Abstract
Background
Group 3 pulmonary hypertension (PH) describes a subpopulation of patients with PH due to chronic lung disease and/or hypoxia, with chronic obstructive pulmonary disease (COPD) and interstitial lung disease (ILD) being two large subgroups. Claims database studies provide insights into the real-world treatment patterns and outcomes among these patients. However, claims data do not provide sufficient detail to assign the clinical subtype of PH required for identifying these patients.
Methods
A panel of PH clinical experts and researchers was convened to discuss methodologies to identify patients with Group 3 PH associated with COPD or ILD in retrospective claims databases. To inform the discussion, a literature review was conducted to identify claims-based studies of Group 3 PH associated with COPD or ILD published from 2010 through June 2020.
Results
Targeted title and abstract review identified 11 claims-based studies and two conference abstracts (eight based in the United States [US] and five conducted outside the US) that met search criteria. Based on insights from the panel and literature review, the following components were detailed across studies in the identification of Group 3 PH associated with COPD and ILD: (a) COPD or ILD identification, (b) PH identification, (c) defining the sequence between COPD/ILD and PH, and (d) other PH Group and Group 3 PH exclusions.
Conclusion
This article provides recommended approaches and considerations for identifying and studying patients with Group 3 PH associated with COPD or ILD using administrative claims data that provide the foundation for future validation studies.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Pulmonary hypertension (PH) is a condition characterized by elevation in pulmonary artery pressures of varying etiologies, which may result in substantial morbidity. PH is clinically classified into five groups, defined by the World Symposium on PH, according to differing pathological findings between groups, including underlying cause of disease, clinical presentation, and hemodynamic characteristics [1]. PH clinical classifications are also used by healthcare providers for medical management and by the Food and Drug Administration (FDA) for labeling of new drugs approved for the treatment of PH [1, 2].
Group 3 PH describes a subpopulation of patients with PH due to chronic lung disease and/or hypoxia. In particular, chronic obstructive pulmonary disease (COPD) and interstitial lung disease (ILD) are two large subgroups of chronic lung disease patients who often develop PH [2, 3]. In COPD, the prevalence rate ranges from 30 to 70% [4]. Because ILD is composed of multiple lung diseases, its prevalence is difficult to estimate. In idiopathic pulmonary fibrosis (IPF), the most common type of ILD, prevalence rates of PH range from 8 to 15% at initial diagnosis to 46% at evaluation for lung transplant and 86% at the time of transplant [5,6,7,8,9,10]. Wide ranges in prevalence can be due to heterogeneity in the definitions for PH, diagnostic modalities for PH, differences in patient populations, physiologic characteristics, and severity of underlying lung disease [4, 11].
Administrative claims are generated following healthcare utilization for the purposes of payment. Medical documentation is converted to standardized codes using uniform coding systems. The International Classification of Diseases (ICD), a medical classification system, is the international standard for reporting diseases and health conditions. In the US, the ICD, 9th Revision, Clinical Modification (ICD-9-CM) and ICD, 10th Revision, Clinical Modification (ICD-10-CM) provide a system of diagnostic codes assigned for each encounter.
ICD-9-CM diagnostic codes provide the level of detail to indicate PH but are not specific to groups of PH defined by World Symposium on PH. All groups of PH due to lung disease are generally coded under the same four-digit codes. The newer ICD-10-CM, implemented in October 2017, provides five-digit codes for PH with greater differentiation but was not required for billing and reimbursement until October 2018 [12]. The designated code for Group 3 PH is I27.23, and it remains to be seen how frequently and accurately the utilization of this code will be.
While collected for billing purposes, claims data can provide real-world evidence outside the setting of a clinical trial about the treatment patterns, risk factors, patient outcomes, healthcare resource utilization, and costs for patients covered by health insurance plans. However, using diagnostic codes in claims databases for the identification of patients with Group 3 PH associated with lung disease conditions like COPD and ILD is not sufficient. Additional considerations must be applied to patient identification to reduce misclassification and improve the usefulness of claims data to understand the medical management and health-related outcomes of patients with these conditions.
Recently, a focused review provided recommended algorithms for the identification of patients with pulmonary arterial hypertension (Group 1 PAH) [13]. Others have explored the use of machine-learning approaches to reduce selection bias in patient identification [14]. Our goal is to provide readers with information to determine the most appropriate methodology for claims-based patient selection under different types of research questions about Group 3 PH associated with COPD or ILD.
Methods
A panel of US-based healthcare providers and researchers with expertise in PH was convened to discuss methodologies to identify patients with Group 3 PH associated with COPD or ILD in retrospective claims databases. Panel members included US-based practicing pulmonologists (n = 2), a nurse (n = 1), pharmacists (n = 6) with expertise in PH and backgrounds in public health and/or claims-based analysis, and researchers (n = 2) with expertise in pharmacoeconomics, epidemiology, and claims-based analysis.
We conducted a literature review to identify studies and explore considerations when using claims-based data to identify patients with Group 3 PH associated with COPD or ILD. The recommendations in this article are those of the authors convened for the discussion and are based on group consensus.
The literature review utilized EMBASE and MEDLINE (via EMBASE) to identify English language articles published from 2010 through June 2020, on adult patient population, including both US and international studies. We looked for claims-based studies, retrospective studies, or healthcare management-related studies, focused on both PH and lung diseases causing Group 3 PH, or that specifically mentioned Group 3 PH. The search terms for lung diseases and PH were derived from several sources [3, 15] and decided upon by the panel. Search terms were required in the title or abstract (Supplemental Table 1). We also referred to a similar effort around conceptualization of Group 1 PAH in order to provide further support [13].
Additionally, we reviewed abstracts from 2018 through 2020 from the American Thoracic Society, the American College of Chest Physicians, and the Pulmonary Vascular Research Institute to identify relevant studies.
Results
The broad literature search strategy resulted in 2,646 potential observational studies in patients with Group 3 PH associated with COPD or ILD (Supplemental Table 1). The targeted title and abstract review identified 11 studies and 2 conference abstracts for claims-based studies focusing on Group 3 PH associated with COPD or ILD; 8 studies based in the US and 5 non-US (Table 1) (Supplemental Fig. 1).
The focus of these studies varied. Four studies evaluated measures across multiple PH groups or Group 3 PH in general [16,17,18,19]. There were 3 studies on patients with COPD [20,21,22]: 2 studies each in ILD [23, 24] and IPF [25, 26] and 1 study in systemic sclerosis classified as Group 3 PH [27]. One study looked at PH in ILD, COPD, and combined ILD and COPD [28].
Most studies (n = 9) claimed to be identifying Group 3 PH and included steps in their methodology to filter for these patients (such as ensuring that PH occurred after COPD/ILD or excluded non-Group 3 PH) [16,17,18,19,20,21,22, 27, 28]. Other studies took additional steps in their methodology but did not specifically claim to identify Group 3 PH [26] or reported PH as a comorbidity to the lung disease [23,24,25].
In the following discussion section, we outline key components to consider in the development of an algorithm for Group 3 PH associated with COPD or ILD. For each, we outline the findings from the literature search that relate to the component followed by our summary and recommendations.
Discussion
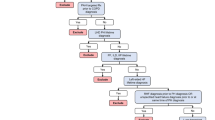
There are several components to consider when choosing an algorithm. The methods used in the published literature for identifying adult patients with Group 3 PH associated with COPD or ILD relied on the following considerations: (a) identification of COPD or ILD, (b) identification of PH, (c) defining the sequence between COPD/ILD and PH diagnoses, and (d) other PH groups or other Group 3 PH exclusions. We provide a summary of the published literature findings followed by considerations as to the impact of how restricting or relaxing the criteria for each of these components can impact the diagnostic performance of the algorithm. Components of the recommended algorithm are provided in Fig. 1.
Identification of COPD or ILD
Findings in the Literature
The criteria used for the identification of COPD and ILD within the examined studies included (a) diagnosis codes, (b) relevant procedures, and (c) claims for medication. All 13 studies used diagnosis codes to identify patients with COPD or ILD, with 11 studies reporting ICD-9-CM codes [16,17,18,19,20,21,22, 24,25,26, 28] and 5 reporting ICD-10-CM codes [19, 20, 23, 27, 28].
Except for one COPD study conducted in the US that required the COPD diagnosis to be in the primary position [21], the remaining studies allowed the COPD or ILD diagnosis codes to be in either the primary or secondary position. Twelve studies identified patients from either inpatient or outpatient claims [16,17,18,19,20,21,22,23, 25,26,27,28] and the one remaining study used inpatient claims only [24]. In addition, 6 of the 12 studies using outpatient claims included a requirement for multiple claims (i.e., ≥ 2 claims) with a specified diagnosis [17, 20,21,22, 26, 28].
Only two studies utilized procedures specific to COPD or ILD to identify patients (i.e., pulmonary function tests in patients evaluated for ILD). Lautsch et al. excluded patients with prior lung transplant, while Frank et al. required patients to have at least one diagnostic procedure including bronchoscopy, lung computerized tomography (CT), pulmonary function testing, or assessment of autoantibodies [20, 23]. Two additional studies utilized procedures specific to COPD or ILD in sensitivity or subset analyses, including lung biopsy and CT [25, 26]. Requiring a procedure for COPD or ILD identification resulted in a significant drop in case count but did not change the general outcome of the studies.
Summary and Recommendations
After reviewing codes across studies, recommended diagnosis codes for COPD and ILD are provided in Table 2. While the US and non-US studies utilized similar ICD-9-CM codes for COPD and ILD, they differed more frequently when it came to ICD-10-CM. The US studies used codes down to the fifth digit, whereas non-US studies tended to use the higher, more general code down to the fourth digit. Thus, corresponding codes should be verified if international versions of ICD are used.
In addition, we recommend searching for COPD or ILD diagnoses in the primary or secondary position. When PH symptom exacerbations are due to underlying COPD or ILD, PH may be the primary reason for utilization diagnosis. We also recommend using both inpatient and outpatient claims unless doing so would affect study objectives (e.g., a study looking at hospital readmissions). When outpatient codes are used, we recommend using ≥ 2 to reduce the likelihood that a single diagnostic claim is used for patient identification.
We also note that PH is a complication of connective tissue disease and can be due to mechanisms other than ILD. Thus, caution should be considered with including connective tissue disease-related codes in the identification of ILD. We specifically recommend limiting diagnostic codes to connective tissue disease with respiratory or lung involvement. We also suggest alternative methods for the assessment of patients whose underlying conditions overlap across PH classification, such as analyzing patients meeting criteria for multiple PH groups separately.
Based on the findings from the evaluated studies, we do not recommend using any COPD/ILD-related procedure codes or medication claims for patient identification.
Identification of PH
Findings in the Literature
The criteria used for the identification of PH within examined studies can be classified into the following: (a) diagnosis codes, (b) relevant procedures, (c) claims for medication, and (d) exclusions. All 13 studies used ICD-9-CM and ICD-10-CM diagnosis codes to identify patients with Group 3 PH associated with COPD or ILD. The majority (n = 11) [16,17,18,19,20,21,22, 24,25,26, 28] contained ICD-9-CM codes for patient identification, while six contained ICD-10-CM codes [19, 20, 22, 23, 27, 28]. No studies required the PH diagnosis to be in the primary position. Twelve studies identified patients from either inpatient or outpatient claims [16,17,18,19,20,21,22,23, 25,26,27,28] and the remaining one study used inpatient claims only [24]. The use of additional criteria such as a requirement for multiple claims was reported in five studies [16, 18, 20, 22, 28].
Only one study utilized PH-related procedures (i.e., right heart catheterization [RHC] or echocardiogram) in identification of patients with PH [16], and no studies required claims for any PH-related medications except Kim et al., which focused on appropriate use of phosphodiesterase-5 inhibitors (PDE5is) [17].
Summary and Recommendations
As all studies included PH-related diagnosis codes and there was general consensus on these codes across studies, we recommend utilizing a limited list of consensus codes (Table 3). Corresponding codes should be verified if international versions of the ICD are used. We recommend that PH diagnosis be in the primary or secondary position, as COPD or ILD associated with PH symptom exacerbation may be reported in the primary position. We also recommend using both inpatient and outpatient claims unless the study objectives are limited to one or the other. When outpatient codes are used, require at least two to reduce the likelihood that a single diagnostic claim is used for patient identification and specify a minimum time frame of at least 30 days for how far apart the outpatient claims need to be in order to qualify.
We generally do not suggest utilizing PH-related procedures. While the guideline-driven practice is to confirm cases by means of RHC, studies suggest that less than two-fifths of patients with PH have an RHC prior to diagnosis [16, 19] within 3 months before or after medication [29] or within 12 months following diagnosis [30]. We also note that claims data indicate if a patient received a diagnostic test but do not include test results, thus utilizing echocardiography, which is non-specific to Group 3 PH, would not improve the sensitivity of patient identification. Thus, we recommend limiting the use of procedure codes for sensitivity analysis in patient identification or to limit the study population when a purer cohort is required. Lastly, we do not suggest utilizing PH-related medications for patient identification, as therapies used in Group 1 PAH are often used on- and off-label for Group 3 PH associated with both COPD and ILD.
The Sequence of the COPD/ILD and PH Diagnoses
Findings in the Literature
In order to confirm that COPD or ILD was a contributing factor to the development of PH, patients must have developed lung disease prior to PH. Eight of the 13 evaluated studies required the COPD or ILD diagnosis code to be prior to the PH diagnosis code [16, 19,20,21,22, 26,27,28], and one study required an underlying cause of PH (not limited to lung disease) to be documented prior to PDE5i prescription [17]. In fact, of the 8 studies that specifically claimed to be studying Group 3 PH, seven of them included this criterion [16, 19,20,21,22, 27, 28].
Summary and Recommendations
We recommend that PH diagnosis occur after the COPD or ILD diagnosis to align with the natural progression of this disease. Patients should be identified and indexed on their first claim with a PH diagnosis code and require a COPD or ILD diagnosis code in the baseline period at least six to 12 months prior to the PH diagnosis.
Identification of Other PH Group and Other Group 3 PH Conditions
Findings in the Literature
When identifying patients with Group 3 PH, five studies used diagnosis codes to identify and exclude non-Group 3 PH patients [16, 17, 20, 22, 28] and two studies used diagnosis codes to separate patients who met criteria for multiple PH groups from those who met only Group 3 PH [18, 19]. In addition to diagnosis codes, two studies utilized claims for PAH-indicated medications [20] and two studies used procedure codes [16, 28] to exclude patients from Group 3 PH. One study used PDE5i guidelines to assign patients with multiple diagnoses to an “appropriate use” group [17]. Algorithm assignment across PH groups was compared to chart abstraction, resulting in a positive predictive value of 86% for possible inappropriate use across groups of PH.
Summary and Recommendations
We recommend methodology to exclude patients with other PH groups prior to their Group 3 PH using diagnosis codes provided in Table 4. We do not recommend excluding patients with other PH conditions that develop after their Group 3 PH diagnosis, as these are relatively uncommon occurrences and may arise from diagnostic workup rather than diagnostic confirmation, as claims data reflect clinical care provided rather than results. We acknowledge that approximately 34% of patients with PH have overlapping diagnoses [19] and that excluding these patients may bias results for some research objectives, and the combination of multiple comorbidities may render patients vulnerable to developing PH and contribute to poor prognosis [19]. Thus, an alternative method would be to analyze patients meeting criteria for multiple PH groups separately or assign patients to a group based on the objectives of the study [17].
Using diagnosis codes in the identification of Group 3 PH does have some limitations. ICD-9-CM codes do not have specificity of classification for secondary PH, and it was not until ICD-10-CM that diagnosis codes provided for greater clinical classification of secondary PH. In addition, diagnosis codes do not reflect severity of the disease, particularly as they relate to distinguishing between multiple underlying causes of disease. So, for a patient with mild heart disease but severe lung disease that contributes significantly to PH, using real-world claims data may inadvertently misclassify this person as Group 2 PH.
In addition, there are some notable considerations. Given the overlap of secondary PH-related conditions in Group 1 PAH and Group 3 PH, we do not recommend using I27.x codes when developing exclusion criteria. Further, when conducting a study to identify Group 3 PH with COPD only, we recommend all patients with diagnosis codes for connective tissue disease be excluded. Lastly, given the high prevalence of sleep disorder breathing in COPD and ILD, we do not recommend excluding patients with this diagnosis.
None of the examined studies utilized PH-related medications to directly identify Group 3 PH, but one study did exclude patients with evidence of PAH-indicated medications [20].
As inhaled treprostinil is now FDA approved in both Group 1 PAH and Group 3 PH, and it is likely that medications approved for use in Group 1 PAH are being used off-label in Group 3 PH to improve exercise capacity, we do not recommend relying on medication use to identify or exclude patients.
Conclusion
Correctly identifying adult patients with Group 3 PH associated with COPD or ILD in claims-based studies can improve the value of research findings for application in clinical care and population health, the utility of real-world evidence in support of FDA regulatory approvals, and more accurately inform formulary decision-making. When using the recommendations provided herein, care should be taken to consider policy and regulatory changes, such as FDA approvals and updated guidelines and their impact on how patients are identified. While some studies reported that they used validated codes or algorithms for either COPD/ILD or PH, it is important to note that only one reviewed study provided results on the validation of their patient identification algorithm. Future research should be conducted to validate patient identification algorithms, especially the combination of criteria required for the identification of Group 3 PH associated with COPD or ILD.
Data Availability
All data generated or analyzed during this study are included in this published article or the supplementary information files.
Code Availability
Not applicable.
References
Simonneau G, Montani D, Celermajer DS et al (2019) Haemodynamic definitions and updated clinical classification of pulmonary hypertension. Eur Respir J 53(1):1801913. https://doi.org/10.1183/13993003.01913-2018
Mathai SC, Mathew S (2018) Breathing (and coding?) a bit easier: changes to International Classification of Disease Coding for pulmonary hypertension. Chest 154(1):207–218. https://doi.org/10.1016/j.chest.2018.04.004
Nathan SD, Barbera JA, Gaine SP et al (2019) Pulmonary hypertension in chronic lung disease and hypoxia. Eur Respir J 53(1):1801914. https://doi.org/10.1183/13993003.01914-2018
Minai OA, Chaouat A, Adnot S (2010) Pulmonary hypertension in COPD: epidemiology, significance, and management: pulmonary vascular disease: the global perspective. Chest 137(6):39S-51S. https://doi.org/10.1378/chest.10-0087
Hamada K, Nagai S, Tanaka S et al (2007) Significance of pulmonary arterial pressure and diffusion capacity of the lung as prognosticator in patients with idiopathic pulmonary fibrosis. Chest 131(3):650–656. https://doi.org/10.1378/chest.06-1466
Kimura M, Taniguchi H, Kondoh Y et al (2013) Pulmonary hypertension as a prognostic indicator at the initial evaluation in idiopathic pulmonary fibrosis. Respiration 85(6):456–463. https://doi.org/10.1159/000345221
Lederer DJ, Arcasoy SM, Wilt JS, D’Ovidio F, Sonett JR, Kawut SM (2006) Six-minute-walk distance predicts waiting list survival in idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 174(6):659–664. https://doi.org/10.1164/rccm.200604-520OC
Minai OA, Santacruz JF, Alster JM, Budev MM, McCarthy K (2012) Impact of pulmonary hemodynamics on 6-min walk test in idiopathic pulmonary fibrosis. Respir Med 106(11):1613–1621. https://doi.org/10.1016/j.rmed.2012.07.013
Nathan SD, Shlobin OA, Ahmad S, Urbanek S, Barnett SD (2007) Pulmonary hypertension and pulmonary function testing in idiopathic pulmonary fibrosis. Chest 131(3):657–663. https://doi.org/10.1378/chest.06-2485
Shorr AF, Wainright JL, Cors CS, Lettieri CJ, Nathan SD (2007) Pulmonary hypertension in patients with pulmonary fibrosis awaiting lung transplant. Eur Respir J 30(4):715–721. https://doi.org/10.1183/09031936.00107206
Caminati A, Cassandro R, Harari S (2013) Pulmonary hypertension in chronic interstitial lung diseases. Eur Respir Rev 22(129):292–301. https://doi.org/10.1183/09059180.00002713
ICDdata.com (2021) The web’s free 2021 ICD-10-CM/PCS medical coding reference. https://ICD10data.com. Accessed January 11, 2021
Mathai SC, Hemnes AR, Manaker S et al (2019) Identifying patients with pulmonary arterial hypertension using administrative claims algorithms. Ann Am Thorac Soc 16(7):797–806. https://doi.org/10.1513/AnnalsATS.201810-672CME
Ong MS, Klann JG, Lin KJ et al (2020) Claims-based algorithms for identifying patients with pulmonary hypertension: a comparison of decision rules and machine-learning approaches. J Am Heart Assoc 9(19):e016648. https://doi.org/10.1161/JAHA.120.016648
Seeger W, Adir Y, Barberà JA et al (2013) Pulmonary hypertension in chronic lung diseases. J Am Coll Cardiol 62(25):D109-116. https://doi.org/10.1016/j.jacc.2013.10.036
Heresi GA, Platt DM, Wang W et al (2017) Healthcare burden of pulmonary hypertension owing to lung disease and/or hypoxia. BMC Pulm Med 17(1):58. https://doi.org/10.1186/s12890-017-0399-1
Kim D, Lee KM, Freiman MR et al (2018) Phosphodiesterase-5 inhibitor therapy for pulmonary hypertension in the United States. Ann Am Thorac Soc 15(6):693–701. https://doi.org/10.1513/AnnalsATS.201710-762OC
Trammell AW, Shah AJ, Phillips LS, Hart CM (2019) Mortality in US veterans with pulmonary hypertension: a retrospective analysis of survival by subtype and baseline factors. Pulm Circ 9(1):2045894019825763. https://doi.org/10.1177/2045894019825763
Wijeratne DT, Lajkosz K, Brogly SB et al (2018) Increasing incidence and prevalence of World Health Organization Groups 1 to 4 pulmonary hypertension: a population-based cohort study in Ontario. Canada Circ Cardiovasc Qual Outcomes 11(2):e003973. https://doi.org/10.1161/CIRCOUTCOMES.117.003973
Lautsch D, Ramey D, Yang L, Liu X, Rajpathak S, Bajwa E. Current trends in treatment of pulmonary hypertension due to chronic obstructive pulmonary disease (PH-COPD) in the United States [abstract P417]. Abstract and poster presented at: American Thoracic Society 2020; August 5–10; virtual conference
Medrek SK, Sharafkhaneh A, Spiegelman AM, Kak A, Pandit LM (2017) Admission for COPD exacerbation is associated with the clinical diagnosis of pulmonary hypertension: results from a retrospective longitudinal study of a veteran population. COPD 14(5):484–489. https://doi.org/10.1080/15412555.2017.1336209
Wu WT, Chen CY (2020) Protective effect of statins on pulmonary hypertension in chronic obstructive pulmonary disease patients: a nationwide retrospective, matched cohort study. Sci Rep 10(1):3104. https://doi.org/10.1038/s41598-020-59828-0
Frank AL, Kreuter M, Schwarzkopf L (2019) Economic burden of incident interstitial lung disease (ILD) and the impact of comorbidity on costs of care. Respir Med 152:25–31. https://doi.org/10.1016/j.rmed.2019.04.009
Pedraza-Serrano F, Jiménez-García R et al (2019) Characteristics and outcomes of patients hospitalized with interstitial lung diseases in Spain, 2014 to 2015. Medicine (Baltimore) 98(21):e15779. https://doi.org/10.1097/MD.0000000000015779
Collard HR, Chen SY, Yeh WS et al (2015) Health care utilization and costs of idiopathic pulmonary fibrosis in US Medicare beneficiaries aged 65 years and older. Ann Am Thorac Soc 12(7):981–987
Collard HR, Ward AJ, Lanes S, Cortney Hayflinger D, Rosenberg DM, Hunsche E (2012) Burden of illness in idiopathic pulmonary fibrosis. J Med Econ 15(5):829–835. https://doi.org/10.3111/13696998.2012.680553
Butt SA, Jeppesen JL, Torp-Pedersen C et al (2019) Cardiovascular manifestations of systemic sclerosis: a Danish nationwide cohort study. J Am Heart Assoc 8(17):e013405. https://doi.org/10.1161/JAHA.119.013405
Hemnes A, Ventetuolo CE, Manaker S et al (2019) The economic burden of pulmonary hypertension among patients with chronic obstructive pulmonary disease and interstitial lung disease. Am J Respir Crit Care Med 199:A7035
Duarte A, Li Lin Y, Sharma G (2017) Incidence of right heart catheterization in patients initiated on pulmonary arterial hypertension therapies: a population-based study. J Heart Lung Transplant 36(2):220–226. https://doi.org/10.1016/j.healun.2016.07.017
Fischer A, Kong AM, Swigris JJ, Cole AL, Raimundo K (2018) All-cause healthcare costs and mortality in patients with systemic sclerosis with lung involvement. J Rheumatol 45(2):235–241. https://doi.org/10.3899/jrheum.170307
Acknowledgements
Not applicable.
Funding
Funding for this study was provided by United Therapeutics Corporation.
Author information
Authors and Affiliations
Contributions
GH and CK made substantial contributions to the analysis and interpretation of the data and reviewed and revised the work for important intellectual content. AK, KM, MS, BW, HC, and PC made substantial contributions to the design of the study, the acquisition, analysis, and interpretation of data and reviewed and revised the work for important intellectual content. BD, HL, and DSM made substantial contributions to the design of the study, the acquisition, analysis, and interpretation of data and drafting of the work. Each author contributes important intellectual content during manuscript drafting and revision, accepts personal accountability for the author’s own contributions, and agrees to ensure that questions pertaining to the accuracy or integrity of any portion of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
GH and CK do not report payment or services for any aspect of the submitted work. CK reports personal fees for Advisory Board participation and Speakers’ Bureau from United Therapeutics Corporation, Actelion, and Boehringer Ingelheim Pharmaceuticals. AK, KM, MS, HC, PC, and BW are employees of United Therapeutics Corporation, the funding source for this research; they may also hold stock in the company. BD, HL, and DSM are employees of Xcenda, LLC, which received funding support for this research.
Ethical Approval
Ethics approval was not required as this research did not include human subjects.
Informed Consent
Not applicable.
Consent for Publication:
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Heresi, G.A., Dean, B.B., Castillo, H. et al. Identifying Patients with Group 3 Pulmonary Hypertension Associated with COPD or ILD Using an Administrative Claims Database. Lung 200, 187–203 (2022). https://doi.org/10.1007/s00408-022-00521-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-022-00521-6