Abstract
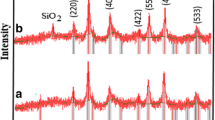
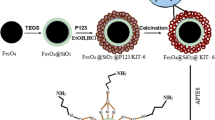
The adsorption of iodine onto silica coated magnetite nanoparticles (im-SCMNPs) that modified with imidazole was investigated for removal of high concentrations of iodine from wastewater. Modified silica magnetite nanoparticles showed high efficiency in removing iodine from wastewater samples. The optimum pH for iodine removal was 7.0–8.0. The adsorption capacity was evaluated using both the Langmuir and Freundlich adsorption isotherm models. The size of the produced magnetite nanoparticles was determined by X-ray diffraction analysis and scanning electron microscopy. Synthesized magnetite nanoparticles showed the high adsorption capacity and would be a good method to increase adsorption efficiency for the removal of iodine in a wastewater treatment process. The Langmuir adsorption capacity (qmax) was found to be 140.84 mg/g of the adsorbent.
Article PDF
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
References
D. I. Kaplan, R. J. Serne, K. E. Parker and I. V. Kutnyakov, Environ. Sci. Technol. 34, 399 (2000). http://dx.doi: 10.1021/es990220g
W. Um, R. J. Serne and K. M. Krupka, Water Res. 38, 2009 (2004). http://dx.doi:10.1016/j.watres.2004.01.026
B. Riebe, S. Dultz and C. Bunnenberg, Appl. Clay Sci. 28, 9 (2005). http://dx.doi:10.1016/j.clay.2004.01.004
M. Sanchez-Polo, J. Rivera-Utrilla, E. Salhi and U. Von Gunten, J. Colloid Interface Sci. 300, 437 (2006). http://dx.doi:10.1016/j.jcis.2006.03.037
P. R. Bhagat, A. K. Pandey, R. Acharyab, A. G. C. Nair, N. S. Rajurkar and A. V. R. Reddy, Talanta, 1313 (2008). http://dx.doi:10.1016/j.talanta.2007.08.035
C. M. Xing, J. P. Deng and W. T. Yang, J. Appl. Polym. Sci. 97, 2026 (2005). http://dx.doi: 10.1002/app.21853
C. C. Lin and J. Inorg. Nucl. Chem. 42, 1093 (1980). http://dx.doi.org/10.1016/0022-1902(80)80416-7
L. dlC. Coo and I. S. Martinez, Talanta 64, 1317 (2004). http://dx.doi.org/10.1016/j.talanta.2004.05.057
L. Kentjono, J. C. Liu, W. C. Chang and C. Irawan, Desalination 262, 280 (2010). http://dx.doi:10.1016/j.desal.2010.06.015
J. W. Lee, D. K. Cha, Y. K. Oh, K. B. Ko and J. S. Song, J. Hazard. Mater. 164, 67 (2009). http://dx.doi.org/10.1016/j.jhazmat.2008.07.147
D. I. Kaplan, R. J. Serne, K. E. Parker and I. V. Kutnyakov, Environ. Sci. Technol. 34, 399 (2000). http://dx.doi.org/10.1021/es990220g
W. Um, R. J. Serne and K. M. Krupka, Water Res. 38, 2009 (2004). http://dx.doi.org/10.1016/j.watres.2004.01.026
B. Riebe, S. Dultz and C. Bunnenberg, Appl. Clay Sci. 28, 9 (2005). http://dx.doi.org/10.1016/j.clay.2004.01.004
M. Sanchez-Polo, J. Rivera-Utrilla, E. Salhi, U. von Gunten, J. Colloid Interface Sci. 300, 437 (2006). http://dx.doi.org/10.1016/j.jcis.2006.03.037
H. Curtius and Z. Kattilparampil, Clay Miner. 40, 455 (2005). http://dx.doi: 10.1180/0009855054040183
A. Henglein, Chem. Rev. 89, 1861 (1989). http://dx.doi:10.1021/cr00098a010
R R. Garro, M. T. Navarro and J. Primo, J. Catal. 233, 342 (2005). http://dx.doi.org/10.1016/j.jcat.2005.04.035
J. G. Hou, Q. Ma, X. Z. Du, H. L. Deng and J. Z. Gao, Talanta 62, 241 (2004). http://dx.doi.org/10.1016/j.talanta.2003.07.003
P. Liang, B. Hu, Z. C. Jiang, Y. C. Qin and T. Y. Peng, J. Anal. At. Spectrom. 16, 863 (2001). http://dx.doi:10.1039/B104050M
T. K. Jain, I. Roy, T. K. De and A. Maitra, J. Am. Chem. Soc. 120, 11092 (1998). http://dx.doi:10.1021/ja973849x
Y. Aoyama, T. Kanamori, T. Nakai, T. Sasaki, S. Horiuchi, S. Sando and T. Niidome, J. Am. Chem. Soc. 125, 3455 (2003). http://dx.doi:10.1021/ja029608t
N. Murthy, Y. X. Thng, S. Schuck, M. C. Xu and J. M. J. Frechet, J. Am. Chem. Soc. 124, 12398 (2002). http://dx.doi:10.1021/ja026925r
X. Xie, X. Zhang, B. Yu, H. Gao, H. Zhang and W. Fei, 280, 164 (2004). http://dx.doi.org/10.1016/j.jmmm.2004.03.008
I. Koh, X. Wang, B. Varughese, L. Isaacs, S. H. Ehrman and D. S. English, J. Phys. Chem. B 110, 1553 (2006). http://dx.doi:10.1021/jp0556310
S. Bucak, D. A. Jones, P. E. Laibinis and T. A. Hatton, Biotechnol. Prog. 19, (2003). http://dx.doi:10.1021/bp0200853
A. Afkhami and R. Moosavi, J. Hazard. Mater. 174, 398 (2010). http://dx.doi.org/10.1016/j.jhazmat.2009.09.066
A. Afkhami, R. Norooz-Asl, J. Colloid Surf. A 346, 52 (2009). http://dx.doi.org/10.1016/j.colsurfa.2009.05.024
X. Q. Liu, Z. Y. Ma, J. M. Xing and H. Z. Liu, J. Magn. Magn. Mater. 270, 1 (2004). http://dx.doi.org/10.1016/j.jmmm.2003.07.006
Afkhami, R. Moosavi and T. Madrakian, Talanta 82, 785 (2010). http://dx.doi.org/10.1016/j.talanta.2010.05.054
M. E. Mahmoud, M. M. El-Essawi, S. A. Kholeif and E. M. I. Fathalla, Anal. Chim. Acta 525, 123 (2004). http://dx.doi.org/10.1016/j.aca.2004.06.032
A. G. S. Prado and C. Airoldi, J. Colloid Interface Sci. 236, 161 (2001). http://dx.doi.org/10.1006/jcis.2000.7401
E. F. Caldin and J. P. Field, J. Chem. Soc. Faraday Trans. 178, 1937 (1982). http://dx.doi:10.1039/F19827801937
M. J. El-Ghomari, R. Mokhlisse, C. Laurence, J. -Y. Le-Questel and M. Berthelot, J. Phys. Org. Chem. 10, 669 (1997). http://dx.doi:10.1002/(SICI)1099-1395(199709)10:9<669
S. R. El-Shabouri, K. M. Emara, P. Y. Khashaba and A. M. Mohamed, Anal. Lett. 31, 1367 (1998). http://dx.doi:10.1080/00032719808002873
H. Greijer, J. Lindgren and A. Hagfeldt, J. Phys. Chem. B 105, 6314 (2001). http://dx.doi:10.1021/jp011062u
D. Cahen, G. Hodes, M. Grätzel, J. F. Guillemoles and I. Riess, J. Phys. Chem. B 104, 2053 (2000). http://dx.doi:10.1021/jp993187t
F. A. Cotton and G. Wilkinson, Advanced Inorganic Chemistry, 5th ed. John Wiley & Sons, New York (1988).
L. Langmuir, J. Am. Chem. Soc. 40, 1361 (1918). http://dx.doi:10.1021/ja02242a004
C. W. Carr, H. Freundlich and K. Sollner, J. Am. Chem. Soc. 63, 693 (1941). http://dx.doi:10.1021/ja01848a015
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visit https://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Madrakian, T., Afkhami, A., Zolfigol, M.A. et al. Application of Modified Silica Coated Magnetite Nanoparticles for Removal of Iodine from Water Samples. Nano-Micro Lett. 4, 57–63 (2012). https://doi.org/10.1007/BF03353693
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03353693