Abstract
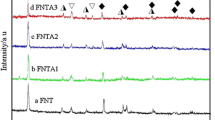
In this paper, Fe-doped TiO2 photocatalyst supported on hollow glass microbeads (Fe-TiO2/beads) is prepared by dip-coating method, which uses hollow glass microbeads as the carriers and tetrabutylorthotitanate [Ti(OC4H9)4] as the raw material. The phase structure, ingredient, morphologies, particle size and shell thickness of the products are characterized by X-ray powder diffraction (XRD), energy-dispersive spectroscopy (EDS) and field emission scanning electron microscope (FESEM). The feasibility of photocatylic degradation of Rhodamine B (RhB) under illumination of UV-vis light is studied. The results show that the core-shell structure catalyst is composed of Fe-doped anatase TiO2 and hollow glass microbeads, and the catalytic activity of the TiO2 is markedly enhanced by Fe ion doping. The optimum concentration of Fe ion is 0.1% (molecular fraction) in the precursor and the photocatalytic activity can be increased to 98% compared with that of the undoped one. The presence of ferrum elements neither influences the transformation of anatase to rutile, nor creates new crystal phases. The possible mechanism of photocatalytic oxidation is also discussed.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
M. R. Hoffmann, S. T. Martin, W. Choi and D. W. Bahnemann, Chem. Rev. 95, 69 (1995). http://dx.doi.org/10.1021/cr00033a004
B. Sun, A. V. Vorontsov and P. G. Smirniotis, Langmuir 19, 3151 (2003). http://dx.doi.org/10.1021/la0264670
A. K. Axelsson and L. J. Dunne, J. Photochem. Photobiol. A: Chem. 144, 205 (2001). http://dx.doi.org/10.1016/S1010-6030(01)00536-6
J. Yu, J. C. Yu, M. K. P. Leung, W. Ho, B. Cheng, X. Zhao and J. Zhao, J. Catal. 217, 69 (2007).
N. Q. Wu, J. Wang, D. N. Tafen, H. Wang and J. G. Zheng, J. Am. Chem. Soc. 132, 6679 (2010). http://dx.doi.org/10.1021/ja909456f
A. Mills, A. Lepre, N. Elliott, S. Bhopal, I. P. Parkin and S. A. O’Neill, J. Photochem. Photobiol. A 160, 213 (2003). http://dx.doi.org/10.1016/S1010-6030(03)00205-3
H. J. Liu, G. G. Liu and X. Y. Shi, Colloids and Surfaces A: Physicochem. Eng. Aspects 363, 35 (2010). http://dx.doi.org/10.1016/j.colsurfa.2010.04.010
R. Erwin, J. P. Daniel and C. Christine, J. Am. Chem. Soc. 131, 18457 (2009). http://dx.doi.org/10.1021/ja907923r
J. G. Yu, J. Yu and J. Zhao, Appl. Catal. B: Environ. 36, 31 (2002). http://dx.doi.org/10.1016/S0926-3373(01)00277-6
C. Natalia, S. Fernando and E. García, J. Phys. Chem. C 112, 1094 (2008). http://dx.doi.org/10.1021/jp0769781
M. Anpo, M. Takeuchi, K. Ikeue and S. Dohshi, Curr. Opin. Solid State Mater. Sci. 6, 381 (2002). http://dx.doi.org/10.1016/S1359-0286(02)00107-9
H. Kisch, L. Zang, C. Lange, W. F. Maier, C. Antonius and D. Meissner, Angew. Chem. Int. Ed. 37, 3034 (1998). http://dx.doi.org/10.1002/(SICI)1521-3773(19981116)37:21$<$3034::AID-ANIE3034$>$3.0.CO;2-2
T. Umebayashi, T. Yamaki, H. Itoh and K. Asai, Appl. Phys. Lett. 81, 454 (2002). http://dx.doi.org/10.1063/1.1493647
R. Asahi, T. Morikawa, T. Ohwaki, K. Aoki and Y. Taga, Science 293, 269 (2001). http://dx.doi.org/10.1126/science.1061051
Y. Yang, X. J. Li, J. T. Chen, L. Y. Wang, J. Photochem. Photobiol. A: Chem. 163, 517 (2004). http://dx.doi.org/10.1016/j.jphotochem.2004.02.008
B. Sun, E. P. Reddy and P. G. Smirniotis, Appl. Catal. B: Environ. 57, 139 (2005). http://dx.doi.org/10.1016/j.apcatb.2004.10.016
E. V. Alexei, F. Yutaka, X. T. Zhang, M. Jin, M. Taketoshi and F. Akira, J. Phys. Chem. B 109, 24441 (2005). http://dx.doi.org/10.1021/jp055090e
S. F. Chen and G. Y. Cao, Desalination 194, 127 (2006). http://dx.doi.org/10.1016/j.desal.2005.11.006
C. Y. Wang, C. Bottcher, D. W. Bahnemann and J. K. Dohrmann, J. Mater. Chem. 13, 2322 (2003). http://dx.doi.org/10.1039/b303716a
P. Kopf, E. Gilbert and S. H. Eberle, J. Photochem. Photobiol. A: Chem. 136, 163 (2000). http://dx.doi.org/10.1016/S1010-6030(00)00331-2
A. Sclafani, L. Palmisano and E. J. Davi, Photochem. Photobiol. A: Chem. 56, 113 (1991).
Y. M. Xu and H. Q. Lu, J. Photochem. Photobiol. A: Chem. 136, 73 (2000). http://dx.doi.org/10.1016/S1010-6030(00)00310-5
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhao, W., Fu, W., Yang, H. et al. Synthesis and Photocatalytic Activity of Fe-doped TiO2 Supported on Hollow Glass Microbeads. Nano-Micro Lett. 3, 20–24 (2011). https://doi.org/10.1007/BF03353647
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03353647