Abstract
Bryozoans are among the most important groups of the Southern Ocean benthic macrofauna, both in terms of species richness and abundance. However, there is a considerable lack of ecological research focused on their distribution patterns and species richness on smaller scale, especially in the soft bottom habitats of Antarctic glacial fjords. The aim of this study was to describe those patterns in the Admiralty Bay. Forty-nine Van Veen grab samples were collected at the depth range from 15 to 265 m, in the summer season of 1979/1980, at three sites distributed along the main axis of the fjord. Among 53 identified species of bryozoans, 32 were recorded in the Admiralty Bay for the first time. The most common and abundant species were Himantozoum antarcticum, Inversiula nutrix and Nematoflustra flagellata. Genera such as Arachnopusia, Cellarinella and Osthimosia were the most speciose taxa. It was demonstrated that depth was important for the distribution of the bryozoans. More than half of the recorded species were found only below 70 m. An influence of glacial disturbance was reflected in the dominance structure of colony growth-forms. The inner region of the fjord was dominated almost entirely by encrusting species, while the diversity of bryozoan growth-forms in less disturbed areas was much higher. In those sites the highest percentage of branched, tuft like species represented by buguliform and flustriform zoaria was observed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Antarctic sessile suspension feeding communities are characterized by a high species richness and diversity (Gili et al. 2006). Bryozoans, ascidians and sponges are a key element of energy transfer from the pelagic zone into the benthic realm of the Southern Ocean (Gili et al. 2001). At least some of the species are able to exploit even very low food concentrations, as those observed during the Antarctic winter (Barnes and Clarke 1995). Bryozoans are also among the most important biomass components of the Southern Ocean benthic communities (Brey and Gerdes 1997).
Suspension feeding macro- and megazoobenthic communities are patchily distributed on dropstones and other types of hard substrata (Gutt and Starmans 1998). Distribution of bryozoan aggregations is also shaped by mineral suspension inflow and iceberg scouring, resulting in lower diversity and abundance, especially in the shallow sublittoral zone (Gutt 2001; Pabis et al. 2011). At greater depths their reduced abundance is explained mostly by lower organic matter supply (Saiz-Salinas et al. 1998).
The total richness of the Southern Ocean bryozoan fauna was estimated at more than 400 species (De Broyer et al. 2011), among which cheilostomatous bryozoans were a dominant and highly endemic group (Griffiths 2010). Most of the research on Antarctic bryozoans were focused on the taxonomy, and the number of newly described species was continuously increasing for the last 30 years (e.g., Lopez Gappa 1986; Hayward 1995; Kukliński and Barnes 2009; Hayward and Winston 2011; Figuerola et al. 2013 and references therein). Some of the most important studies were dedicated to zoogeography (Moyano 2005; Barnes and Griffiths 2008; Barnes and Kukliński 2010), colonization and succession processes (Stanwell-Smith and Barnes 1997; Bowden et al. 2006), as well as biology of particular species (e.g., Barnes 1995a; Barnes and Clarke 1995; Barnes et al. 2006).
The studies concerning species richness and distribution patterns on smaller scale are still relatively scarce and limited to only few Antarctic locations, such as Signy Island (Barnes 1995b; Barnes and Clarke 1995), Terra Nova Bay (Rosso and Sanfilippo 2000) and Bouvet Island (Barnes 2006). Even the Antarctic Peninsula region, one of the most intensively sampled areas in the Antarctic suffers from the scarcity of research concerning bryozoan fauna (Moyano 1979; Winston and Heimberg 1988; Moyano and Cancino 2002; Figuerola et al. 2012). There is still a considerable lack of ecological studies based on the quantitative samples. Moreover, many of the previous research were focused on typical hard bottom rocky habitats. Bryozoans, as a lophophorate organisms, are sensitive to disturbance caused by glacial sedimentation. On the other hand, some of them are considered as robust and can benefit from recent climate-related changes in the Antarctic benthic communities (Barnes and Griffiths 2008). For this reason, there is a need for studies at the sites characterized by high inflow of mineral suspension, especially in glacial fjords, such as Admiralty Bay. This basin belongs to the most comprehensively studied areas in the Antarctic in respect to benthic macrofauna and can be treated as a model ecosystem and reference site for future monitoring activities in the area of the Antarctic Peninsula (Siciński et al. 2011), the region currently facing the most rapid temperature increase in the Southern Hemisphere (Clarke et al. 2007; Walsh 2009). Many groups of benthic fauna in this bay, including polychaetes (e.g., Siciński 2004; Petti et al. 2006; Pabis and Siciński 2010), peracarid crustaceans (e.g., Jażdżewski et al. 1991; Pabis and Błażewicz-Paszkowycz 2011) and echinoderms (e.g., Presler and Figielska 1997; Nonato et al. 2000), were thoroughly analyzed. In contrast, the bryozoans were only scarcely studied (Moyano 1979). Thorough taxonomic inventory of all important benthic groups of macrofauna, and evaluation of their distribution patterns at such sites is essential for further ecological and zoogeographic assessments. Therefore, the quantitative studies presented here fill a gap in the ecological research on this group of organisms, demonstrating their species richness, biomass and distribution, on the soft bottom of the Admiralty Bay.
Materials and methods
Study area
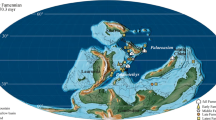
Admiralty Bay is a glacial fjord like embayment of tectonic origin, typical of the Antarctic Peninsula region. This basin covers the area of about 120 000 000 m2. It is located at the south-western part of King George Island. Four main parts are distinguished within this bay, a central basin and three inlets—Ezcurra Inlet, Martel Inlet and MacKellar Inlet. Ezcurra Inlet is a narrow fjord with large glaciers distributed along its coastline (Fig. 1), especially in the innermost region (Braun and Grossmann 2002). Ice disturbance has not been recorded in this semi-closed basin. Smaller growlers affect the bottom only in the intertidal zone and in shallowest sublittoral (down to about 2 m), while icebergs do not enter these fjord. In contrast, the ice disturbance was observed in the central basin and in the Martel Inlet down to 30 m depth (Nonato et al. 2000; Echeverria et al. 2005; Pabis et al. 2011). The total amount of mineral suspension transported every day into the waters of Admiralty Bay was estimated at 2,000,000 kg (Pęcherzewski 1980). Its primary source is crushed clastic material transported into the bay by subglacial streams (Jonasz 1983). A steep gradient of mineral suspension content was observed along the main axis of Ezcurra Inlet. The highest amount of mineral suspension (>0.1 kg/m3) was noted in the innermost parts, close to the glacial termini (in Goulden Cove and Cardozo Cove), and decreases along the axis of the fjord (Fig. 1) toward the mouth of Ezcurra Inlet (about 0.015 kg/m3) and the central basin (even 0.0028 kg/m3) (Pęcherzewski 1980). High water turbidity was recorded in Ezcurra Inlet, especially in the inner glacial bays, and it diminished toward the central basin (Lipski 1987). The sedimentation process was reflected in the character of the bottom sediments of the bay (Fig. 1). The inner, highly disturbed part of the Ezcurra Inlet is characterized by silty clay and clay silt sediments, while its middle and outer regions have sandy clay silt sediments as well as larger amount of the skeletal fractions. The proportion of sandy bottom deposits is higher in the shallow sublittoral of the central basin. In the central basin, more dropstones were also noted (Siciński 2004; Siciński et al. 2011).
Distribution of sampling stations in Admiralty Bay together with characteristics of sediments and suspended matter content in the investigated area. Data on sediments are derived from the analysis by Siciński (2004), while the diagram of mineral suspension content is constructed according to Pęcherzewski (1980)
The character of bottom deposits from samples used in this study was described by Jażdżewski et al. (1986). Their results are congruent with those presented by Siciński (2004). Section III was characterized mostly by muddy deposits. In the section II, sediments were also muddy; however, the number of stations with gravel and stones was higher than in section III. In the shallow sublittoral of central part of the bay (section I), the content of sandy sediments was higher than in two other sections, although gravel and stones were also important element of these bottom area (Table 1).
The inner area of Ezcurra Inlet is characterized by intricate bottom configuration and is separated from the outer part by a conspicuous submerged sill. The outer area has a form of deep trough (Marsz 1983). Waters of this inlet had also lower values of chlorophyll-a content compared with the central basin where those values are very high (up to 0.22 kg/m3). Those differences are especially noticeable from May to November. In the central part of the bay, larger concentrations of chlorophyll-a were found even below the euphotic zone (Tokarczyk 1986). Central basin is the deepest and less disturbed part of the bay that opens to the Bransfield Strait (Braun and Grossmann 2002). It is characterized by low water turbidity, low amount of mineral suspension and higher chlorophyll-a content (Pęcherzewski 1980; Tokarczyk 1986; Lipski 1987).
Sampling
Samples were collected in the 1979/80 austral summer during the 4th Antarctic Expedition of the Polish Academy of Sciences, with use of Van Veen grab (0.09 m2). Forty-nine samples were collected at 18 stations, at depths ranging from 15 to 265 m. Three replicate samples were usually taken at each station, with exception of stations: SI D, SII A, SII E, SII F, SIII B and SIII E where two samples were collected, and SII B (four samples). Three sampling areas were selected. One site was located in the central basin of the bay, close to the Henryk Arctowski Station (section I—14 samples, 15–265 m). The second sampling area was situated in the outer part of Ezcurra Inlet, between Thomas Point and Urbanek Crag (section II—22 samples, 15–260 m). The third investigated area was located in the inner part of Ezcurra Inlet, between Dera Icefall and Dufayel Island (section III—13 samples, 15–70 m) (Fig. 1).
Samples were sieved on 0.5-mm mesh sieves and preserved in 5 % buffered formaldehyde. Bryozoa were identified to the species level, with use of SEM microscope (ZEISS LEO 1430). The samples were examined individually, and the wet weight of each bryozoan species was measured with the accuracy of 0.001 g using the analytical balance of Redwag WTB 200. Abundance and biomass of higher taxa from this set of samples were analyzed by Jażdżewski et al. (1986).
Data analysis
Biomass is an universal indicator of the community character, especially when colonial organisms, such as Bryozoa, are considered (Magurran 2004). For this reason, the analysis was based on the values of total wet weight of every species in each sample. Species richness (S) was also calculated for each sample (Magurran 2004). Differences between the species richness and total biomass values between sampling areas were tested using nonparametric Kruskal–Wallis test. Post hoc testing was done using Dunn’s test in Statistica 6 package. Mean (B) with standard deviation (SD) and maximal (BMax) biomass values as well as frequency of occurrence (F—percentage of samples where a species was found in total number of samples) were calculated for each species in each area, and in the whole material. Frequency of occurrence of bryozoans as a whole and for each of the growth-form type was also calculated. The deepest station from section III was located at the depth of 70 m. To make the data fully comparable, we also compared the species richness and biomass on all three sites taking into account only the samples from the 15–70 m depth range. Each species was also assigned to a type of growth-form following the classification by Stach (1936) and Moyano (1979, 2005). Dominance (percentage of the biomass of a particular group in a total biomass) was calculated for each type of growth-form, in each of the studied sites. Similarity between the samples was calculated using the Bray–Curtis index. Hierarchical agglomerative clustering was performed using group average method. Biomass values (wet weight g/0.09 m2) of all species were square-root transformed before the analysis (Clarke and Warwick 1994).
Results
Species richness and biomass
Fifty-three species of Bryozoa from 24 families were found in the analyzed material. The majority of the species represents the order Cheilostomatida. Five species: Tubulipora tubigera, Idmidronea atlantica, Mecynoecia sp., Disporella canaliculata and Favosipora sp. belong to Cyclostomatida and one, Alcyonidium sp., to Ctenostomatida. Bryozoa were found in 29 of the 49 collected samples. Thirty-two species were recorded in Admiralty Bay for the first time (Table 2). Mean biomass values and frequency of all species were low. Relatively high values in the whole material were noted only for three species: Himantozoum antarcticum (F = 20.4 %, B = 0.2 ± 1.1 g/0.09 m2, BMax = 6.3 g/0.09 m2), Inversiula nutrix (F = 14.2 %, B = 0.08 ± 0.5 g/0.09 m2, BMax = 3.5 g/0.09 m2) and Nematoflustra flagellata (F = 12.2 %, B = 0.04 ± 0.1 g/0.09 m2, BMax = 1.02 g/0.09 m2). Despite the relatively high number of species recorded in this study, the species richness values were low and did not exceed 10 species per sample. The highest mean species richness and biomass values were detected in section I (Fig. 2). Statistically significant differences for both values were found between sections III and II, as well as III and I (Kruskal–Wallis test, Dunn’s test p < 0.05). The results were different in analysis of samples collected at 15–70 m depth range. Mean species richness and biomass were the highest in the section I (Fig. 2); however, no significant differences for both values between all the sites were observed (Kruskal–Wallis test, p < 0.05).
Thirty-three species were found in the central basin (section I). Seventeen of them were recorded only there (Table 1). This area was dominated by H. antarcticum (F = 42.8 %, B = 0.8 ± 2.1 g/0.09 m2) followed by N. flagellata (F = 28.5 %, B = 0.1 ± 0.3 g/0.09 m2) and I. nutrix (F = 28.5 %, B = 0.2 ± 0.9 g/0.09 m2). Two other species: Osthimosia notialis (F = 21.4 %, B = 0.01 ± 0.04 g/0.09 m2) and Orthoporidra stenorhyncha (F = 21.4 %, B = 0.03 ± 0.1 g/0.09 m2) had relatively high frequency in this area. The highest frequency of Bryozoa (F = 85.7 %) was also noted in this part of the bay.
Twenty-six species were found in outer region of Ezcurra Inlet (section II), including eleven species found exclusively here (Table 1). The most frequent and abundant species was H. antarcticum (F = 18.1 %, B = 0.04 ± 0.1 g/0.09 m2). Frequency of Bryozoa in this area was as high as 59.0 % (Table 2).
Only 14 species were found in the inner region of Ezcurra Inlet (section III), and all had very low biomass and frequency. The total frequency of Bryozoa in this area was low (F = 46.1 %). All species had very low biomass and frequency in this area. Seven of them were found only in this part of the bay (Table 2).
Bryozoan growth-forms
Bryozoans of Admiralty Bay were also characterized by a high diversity of the colonial forms (Fig. 3). Eight zoarial growth-forms have been distinguished including: membraniporiform (18 species), adeoniform (11 species), celleporiform (6 species), flustriform (4 species), buguliform (4 species), vinculariform (4 species), cellariform (3 species) and fungiform (3 species).
Central basin (section I) was characterized by a presence of all bryozoan growth-forms. The most important biomass component was buguliform bryozoans (56.4 %, F = 42.8 %), but only one species represented this growth-form. Encrusting (mebraniporiform) bryozoans had also high percentage of biomass and high frequency in this area (18.6 %, F = 57.1). Moreover, membraniporiform bryozoans had the highest species richness in this region. Eight species represented this growth-form. In the section II, diversity of growth-forms was similar. All types found in Admiralty Bay were recorded here. The highest number of species was found for membraniporiform (6 species) and adeoniform form (6 species), followed by a buguliform type (4 species). Buguliform (45.8 %, F = 31.8 %) and flustriform (25.3 %, F = 27.2 %) bryozoans dominated the biomass and had the highest frequency in this part of the fjord. Section III was dominated by encrusting species (Fig. 3). Eleven of 14 species found in this area belong to the membraniporiform growth-form. This group constituted 88.4 % of the biomass and had relatively high frequency (F = 30.7 %).
At the 15–70 m depth range, the total number of species is decreasing along the axis of the fjord from the section III to section I. The number of encrusting species is very high in the inner area (section III) and much lower in other two areas where diversity of the growth-forms was higher (Fig. 4).
Bathymetric distribution
The majority of collected bryozoan species were recorded in single samples, and it was difficult to describe their depth range in Admiralty Bay. Some taxa occurred patchily in the studied depth range (Fig. 5). The species with the widest bathymetrical range were as follows: Aimulosia australis, Antarctothoa antarctica, I. nutrix, H. antarcticum, Harpecia spinosissima, O. stenorhyncha and N. flagellata. Ten species were found only in shallow sublittoral, down to 40 m. Among the species found only in the shallowest areas, seven were assigned to membraniporiform growth-form. This growth-form was also characteristic of three species with the widest bathymetric range: A. australis, A. antarctica and I. nutrix. Ten species were recorded only in the deeper sublittoral, below 220 m (Fig. 5).
Similarity of fauna
No faunal groupings were observed in Admiralty Bay (Fig. 6). Samples taken from different depths and sampling areas were mixed in the analysis. Even if some groups were distinguished, the similarity was very low (about 10 %). Samples from section III were clustered within different groups or did not grouped with any other sample.
Discussion
Environmental gradients associated with intensity of glacial disturbance (high sedimentation rates, silting of bottom sediments) and the depth are typical of the polar fjords (Görlich et al. 1987; Siciński 2004; Włodarska-Kowalczuk and Pearson 2004; Grzelak and Kotwicki 2011). However, the distribution patterns of particular taxonomic groups of macrofauna can differ even in the same basin (Siciński 2004; Pabis and Błażewicz-Paszkowycz 2011). Those differences can be highly pronounced when various ecological groups are compared, e.g., small size, burrowing infauna versus large, filter-feeding epibenthos (Jażdżewski et al. 1986; Siciński 2004).
It was unexpected that at the depth range from 15 to 70 m, which is more vulnerable to disturbance, there was no significant difference in bryozoan species richness for the investigated sites. An increase in biodiversity along the fjord axis from the inner part to the central basin was observed for polychaetes, and peracarid crustaceans in Ezcurra Inlet (Siciński 2004; Pabis and Błażewicz-Paszkowycz 2011; Jażdżewska unpublished results). Difference in community structure between the investigated sites was found only in the species richness of bryozoan growth-forms. Encrusting species strongly dominated the inner area (section III). Distribution and composition of bryozoan growth-forms can be a good indicator of environmental conditions (Amini et al. 2004). High dominance of encrusting (membraniporiform) bryozoans in the disturbed inner region of Ezcurra Inlet, as well as low diversity of growth-forms can be explained by an influence of sedimentation inflow of glacial origin. Those bryozoans are considered the most opportunistic ones, and they can be found on various types of substrates (Amini et al. 2004). Encrusting species are very common in the shallow, intertidal rocky areas, influenced by wave action and other types of disturbance (Kukliński 2009). Moyano (1979) noted high dominance of encrusting forms at sites affected by volcanic disturbance in Port Foster. Branching bryozoans (adeoniform species) or bushy tufts (buguliform and flustriform species) are more vulnerable to high amount of inorganic suspension. Their abundance often increases with depth and with the increasing distance from the source of disturbance (Boyer et al. 1990; Barnes 1995b; Rosso and Sanfilippo 2000; Kukliński et al. 2005). Higher content of inorganic suspension in the waters around section III (Pęcherzewski 1980) can clog their filtering apparatus (Moore 1977).
Depth was also important for the distribution of bryozoans in Admiralty Bay. Most of the species (Fig. 5) were found only below 70 m. Kukliński et al. (2005) pointed that aside glacial disturbance in the Arctic fjords, the depth was also influencing bryozoan assemblages. However, Antarctic bryozoans are mostly eurybathic (Barnes 1995c; Lopez-Fe 2005; Barnes and Kukliński 2010; Figuerola et al. 2013). This fact might be associated with a deep-water origin of that fauna (Barnes and Kukliński 2010). Nevertheless, on smaller scale, in the semi-closed fjords and glacial bays, the bathymetric distribution of many species can be different and might be shaped by sedimentation inflow and other factors influencing bottom communities in the shallower areas. Deeper sublittoral of Admiralty Bay is characterized by relatively stable environmental conditions what support a higher richness and diversity of benthic communities (Siciński et al. 2011). Higher species richness of bryozoans in the sections I and II, which are deeper and less disturbed than the section III, could also result from the higher microhabitat diversity created by branching forms of Bryozoa, which may constitute an additional, three-dimensional substrate for the other bryozoan species (Barnes 1994). H. antarcticum and N. flagellata serve as a substrate for many other species recorded in South Bay (Moyano and Cancino 2002). The number of larger dropstones available for colonization and growth of branched bryozoans is clearly low in the section III (Fig. 1; Table 1), and stones can be buried due to higher sedimentation.
Clearly defined faunal assemblages of bivalves, polychaetes or amphipods were often described in the polar fjords and can be associated with depth, distance from the glaciers or sediment type (Siciński 2004; Włodarska-Kowalczuk and Pearson 2004; Jażdżewska unpublished results). Similar patterns were also found for the bryozoan fauna in the Svalbard fjords, where species composition was associated with depth and distance from the glaciers (Kukliński et al. 2005). High level of patchiness resulted in a lack of well-defined assemblages of bryozoan fauna in Admiralty Bay. Most of the species recorded during our study had very low frequency of occurrence. The distribution of particular species is highly irregular. Many bryozoans occurred in only a single patch. This pattern can be associated with recruitment and colonization processes in a glacial fjord like the Admiralty Bay. The only possible substrate for the sessile species in the studied soft bottom is various size stones, randomly distributed, mainly in the less disturbed central part of the bay (Marsz 1983; Siciński 2004). It was most probably the main reason for the lack of apparent zonation in the distribution of bryozoans species in the Admiralty Bay. The distribution of many species might be explained by a single colonization of the dropstones. These dropstones may be treated as stepping stones in colonization of the muddy sediments which are otherwise unsuitable for sessile species (Kukliński 2005) and a founder effect can occur here. Competition for a very limited space is also high in such environment (Barnes and Kukliński 2005).
In the material studied by us, most of the species were rare and only a few of them were widely distributed in the investigated sites, as well as in wide bathymetric range. Species such as H. antarcticum and N. flagellata can feed even during winter when food concentration is minimal (Barnes and Clarke 1994; Sanderson et al. 1994; Barnes and Clarke 1995). This ability can explain their relatively wide distribution in the Admiralty Bay. Moreover, N. flagellata is a fast growing species and probably does not show seasonal changes in growth rate (Barnes 1995a).
Generally, the biomass values recorded in our study were very low. This result surprised, especially if compared with the bryozoan biomass values recorded in the central basin of the Admiralty Bay, in 40–380 m depth range (Pabis et al. 2011; Pabis and Siciński 2012). Moreover, bryozoans can constitute up to 14 % of the macrozoobenthos biomass at some Southern Ocean sites (Winston and Heimberg 1988). Although, the biomass of sessile suspension feeders in the earlier studies done in the inner and middle part of Ezcurra Inlet was very low (Pabis et al. 2011). In our study, higher biomass was noted only in central basin of the bay (section I); however, the mean value was still low 1.6 ± 2.7 g/0.09 m2. Nevertheless, this value of bryozoan biomass, higher than in two other sections could be linked with a very low mineral suspension content (Pęcherzewski 1980) and a higher food availability in this area (Tokarczyk 1986); however, differences between three studied areas at the depth range from 15 to 70 m were not statistically significant. Relatively low bryozoan biomass in shallower areas of section I could be associated with an influence of ice disturbance, which creates an important boundary for sessile benthos in the central basin of Admiralty Bay at depth of about 30 m (Nonato et al. 2000; Echeverria et al. 2005; Pabis et al. 2011).
Conclusions
This study is the first analysis of the bryozoan community of the Antarctic glacial fjord based on the large set of quantitative samples. It demonstrates that depth is important in shaping the bryozoan community in this basin. Almost 55 % of all species recorded were found only below 70 m. In shallower sublittoral (15–70 m depth), which is the most vulnerable to disturbance, there was no difference in species richness between all three sites located along the axis of the fjord. The influence of glacial disturbance was visible only in the dominance structure and diversity of the bryozoan growth-forms. The inner area characterized by silty clay sediments and high mineral suspension content in water was strongly dominated by encrusting species. It showed that the composition of bryozoan growth-forms can be a better indicator of glacial disturbance than species richness itself. The distribution of the bryozoan species in Admiralty Bay was characterized by a strong patchiness. In the soft bottom habitat of this fjord, those sessile suspension feeders can colonize only randomly distributed dropstones, what explains lack of clearly defined assemblages of bryozoan fauna.
Further studies of bryozoan communities from Antarctic fjords should be focused on the influence of suspension inflow on the community structure and should cover wider bathymetric range, as well as include the glacial bays located in the innermost part of the fjords. There is also a need for studies on colonization and succession processes in these disturbed bottom areas. Subsequent research should also address the problem of temporal changes in the diversity and species richness of the bryozoan communities. Repeated sampling at appropriate time scales (20–30 years) can be useful for detection of possible temporal variability associated with a climate change. Similar studies have already been done for some Arctic sites (Kędra et al. 2010; Węsławski et al. 2010) and demonstrated significant changes in the benthic community structure and diversity. Bryozoans are important ecological indicators and can be used in the assessments of the long-term environmental changes. Climate-related changes in the bryozoan growth rates have been recently noted in the Southern Ocean (Barnes et al. 2006, 2011), and we can also expect shifts in the distribution patterns and diversity of their communities. Such research should be planned in locations characterized by comprehensive benthic studies and availability of data collected in the period when climate warming was not so strongly pronounced as nowadays, in the 1970s and 1980s of the twentieth century. Admiralty Bay as a model fjord basin and because of its extensive research history of benthic fauna offers the possibility for such comparisons.
References
Amini ZZ, Adabi MH, Burret CF, Quilty PG (2004) Bryozoan distribution and growth form associations as a tool in environmental interpretation, Tasmania, Australia. Sediment Geol 167:1–15. doi:10.1016/j.sedgeo.2004.01.010
Barnes DKA (1994) Communities of epibiota on two erect species of Antarctic Bryozoa. J Mar Biol Assoc UK 74:863–872. doi:10.1017/S0025315400043927
Barnes DKA (1995a) Seasonal and annual growth in erect species of Antarctic bryozoans. J Exp Mar Biol Ecol 188:181–198. doi:10.1016/0022-0981(95)00003-A
Barnes DKA (1995b) Sublittoral epifaunal communities at Signy Island, Antarctica. II. Below the ice–foot zone. Mar Biol 121:565–572. doi:10.1007/BF00349467
Barnes DKA (1995c) Sublittoral epifaunal communities at Signy Island, Antarctica. I. The ice–foot zone. Mar Biol 121:555–563. doi:10.1007/BF00349466
Barnes DKA (2006) A most isolated benthos: coastal bryozoans of Bouvet Island. Polar Biol 29:114–119. doi:10.1007/s00300-005-0015-3
Barnes DKA, Clarke A (1994) Seasonal variation in the feeding activity of four species of Antarctic bryozoans in relation to environmental factors. J Exp Mar Biol Ecol 181:117–133. doi:10.1016/0022-0981(94)90108-2
Barnes DKA, Clarke A (1995) Seasonality of feeding activity in Antarctic suspension feeders. Polar Biol 15:335–340. doi:10.1007/BF00238483
Barnes DKA, Griffiths HJ (2008) Biodiversity and biogeography of southern temperate and polar bryozoans. Global Ecol Biogeogr 17:84–99. doi:10.1111/j.1466-8238.2007.00342.x
Barnes DKA, Kukliński P (2005) Bipolar patterns of intraspecific competition in bryozoans. Mar Ecol Prog Ser 285:75–87. doi:10.3354/meps285075
Barnes DKA, Kukliński P (2010) Bryozoans of the Weddell Sea continental shelf, slope and abyss: did marine life colonize the Antarctic shelf from deep water, outlying islands or in situ refugia following glaciations? J Biogeogr 37:1648–1656. doi:10.1111/j.1365-2699.2010.02320.x
Barnes DKA, Webb K, Linse K (2006) Slow growth of Antarctic bryozoans increases over 20 years and is anomalously high in 2003. Mar Ecol Prog Ser 314:187–195. doi:10.3354/meps314187
Barnes DKA, Kukliński P, Jackson JA, Keel GW, Morley SA, Winston JE (2011) Scott’s collections help reveal accelerating marine life growth in Antarctica. Curr Biol 21:147–148. doi:10.1016/j.cub.2011.01.033
Bowden DA, Clarke A, Peck LS, Barnes DKA (2006) Antarctic sessile marine benthos: colonization and growth on artificial substrata over three years. Mar Ecol Prog Ser 316:1–16. doi:10.3354/meps316001
Boyer M, Matricardi G, Pisano E (1990) Zoarial forms in the development of a bryozoan community. Anales Biol 16:155–162
Braun M, Grossmann H (2002) Glacial changes in the areas of Admiralty Bay and Potter Cove, King George Island, maritime Antarctica. In: Beyer L, Bolter M (eds) Geoecology of the Antarctic ice–free coastal landscapes. Springer, Berlin, pp 75–90
Brey T, Gerdes D (1997) Is Antarctic benthic biomass really higher than elsewhere? Antarct Sci 9:266–267. doi:10.1017/S0954102097000357
Clarke KR, Warwick RM (1994) Change in marine communities: an approach to statistical analysis and interpretation. Natural Environment Research Council, Plymouth
Clarke A, Murphy EJ, Meredith MP, King JC, Peck LS, Barnes DKA, Smith RC (2007) Climate change and the marine ecosystem of the western Antarctic Peninsula. Philos Trans R Soc B 362:149–166. doi:10.1098/rstb.2006.1958
De Broyer C, Danis B, Allcock L, Angel M, Arango C, Artois T, Barnes D, Bartsch I, Bester M, Blachowiak-Samolyk K, Błażewicz M, Bohn J, Brandt A, Brandao SN, David B, de Salas M, Eleaume M, Emig C, Fautin D, George KH, Gillan D, Gooday A, Hopcroft R, Jangoux M, Janussen D, Koubbi P, Kouwenberg J, Kuklinski P, Ligowski R, Lindsay D, Linse K, Longshaw M, Lopez-Gonzalez P, Martin P, Munilla T, Muhlenhardt-Siegel U, Neuhaus B, Norenburg J, Ozouf-Costaz C, Pakhomov E, Perrin W, Petryashov V, Pena-Cantero AL, Piatkowski U, Pierrot-Bults A, Rocka A, Saiz-Salinas J, Salvini-Plawen L, Scarabino V, Schiaparelli S, Schrodl M, Schwabe E, Scott F, Siciński J, Siegel V, Smirnov I, Thatje S, Utevsky A, Vanreusel A, Wiencke C, Woehler E, Zdzitowiecki K, Zeidler W (2011) How many species in the Southern Ocean? Towards a dynamic inventory of the Antarctic marine species. Deep-Sea Res Pt II 58:5–17. doi:10.1016/j.dsr2.2010.10.007
Echeverria CA, Paiva PC, Alves VC (2005) Composition and biomass of shallow benthic megafauna during an annual cycle in Admiralty Bay, King George Island, Antarctica. Antarct Sci 17:312–318. doi:10.1017/S0954102005002762
Figuerola B, Monleón-Getino T, Ballesteros M, Avila C (2012) Spatial patterns and diversity of bryozoan communities from the Southern Ocean: South Shetland Islands, Bouvet Island and Eastern Weddell Sea. Syst Biodiv 10:109–123. doi:10.1080/14772000.2012.668972
Figuerola B, Ballesteros M, Avila C (2013) Description of a new species of Reteporella (Bryozoa: Phidoloporidae) from the Weddell Sea (Antarctica) and the possible functional morphology of avicularia. Acta Zool-Stockholm 94:66–73. doi:10.1111/j.1463-6395.2011.00531.x
Gili JM, Coma R, Orejas C, Lopez-Gonzales PJ, Zabala M (2001) Are Antarctic suspension-feeding communities different from those elsewhere in the world? Polar Biol 24:473–485. doi:10.1007/s003000100257
Gili JM, Arntz WE, Palanques A, Orejas C, Clarke A, Dayton PK, Isla E, Teixido N, Rossi S, Lopez-Gonzalez PJ (2006) A unique assemblage of epibenthic sessile suspension feeders with archaic features in the high-Antarctic. Deep Sea Res Pt II 53:1029–1052. doi:10.1016/j.dsr2.2005.10.021
Görlich K, Węsławski JM, Zajączkowski M (1987) Suspension settling effect on macrobenthos biomass distribution in the Hornsund fjord, Spitsbergen. Polar Res 5:175–192. doi:10.1111/j.1751-8369.1987.tb00621.x
Griffiths HJ (2010) Antarctic marine biodiversity—what do we know about the distribution of life in the Southern Ocean? PLoS One 5:e11683. doi:10.1371/journal.pone.0011683
Grzelak K, Kotwicki L (2011) Meiofaunal distribution in Hornsund fjord, Spitsbergen. Polar Biol 35:269–280. doi:10.1007/s00300-011-1071-5
Gutt J (2001) On the direct impact of ice on marine benthic communities, review. Polar Biol 24:553–564. doi:10.1007/s003000100262
Gutt J, Starmans A (1998) Structure and biodiversity of megabenthos in the Weddell and Lazarev Seas (Antarctica): ecological role of physical parameters and biological interactions. Polar Biol 20:229–247. doi:10.1007/s003000050300
Hayward PJ (1995) Antarctic cheilostomatous Bryozoa. Oxford University Press, Oxford
Hayward PJ, Winston JE (2011) Bryozoa collected by the United States Antarctic Research Program: new taxa and new records. J Nat Hist 45:2259–2338. doi:10.1080/00222933.2011.574922
Jażdżewski K, Jurasz W, Kittel W, Presler E, Presler P, Siciński J (1986) Abundance and biomass estimates of the benthic fauna in Admiralty Bay, King George Island, South Shetland Islands. Polar Biol 6:5–16
Jażdżewski K, Teodorczyk W, Siciński J, Kontek B (1991) Amphipod crustaceans as an important component of zoobenthos of the shallow Antarctic sublittoral. Hydrobiologia 223:105–117. doi:10.1007/BF00446235
Jonasz M (1983) Particulate matter in Ezcurra Inlet: concentration and size distributions. Oceanologia 15:65–74
Kędra M, Włodarska-Kowalczuk M, Węsławski JM (2010) Decadal change in macrobenthic soft-bottom community structure in a high Arctic fjord (Kongsfjorden, Svalbard). Polar Biol 33:1–11. doi:10.1007/s00300-009-0679-1
Kukliński P (2005) Bryozoan mode of life in the High Arctic dynamic fjordic environment. In: Moyano G, Hugo I, Cancino JM, Wyse Jackson PN (eds) Bryozoan Studies 2004. A.A. Balkema Publishers, London, pp 153–161
Kukliński P (2009) Ecology of stone-encrusting organisms in the Greenland Sea—a review. Polar Res 28:222–237. doi:10.1111/j.1751-8369.2009.00105.x
Kukliński P, Barnes DKA (2009) A new genus and three new species of Antarctic cheilostome Bryozoa. Polar Biol 32:1251–1259. doi:10.1007/s00300-009-0621-6
Kukliński P, Gulliksen B, Lonne OJ, Węsławski JM (2005) Composition of bryozoan assemblages related to depth in Svalbard fjords and sounds. Polar Biol 28:619–630. doi:10.1007/s00300-005-0726-5
Lipski M (1987) Variations of physical conditions, nutrients and chlorophyll a contents in Admiralty Bay (King George Island, South Shetland Islands, 1979). Pol Polar Res 8:307–332
Lopez Gappa JJ (1986) A new bryozoans genus from the Weddell Sea, Antarctica. Polar Biol 6:103–105. doi:10.1007/BF00258260
Lopez-Fe CM (2005) Cheilostomate Bryozoa of the Bellingshausen Sea (Western Antarctica): a preliminary report of the results of the ‘Bentart 2003’ Spanish expedition. In: Moyano G, Hugo I, Cancino JM, Wyse Jackson PN (eds) Bryozoan studies 2004. A.A. Balkema Publishers, London, pp 173–179
Magurran AE (2004) Measuring biological diversity. Blackwell, Carlton
Marsz A (1983) From surveys of the geomorphology of the shores and bottom of the Ezcurra Inlet. Oceanologia 15:209–220
Moore PG (1977) Inorganic particulate suspensions in the sea and their effects on marine animals. Oceanogr Mar Biol Annu Rev 15:225–363
Moyano HI (1979) Bryozoa from Antarctic Bays: some ecological aspects. In: Larwood GP, Abbott MB (eds) Advances in bryozoology, systematic association special, vol 13. Academic Press, London, pp 383–402
Moyano HI (2005) Scotia Arc bryozoans from LAMPOS expedition: a narrow bridge between two different faunas. Sci Mar 69:103–112
Moyano HI, Cancino JM (2002) Bryozoa de aguas someras en Bahia South, Doumer Island, Antarctic Peninsula. Gayana 66:119–127
Nonato EF, Brito TAS, De Paiva PC, Petti MAV, Corbisier TN (2000) Benthic megafauna of the nearshore zone of Martel Inlet (King George Island, South Shetland Islands, Antarctica): depth zonation and underwater observations. Polar Biol 23:580–588. doi:10.1007/s003000000129
Pabis K, Błażewicz-Paszkowycz M (2011) Distribution and diversity of cumacean assemblages in Admiralty Bay, King George Island. Pol Polar Res 32:341–354. doi:10.2478/v10183-011-0024-6
Pabis K, Siciński J (2010) Polychaete fauna associated with holdfasts of the large brown alga Himantothallus grandifolius in Admiralty Bay, King George Island, Antarctic. Polar Biol 33:1277–1288. doi:10.1007/s00300-010-0816-x
Pabis K, Siciński J (2012) Is polychaete diversity in the deep sublittoral of an Antarctic fjord related to habitat complexity? Pol Polar Res 33:181–197. doi:10.2478/v10183-012-0009-0
Pabis K, Siciński J, Krymarys M (2011) Distribution patterns in the biomass of macrozoobenthic communities in Admiralty Bay (King George Island, South Shetlands, Antarctic). Polar Biol 34:489–500. doi:10.1007/s00300-010-0903-z
Pęcherzewski K (1980) Distribution and quantity of suspended matter in Admiralty Bay (King George Island, South Shetland Islands. Pol Polar Res 1:75–82
Petti MAV, Nonato EF, Skowronski RSP, Corbisier TN (2006) Bathymetric distribution of the meiofaunal polychaetes in the nearshore zone of Martel Inlet, King George Island, Antarctica. Antarct Sci 18:163–170. doi:10.1017/S0954102006000186
Presler P, Figielska E (1997) New data on the Asteroidea of Admiralty Bay, King George Island, South Shetland Islands. Pol Polar Res 18:107–117
Rosso A, Sanfilippo R (2000) Shallow-water bryozoans and serpuloideans from the Ross Sea (Terra Nova Bay, Antarctica). In: Faranda FM, Griglielmo L, Lauora A (eds) Ross sea ecology. Springer, Berlin, pp 515–525
Saiz-Salinas JI, Ramos A, Munilla T, Rauschert M (1998) Changes in the biomass and dominant feeding mode of benthic assemblages with depth off Livingston Island (Antarctica). Polar Biol 19:424–428. doi:10.1007/s003000050269
Sanderson WG, Thorpe JP, Clarke A (1994) A preliminary study of feeding rates in the Antarctic cheilostome bryozoan Himantozoum antarticum. In: Hayward PJ, Ryland JS, Taylor PD (eds) Biology and palaeobiology of bryozoans. Olsen and Olsen, Fredensborg, pp 167–170
Siciński J (2004) Polychaetes of Antarctic sublittoral in the proglacial zone (King George Island, South Shetland Islands). Pol Polar Res 25:67–96
Siciński J, Jażdżewski K, De Broyer C, Presler P, Ligowski R, Nonato EF, Corbisier TN, Petti MAV, Brito TAS, Lavrado HP, Błażewicz-Paszkowycz M, Pabis K, Jażdżewska A, Campos LS (2011) Admiralty Bay Benthos diversity—a census of a complex polar ecosystem. Deep-Sea Res Pt II 58:30–48. doi:10.1016/j.dsr2.2010.09.005
Stach LW (1936) Correlation of zoarial form with habitat. J Geol 44:60–75
Stanwell-Smith D, Barnes DKA (1997) Benthic community in Antarctica: recruitment and growth on settlement panels at Signy Island. J Exp Mar Biol Ecol 212:61–79. doi:10.1016/S0022-0981(96)02754-2
Tokarczyk R (1986) Annual cycle of chlorophyll α in Admiralty Bay 1981–1982 (King George Island, South Shetland). Pol Arch Hydrobiol 3:177–188
Walsh JE (2009) A comparison of Arctic and Antarctic climate change, present and future. Antarct Sci 21:179–188. doi:10.1017/S0954102009001874
Węsławski JM, Wiktor J, Kotwicki L (2010) Increase in biodiversity in the arctic rocky litoral, Sorkappland, Svalbard, after 20 years of climate warming. Mar Biodiv 40:123–130. doi:10.1007/s12526-010-0038-z
Winston JE, Heimberg BF (1988) The role of bryozoans in the benthic community at Low Island, Antarctica. Antarct J 21:188–189
Włodarska-Kowalczuk M, Pearson TH (2004) Soft-bottom macrobenthic faunal associations and factors affecting species distribution in an Arctic glacial fjord (Kongsfjord, Spitsbergen). Polar Biol 27:155–167. doi:10.1007/s00300-003-0568-y
Acknowledgments
The study was supported by a grant of Polish Ministry of Science and Higher Education No. 51/N-IPY/2007/0 as well as Census of Antarctic Marine Life Project. Krzysztof Pabis was also partially supported by University of Lodz internal funds. This research was also supported by the Polish Geological Institute-National Research Institute during the realization of the project numbered 40.2900.0903.18.0 titled “Bryozoan assemblage of Admiralty Bay—richness, diversity and abundance.” Urszula Hara is deeply grateful to Leszek Giro (Micro-area Analyses Laboratory at the Polish Geological Institute-National Research Institute, Warsaw), for providing SEM assistance during the project. We also want to thank two anonymous reviewers for their suggestions that helped us improve this article. Thanks are also due to Magdalena Błażewicz-Paszkowycz for language correction and polishing the final version of the manuscript.
Conflict of interest
None of the co-authors have any sort of concern in the submission of this manuscript to the journal Polar Biology. There is no conflict of interest with any financial organization regarding the material discussed in the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution License which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
About this article
Cite this article
Pabis, K., Hara, U., Presler, P. et al. Structure of bryozoan communities in an Antarctic glacial fjord (Admiralty Bay, South Shetlands). Polar Biol 37, 737–751 (2014). https://doi.org/10.1007/s00300-014-1474-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-014-1474-1