Abstract
Introduction
Cyclooxygenases (COXs) play important roles during inflammation. While reports on COX-2 function in inflammation preceded those on COX-1, it is now well established that both isoforms participate in this process. During inflammation, COX expression was reported in inflammatory leukocytes, but much less is known about their presence in tissue- resident leukocytes. The aim was thus to verify the expression and activity of the COX isoforms in resident peritoneal mast cells and macrophages during acute peritonitis.
Materials and Methods
Zymosan peritoneal inflammation was induced in C57BL/6J mice and COX-1 and COX-2 expression was evaluated by RT-PCR (mRNA level) and immunocytochemistry (protein level). COX activity was assessed by a specific assay and prostaglandin production by ELISA. Furthermore, some mice were selectively depleted of either peritoneal mast cells or macrophages and then COX activity was determined.
Results
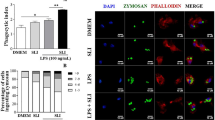
The study revealed that both COXs are expressed/active at the peak of inflammation, but COX-2 predominates during resolution. The expressions of the COXs were detectable in both populations of resident peritoneal leukocytes. In peritoneal macrophages both isoforms were active even during the late phases of peritonitis and the cells significantly contributed to PGE2 and PGD2 synthesis. The most striking observation was that resident macrophages are critical for PGD2 production during the resolution of inflammation.
Conclusions
This study documents that both COX isoforms participate in all stages of acute inflammation and that tissue-resident leukocytes, especially macrophages, are important sites of COX-1/COX-2 expression and prostaglandin synthesis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- ANOVA:
-
one-way analysis of variance
- cDNA:
-
complementary deoxyribonucleic acid
- Cl2MDP:
-
dichloromethylene diphosphonate
- DAB:
-
3’,3’-diaminobenzidine tetrahydrochloride
- EDTA:
-
ethylenediaminetetraacetic acid
- ELISA:
-
enzyme-linked immunosorbent assay
- HPRT:
-
hypoxanthine phosphoribosyltransferase
- IgG:
-
immunoglobulin G
- IL-10:
-
interleukin 10
- MC x :
-
mast cell-depleted mice
- Mϕx:
-
macrophage-depleted mice
- mRNA:
-
messenger ribonucleic acid
- TMPD:
-
oxidized N,N,N’,N’-tetramethyl-p-phenylenediamine
- Tris-HCl:
-
tris(hydroxymethyl)aminomethane and concentrated hydrochloric acid
- PGD2 :
-
prostaglandin D2
- PGE2 :
-
prostaglandin E2
- PGI2 :
-
prostaglandin I2
- RT-PCR:
-
reverse transcriptase polymerase chain reaction
- SD:
-
standard deviation
- PTL:
-
total peritoneal leukocyte
References
Ajuebor MN, Das AM, Virag L et al (1999) Role of resident peritoneal macrophages and mast cells in chemokine production and neutrophil migration in acute inflammation: evidence for an inhibitory loop involving endogenous IL-10. J Immunol 162: 1685–1691
Das AM, Flower RJ, Perretti M (1997) Eotaxin-induced eosinophil migration in the peritoneal cavity of ovalbumin-sensitized mice: mechanism of action. J Immunol 159: 1466–1473
Doherty NS, Poubelle P, Borgeat P et al (1985) Intraperitoneal injection of zymosan in mice induces pain, inflammation and the synthesis of peptidoleukotrienes and prostaglandin E2. Prostaglandins 30: 769–789
Fu JY, Masferrer JL, Seibert K et al (1990) The induction and suppression of prostaglandin H2 synthase (cyclooxygenase) in human monocytes. J Biol Chem 265: 16737–16740
Gilroy DW, Colville-Nash PR, Willis D et al (1999) Inducible cyclooxygenase may have anti-inflammatory properties. Nat Med 5: 698–701
Gilroy DW, Tomlinson A, Willoughby DA (1998) Differential effects of inhibitors of cyclooxygenase (cyclooxygenase 1 and cyclooxygenase 2) in acute inflammation. Eur J Pharmacol 355: 211–217
Hamberg M, Samuelsson B (1973) Detection and isolation of an endoperoxide intermediate in prostaglandin biosynthesis. Proc Natl Acad Sci USA 70: 899–903
Kolaczkowska E, Arnold B, Opdenakker G (2008a) Gelatinase B/MMP-9 as an inflammatory marker enzyme in mouse zymosan peritonitis: comparison of phase-specific and cell-specific production by mast cells, macrophages and neutrophils. Immunobiology 231: 109–124
Kolaczkowska E, Barteczko M, Plytycz B et al (2008b) Role of lymphocytes in the course of murine zymosan-induced peritonitis. Inflamm Res 57: 272–278
Kolaczkowska E, Chadzinska M, Scislowska-Czarnecka A et al (2006a) Gelatinase B/matrix metalloproteinase-9 contributes to cellular infiltration in a murine model of zymosan peritonitis. Immunobiology 211: 137–148
Kolaczkowska E, Chadzinska M, Seljelid R et al (2001a) Strain differences in some immune parameters can be obscured by circadian variations and laboratory routines: studies of male C57BL/6J, Balb/c and CB6 F1 mice. Lab Anim 35: 91–100
Kolaczkowska E, Lelito M, Kozakiewicz E et al (2007) Resident peritoneal leukocytes are important sources of MMP-9 during zymosan peritonitis: superior contribution of macrophages over mast cells. Immunol Lett 113: 99–106
Kolaczkowska E, Plytycz B, Arnold B et al (2009) Increased cyclooxygenase activity impairs apoptosis of inflammatory neutrophils in mice lacking gelatinase B/matrix metalloproteinase-9. Immunology 128: e262–e274
Kolaczkowska E, Scislowska-Czarnecka A, Chadzinska M et al (2006b) Enhanced early vascular permeability in gelatinase B (MMP-9)-deficient mice: putative contribution of COX-1-derived PGE2 of macrophage origin. J Leukoc Biol 80: 125–132
Kolaczkowska E, Seljelid R, Plytycz B (2001b) Role of mast cells in zymosan-induced peritoneal inflammation in Balb/c and mast cell-deficient WBB6F1 mice. J Leukoc Biol 69: 33–42
Kolaczkowska E, Shahzidi S, Seljelid R et al (2002) Early vascular permeability in murine experimental peritonitis is co-mediated by resident peritoneal macrophages and mast cells: crucial involvement of macrophage-derived cysteinylleukotrienes. Inflammation 26: 61–71
Langenbach R, Morham SG, Tiano HF et al (1995) Prostaglandin synthase 1 gene disruption in mice reduces arachidonic acid-induced inflammation and indomethacin-induced gastric ulceration. Cell 83: 483–492
Majno G, Joris I (2004) Cells, tissues, and disease: principles of general pathology. Blackwell, Oxford
Masferrer JL, Zweifel BS, Seibert K et al (1990) Selective regulation of cellular cyclooxygenase by dexamethasone and endotoxin in mice. J Clin Invest 86: 1375–1379
Miller SB (2006) Prostaglandins in health and disease: an overview. Semin Arthritis Rheum 36: 37–49
Morham SG, Langenbach R, Loftin CD et al (1995) Prostaglandin synthase 2 gene disruption causes severe renal pathology in the mouse. Cell 83: 473–482
Rao TS, Currie JL, Shaffer AF et al (1994) In vivo characterization of zymosan-induced mouse peritoneal inflammation. J Pharmacol Exp Ther 269: 917–925
Sheibanie AF, Yen JH, Khayrullina T et al (2007) The proinflammatory effect of prostaglandin E2 in experimental inflammatory bowel disease is mediated through the IL-23—>IL-17 axis. J Immunol 178: 8138–8147
Simmons DL, Botting RM, Hla T (2004) Cyclooxygenase isozymes: the biology of prostaglandin synthesis and inhibition. Pharmacol Rev 56: 387–437
Straus DS, Pascual G, Li M et al (2000) 15-deoxy-delta 12,14-prostaglandin J2 inhibits multiple steps in the NF-kappa B signaling pathway. Proc Natl Acad Sci USA 97: 4844–4849
Tchougounova E, Lundequist A, Fajardo I et al (2005) A key role for mast cell chymase in the activation of pro-matrix metalloprotease-9 and pro-matrix metalloprotease-2. J Biol Chem 280: 9291–9296
Ueno N, Taketomi Y, Koga K et al (2008) A negative regulator of delayed prostaglandin D2 production in mouse mast cells. Biochim Biophys Acta 1781: 415–421
van Rooijen N, Sanders A (1994) Liposome mediated depletion of macrophages: mechanism of action, preparation of liposomes and applications. J Immunol Methods 174: 83–93
Vane JR (1971) Inhibition of prostaglandin synthesis as a mechanism of action for aspirin-like drugs. Nat New Biol 231: 232–235
Warner TD, Mitchell JA (2004) Cyclooxygenases: new forms, new inhibitors, and lessons from the clinic. FASEB J 18: 790–804
Willoughby DA, Moore AR, Colville-Nash PR (2000) COX-1, COX-2, and COX-3 and the future treatment of chronic inflammatory disease. Lancet 355: 646–648
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is published under an open access license. Please check the 'Copyright Information' section either on this page or in the PDF for details of this license and what re-use is permitted. If your intended use exceeds what is permitted by the license or if you are unable to locate the licence and re-use information, please contact the Rights and Permissions team.
About this article
Cite this article
Kolaczkowska, E., Goldys, A., Kozakiewicz, E. et al. Resident peritoneal macrophages and mast cells are important cellular sites of COX-1 and COX-2 activity during acute peritoneal inflammation. Arch. Immunol. Ther. Exp. 57, 459–466 (2009). https://doi.org/10.1007/s00005-009-0053-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00005-009-0053-6