Abstract
Background
Immune checkpoint inhibitors (ICIs) have shown modest antitumor activity in unselected advanced sarcomas. Histology driven approach to patient selection is the current standard for off-label anti-programmed cell death 1 (PD1) immunotherapy use.
Methods
We retrospectively reviewed the clinical characteristics and outcomes of patients with advanced sarcoma who were treated with off label anti-PD1 immunotherapy at our center.
Results
A total of 84 patients with 25 histological subtypes were included. Nineteen patients (23%) had a cutaneous primary tumor site. Eighteen patients (21%) were classified as having clinical benefit, including 1 patient with complete response, 14 with partial response, and 3 with stable disease lasting over 6 months with previously progressive disease. Cutaneous primary site location was associated with higher clinical benefit rate (58% vs. 11%, p < 0.001), longer median PFS (8.6 vs. 2.5 months, p = 0.003) and OS (19.0 vs. 9.2 months, p = 0.011), compared to non-cutaneous primary. Patients with histological subtypes that pembrolizumab is indicated per current National Comprehensive Cancer Network guidelines had modestly higher rate of clinical benefit versus other histologies, however, the difference was statistically insignificant (29% vs. 15%, p = 0.182) and no statistically significant difference in PFS or OS was observed between these groups. Immune-related adverse events were more frequently seen among patients with clinical benefit (72% vs. 35%, p = 0.007).
Conclusions
Anti-PD1-based immunotherapy is highly efficacious in advanced sarcomas of cutaneous primary site. Cutaneous primary site location is a stronger predictor of ICI response than histologic subtype and should be accounted for in treatment guidelines and clinical trial design.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Immune checkpoint inhibitors (ICIs) targeting programmed cell death 1 (PD-1) or PD-ligand-1 (PD-L1), have led to remarkable outcomes in many cancer types. Anti-PD1 therapy has been particularly effective in virtually every cutaneous malignancy in which this therapy class has been evaluated formally, likely in part due to the stigmata of a high ultraviolet (UV)-mutational burden that is common across these cancer types.[1,2,3] In contrast, reports from clinical trials with pembrolizumab or nivolumab as either monotherapy or in combination with ipilimumab have shown only modest antitumor activity in unselected advanced sarcomas, with response rates of roughly 10–20% when including all histological subtypes.[4,5,6,7]. Responses to ICIs are noted to be histology-dependent, with higher response rates reported in undifferentiated pleomorphic sarcoma (UPS),[6] alveolar soft part sarcoma (ASPS),[8, 9] cutaneous angiosarcoma,[10] and Kaposi sarcoma,[11, 12] etc. However, because of the diversity and rarity of sarcoma, most clinical trials have varied histology specific representation and small sample sizes.[13] Current NCCN guidelines[14, 15] reflect a histology-driven approach to patient selection for anti-PD1 immunotherapy in sarcoma, with pembrolizumab being listed as an off-label consideration for later lines of therapy for selected sarcoma histologic subtypes including myxofibrosarcoma, UPS, cutaneous angiosarcoma and undifferentiated sarcomas and as a preferred therapy option for ASPS. As in all solid tumors, high mutational burden (≥ 10 mutations/Mb) is also considered by guidelines as an indication to consider anti-PD1 therapy for patient that are refractory to other standard treatments.
In this study, we reviewed the clinical characteristics and outcomes of patients who were treated with ICI for advanced sarcoma at our institution and sought to explore the factors that are associated with clinical benefit from ICI. We hypothesized that a cutaneous primary site location would predict for higher chance of ICI benefit.
Materials and methods
We retrospectively reviewed all patients with advanced sarcoma who were treated with off-label pembrolizumab or nivolumab at Moffitt Cancer Center (MCC). We did not include patients treated with this agent class in the context of a clinical trial. All patients who received at least one dose of ICI were included in this study. This study was reviewed and approved by the Institutional Review Board at MCC.
Treatment response was assessed according to Response Evaluation Criteria in Solid Tumors (RECIST) 1.1. Patients with best response of complete response (CR), partial response (PR) or stable disease (SD) for at least 6 months in previously progressive disease were considered having clinical benefit from ICI. Overall survival (OS) was defined as the time interval from the initiation of ICI to death from any cause. Progression-free survival (PFS) was defined as the time interval from the initiation of ICI to disease progression or death. Adverse events were graded based on Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0.
Statistical analysis
Statistical significance between groups was analyzed using the chi-square tests or Fisher’s exact test for categorical variables and the Student’s t-test or Mann–Whitney U non-parametric test for continuous variables. The estimated OS and PFS were derived using the Kaplan–Meier method and compared by the Mantel-Cox log-rank test. The statistical analyses were performed using IBM SPSS Statistics for Windows, version 28.0 (IBM Corp, Armonk, NY). Kaplan–Meier survival curves were generated in R (version 4.2.1; www.r-project.org), using the ‘survminer’ package and the ‘ggsurvplot’ function. All reported p values were two-sided. The level of significance was set at p < 0.05.
Results
Clinical characteristics
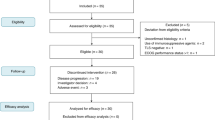
A total number of 88 patients who received pembrolizumab or nivolumab for advanced sarcoma between November 2015 and July 2022 were identified. Four patients were excluded for further analysis due to the lack of adequate information for outcome evaluation, generally due to lack of follow-up and/or transfer of care to an alternate location. The clinical characteristics of the remaining 84 patients are summarized in Table 1.
There were 52 men and 32 women with a median age of 64 years (range: 17–91 years). Among the 25 histological subtypes of sarcoma included in this study, the most common were UPS (19, including 4 pleomorphic dermal sarcomas [PDS]), angiosarcoma (15, including 12 cutaneous), dedifferentiated liposarcoma (DDLS, 9), ASPS (5), and conventional chondrosarcoma (5). The primary tumor of 19 patients (23%) was cutaneous/dermal sarcoma, including angiosarcoma (12), UPS/PDS (4), classic Kaposi sarcoma (CKS, 2), and a dermal fibrosarcoma (1). Two patients had radiation-associated tumors, one non-cutaneous angiosarcoma and one osteosarcoma. At the time of ICI initiation, 76 patients (90%) had distant metastasis, and the remaining 8 (10%) had locally or regionally advanced disease. PD-L1 IHC status was known in 38 patients, 19 of whom (50%) were considered as PD-L1 positive. Ten patients had information on MSI status, only one was MSI-high with a known diagnosis of Lynch syndrome. TMB was assessed in 5 patients, only one had high TMB over 10 mutations per megabase (54 muts/Mb).
The median number of prior systemic therapy was 1 (range: 0–13). ICI was given as first line in 24 patients (29%), second line in 28 patients (33%), and third line and above in 32 patients (38%). The majority of patients received single agent pembrolizumab (75) while pembrolizumab was combined with eribulin (two DDLSs and one leiomyosarcoma), Lenvatinib (two leiomyosarcomas), and axitinib (one ASPS). For the remaining 3 patients, nivolumab was given alone (2, DDLS and gastrointestinal stromal tumor [GIST]) or in combination with ipilimumab (1, GIST). The median number of ICI administered was 5 doses (range: 1–38).
Outcome
Best response was CR in one patient with cutaneous angiosarcoma (1%), PR in 14 patients (17%, 2 cutaneous KS, 2 cutaneous UPS/PDS and 2 UPS of other sites, 2 cutaneous angiosarcomas and 1 angiosarcoma of other site, 1 ASPS, 1 DDLS, 1 dermal fibrosarcoma, 1 pleomorphic rhabdomyosarcoma, and 1 SMARCA4-deficient thoracic sarcoma), SD in 13 patients (15%), and PD in 56 patients (67%). The median PFS and OS was 2.7 months (95% confident interval [CI]: 2.4–3.1 months) and 12.5 months (95% CI: 7.7–17.3 months), respectively (Table S1, Fig. 1). irAEs of any grade were seen in 36 patients (43%), 11 of them (13%) had grade 3/4 irAEs.
Eighteen patients (21%) were classified as having clinical benefit, including the 1 CR, 14 PRs, and 3 patients with SD > 6 months with previously progressive disease (2 cutaneous UPS/PDS, 1 cutaneous angiosarcoma). Of the 3 patients with SD classified as having clinical benefit, all were progressing at treatment initiation, all experienced clinical tumor regression not meeting the RECIST threshold for PR, and duration of response was 8 months in one patient and is ongoing at 16 + and 45 + months in the other two. All 18 patients classified as having clinical benefit received single agent anti-PD1. The median PFS and OS among the patients with clinical benefit were 32.7 months (no estimation on 95% CI) and not reached, respectively, whereas the median PFS and OS among those without clinical benefit were 2.2 months (95% CI: 1.8–2.7 months) and 8.8 months (95% CI: 6.5–11.2 months), respectively (Table S1). irAEs were more frequently seen among patients with clinical benefit (72% vs. 35%, p = 0.007, Table 1).
There were more patients with cutaneous sarcoma who experienced clinical benefit compared to those with primary tumor of other sites (11/19 [58%] vs. 7/65 [11%], p < 0.001, Table 1). The median PFS (8.6 months vs. 2.5 months, p = 0.003) and OS (19.0 months vs. 9.2 months, p = 0.011) were significantly longer among patients with cutaneous sarcoma than those with non-cutaneous primary (Table S1, Fig. 1).
Patients with the histological subtypes that pembrolizumab may be indicated per current NCCN guideline, i.e., UPS, cutaneous angiosarcoma, ASPS and myxofibrosarcoma in this study, had trend towards higher rate of ICI clinical benefit, however, the difference was not statistically significant (11/38 [29%] vs. 7/46 [15%], p = 0.182, Table 1). Similarly, there was non-statistically significant tendency of patients to have a greater chance of clinical benefit for those with locoregional disease only, positive PD-L1, or no or fewer prior systemic therapies. No statistically significant difference in PFS or OS was observed based on above factors (Table S1). Better Eastern Cooperative Oncology Group (ECOG) performance status (PS) at the initiation of ICI was associated with longer OS (16.3 months in PS 0 vs. 12.8 months in PS 1 vs. 4.5 months in PS 2, p = 0.024).
Discussion
We report our institutional experience with off-label anti-PD1-based immunotherapy use in advanced cases of sarcoma. In 84 patients representing 25 different sarcoma histologic subtypes, we report an overall clinical benefit rate of 21% in our cohort. While we noted a modest enrichment for treatment benefit if applying a histology-based, NCCN guideline-like patient selection criteria, our most striking observation was that cutaneous primary site location was highly predictive of ICI benefit, with a clinical benefit rate of 58% (11 of 19) based on this single clinical factor.
The benefit of ICI therapy in 21% of our overall cohort is congruent with prior reports,[4, 6, 7] though slightly on the high end of expected benefit for single-agent anti-PD1 in unselected sarcoma patients. This is very likely explained by a biased patient selection towards off-label anti-PD1 use in sarcoma histologies that are felt to be more immunotherapy responsive based on prior reports.[6, 8,9,10,11,12] Notably, almost half of our cohort (38 of 84, 45%) is represented by NCCN-guideline suggested, “anti-PD1 responsive” histologic subtypes.
Most notably, we observe a signal for oncologic benefit for anti-PD1 therapy in sarcomas from a cutaneous primary site, with an overall 58% clinical benefit rate in this subset of patients across several sarcoma histologic subtypes. This observation is concordant with prior reports in angiosarcoma, [10, 16, 17] but to our knowledge has not been as well appreciated in other sarcoma histologic types such as undifferentiated sarcomas [18]. Presumably, higher mutational burden due to the genomic stigmata of UV mutagenesis as has been reported in several cutaneous sarcoma subtypes [18, 19] might in part explain the higher response rate of cutaneous sarcomas to this drug class. The observation of high ICI benefit rate related to cutaneous primary site has particular importance for UPS, a sarcoma subtype in which emphasis has been placed for checkpoint inhibitor clinical trials and clinical practice. All four of the cutaneous UPS (a.k.a. pleomorphic dermal sarcomas) in our cohort experienced clinical benefit from anti-PD1 therapy, as opposed to only 2 of 15 (13%) UPS tumors from other primary locations.
Our study has several limitations. Due to the rarity of sarcomas, the sample size of our cohort is small, although it represents a relatively large single institutional study. The retrospective design also makes it difficult to draw definitive conclusions. In addition, the low percentage of NGS and PD-L1 IHC tests performed in our study limited our ability to evaluate TMB and PD-L1 expression as potential predictive biomarkers.
Our findings have important implications for anti-PD1 therapy and anti-PD1 based clinical trials in sarcoma patients. First, the magnitude of overall benefit of anti-PD1 therapy in cutaneous sarcomas that we observed compares favorably to other cutaneous malignancies such as melanoma in which this agent class has clearly become a preferred first line treatment option. Further, it is well-known that deferring checkpoint inhibitor therapy to later lines of therapy in other immunotherapy-responsive cutaneous malignancies is detrimental to outcomes for the general patient population, even when the alternative first line therapy has significant disease efficacy such as BRAF/MEK inhibitor therapy in melanoma [20] or cytotoxic chemotherapy in Merkel cell carcinoma [2, 21]. It would therefore stand to reason that anti-PD1 based immunotherapy should be evaluated as first line therapy for unresectable or metastatic cutaneous sarcomas, even when alternative options with anti-tumor efficacy exist, e.g. taxane-based cytotoxic chemotherapy for cutaneous angiosarcoma. Second, for ICI clinical trial design in sarcomas, we believe that it is imperative that cutaneous primary site location be considered at a minimum as a predefined stratification variable for randomization and for outcome reporting. Currently, we observe that this approach is being increasingly adopted for angiosarcoma but has not been widely considered for “undifferentiated sarcoma” trials. Specifically for UPS/undifferentiated sarcoma, we also believe it essential for any ICI trial to attempt NGS to help rule out a UV-mutation signature, the finding of which could only be present if the primary site was dermal or if the tumor was a misdiagnosed spindle cell variant of melanoma.
In summary, we review our institutional experience with off-label anti-PD1 immunotherapy in advanced sarcoma patients. We observe striking efficacy of this drug class in sarcomas with cutaneous primary site location, rivaling that of other cutaneous malignancies in which checkpoint inhibitor therapy is the well-established first line therapy. If confirmed in follow-up reports, our findings have profound implications for patient selection for anti-PD1 therapy and for ICI clinical trial design for sarcoma histologic subtypes that can originate in the dermis, such as undifferentiated pleomorphic sarcomas.
Abbreviations
- ASPS:
-
Alveolar soft part sarcoma
- CKS:
-
Classic Kaposi sarcoma
- CTCAE:
-
Common Terminology Criteria for Adverse Events
- CR:
-
Complete response
- DDLS:
-
Dedifferentiated liposarcoma
- ECOG:
-
Eastern Cooperative Oncology Group
- GIST:
-
Gastrointestinal stromal tumor
- ICI:
-
Immune checkpoint inhibitor
- irAEs:
-
Immune-related adverse events
- IHC:
-
Immunohistochemistry
- MSI:
-
Microsatellite instability
- MCC:
-
Moffitt Cancer Center
- NCCN:
-
National Comprehensive Cancer Network
- NGS:
-
Next-generation sequencing
- OS:
-
Overall survival
- PR:
-
Partial response
- PS:
-
Performance status
- PDS:
-
Pleomorphic dermal sarcomas
- PD-1:
-
Programmed cell death 1
- PD-L1:
-
Programmed cell death ligand-1
- PFS:
-
Progression-free survival
- PD:
-
Progressive disease
- RECIST:
-
Response Evaluation Criteria in Solid Tumors
- SD:
-
Stable disease
- TMB:
-
Tumor mutational burden
- UV:
-
Ultraviolet
- UPS:
-
Undifferentiated pleomorphic sarcoma
- WHO:
-
World Health Organization
References
Hall ET, Fernandez-Lopez E, Silk AW, Dummer R, Bhatia S (2020) Immunologic characteristics of nonmelanoma skin cancers: implications for immunotherapy. Am Soc Clin Oncol Educ Book 40:1–10. https://doi.org/10.1200/EDBK_278953
Knepper TC, Montesion M, Russell JS et al (2019) The genomic landscape of merkel cell carcinoma and clinicogenomic biomarkers of response to immune checkpoint inhibitor therapy. Clin Cancer Res 25:5961–5971. https://doi.org/10.1158/1078-0432.CCR-18-4159
Dousset L, Poizeau F, Robert C et al (2021) Positive association between location of melanoma, ultraviolet signature, tumor mutational burden, and response to anti-PD-1 therapy. JCO Precis Oncol. https://doi.org/10.1200/PO.21.00084
Zhou M, Bui N, Bolleddu S, Lohman M, Becker HC, Ganjoo K (2020) Nivolumab plus ipilimumab for soft tissue sarcoma: a single institution retrospective review. Immunotherapy 12:1303–1312. https://doi.org/10.2217/imt-2020-0155
Zhu MMT, Shenasa E, Nielsen TO (2020) Sarcomas: Immune biomarker expression and checkpoint inhibitor trials. Cancer Treat Rev. 91:102115. https://doi.org/10.1016/j.ctrv.2020.102115
Tawbi HA, Burgess M, Bolejack V et al (2017) Pembrolizumab in advanced soft-tissue sarcoma and bone sarcoma (SARC028): a multicentre, two-cohort, single-arm, open-label, phase 2 trial. Lancet Oncol 18:1493–1501. https://doi.org/10.1016/S1470-2045(17)30624-1
D’Angelo SP, Mahoney MR, Van Tine BA et al (2018) Nivolumab with or without ipilimumab treatment for metastatic sarcoma (Alliance A091401): two open-label, non-comparative, randomised, phase 2 trials. Lancet Oncol 19:416–426. https://doi.org/10.1016/S1470-2045(18)30006-8
Groisberg R, Hong DS, Behrang A et al (2017) Characteristics and outcomes of patients with advanced sarcoma enrolled in early phase immunotherapy trials. J Immunother Cancer 5:100. https://doi.org/10.1186/s40425-017-0301-y
Wilky BA, Trucco MM, Subhawong TK et al (2019) Axitinib plus pembrolizumab in patients with advanced sarcomas including alveolar soft-part sarcoma: a single-centre, single-arm, phase 2 trial. Lancet Oncol 20:837–848. https://doi.org/10.1016/S1470-2045(19)30153-6
Florou V, Rosenberg AE, Wieder E et al (2019) Angiosarcoma patients treated with immune checkpoint inhibitors: a case series of seven patients from a single institution. J Immunother Cancer 7:213. https://doi.org/10.1186/s40425-019-0689-7
Zer A, Icht O, Yosef L et al (2022) Phase II single-arm study of nivolumab and ipilimumab (Nivo/Ipi) in previously treated classical Kaposi sarcoma (cKS). Ann Oncol 33:720–727. https://doi.org/10.1016/j.annonc.2022.03.012
Delyon J, Biard L, Renaud M et al (2022) PD-1 blockade with pembrolizumab in classic or endemic Kaposi’s sarcoma: a multicentre, single-arm, phase 2 study. Lancet Oncol 23:491–500. https://doi.org/10.1016/S1470-2045(22)00097-3
Saerens M, Brusselaers N, Rottey S, Decruyenaere A, Creytens D, Lapeire L (2021) Immune checkpoint inhibitors in treatment of soft-tissue sarcoma: a systematic review and meta-analysis. Eur J Cancer 152:165–182. https://doi.org/10.1016/j.ejca.2021.04.034
Network NCC (2022) Soft Tissue Sarcoma (Version 2.2022). https://www.nccn.org/professionals/physician_gls/pdf/sarcoma.pdf. 2022
Network NCC (2022) Bone Cancer (Version 2.2023). https://www.nccn.org/professionals/physician_gls/pdf/bone.pdf. 2022
Rosenbaum E, Antonescu CR, Smith S et al (2022) Clinical, genomic, and transcriptomic correlates of response to immune checkpoint blockade-based therapy in a cohort of patients with angiosarcoma treated at a single center. J Immunother Cancer. https://doi.org/10.1136/jitc-2021-004149
Wagner MJ, Othus M, Patel SP et al (2021) Multicenter phase II trial (SWOG S1609, cohort 51) of ipilimumab and nivolumab in metastatic or unresectable angiosarcoma: a substudy of dual anti-CTLA-4 and anti-PD-1 blockade in rare tumors (DART). J Immunother Cancer. https://doi.org/10.1136/jitc-2021-002990
Helbig D, Klein S (2022) Immune checkpoint inhibitors for unresectable or metastatic pleomorphic dermal sarcomas. Front Oncol. 12:975342. https://doi.org/10.3389/fonc.2022.975342
Painter CA, Jain E, Tomson BN et al (2020) The Angiosarcoma project: enabling genomic and clinical discoveries in a rare cancer through patient-partnered research. Nat Med 26:181–187. https://doi.org/10.1038/s41591-019-0749-z
Atkins MB, Lee SJ, Chmielowski B et al (2022) Combination Dabrafenib and Trametinib Versus Combination Nivolumab and Ipilimumab for Patients With Advanced BRAF-Mutant Melanoma: The DREAMseq Trial-ECOG-ACRIN EA6134. J Clin Oncol. https://doi.org/10.1200/JCO.22.01763
Kaufman HL, Russell JS, Hamid O et al (2018) Updated efficacy of avelumab in patients with previously treated metastatic Merkel cell carcinoma after >/=1 year of follow-up: JAVELIN Merkel 200, a phase 2 clinical trial. J Immunother Cancer 6:7. https://doi.org/10.1186/s40425-017-0310-x
Funding
Funding for this study was provided by institutional support and the AmandaLee Sarcoma Fund, Moffitt Cancer Center. The authors confirm that the data supporting the findings of this study are available within the article.
Author information
Authors and Affiliations
Contributions
The authors confirm contribution to the paper as follows: study conception and design: ASB; data collection: RM, JS, DM, SL, ASB; analysis and interpretation of results: RM, JW, DM, SL, LC, MF, DRR, MD, ASB; draft manuscript preparation: RM, ASB. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Confilct of interest
DM: speakers bureau: Jazz Pharmaceuticals; advisory board: Pfizer, Eli Lilly, and Takeda Pharmaceuticals; consultant: Dedham Group. JS: consultant: Iovance. DRR: advisory board: Springworks and Eisai. ASB: advisory board: Bayer and Deciphera. All stated disclosures are outside the scope current work. All other authors report no financial disclosures.
Ethical approval.
This study was reviewed and approved by the Institutional Review Board at MCC. The authors consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Miao, R., Swank, J., Melzer, D. et al. Anti-PD-1 therapy in advanced sarcomas: is cutaneous primary site a stronger predictor of response than histologic subtype?. Cancer Immunol Immunother 72, 2521–2527 (2023). https://doi.org/10.1007/s00262-023-03387-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-023-03387-6