Abstract
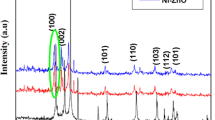
Zinc oxide (ZnO) nanoparticles of varying sizes (20, 44 and 73 nm) have been successfully synthesized by a hybrid electrochemical-thermal method using aqueous sodium bicarbonate electrolyte and sacrificial Zn anode and cathode in an undivided cell under galvanostatic mode at room temperature. The as-synthesized product was characterized by X-ray diffraction (XRD), X-ray photoelectron spectra (XPS), Scanning electron microscopy along with Energy dispersive analysis of X-ray (SEM/EDAX), Transmission electron microscopy (TEM), Ultra Violet - Diffuse reflectance spectroscopic methods (UV-DRS). and UV-DRS spectral methods. The as-synthesized compound were single-crystalline and Rietveld refinement of calcined samples exhibited hexagonal (Wurtzite) structure with space group of P63mc (No.186). The band gaps for synthesized ZnO nanoparticles were 3.07, 3.12 and 3.13 eV, respectively, based on the results of diffuse reflectance spectra (DRS). The electrochemically synthesized ZnO powder was used as photocatalysts for UV-induced degradation of Methylene blue (MB). Photodegradation was also found to be function of exposure time and dye solution pH. It has been found that as-synthesized powder has excellent photocatalytic activity with 92% degradation of MB, indicating ZnO nanoparticles can play an important role as a semiconductor photocatalyst.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Y. H. Zheng, L. Zheng, Y. Y. Zhan, X. Y. Lin, Q. Zheng and K. Wei, Inorg. Chem. 46, 6980 (2007). http://dx.doi.org/10.1021/ic700688f
G. Colon, M. C. Hidalgo, J. A. Navio, E. P. Melian, O. G. Diaz and J. M. Dona, Appl. Catal. B Environ 78, 176 (2008). http://dx.doi.org/10.1016/j.apcatb.2007.09.019
H. G. Kim, P. H. Borse, W. Choi and J.S. Lee, Angew. Chem. Int. Ed 44, 4585 (2005). http://dx.doi.org/10.1002/anie.200500064
G. Marci, V. Augugliaro, M. J. Lopez-Munoz, C. Martin, L. Palmisano, V. Rives, M. Schiavello, R.J.D. Tilley and A. M. Venezia, J. Phys. Chem. B 105, 1026, 1033 (2001).
M. R. Hoffmann, S. T. Martin, W. Y. Choi and D. W. Bahnemann, Chem. Rev. 95, 69 (1995). http://dx.doi.org/10.1021/cr00033a004
T. Sehili, P. Boule and J. Lemaire, J. Photochem, Photobiol. A. Chem. 50, 103 (1989).
J. Villasenor, P. Reyes and G. Pecchi, J. Chem. Technol. Biotechnol. 72, 105 (1998). http://dx.doi.org/10.1002/(SICI)1097-4660(199806)72:2<105::AID-JCTB883>3.0.CO;2-0
M. D. Driessen, T. M. Miller and V. H. Grassian, J. Mol. Catal. A. Chem. 131, 149 (1998). http://dx.doi.org/10.1016/S1381-1169(97)00262-8
M. A. Behnajady, N. Modirshahla and R. Hamzavi, J. Hazard. Mater. B 133, 226 (2006). http://dx.doi.org/10.1016/j.jhazmat.2005.10.022
M. H. Huang, S. Mao, H. Feick, H. Q. Yan, Y. Y. Wu, H. Kind, E. Weber, R. Russo and P. D. Yang, Science 292, 1897 (2001). http://dx.doi.org/10.1126/science.1060367
E. Comini, G. Faglia, G. Sberveglieri, Z. W. Pan and Z. L. Wang, Appl. Phys. Lett. 81, 1869 (2003). http://dx.doi.org/10.1063/1.1504867
D. Han, X. L. Ren, D. Chen, F. Q. Tang, D. Wang and J. Ren, Photogr. Sci. Photochem. 23, 414 (2005).
X. Y. Kong and Z. L. Wang, Nano. Lett. 3, 1625 (2003). http://dx.doi.org/10.1021/nl034463p
S. J. Li, Z. C. Ma, J. Zhang and J. Z. Liu, Catal. Commun. 9, 1482 (2008). http://dx.doi.org/10.1016/j.catcom.2007.12.016
Q. Zhang, W. Fan and L. Gao, Appl. Catal. B- Environ. 76, 168 (2007). http://dx.doi.org/10.1016/j.apcatb.2007.05.024
L. Q. Jing, B. F. Xin, F. L. Yuan, L. P. Xue, B. Q. Wang and H. G. Fu, J. Phys. Chem. B 110, 17860 (2006). http://dx.doi.org/10.1021/jp063148z
N. Pradhan, A. Pal and T. Pal, Langmuir 17, 1800 (2001). http://dx.doi.org/10.1021/la000862d
C. A. K. Gouvea, F. Wypych and S. G. Moraes, Chemosphere 40, 433 (2000). http://dx.doi.org/10.1016/S0045-6535(99)00313-6
K. R. Lee, S. Park, K. W. Lee and J. H. Lee, J. Mater. Sci. Lett. 22, 65 (2003). http://dx.doi.org/10.1023/A:1021738526590
R. Ullah and J. Dutta, 2nd Inter. Conf. Emerging Tech. 353 (2006).
H. M. Deng, J. Ding, Y. Shi, X. Y. Liu and J. Wang, J. Mater. Sci. 36, 3273 (2001). http://dx.doi.org/10.1023/A:1017902923289
X. Jiaqiang, P. Qingyi, S. Yuan and L. Zhanchai, Chin. J. Inorg. Chem. 14, 355 (1998). (in Chinese)
W. Wenliang, L. Dongsheng, H. Xiangyang, S. Zhenmin, W. Jiwu and Z. Caihua, Chem. Res. Appl. 13, 157 (2001). (in Chinese)
N. Faal Hamedani and F. Farzaneh, J. Sci. Islamic. Rep. of Iran 17, 231 (2006).
K.J. Rao, K. Mahesh and S. Kumar, Bull. Mater. Sci. 28, 19 (2005). http://dx.doi.org/10.1007/BF02711166
M. Vafaee and M.S. Ghamsari, Mater. Lett. 61, 3265 (2007). http://dx.doi.org/10.1016/j.matlet.2006.11.089
K. G. Chandrappa, T. V. Venkatesha, K. Vathsala and C. Shivakumara, J. Nanopart. Res. 12, 2667 (2010). http://dx.doi.org/10.1007/s11051-009-9846-0
Y. GaoQing, J. Huanfeng, L. Chang and L. ShiJun, J. Cryst. Growth 303, 400 (2007).
E. Boschke, U. Bohmer, J. Lange, M. Constapel, M. Schellentrager and T. Bley, Chemosphere 67, 2163 (2007). http://dx.doi.org/10.1016/j.chemosphere.2006.12.041
H. Wang, C. Xie, W. Zhang, S. Cai, Z. Yang and Y. Gui, J. Hazard. Mater. 141, 645 (2007). http://dx.doi.org/10.1016/j.jhazmat.2006.07.021
R. Y. Hong, J. H. Li, L. L. Chen, D. Q. Liu, H. Z. Li, Y. Zheng and J. Ding, Powder Technol. 189, 426 (2009). http://dx.doi.org/10.1016/j.powtec.2008.07.004
R. M. Trommer, A. K. Alves and C. P. Bergmann, J. Alloys Compd. 491, 296 (2010). http://dx.doi.org/10.1016/j.jallcom.2009.10.147
R. Xiangling, H. Dong, C. Dong and T. Fangqiong, Mater. Res. Bull. 42, 807 (2007). http://dx.doi.org/10.1016/j.materresbull.2006.08.030
P. Scherrer, Nachr. Ges. Wiss. Gottingen Math. Phys. 2, 98 (1918).
Siqingaowa, Zhaorigetu, Y. Hongxia and Garidi, Front. Chem. China 3, 277 (2006). http://dx.doi.org/10.1007/s11458-006-0036-7
L. P. Berube and G. L. Esperance, J. Electrochem. Soc. 136, 2314 (1989). http://dx.doi.org/10.1149/1.2097318
M. L. Curri, R. Comparelli, P. D. Cozzoli, G. Mascolo and A. Agostiano, Mater. Sci. Eng. C 23, 285 (2003). http://dx.doi.org/10.1016/S0928-4931(02)00250-3
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chandrappa, K.G., Venkatesha, T.V. Electrochemical Synthesis and Photocatalytic Property of Zinc Oxide Nanoparticles. Nano-Micro Lett. 4, 14–24 (2012). https://doi.org/10.1007/BF03353686
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03353686