Abstract
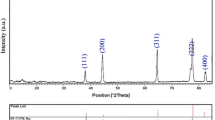
A facile and completely green route to synthesize Au nanoparticles by mixing the Au(III) ions-dissolved rice wine and soda at a slightly elevated temperature in the absence of extra protective agents was developed. Rice wine was used as a solvent and a reducing agent. Also, soda not only functioned as a protective agent but also played a role of base catalyst. No extra protective agents are needed. From the analyses of UV/VIS absorption spectra, TEM, and XRD patterns, the formation of Au nanoparticles was recognized. The appropriate pH and temperature were around 6.5 and 25–55°C, respectively. The resultant solution was quite stable. No precipitation occurred even after several months. In addition, rice wine and soda are often used as the additives of drinks or foods, and are easily obtained in our daily life. So, it becomes possible that people can easily make the Au nanoparticles at home by the route proposed in this work.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
M. Poliakoff, P. Anastas,Nature, 2001,413, 257
M. Poliakoff, J.M. Fitzpatrick, T.R. Farren, P.T. Anastas,Science, 2002,297, 807
R.A. Gross, B. Kalra,Science, 2002,297, 803
J.M. DeSimone,Science, 2002,297, 799
P. Raveendran, J. Fu, S.L. Wallen,J. Am. Chem. Soc., 2003,125, 13940
M.C. Daniel, D. Astruc,Chem. Rev., 2004,104, 293
M. Haruta,Cattech, 2002,6, 102–115
M. Zayats, A.B. Kharitonov, S.P. Pogorelova, O. Lioubashevski, E. Katz, I. Willner,J. Am. Chem. Soc., 2003,125, 16006
W. Jahn,J. Struct. Biol., 1999,127, 106
S.A. Maier, M.L. Brongersma, P.G. Kik, S. Meltzer, A.A.G. Requicha, H.A. Atwater,Adv. Mater., 2001,13, 1501
D.A. Handley,Colloidal Gold: Principles, Methods, and Applications, Hayat M. A. Ed., Academic Press: San Diego, 1989; Vol. 1, Chapter 2
C. Mangeney, F. Ferrage, I. Aujard, V. Marchi-Artzner, L. Lullien, O. Ouari, E.D. Rékaï, A. Laschewsky, I. Vikholm, J.W. Sadowski,J. Am. Chem. Soc., 2002,124, 5811
M. Schulz-Dobrick, K.V. Sarathy, M. Jansen,J. Am. Chem. Soc., 2005,127, 12816
M. Aslam, L. Fu, M. Su, K. Vijayamohanan, V. P. Dravid,J. Mater. Chem., 2004,14, 1795
J. Liu, G. Qin, P. Raveendram, Y. Ikushima,Chem. Eur. J., 2006,12, 2131
S. Si, R.R. Bhattacharjee, A. Banerjee, T.K. Mandal,Chem. Eur. J., 2006,12, 1256
M.N. Nadagouda, R.S. Varma,Green Chem., 2006,8, 516
P. Raveendran, J. Fu, S.L. Wallen,Green Chem., 2006,8, 34
B. Ankamwar, C. Damle, A. Ahmad, M. Sastry, J. Nanosci.Nanotech., 2005,5, 1665
H. Hirai, Y. Nakao, N. Toshima,J. Macromol. Sci. Chem., 1979,A13, 727
J. Chatt, B.L. Shaw, A.E. Field,J. Chem. Soc., 1964, 3466
K.S. Lee, M.A. El-Sayed,J. Phys. Chem. B, 2005,109, 20331
J.C. Hulteen, C.J. Patrissi, D.L. Miner, E.R. Crosthwait, E.B. Oberhauser, C.R. Martin,J. Phys. Chem. B, 1997,101, 7727
Z.Y. Huang, G. Mills, B. Hajek,J. Phys. Chem., 1993,97, 11542
Author information
Authors and Affiliations
Corresponding author
Additional information
Mr. Chien-Chen Wu currently works as a PhD student in the laboratory of Prof. Chen in Department of Chemical Engineering at National Cheng Kung University (Taiwan). His research focuses on the synthesis of nano-structured materials and their applications in catalyst and electrochemistry.
Dr. Dong-Hwang Chen is a professor of Chemical Engineering Department at National Cheng Kung University (Taiwan). His research works include the synthesis of nanoparticles, the fabrication of composite nanoparticles and thin films, the development of nanomaterials for electromagnetic wave absorption, as well as the fabrication and applications of functionalized magnetic nanocarriers in separation, catalysis, and biomedicine.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Wu, CC., Chen, DH. A facile and completely green route for synthesizing gold nanoparticles by the use of drink additives. Gold Bull 40, 206–212 (2007). https://doi.org/10.1007/BF03215582
Issue Date:
DOI: https://doi.org/10.1007/BF03215582