation found the relationship between variables and then logistic regression to predict the most dependent factor. P-value was considered significant if it was ≤ 0.05.
Results
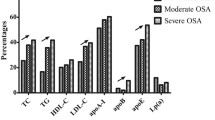
A total of 120 patients were included in the study. Most cases were severe OSA (70%). In severe obstructive sleep apnea, the mean age was 44.04 with SD 9.49, males were 92.9%, and the mean BMI was 36.63 with SD 7.79. LDL was positively correlated with AHI and DI (significantly); triglycerides were positively correlated with AHI, DI, and min oxygen saturation (significantly); and HDL was negatively correlated with AHI and DI significantly.
Among lipid profile variables, LDL was the most predictor for AHI severity.
(1) Comparison between mild, moderate, and severe levels of ASA according to different variables
Mild (14) | Moderate (22) | Severe (84) | ANOVA | p-value | ||||
|---|---|---|---|---|---|---|---|---|
Mean | SD | Mean | SD | Mean | SD | |||
Age | 43.79 | 14.59 | 45.95 | 8.72 | 44.04 | 9.49 | 0.343 | 0.71 |
BMI | 31.67 | 4.64 | 33.85 | 3.86 | 36.63 | 7.79 | 3.87 | 0.024* |
DI | 15.16 | 13.82 | 22.14 | 10.82 | 68.17 | 28.01 | 50.34 | < 0.001** |
MIN DES | 82.5 | 7.87 | 79.68 | 6.79 | 76.0 | 8.03 | 5.29 | 0.006** |
LDL | 105.63 | 36.68 | 114.59 | 25.76 | 141.66 | 29.05 | 13.96 | < 0.001** |
HDL | 45.79 | 9.45 | 39.31 | 10.90 | 38.46 | 6.19 | 5.52 | 0.005** |
TG | 138.9 | 59.24 | 175.96 | 73.05 | 199.96 | 100.13 | 2.89 | 0.059 |
(2) Correlation between LDL, HDL, TG, and different variables
LDL | HDL | TG | ||||
|---|---|---|---|---|---|---|
r | P | r | P | r | P | |
BMI | − 0.156 | 0.088 | − 0.157 | 0.087 | 0.05 | 0.588 |
AHI | 0.416 | < 0.001** | − 0.278 | 0.002** | 0.334 | 0.001** |
DI | 0.265 | 0.003** | − 0.248 | 0.006** | 0.335 | < 0.001** |
MIN DES | − 0.023 | 0.802 | 0.037 | 0.684 | − 0.213 | 0.019* |
(3) Logistic regression to predict the most dependent factor affecting AHI
AHI | Exp (b) | P | LL 95% CI | UL 95% CI |
|---|---|---|---|---|
BMI | 1.11 | 0.01* | 1.02 | 1.20 |
LDL | 1.04 | < 0.001** | 1.02 | 1.07 |
HDL | 0.968 | 0.407 | 0.895 | 1.05 |
TG | 1.0 | 0.245 | 0.997 | 1.01 |
Discussion
Preceding research found a significant association between obstructive sleep apnea severity and lipid profile defects [6, 7]. Extremely raised cholesterol or triacylglycerols is accompanied by increased cardiovascular risk, chiefly due to the rapidity of the atherosclerotic process. However, it is found that aberrant lipid elimination may be accountable for this process due to the reduced action of lipoprotein lipase and consequent confirmation of LDL subclass B, which by oxidation causes rapid vascular damage [8].
The action of lipoprotein lipase is maintained by insulin and lowered by epinephrin and cortisol. This caused different research to highlight the role of insulin resistance and increased sympathetic activity as major pathological mechanisms related to obstructive sleep apnea [9, 10]. Similarly, diverse animal models found that the existence and severity of intermittent hypoxia are significantly correlated to hyperlipidemia and liver oxidative stress, causing raised lipolysis and diminished lipoprotein clearance [11].
The current work aimed to correlate lipid abnormalities with the severity of obstructive sleep apnea. This study on 120 patients with newly diagnosed obstructive sleep apnea showed a significant correlation between lipid abnormalities and obstructive sleep apnea severity. In our study, lipid abnormalities associated with OSA severity were triglycerides, high-density lipoprotein (HDL), and low-density lipoprotein (LDL). These lipid abnormalities were more obvious in patients with severe obstructive sleep apnea (AHI ≥ 30 per h). LDL was positively correlated with the apnea–hypopnea index and desaturation index (in statistically significant value).
Triglycerides were positively correlated with the apnea-hypopnea index, desaturation index, and minimum oxygen desaturation. HDL was negatively correlated with AHI and DI (in statistically significant value). In the current work, LDL was the most predictor variable for AHI severity. Guscoth et al. (2021) found a relationship between triglycerides and the severity of obstructive sleep apnea even in patients without central adiposity which was highly significant [12].
The chief mechanisms included in increased cholesterol and triglyceride levels in patients with OSA have increased hepatic triglyceride and cholesterol production, decreased clearance by lipase activity inhibition, and raised free fatty acid mobilization from adipose tissue [13]. Intermittent hypoxia commences overexpression of significant transcription factors in the course of cholesterol and triglyceride biosynthesis [14].
Patients with insulin resistance showed increased lipid levels especially those with severe forms of OSA [15]. In a study done by Togeiro et al. (2013), triglyceride levels were remarkable as a solid index of periodic hypoxia, solely related to AHI [16].
Popadi et al. (2022) found a substantial relationship between HDL with OSA severity, implying that patients with decreased HDL levels have increased apnea–hypopnea index (AHI) [17]. Regarding the defensive cardiovascular effects of high-density lipoproteins (HDL), it is found that definite subfractions of HDL, particularly small HDL subfractions involving small LDL3–7 and small HDL8–10, can have atherogenic consequences in patients with OSA [18].
Alternately, bigger HDL subfractions can have disabled antioxidant action due to raised HDL lipid peroxide degree and diminished serum paraoxonase-1 (PON1) activity, which are the two chief elements causing the antioxidant activity of HDL [19]. This could be a conceivable illustration of insufficient proof of the defensive effects of HDL in patients with OSA, even in patients with average HDL levels. Cases with diminished antioxidant HDL activity similarly had raised tumor necrosis factor-α (TNF-α) and intercellular adhesion molecule 1 (ICAM-1) values denoting raised systemic inflammation in severe OSA [20]. Research by Koseoglu et al. (2018) implied the compelling role of monocyte to HDL ratio as a possible criterion of OSA severity and its relationship with raised cardiovascular risk [21].
Conclusion
Patients with risk factors for severe forms of OSA, including gender and body mass index, should promptly be evaluated for OSA for management and to reduce the risk of various complications. Also, patients diagnosed with obstructive sleep apnea, especially severe ones, should be screened for lipid profiles to correct any abnormality and protect from long-term harmful effects of their increased level.
The drawbacks of this study were a small sample size, and not discussing hyperglycemia and HTN besides lipid profile.
Availability of data and materials
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
Abbreviations
- OSA:
-
Obstructive sleep apnea
- LDL:
-
Low-density lipoprotein
- HDL:
-
High-density lipoprotein
- PSG:
-
Polysomnography
- AHI:
-
Apnea-hypopnea index
- BMI:
-
Body mass index
- CBC:
-
Complete blood count
- ESR:
-
Erythrocyte sedimentation rate
- ODI:
-
Oxygen desaturation index
References
Sforza E, Roche F (2016) Chronic intermittent hypoxia and obstructive sleep apnea: an experimental and clinical approach. Hypoxia (Auckl) 4:99–108. https://doi.org/10.2147/HP.S1030
Morsy NE, Farrag NS, Zaki NFW, Badawy AY, Abdelhafez SA, ElGilany AH et al (2019) Obstructive sleep apnea: personal, societal, public health, and legal implications. Rev Environ Health 34(2):153–169. https://doi.org/10.1515/reveh-2018-0068
Peppard PE, Young T, Barnet JH, Palta M, Hagen EW, Hla KM (2013) Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol 177(9):1006–1014. https://doi.org/10.1093/aje/kws342
Semelka M, Wilson J, Floyd R (2016) Diagnosis and treatment of obstructive sleep apnea in adults. Am Fam Physician 94(5):355–360. https://doi.org/10.1037/e676562012-001
Arnaud C, Dematteis M, Pepin JL, Baguet JP, Lévy P (2009) Obstructive sleep apnea, immuno-inflammation, and atherosclerosis. Semin Immunopathol 31(1):113–125. https://doi.org/10.1007/s00281-009-0148-5
Gündüz C, Basoglu OK, Hedner J, Zou D, Bonsignore MR, Hein H et al (2018) Obstructive sleep apnoea independently predicts lipid levels: data from the European Sleep Apnea Database. Respirology 23(12):1180–1189. https://doi.org/10.1111/resp.13372
Gündüz C, Basoglu OK, Hedner J, Bonsignore MR, Hein H, Staats R et al (2019) Hyperlipidemia prevalence and cholesterol control in obstructive sleep apnea: data from the European Sleep Apnea Database (ESADA). J Intern Med 286(6):676–688. https://doi.org/10.1111/joim.12952
Mirrakhimov AE, Ali AM (2013) Pathobiology of obstructive sleep apnea-related dyslipidemia: focus on the liver. ISRN Cardiol 2013:687069. https://doi.org/10.1155/2013/687069
da Jung W, Hwang SH, Lee YJ, Jeong DU, Park KS (2016) Oxygen desaturation index estimation (through unconstrained cardiac sympathetic activity assessment using three ballisto cardiographic systems. Respiration 92(2):90–97. https://doi.org/10.1159/000448120
Almendros I, García-Río F (2017) Sleep apnoea, insulin resistance and diabetes: the first step is in the fat. Eur Respir J 49(4):1700179. https://doi.org/10.1183/13993003.00179-2017
Drager LF, Jun JC, Polotsky VY (2010) Metabolic consequences of intermittent hypoxia: relevance to obstructive sleep apnea. Best Pract Res Clin Endocrinol Metab 24(5):843–851. https://doi.org/10.1016/j.beem.2010.08.011
Guscoth LB, Appleton SL, Martin SA, Adams RJ, Melaku YA, Wittert GA (2021) The association of obstructive sleep apnea and nocturnal hypoxemia with lipid profiles in a population-based study of community-dwelling Australian men. Nat Sci Sleep 13:1771–1782. https://doi.org/10.2147/NSS.S327478
Drager LF, Tavoni TM, Silva VM, Santos RD, Pedrosa RP, Bortolotto LA et al (2018) Obstructive sleep apnea and effects of continuous positive airway pressure on triglyceride-rich lipoprotein metabolism. J Lipid Res 59(6):1027–1033. https://doi.org/10.1194/jlr.M083436
Li J, Grigoryev DN, Ye SQ, Thorne L, Schwartz AR, Smith PL et al (2005) Chronic intermittent hypoxia upregulates genes of lipid biosynthesis in obese mice. J Appl Physiol 99(5):1643–1648. https://doi.org/10.1152/japplphysiol.00522.2005
Liu A, Cardell J, Ariel D, Lamendola C, Abbasi F, Kim SH et al (2015) Abnormalities of lipoprotein concentrations in obstructive sleep apnea are related to insulin resistance. Sleep 38(5):793–799. https://doi.org/10.5665/sleep.4678
Togeiro SM, Carneiro G, Ribeiro Filho FF, Zanella MT, Santos-Silva R, Taddei JA et al (2013) Consequences of obstructive sleep apnea on metabolic profile: a population-based survey. Obes (Silver Spring) 21(4):847–851. https://doi.org/10.1002/oby.20288
Popadic V, Brajkovic M, Klasnja S(2022) Correlation of dyslipidemia and inflammation with obstructive sleep apnea severity 13. https://doi.org/10.3389/fphar.2022.897279
Kollar B, Siarnik P, Hluchanova A, Klobucnikova K, Mucska I, Turcani P et al (2021) The impact of sleep apnea syndrome on the altered lipid metabolism and the redox balance. Lipids Health Dis 20(1):175. https://doi.org/10.1186/s12944-021-01604-8
Yadav R, France M, Aghamohammadzadeh R, Liu Y, Hama S, Kwok S et al (2014) Impairment of high-density lipoprotein resistance to lipid peroxidation and adipose tissue inflammation in obesity complicated by obstructive sleep apnea. J Clin Endocrinol Metab 99(9):3390–3398. https://doi.org/10.1210/jc.2013-3939
Pak VM, Grandner MA, Pack AI (2014) Circulating adhesion molecules in obstructive sleep apnea and cardiovascular disease. Sleep Med Rev 18(1):25–34. https://doi.org/10.1016/j.smrv.2013.01.002
Inonu Koseoglu H, Pazarli AC, Kanbay A, Demir O (2018) monocyte count/HDL cholesterol ratio and cardiovascular disease in patients with obstructive sleep apnea syndrome: clinical and applied thrombosis/hemostasis 24(1) 139–144. ª The Author(s) 2016 Reprints and permission: sagepub.com/journalsPermissions.nav DOI: https://doi.org/10.1177/1076029616677803journals.sagepub.com/home/cat
Acknowledgements
The authors are grateful to the medical staff at the Department of Chest Diseases at Benha University Hospital for their kind help and support throughout the study.
Funding
No financial support was relevant to this study.
Author information
Authors and Affiliations
Contributions
Conceptualization, RMH and MAE. Methodology, RMH, MAE, RSR, and HEN. Writing—original draft preparation, RMH, MAE, and RSR. Writing—review and editing, RMH, statistical analysis and supervision. RMH, MAE, HEN, and RSR. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent for participation
This work was approved by the Research Ethics Committee (MoHP No.: 0018122017/Certificate No.: 1017).
Consent for publication
Institutional consent was taken from the Ethical Committee of the Benha Faculty of Medicine.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hendy, R.M., Elawady, M.A., Nasr, H.E. et al. Correlation of obstructive sleep apnea severity and lipid profile level. Egypt J Bronchol 18, 7 (2024). https://doi.org/10.1186/s43168-024-00260-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s43168-024-00260-7