Abstract
This publication presents an unusual behavior of a comatulid crinoid that was tentatively attributed to Heliometria glacialis (Echinodermata, Crinoidea, Heliometrinae). This unstalked crinoid was observed clinging on Hyas sp. crabs in the inner part of the Spitsbergen fjords heavily loaded with suspension runoff. In situ observations were made using a 16-h and 45-min time-lapse sequence showing the decompositions of Atlantic cod. During the exposure, apart from typical organisms such as carrion crustaceans, crabs, and starfish, three individuals of crinoids were observed. One of the crinoids was clinging to the edge of the platform for most of the exposure. The remaining two individuals were observed attached to the carapace of Hyas sp. crabs for 6 h 55 min. It was also found, based on analysis of the video material from 202 locations examined in the summer season between 2015 and 2020 using a drop camera, two additional locations showing comatulid crinoids. Both locations in the Ekmanfjorden and in the interior of the Vanmijenfjorden are areas under the strong influence of a glacier meltwater plum or a glacial river runoff. So far, comatulid crinoids have been found mainly in locations outside of the influence of turbid waters from fjords. We interpret this new behavior within comatulid crinoids as an adaptation that help colonize new regions gradually exposed by the retreating glaciers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Due to the global warming, the European Arctic is one of the most changing marine ecosystems in the world (IPCC 2022). The retreat of sea ice, and tidal glaciers (de- icing), associated with increased advection of Atlantic waters is especially visible in coastal waters and fjords of the Svalbard archipelago (Søreide et al. 2021). This rapid change creates unique opportunity to observe how organisms adapt to new environment and emerging niches.
Particularly not only interesting, but also difficult to study are areas located in the direct vicinity of a tidewater glacier or a glacial river. Extreme sedimentation rates create exceptionally soft sediment that does not allow the use of traditional drop cameras with triggering by a weight hitting the bottom. Visibility in such places often reaches 20 cm and less.
Rapidly melting tidal glaciers on Svalbard discharge huge amount of freshwater, fine, and coarse mineral particles in the vicinity of the glacier front (Lydersen et al. 2014). In areas of high mineral sedimentation rates, colonization by the epibenthic fauna requires an adapted behavior to prevent sinking into the sediment, avoid clogging of filtering apparatus, and being buried under massive rain of particles. Typical adaptations are to be mobile (fast swimming crustaceans) or very small and light (minute bivalves)—(Wlodarska-Kowalczuk and Pearson 2004) or being able to turn around and shake the sediment off—like colonial bryozoan Alcyonidium disciformae (Kukliński and Porter 2004). Other option is to use large dropstones as a platform above the soft substrate or larger mobile species as a vehicle like organisms colonizing hermit crab shells (Balazy et al. 2015).
Our aim was to study newly exposed areas of coastline and seabed and check how these are becoming colonized by fauna (ACCES project—see Søreide et al 2021). Recording time-lapse videos from the deglaciated fjords, we have observed an unusual behavior of the filter feeder crinoid in the turbid coastal environment, which is a new observation of the presumed adaptation helping the colonization of new regions.
There are two species of unstalked crinoids known from Spitsbergen coast (Clark 1970); however, only one, Heliometra glacialis, is regarded as relatively common and widely distributed on the outer shelf of Barents and Greenland seas—(Dyer et al. 1984; Graeve et al. 1997; Schander et al. 2010). The most typical bottom for this species is gravel mixed with mud and sand, but it has also been found only on sand, silt, and loose stones (Clark and Clark McGown 1967). Present records of this species typically placed it on mixed to hard bottom out of the fjords turbid waters, so occurrence of Heliometra glacialis within inner fjord basin comes as a surprise.
Research area (environmental setting and location)
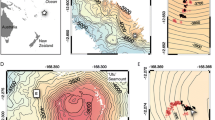
The research area is the western fjords of Spitsbergen. The Ekmanfjorden is the northern branch of the large centrally located Isfjorden fjord system (Fig. 1). It ends with two bays. One of them is Brevika, ends with a glacier flowing directly into the water, while the other is Mudderbukta ends with a river delta that drains water from glaciers ending on land. The place is heavily loaded with suspension runoff at least periodically since June–August (Fig. 1). This is visible on the water surface in the form of brick-red plumes, which is the result of the erosion of Devonian sandstones lying in the area (Hjelle 1993; Kavan et al. 2022). Median size of suspended particle near glacier area is much bigger than in inner and outer parts of the fjords (Sagan and Darecki 2018). The median size of suspended particles increases in water column due to sedimentation and flocculation processes (Szczuciński and Zajączkowski 2013; Moskalik et al. 2018). In winter, the fjord freezes and the ice recedes at the beginning of June (Kavan et al. 2022). Van Mijenfjord is a medium-sized fjord located south of Isfjord. The station where the presence of unstalked crinoids was confirmed is situated in the inner part of the fjord between Rindersbukta with a glacier still actively flowing into the bay and Sveabukta, where suspension from the great glacial river Kjellstromdalen is discharged. The innermost part of the fjord usually freezes in winter. The image material was collected in the middle of the ablation season (Holmes et al. 2019) when the sedimentation rate is very high. In Kongsfjorden, the sedimentation rate reaches 20,000 g m−2 year−1 in the area of the ice front (Svendsen et al. 2002). During the rest of the year the activity of the glacier and therefore the sedimentation rate is much lower. On the basis of research in Adolfbukta, it was found that in the autumn, the particulate matter flux to the bottom decreases by an order of magnitude compared to summer while the concentration of suspended particulate matter (SPM) decreases only twice. The authors explain this phenomenon with less effective flocculation (Szczuciński and Zajączkowski 2013).
General map of Spitsbergen (at left) with locations of the drop camera stations and detailed satellite views figured at right. Red dots: drop camera stations, green dots: stations in which the presence of unstalked crinoids was recorded (DK1 and DK2), red asterisk: Lander station. The satellite images were taken two days after the Lander experiment and show plumes of sediment reaching the station areas
Materials and methods
Time-lapse cameras with lighting according to Balazy et al. (2018) were mounted on an adapted Lander structure previously used to take underwater vertical photos. The device was lowered to the seabed from the RV Oceania at the selected location (Fig. 1) at a depth of 35 m. The ship then sailed to another position minimizing its impact. Lander is a construction made of stainless steel, weighted with lead to minimize tipping when lowering to the bottom. Modification of the Lander consisted mainly in building special clamps for two sets of cameras and adding a platform at the height of 30 cm for attaching the bait. Due to the often very hydrated and soft sediment in the inner parts of the Spitsbergen fjords, the heavy lander was sinking into the sediment, and as a result, the bait platform was at the bottom level. Classical benthic gear (box cores) use to sink in the sediment as well. A drop camera with a preview or specially prepared time-lapse video equipment left for a short time up to several days (buried by sediment) will work best. One of the cameras was mounted at an angle, the other directly above the bait. The bait was half Atlantic cod. The exposure time was 16 h 45 min. The camera mounted at an angle worked with an interval of 5 min, which allowed to obtain 205 photos. The vertically mounted camera worked with an interval of 15 min, which allowed to obtain 67 photos. The vertical camera images had a defect due to a trapped air bubble in front of the lens but were still usable. The material was recorded in both JPG and RAW format, which allowed to easily and without loss of quality improve the exposure of photos. From the captured material, a time-lapse movie (15 fps—frame per second) was made showing the course of the entire experiment. Drop camera photos were taken according to the methodology described by Deja et al. 2019 in the summer season between 2015 and 2020 from the research vessel RV Oceania. In total, material was collected from 202 locations. The bottom was filmed with a camera for 10 min. In the studied locations, CTD (Condutivity–Temperture–Depth) profiles were also made to check the temperature and salinity in the bottom layer.
Results
After 55 min, the first carrion crustaceans appeared on the bait. Mainly Onisimus sp. and Orchomenella minuta. At the same time, they constituted the main part of the attracted invertebrates. After two hours and 55 min, the first representative of crabs attributed to Hyas sp. was recorded. Several large scavenger crustaceans Anonyx nugax were also attracted, but in very small numbers (only a few individuals). The bait was also visited once by a starfish, probably Urasterias lincki. After 4 h and 35 min from the beginning of the time-lapse sequence, one unstalked crinoid from the Comatulida order attributed to Heliometra glacialis was recorded. After the analysis of subsequent frames, it can be seen that the this unstalked crinoid was clinging to the platform edge (Fig. 2) and remained in this position until the Lander was pulled out, i.e., over 12 h. In the eighth hour of exposure, a second specimen of Heliometra glacialis was registered. It was clinging to the carapace of a crab Hyas sp. For the next hours, this Hyas sp. crab with a crinoid attached remained visible in the camera field, and at 14 h a second Hyas sp. crab also with a crinoid on its carapace appears (Fig. 2). Until Lander was brought to the surface, the crabs with attached crinoids were feeding on the exposed bait. For a total of 6 h 55 min (131 frames), the assemblage crabs/crinoid or only crinoid arms were recorded in the field of view (which meant that the crab was feeding out of frame on an invisible fragment of the bait).
Photographs showing Heliometra glacialis recorded during bait experiment on Lander station. 1a—A crinoid attached with its cirri to the edge of the platform. 2a—Specimen with cirri clinging on Hyas sp. carapace. 2b—Arm of a second crinoid also attached to a nearby crab. 3–4—photographs from the horizontal camera, better showing the inter-relationships crinoid/crab
The analysis of the video material from the drop camera showed that comatulid crinoid was also recorded in two additional locations out of the 202 surveyed places. Both locations (DK1, DK2) are located in the inner part of the fiords and are similar in terms of hydrological conditions (Fig. 1). Temperature and salinity in the bottom layer for station DK1 (72 m depth) are (− 1.45 °C; 34.27) and those for station DK2 (42 m depth) are (2.7 °C; 33.7), respectively. On the surface layer, temperature varies from 4.59 °C on DK1 station to 8.03 on DK2, and salinity varies from 28.49 to 16.21 respectively. However, none of the comatulid crinoid observed in these additional locations was seen clinging on a crab.
Observations of crinoids in other glacial bays, the innermost parts of the fjords of western Spitsbergen were confirmed using a drop camera (Fig. 3) and other common echinodermata (starfish and brittle stars) were found in large numbers within the glacier basin and glacier front (Deja unpublished data).
Specimens of Heliometria glacialis registered in the summer of 2020 in Vanmijenfjorden at DK1 station (1;2) at a 72 m depth and in Ekmanfjorden at DK2 station (3;4) at a 42 m depth. Photographs from Ekamnfjorden are not to scale due to very difficult hydrological conditions (strong currents and high suspension)
Discussion
Comatulids and other invertebrate suspension feeders often colonize elevations on the seabed (Meyer 1980; Messing et al. 1990; Messing 1997, 2004; Meyer et al. 2014, 2021; Meyer and Sweetman 2015). At such heights, the flow of current is faster and more laminar, thanks to which these organisms get a feeding advantage (Genin et al. 1986; Meyer-Kaiser et al. 2021). We observed one crinoid attached to the edge of the platform and the other two attached to the carapace of a crab (Fig. 2, Online Resource—supplementary time-lapse video material). An experiment conducted in the LTER (long-Term Ecological Research) area of HAUSGARTEN, Meyer-Kaiser et al. (2019) showed that on abandoned steel structure the number of comatulid crinoids attributed to Poliometra prolixa was significantly higher than at the bottom surface. In Fram Strait where the hard substrate is limited, P. prolixa has been frequently observed on stones and as an epibiont on sponges (Meyer et al. 1984, 2014, 2016; Schulz et al. 2010). Bottom currents are essential for benthic organisms to disperse, reproduce, and feed (Eléaume et al. 2011). Dense aggregations of Heliometra have been noted on the elevations of sea bed in oceanic ridge—Mohn area in N Atlantic or on the large colonies of sponges (Schander et al. 2010). In the glacial bay, the hard substrate is also very limited, which may explain to some extent the association with crabs, and the attachment of one specimen to the edge of the platform (Fig. 2). To the best of our knowledge, no similar observation has ever been recorded. Crabs from the Majoidea superfamily, which are also represented by the observed Hyas sp., are known for decorating the carapace. An association between Hyas araneus and Hyas coarctatus with the sea anemones Cribrinopsis similis, Hormathia nodosa, and Urticina crassicornis was previously observed in the study area (Balazy et al. 2014). There are only a few reports of epibiont relationships between echinodermata and sea crabs in general (Ng and Jeng 1999; Lee and Ko 2009). Relationships among Pilumnidae, Portunidae are mainly described. Associations with crinoids (Harrovia albolineata, Harrovia japonica, Tiaramedon spinosum) have also been recorded, but none of these observations concern the Arctic regions.
The dropstone melted from the growler will be quickly buried, but the highly mobile large crab gets rid of the suspended matter, while being a convenient substrate for the crinoids. The notion that Echinodermata prefer non-turbid waters often mentioned in the literature is a misleading generalization (Meyer 1980). Many Echinodermata, including crinoids, live in at least periodically cloudy environments (Meyer 1973, 1980). Such a periodically cloudy environment is precisely the glacier bay, the innermost parts of the fjords.
Comatulids in Svalbard were mainly observed in the deep water region (Anisimova and Cochrane 2003; Meyer-Kaiser et al. 2021), the northern Svalbard shelf (Meyer et al. 2015), Wijdefjorden in the northern part (Gulliksen and Svensen 2004). Heliometra glacialis generally prefers open waters and has rarely been observed in bays or fjords. However, its presence was found in Gronfjord (small branch in the entrace of Isfjord) (Clark and Clark McGown 1967) and a study conducted over 100 years ago (Hofsten 1915) also found isolated occurrences of Heliometria glacialis in Isfjord. The temperature range in which the species was recorded ranges from − 1.90 to 5.80 °C, while the salinity ranges from 32.2 to 34.9. (Clark and Clark 1967) which is consistent with the temperature and salinity values measured during our study.
The area of the fjords, especially the inner parts, still have much to offer and can significantly expand our knowledge about the behavior, interactions, and functioning of benthic organisms in this changing environment.
The melting tidal glacier forms environment that can offer benefits and not only difficulties. First of all, there is massive rain of marine plankton killed at the surface by the osmotic shock (Węsławski and Legeżyńska 1998; Zajaczkowski and Legezyńska 2001). This large and predictable food source sustains rich community of mobile carrion feeders and occasionally also krill that is able to collect phytoplankton food from the sediment (Hirche et al. 2016; Deja et al. 2019). This may be not only a convenient niche for crinoids, but also for the broadly defined megafauna throughout the fjord, also because that large benthic predators such as Atlantic cod avoid such environments (Szczucka et al. 2017).
Crinoids seems to use the uncommon method—crab riding, to avoid the problem with unstable bottom and still use the benefits of glacial front. However, it should be taken into account that crinoids can move on their own, and also might be transported with currents to places behind their usual optimal habitats. There the crab riding might be a useful survival strategy.
Conclusion
During recording time laps videos, we observed the presence of crinoids in the inner part of deglaciated fjords, under strong influence of turbid water from glacier meltwater. Known records of this species typically place it on mixed to hard bottom out of the fjords turbid waters, so our finding of Heliometra within inner fjord basin comes as a surprise. In addition, we have observed two specimens of the comatulid crinoid Heliometra glacialis clinging on crabs. We interpret this yet undescribed behavior as an adaptation to colonization of new habitats and in this case to avoid the problem with unstable bottom and still use the benefits of glacial bay.
An alternative interpretation could be: fjords are not optimal habitats for comatulids but some individuals, transported by currents or by chance end up in there, some are lucky enough to find elevated substrate on which to cling, some of these substrates are crabs and this is possible because this crab accepts symbiotic relationships. This interpretation takes into account all of the data available and points at the idea that this is not an “adaptation” (this needs to be defined) but some sort of colonization by chance that is expected in animal populations, especially in populations composed of many individuals such as Heliometra.
References
Anisimova NA, Cochrane SJ (2003) An annotated checklist of the echinoderms of the Svalbard and Franz Josef Land archipelagos and adjacent waters. Sarsia 88:113–135. https://doi.org/10.1080/00364820310000102
Balazy P, Kuklinski P, Sanamyan N (2014) Hyas spp. crabs and sea anemones—new species associations from Svalbard. Mar Biodivers 44:161–162. https://doi.org/10.1007/s12526-014-0203-x
Balazy P, Kuklinski P, Wlodarska-Kowalczuk M et al (2015) Hermit crabs (Pagurus spp.) at their northernmost range: Distribution, abundance and shell use in the European Arctic. Polar Res 34:1–17. https://doi.org/10.3402/polar.v34.21412
Balazy P, Kuklinski P, Berge J (2018) Diver deployed autonomous time-lapse camera systems for ecological studies. J Mar Eng Technol 17:137–142. https://doi.org/10.1080/20464177.2017.1357164
Clark AM (1970) Echinodermata, Crinoidea. Marine invertebrates of Scandinavia. Universitetsforlaget, Oslo, pp 1–55
Clark AH, Clark AM (1967) A monograph of the existing crinoids 1(5). US Nat Mus Bull 82:1–860
Deja K, Ormańczyk M, Dragańska-Deja K (2019) Plankton or benthos: where krill belongs in Spitsbergen fjords? (Svalbard Archipelago, Arctic). Polar Biol 42:1415–1430. https://doi.org/10.1007/s00300-019-02524-1
Dyer MF, Cranmer GJ, Fry PD, Fry WG (1984) The distribution of benthic hydrographic indicator species in Svalbard Waters, 1978–1981. J Mar Biol Assoc United Kingdom 64:667–677. https://doi.org/10.1017/S0025315400030332
Eléaume M, Beaman RJ, Griffiths HJ et al (2011) Near-bottom current direction inferred from comatulid crinoid feeding postures on the Terre Adélie and George V shelf, East Antarctica. Deep Res Part II Top Stud Oceanogr 58:163–169. https://doi.org/10.1016/j.dsr2.2010.05.023
Genin A, Dayton PK, Lonsdale PF, Spiess FN (1986) Corals on seamount peaks provide evidence of current acceleration over deep-sea topography. Nature 322:59–61. https://doi.org/10.1038/322059a0
Graeve M, Kattner G, Piepenburg D (1997) Lipids in arctic benthos: does the fatty acid and alcohol composition reflect feeding and trophic interactions? Polar Biol 18:53–61. https://doi.org/10.1007/s003000050158
Gulliksen B, Svensen E (2004) Svalbard and life in polar oceans. KOM Forlag, Oslo
Hirche HJ, Laudien J, Buchholz F (2016) Near-bottom zooplankton aggregations in Kongsfjorden: implications for pelago–benthic coupling. Polar Biol 39:1897–1912. https://doi.org/10.1007/s00300-015-1799-4
Hjelle A (1993) Geology of Svalbard. Norsk Polarinstitutt, Oslo
Hofsten N (1915) Die Echinodermen des Eisfjords. In: Zoologische ergebnisse der schwedischen expedition nach Spitzbergen 1908. Almqvist & Wiksells Boktryckeri, Stockholm
Holmes FA, Kirchner N, Kuttenkeuler J et al (2019) Relating ocean temperatures to frontal ablation rates at Svalbard tidewater glaciers: insights from glacier proximal datasets. Sci Rep 9:1–11. https://doi.org/10.1038/s41598-019-45077-3
IPCC (2022) Climate change 2022: mitigation of climate change. Contribution of Working Group III to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge, UK and New York, NY, USA
Kavan J, Wieczorek I, Tallentire GD et al (2022) Estimating suspended sediment fluxes from the largest glacial lake in Svalbard to Fjord system using sentinel-2 data: trebrevatnet case study. Water (switzerland) 14:1–14. https://doi.org/10.3390/w14121840
Kukliński P, Porter JS (2004) Alcyonidium disciforme: an exceptional Arctic bryozoan. J Mar Biol Assoc United Kingdom 84:267–275. https://doi.org/10.1017/S0025315404009130h
Lee S-H, Ko H-S (2009) First record of crinoid symbiotic crab, Harrovia japonica (Decapoda: Brachyura: Pilumnidae) from Korea. Anim Syst Evol Divers 25:125–128. https://doi.org/10.5635/kjsz.2009.25.1.125
Lydersen C, Assmy P, Falk-Petersen S et al (2014) The importance of tidewater glaciers for marine mammals and seabirds in Svalbard, Norway. J Mar Syst 129:452–471. https://doi.org/10.1016/j.jmarsys.2013.09.006
Messing CG (1997) Living comatulids. In: Waters JA, Maples CG (eds) Geology of echinoderms. The paleontological society papers, 3. Carnegie Museum of Natural History, Pittsburgh, pp 3–30
Messing CG (2004) Biozonation on deep-water carbonate mounds and associated hardgrounds along the western margin of Little Bahama Bank, with notes on the Caicos Platform Island Slope. Proc 11th Symp Geol Bahamas Other Carbonate Reg 107–115
Messing CG, Neumann AC, Lang JC (1990) Biozonation of deep-water lithoherms and associated hardgrounds in the northeastern Straits of Florida. Palaios 5:15–33. https://doi.org/10.2307/3514994
Meyer DL (1973) Feeding behavior and ecology of shallow-water unstalked crinoids (echinodermata) in the Caribbean Sea. Mar Biol 22:105–129. https://doi.org/10.1007/BF00391776
Meyer DL (1980) Ecology and biogeography of living classes. Notes a Short Course Stud Geol 3:1–14. https://doi.org/10.1017/s0271164800000051
Meyer KS, Sweetman AK (2015) Observation of a living macroalga at 166 m in a high Arctic fjord. Mar Biodivers Rec 8:e58. https://doi.org/10.1017/S175526721500038X
Meyer DL, LaHaye CA, Holland ND et al (1984) Time-lapse cinematography of feather stars (Echinodermata: Crinoidea) on the Great Barrier Reef, Australia: demonstrations of posture changes, locomotion, spawning and possible predation by fish. Mar Biol 184:179–184
Meyer KS, Soltwedel T, Bergmann M (2014) High biodiversity on a deep-water reef in the eastern Fram Strait. PLoS ONE 9:1–16. https://doi.org/10.1371/journal.pone.0105424
Meyer KS, Sweetman AK, Young CM, Renaud PE (2015) Environmental factors structuring Arctic megabenthos-a case study from a shelf and two fjords. Front Mar Sci. https://doi.org/10.3389/fmars.2015.00022
Meyer KS, Wagner JKS, Ball B et al (2016) Hyalinoecia artifex: Field notes on a charismatic and abundant epifaunal polychaete on the US Atlantic continental margin. Invertebr Biol 135:211–224. https://doi.org/10.1111/ivb.12132
Meyer DL, Veitch M, Messing CG, Stevenson A (2021) Crinoid feeding strategies: new insights from subsea video and time-lapse. Cambridge University Press, Cambridge
Meyer-Kaiser K, Bergmann M, Soltwedel T, Klages M (2019) Recruitment of Arctic deep-sea invertebrates Results from a long-term. Limnol Oceanogr 64:1924–1938
Meyer-Kaiser K, Smith A, Soltwedel T (2021) Ontogenetic development of the crinoid Poliometra prolixa in the Arctic deep sea. Invertebr Biol 140:12331. https://doi.org/10.1111/ivb.12331
Moskalik M, Ćwiąkała J, Szczuciński W et al (2018) Spatiotemporal changes in the concentration and composition of suspended particulate matter in front of Hansbreen, a tidewater glacier in Svalbard. Oceanologia 60:446–463. https://doi.org/10.1016/j.oceano.2018.03.001
Ng PKL, Jeng MS (1999) The brachyuran crabs (Crustacea: Decapoda: Eumedonidae and Portunidae) symbiotic with echinoderms in Taiwan. Zool Stud 38:268–274
Sagan S, Darecki M (2018) Inherent optical properties and particulate matter distribution in summer season in waters of Hornsund and Kongsfjordenen, Spitsbergen. Oceanologia 60:65–75. https://doi.org/10.1016/j.oceano.2017.07.006
Schander C, Rapp HT, Kongsrud JA et al (2010) The fauna of hydrothermal vents on the Mohn Ridge (North Atlantic). Mar Biol Res 6:155–171. https://doi.org/10.1080/17451000903147450
Schulz M, Bergmann M, von Juterzenka K, Soltwedel T (2010) Colonisation of hard substrata along a channel system in the deep Greenland Sea. Polar Biol 33:1359–1369. https://doi.org/10.1007/s00300-010-0825-9
Søreide JE, Pitusi V, Vader A et al (2021) Environmental status of Svalbard coastal waters: coastscapes and focal ecosystem components (SvalCoast). State Environ Sci Svalbard Rep 2020:143–174. https://doi.org/10.5281/zenodo.4293849
Svendsen H, Beszczynska-Møller A, Hagen JO et al (2002) The physical environment of Kongsfjorden—Krossfjorden, an Arctic fjord system in Svalbard. Polar Res 21:133–166. https://doi.org/10.1111/j.1751-8369.2002.tb00072.x
Szczuciński W, Zajączkowski M (2013) Factors controlling downward fluxes of particulate matter in glacier-contact and non-glacier contact settings in a subpolar Fjord (Billefjorden, Svalbard). Sediments Morphol Sediment Process Cont Shelves. https://doi.org/10.1002/9781118311172.ch18
Szczucka J, Hoppe Ł, Schmidt B, Fey DP (2017) Acoustical estimation of fish distribution and abundance in two Spitsbergen fjords. Oceanologia 59:585–591. https://doi.org/10.1016/j.oceano.2017.04.007
Węsławski JM, Legeżyńska J (1998) Glaciers caused zooplankton mortality? J Plankton Res 20:1233–1240. https://doi.org/10.1093/plankt/20.7.1233
Wlodarska-Kowalczuk M, Pearson TH (2004) Soft-bottom macrobenthic faunal associations and factors affecting species distributions in an Arctic glacial fjord (Kongsfjord, Spitsbergen). Polar Biol 27:155–167. https://doi.org/10.1007/s00300-003-0568-y
Zajaczkowski MJ, Legezyńska J (2001) Estimation of zooplankton mortality caused by an Arctic glacier outflow. Oceanologia 43:341–351. https://doi.org/10.1016/j.jmarsys.2013.09.006
Acknowledgements
We are grateful to the Piotr Bałazy for sharing their knowledge and experience, valuable hints, and suggestions for custom designing a time-lapse system for our research. This research was supported by funds from the European Union’s Horizon 2020 research and innovation programme (GA No 776617) under the BiodivScen call (research project no. 2018/28/Z/NZ8/00079, acronym: ACCES, research funding institution: National Science Centre, Poland), National Science Centre, Poland project no. 2015/19/N/ST10/01659 and National Science Centre, Poland project no. 2018/29/B/NZ8/02340. We appreciate the constructive comments from the reviewers Michel Roux and Marc Eléaume that improved the manuscript considerably.
Author information
Authors and Affiliations
Contributions
KD conducted the experiments, analyzed the data, and designed and wrote the manuscript. KDD wrote and edited the manuscript. JMW reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors are required to disclose financial or non-financial interests that are directly or indirectly related to the work submitted for publication. Please refer to “Competing Interests and Funding” below for more information on how to complete this section.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (MP4 69892 KB)
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Deja, K., Dragańska-Deja, K. & Wesławski, J.M. New strategies for the new environment in Spitsbergen fjords (Arctic). Scattering of the feather star Heliometra glacialis (Echinodermata, unstalked crinoid) clinging to a crab. Polar Biol 46, 1137–1143 (2023). https://doi.org/10.1007/s00300-023-03171-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-023-03171-3