Abstract
Herbivores select host plants depending on plant quality and the presence of predators and competitors. Competing herbivores change host plant quantity through consumption, but they can also change plant quality through induction of plant defences, and this affects the performance of herbivores that arrive later on the plant. Some herbivores, such as the spider mite Tetranychus evansi, do not induce, but suppress plant defences, and later-arriving herbivores can profit from this suppression. It has been suggested that the dense web produced by this spider mite serves to prevent other herbivores to settle on the plant and benefit from the suppressed defences. Here, we confirmed this by studying the preference and performance of the whitefly Bemisia tabaci, a generalist herbivorous pest. To disentangle the effects through changes in plant defences from the effects of spider-mite web, we included treatments with a strain of the closely-related web-producing spider mite T. urticae, which induces plant defences. Whiteflies did perform worse on plants with defences induced by T. urticae, but, in contrast to other herbivores, did not perform better on plants with defences suppressed by T. evansi. Moreover, the web of both spider mites reduced the juvenile survival of whiteflies, and whiteflies avoided plants that were covered with web. Hence, whitefly performance was not only affected by plant quality and induced plant defences, but also through the web produced by spider mites, which thus serves to protect against potential competitors, especially when these could profit from the suppression of plant defences by the mites.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Plants have evolved several mechanisms to defend themselves against herbivore attacks. They possess so-called constitutive defences, present continuously, but they will also mount so-called induced defences after being attacked by herbivores (Karban and Carey 1984; Dicke and Sabelis 1988; Turlings et al. 1990; Karban and Baldwin 1997). Both constitutive and induced defences can have direct effects on the herbivores, e.g. by way of production of anti-nutritive or toxic compounds (Duffey and Stout 1996; Howe and Jander 2008; Karban and Baldwin 1997; Ohgushi 2005), or have indirect effects on herbivores through interactions of the plant with the natural enemies of the herbivores (Price et al. 1980; Dicke and Sabelis 1988; Turlings et al. 1990; Heil 2008; Kant et al. 2015). When induced, these defences can have negative effects on later-arriving conspecific and heterospecific herbivores (Karban and Carey 1984; Karban and Baldwin 1997; Poelman et al. 2008), but they can also have positive effects (Karban and Baldwin 1997; Bruessow et al. 2010; Peñaflor et al. 2019).
Some herbivores do not induce plant defences but suppress them (Musser et al. 2002; Zarate et al. 2007; Lawrence et al. 2008; Sarmento et al. 2011a; Alba et al. 2012; Takemoto et al. 2013; Kant et al. 2015; Godinho et al. 2016; Villarroel et al. 2016; De Lange et al. 2020). For example, the phytophagous tomato red spider mite Tetranychus evansi suppresses direct plant defences: unlike most herbivores, including the closely related spider mite Tetranychus urticae, T. evansi causes a reduction of defence compounds to levels below those found in undamaged tomato plants (Sarmento et al. 2011a; Alba et al. 2015; de Oliveira et al. 2016). As a result, conspecifics and other herbivores such as the spider mites T. urticae and Tetranychus ludeni have increased performance on plants in which defences are suppressed by T. evansi (Sarmento et al. 2011a; de Oliveira et al. 2016, 2019; Schimmel et al. 2017a). Tetranychus evansi appears to have strategies to prevent these competitors from taking advantage of the suppressed plant defences by covering the plant surface with a dense web that prevents other herbivores from feeding on the plant (Ferragut and Escudero 1999; Sarmento et al. 2011b; Sato et al. 2016; Blaazer et al. 2018; Clemente et al. 2018). Spider mites from the family Tetranychidae are well known for covering their colonies on plants with silken web (Gerson 1985). This web can protect against natural enemies and prevent the colonization of the plant by competitors (Gerson 1985; Sabelis and Bakker 1992).
Tetranychus evansi originates from South America, but spread to Africa and Mediterranean Europe, where it now poses a serious threat to tomato crops (Navajas et al. 2013). Although the invasion of T. evansi into Europe is suspected to have changed the composition of the phytophagous mite communities on various plant species (Ferragut et al. 2013), it is not known how the suppression of direct plant defences by T. evansi and the production of web affect other herbivores that attack the same plants, except for the spider mites T. urticae and T. ludeni (Sarmento et al. 2011a; Alba et al. 2015; Schimmel et al. 2017b; Godinho et al. 2020). Among the herbivores that can co-occur with T. evansi on tomato plants, the whitefly Bemisia tabaci stands out as being an important pest of tomato plants worldwide (Oliveira et al. 2001). Interactions among B. tabaci and other herbivores on a shared host plant have been studied to some extent (Inbar et al. 1999; Mayer et al. 2002; Inbar and Gerling 2008; Nombela et al. 2009; Zhang et al. 2011, 2014; Tan et al. 2014), but there is no study on the performance and preference of B. tabaci for plants infested with T. evansi. We therefore tested to what extent plant-mediated facilitation (via defence suppression by T. evansi) and interference (via web production by T. evansi) determine host plant selection and performance of B. tabaci on a shared host. To tease apart the effects of induced or suppressed plant defences from those of the spider-mite web, we included a line of T. urticae which also produces web but induces plant defence responses.
Material and methods
Rearing methods
Tomato plants (Solanum lycopersicum, variety Santa Clara I-5300) were grown in pots (2 L) using a commercial substrate based on pinus bark, peat and expanded vermiculite, enriched with macro- and micronutrients (Tropstrato HT, Vida Verde, Mogi Mirim, São Paulo, Brazil). The plants were kept inside a greenhouse (25 ± 5 °C), where they were fertilized with NPK (4-14-8) plus superphosphate and watered as needed. All experiments were performed with tomato plants with six completely developed leaves, with each leaf having 5–7 leaflets. Both T. urticae and T. evansi were originally collected from tomato plants on the campus of the Federal University of Viçosa (Sarmento et al. 2011b). This strain of T. urticae induces both salicylic acid and jasmonic acid-related defences in tomato, whereas T. evansi suppresses both (Sarmento et al. 2011a). They were reared on detached tomato leaves, with the petiole of the tomato leaves inserted in a plastic tube filled with water to maintain leaf turgor. The tubes with the tomato leaves were kept in plastic trays inside a larger tray filled with water to prevent mite escapes and invasion of other arthropods. Clean tomato leaves were added to the inner tray as needed, allowing the mites to move from old to new leaves. Old leaves were removed from the cultures and the trays were cleaned once every two weeks. The cultures were kept in a room at 25 ± 2 °C, 80 ± 10% relative humidity and 12 h of light.
The whitefly Bemisia tabaci was one of the most frequently captured herbivores on clean plants and plants with T. evansi and T. urticae (Dias 2017). It was also collected on the campus of the Federal University of Viçosa and was reared on entire tomato plants of the same variety as above. The plants were maintained inside a cage (60 × 60 × 120 cm) with fine mesh (70 µm). A clean tomato plant with at least four completely developed leaves was added to the culture every two weeks. The oldest plants were removed from the cage once a month. The cage with the culture was kept in an open area under a roof under natural climate conditions (average temperature 20.5 °C; average minimum temperature 16.3 °C; average maximum temperature 27.4 °C; average humidity 76.7%).
Induction and suppression of plant defences
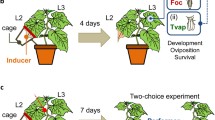
Plants were either infested with spider mites or were left clean. To infest the plants with spider mites, all leaflets of all six leaves of the tomato plants were infested with 40 adult female spider mites, either T. urticae or T. evansi, collected randomly from the cultures. Subsequently, they were incubated with the spider mites inside a greenhouse (25 ± 5 °C) in separate cages on separate benches to avoid cross contamination for four days, which is sufficient time for the mites to induce or suppress plant defences (de Oliveira et al. 2016). To assess the effects of the web produced by spider mites, the web was carefully removed from some plants infested with spider mites with a soft brush three days after infestation, which also resulted in the removal of a few spider mites and possibly trichomes. This resulted in five plant treatments: (1) plants infested with T. evansi with their web; (2) plants infested with T. evansi without web; (3) plants infested with T. urticae with web; (4) plants infested by T. urticae without web; and (5) uninfested plants (Control). We used one plant of the treatments 1, 3, and 5 for assessing the effects of plant treatments (see next section) and one plant of each treatment for host plant choice (see subsequent section).
Validation of plant defence induction and suppression
To ensure that plant infestations with T. evansi and T. urticae indeed affected plant defences in the predicted manner (Alba et al. 2015; Schimmel et al. 2017a; Knegt et al. 2020), we measured the performance of T. evansi on leaf discs from plants infested with T. evansi, plants infested with T. urticae and clean plants as above. This was done at various times throughout this investigation, always at the same time as replicates of the experiments on host plant choice and whitefly performance described below and always with the same batch of plants, thus verifying that plant defences were induced or suppress throughout all experiments. Four to five leaf discs (Ø = 1.5 cm) were cut from the fifth and sixth leaves of plants infested with T. evansi or T. urticae, all with web present, and from clean plants, with 5 plants per treatment. The discs were kept in Petri dishes on water-saturated cotton wool to keep them fresh. Adult spider mites, nymphs, eggs and their web were removed from the discs with a soft brush. The plants from which these discs were taken were discarded. Subsequently, the discs were used to measure the oviposition rate of T. evansi with a method described by Sarmento et al. (2011a). Oviposition by spider mites is a reasonable stand-in measure for fitness in local populations on a plant (Sabelis 1991), and we previously showed that it is correlated with induction and suppression of plant defences (Kant et al. 2004; Sarmento et al. 2011a). In short, a female of T. evansi, aged 12 days since egg hatching, was released on each leaf disc, incubated at 25 (± 2)°C and her eggs were counted daily during three days. The first day was excluded from further analysis to avoid effects of previous diet (Sabelis 1990). Oviposition rates of day two and three were first averaged per plant, resulting in 5 averages per treatment, and these averages were analysed with a generalized linear model (GLM) with plant treatment as factor and a Gaussian error distribution (identity link). Contrasts among treatments were assessed using least-squares means with a Tukey correction for multiple comparisons (function lsm of the lsmean package, Lenth 2016). All statistical analyses were done with R (R Core Team 2019).
Host plant choice
Whiteflies were offered a choice among plants with the five different treatments as outlined above. After the spider mite web was removed from plants of the respective treatments (2 and 4, above), one plant of each treatment (so five plants in total) was placed inside a cage consisting of a frame (160 × 160 × 120 cm) covered with a fine mesh and were allowed to acclimatize until the next day. The five plants were placed in a pentagon with a radius of 80 cm, and a distance between neighbouring plants of 94 cm. The cage could be opened on all sides, which facilitated access to the plant and was positioned outdoors under natural conditions of light, humidity and temperature, because tomato plants in the region are usually grown outdoors. The average temperature during the experiment was 17.9 °C (s.e. 0.6), relative humidity was 74% (s.e. 1.81), and it did not rain (Universidade Federal de Viçosa—UFV 2022). The area was delimited at one side by a building and by native vegetation and cultivated plants at the other sides. This experiment was replicated five times through time, each with different plants and whiteflies.
On the day before the experiment at 18 h, 100 female whiteflies were collected individually from the culture in a pipette tip. Their gender was checked under a binocular microscope and males were discarded. Each tip was closed with a metal thumbtack (Meng et al. 2006) and stored in a pipette tip box. The morning after the plants had been placed in the cage and the whiteflies had been collected, the pipette tip box was placed in the centre of the arena. A magnetic strip was used to simultaneously open all pipette tips to release the whiteflies with minimal disturbance (Meng et al. 2006) at 9 h. Previous tests showed that almost all the whiteflies left the tips to find a host plant after four hours (CR Dias, personal observation). Therefore, we counted the whiteflies that arrived on plants by careful visual inspection of the under- and upper-side of each leaf after four and 24 h. The whiteflies observed on the plants were not recaptured after 4 h in order to observe whether their distribution over the host plants changed during the experiment. Thus, the second observation included individuals that remained on plants since the previous evaluation, arrived later on the plants, or changed plants between the assessments. The log-transformed numbers of whiteflies observed on plants after four and 24 h were analysed using a linear mixed effects model (function lme of the nlme package, Bates et al. 2015) with treatment, time of evaluation (four and 24 h after release) and their interaction as fixed factors, and replicate and position of the plants as non-nested random factors. The interaction of plant treatment and time was not significant, indicating that the distribution of whiteflies over the plants did not change significantly between 4 and 24 h. Because whiteflies were not removed when counting them after 4 h, the numbers of whiteflies on the plants after 4 h and 24 h were not independent, and we therefore only present the analysis of numbers of whiteflies on the plants after 24 h. Contrasts among treatments were assessed through model simplification by combining factor levels and using the anova function of R to compare models. Plants of different treatments were in different positions in each replicate, so all treatments occupied all positions to correct for possible directionality of external cues such as direct sunlight or shade.
Because the whiteflies mainly chose plants without spider-mite web in the previous experiment, we could not determine whether there was a difference in preference between plants infested with T. evansi or T. urticae plus their web. We therefore gave whiteflies a choice between four plants with two treatments in a subsequent experiment; two plants infested with T. evansi, and two plants infested with T. urticae, in both treatments the web was left on the plants. These plants were placed equidistantly in a square (L = 80 cm) inside the cage (as above) and the experiment was replicated four times with different sets of plants and whiteflies. Again, care was taken that plants of each treatment occupied a different position in each replicate to control any unforeseen directionality in the searching behaviour of the whiteflies (Janssen 1999). This experiment was repeated four times, each with different plants and whiteflies (average temp. 22.1 °C, s.e. 0.71, rel. humidity 72.8%, s.e. 4.6). There was some light rain during one of the replicates.
Whitefly survival and development
We conducted three experiments to assess how the infestation of plants with T. evansi or T. urticae and the presence of their web affected the juvenile development and survival of whiteflies. We assessed whitefly performance on tomato plants that had received one of three treatments: (1) plants infested with T. evansi; (2) plants infested with T. urticae; and (3) clean plants. In the first experiment, plants were infested with 40 adult female spider mites per leaflet as above. This resulted in heavy spider mite damage during the experiment, hence, performance of the whiteflies was not only affected indirectly through plant defences, but also through competition for plant resources of whiteflies with spider mites and through the web produced by spider mites. Therefore, we used a lower infestation of ten adult female spider mites per leaflet in the second and third experiments. In all three experiments, a circle (Ø = 1.5 cm) of non-drying glue (BioStop Cola®, Biocontrole SA, São Paulo) was painted on the abaxial surface of each leaflet of the fifth and sixth leaves of each plant on the fourth day after plant infestation, taking care that mites and their web were included in this circle. Subsequently, one whitefly crawler (one-day old mobile stage) was released in the centre of each glue circle so that its movements were confined and it could be found back easily on subsequent days. The plants with the mites and the whiteflies were incubated in a climate-controlled room as above (25 ± 2 °C, 80 ± 10% relative humidity and 12 h of light).
In the third experiment, plants were infested with ten spider mites per leaflet as above, but spider mite web was excluded from the leaf surface on which the whiteflies developed. This was done by painting the glue circle on the abaxial surface of each leaflet before infestation with spider mites. The spider mites released on the plants could thus walk, lay eggs and produce web on the whole plant except inside the circle. Hence, the whitefly crawler that was placed in the centre of these circles four days after plant infestation could develop on a plant on which spider mites were present, but without physical contact with them or their web. This served to measure the effects of induced or suppressed plant defences on whitefly development and survival without the effects of spider-mite web.
Replicates consisted of a plant with ten arenas (for plants infested with 40 spider mites and ten spider mites without web) or five arenas (for plants infested with ten spider mites with web) and we evaluated 4 plants per treatment. The plants were checked 24 h later to verify that crawlers were still inside the glue circle to eliminate losses of whiteflies due to manipulation by the experimenters. The juveniles were observed daily until all of them reached adulthood or died. In treatments with web, we scored the number of dead adults that got trapped in the spider mite web. The numbers of dead juveniles plus adults that were caught in the web were first summed per plant and proportions of these totals were then analysed with a GLM with a quasi-binomial error distribution (logit link). The mean developmental time of whitefly juveniles was first averaged per plant, and these averages were compared among plant treatments within each experiment with a GLM with a Gaussian error distribution (identity link). Contrasts among treatments were assessed as above. Because the three experiments were carried out in different periods for logistical reasons, the results could not be compared among experiments, but the effects of spider mites and their web were assessed through comparison of the performance on infested plants with or without web with that on clean plants.
Results
Validation of plant defence induction and suppression
The oviposition rate of T. evansi differed significantly with plant treatment (Fig. 1, GLM: F2,12 = 135.5, P < 0.0001). Compared to oviposition on clean plants, oviposition was higher on leaf discs of plants previously attacked by T. evansi and was lower on leaf discs from plants previously attacked by T. urticae (Fig. 1), as was found before for plants with induced and suppressed defences (Kant et al. 2004; Sarmento et al. 2011a). Hence, the experimental manipulation of plant defences through the previous infestation of the plants by the defence-inducing strain of T. urticae and by the defence-suppressing T. evansi was successful.
Host plant choice
In the experiment where B. tabaci could choose among clean plants, plants infested with T. evansi or with T. urticae, either with or without their web, numbers of whiteflies recaptured on plants differed significantly among treatments (Fig. 2a, LME: LR = 13.0, d.f. = 4, P = 0.012). The numbers of whiteflies on clean plants and plants infested with T. evansi without web were higher than on the plants with web of either T. evansi or T. urticae and on plants with T. urticae but without web (Fig. 2a).
Average (± SE) number of B. tabaci recaptured on plants that were treated in several ways. a Clean plants, plants infested with T. evansi without its web (T. evansi–web), plants infested with T. urticae without web (T. urticae–web), plants infested with T. evansi with web (T. evansi + web) and plants infested with T. urticae with web (T. urticae + web), after 24 h. b Plants infested with T. evansi or with T. urticae, both with web. Different letters above bars indicate significant differences in the average number of whiteflies observed on each plant (contrast after an LME, P < 0.05)
Because the numbers of whiteflies recaptured on plants with T. urticae or T. evansi with their web was low (Fig. 2a), it was not possible to discern whether whiteflies had a preference for either of the two. We therefore offered whiteflies a choice between plants infested with T. urticae or with T. evansi, both with web, but there was no significant difference in the numbers of whiteflies on plants with T. urticae or T. evansi (Fig. 2b, LME, LR = 1.64, d.f. = 1, P = 0.20).
Whitefly survival and development
When plants were infested with 40 spider mites per leaflet and covered by their web, mortality of B. tabaci differed significantly among treatments (Fig. 3a first bars, GLM, F2,9 = 31.2, P < 0.001). Mortality was significantly lower on clean plants than on plants infested either with T. urticae or T. evansi (Fig. 3a). Several adults that emerged from infested plants were found trapped in the spider mite web (Fig. 3a). The mean developmental time of B. tabaci was also significantly affected by the treatments (Fig. 3b, first group of bars, GLM: F2,8 = 7.36, P = 0.015), with whitefly juveniles developing slower into adults on plants infested with spider mites than on clean plants (Fig. 3b). None of the 40 whitefly immatures per treatment went missing.
a Proportion mortality (± 95% CI) of B. tabaci, until dispersing adult. Confidence intervals were calculated with the Jeffreys method (package DescTools in R, Signorell 2020). b Mean developmental time (± SE, days) of B. tabaci juveniles from first nymphal stage until adulthood. White bars: uninfested plants; light grey bars: plants infested with T. evansi; dark grey bars: plants infested with T. urticae. Groups of bars correspond to levels of infestation of plants with T. evansi or T. urticae (numbers of spider mites per leaflet) and the presence/absence of spider mite web. Different letters above bars indicate significant differences within each group of bars (contrast after GLM, P < 0.05), hatched parts of the bars in a indicate proportion mortality due to adults being caught in the spider mite web
When plants were infested with 10 spider mites per leaflet and covered with web, mortality again differed significantly among treatments (Fig. 3a, second groups of bars, GLM, F2,9 = 11.0, P = 0.004), and was again significantly lower on clean plants than on plants infested either with T. urticae or T. evansi (Fig. 3a). Again, adults that emerged from infested plants were found trapped in the spider mite web (Fig. 3a). The mean juvenile developmental period also varied significantly with plant treatment (Fig. 3b second group of bars, GLM, F2,9 = 5.14, P = 0.03), with development being faster on plants infested with T. urticae than on plants infested with T. evansi, whereas development on clean plants did not differ from that with both other treatments (Fig. 3b). During the experiment, 2 juvenile whiteflies out of 20 went missing per treatment.
When plants were infested with 10 spider mites per leaflet and the whiteflies were not covered with web, mortality was much lower than in the treatments with web (Fig. 3a), but again differed significantly among treatments (Fig. 3a third group of bars, GLM: F2,9 = 5.81, P = 0.024). Mortality was higher on plants infested with T. urticae than on clean plants and plants infested with T. evansi (Fig. 3a). The mean developmental time of B. tabaci juveniles did not differ significantly among treatments (Fig. 3b third group of bars, GLM, F2,9 = 0.30, P = 0.75). One juvenile whitefly out of 40 went missing per treatment.
Discussion
Herbivores often avoid plants with other herbivores that induce plant defences, causing reduced performance of the later-arriving herbivores (Karban and Carey 1984; Karban and Baldwin 1997; Pallini et al. 1997; Poelman et al. 2008; Tan and Liu 2014). In contrast, herbivores may be attracted to plants with other herbivores because the interactions of the plant with these other herbivores result in lower direct or indirect defence against herbivores arriving later (Shiojiri et al. 2002; Rodriguez-Saona et al. 2005). We show here that both plant-mediated effects via induction and suppression of defences by spider mites and effects via spider-mite web affected the host plant choice and performance of the whitefly B. tabaci. Without web, this whitefly preferred clean plants and plants attacked by T. evansi over plants attacked by T. urticae (Fig. 2), the latter having induced defences (Fig. 1). Whiteflies settled less on plants covered with web of either of the two spider mites. This shows that both spider-mite induced plant defences and spider mite web affected whitefly host plant choice. Below, we discuss the effects of plant defences, the effects of spider-mite web and their combined effects on host plant choice and performance of the whiteflies.
To evaluate the effects of plant defences, we compared whitefly preference and performance on plants without web with those on clean plants. The whiteflies settled preferentially on clean plants and plants attacked by T. evansi without web (Fig. 2). This coincided with the higher juvenile survival on clean plants and plants attacked by T. evansi than on plants attacked by T. urticae (Fig. 3a, without web), suggesting that differences in host plant preference were determined by differences in plant defences. Juvenile development did not differ among plants with the three different treatments (Fig. 3b, without web), suggesting that development is not affected by differences in plant defences. Tomato plant defences induced by T. urticae thus negatively affected plant preference and performance of B. tabaci, which is consistent with earlier studies showing that such defences negatively affect conspecifics and several other spider mites (Sarmento et al. 2011a; de Oliveira et al. 2016; Godinho et al. 2016). The performance and preference of B. tabaci on plants with defences suppressed by T. evansi did not differ significantly from that on clean plants (Fig. 3, without web), suggesting that this herbivore does not benefit from the defence suppression. Earlier studies showed increased performance of several species of spider mites on plants with defences suppressed by T. evansi in the absence of its web (Sarmento et al. 2011a; Godinho et al. 2016; Oliveira et al. 2016), hence, our results show that not all herbivores benefit from this defence suppression. Bemisia tabaci is known to suppress defences of the jasmonic acid (JA) pathway, probably through induction of antagonistic salicylic acid (SA) defences (Zarate et al. 2007; Walling 2009; Zhang et al. 2009; Xu et al. 2019). Because the strain of T. urticae used here induces JA- and SA-related defences in tomato (Sarmento et al. 2011a), apparently the whiteflies cannot suppress these induced defences. The down-regulation of defences by T. evansi could in theory benefit the whiteflies since they could reduce their efforts to suppress JA-related defences and thereby increase performance. For example, the performance of T. evansi on leaf tissue in which defences were suppressed by a previous infestation by conspecifics was higher than on leaf tissue where they had to suppress the defences themselves (Sarmento et al. 2011a, b). However, we did not observe B. tabaci to benefit from the presence of T. evansi, suggesting that the whiteflies constitutively suppress JA-defences or that our measurements were not sensitive enough to reveal the costs of suppression of JA-related defences.
Spider mites from the family Tetranychidae are known for their production of web (Gerson 1985) and this is thought to have several functions, including preventing predators and competitors from colonising the plant (Sabelis and Bakker 1992; Morimoto et al. 2006; Sabelis et al. 2009). Tetranychus evansi produces very dense web (Ferragut and Escudero 1999), much denser than does T. urticae, possibly to avoid boosting the performance of competitors through the suppressed plant defences (Sarmento et al. 2011b; Blaazer et al. 2018). Our results show that B. tabaci had lower preference for plants covered with spider-mite web than for plants without web and that juvenile survival of B. tabaci on these plants was lower than on clean plants or plants attacked by T. evansi but without web (Fig. 2, 3a). This suggests that the web indeed serves to protect plants from being invaded by competing heterospecific herbivores, confirming earlier results (Sarmento et al. 2011b; Blaazer et al. 2018). For T. evansi, this was further confirmed by the observation of increased web production when it perceived cues of competitors (Sarmento et al. 2011b).
Taken together, the combined effects of induced and suppressed plant defences and the web produced by spider mites serve to reduce competition of the mites with whiteflies. Tetranychus evansi suppresses plant defences, but B. tabaci does not seem to profit from this. Moreover, the web produced by T. evansi prevents the whiteflies to settle on plants with the mites. In the case of T. urticae, the whiteflies avoided plants with this spider mite. Although the presence of web of T. urticae seems to cause higher mortality of whiteflies than the induction of plant defences by this spider mite (Fig. 3a), the whiteflies did avoid plants with or without web of T. urticae to a similar extent (Fig. 2a). This suggests that the whiteflies may avoid these plants based on other cues than the presence of spider-mite web, perhaps using the plant volatiles induced by attacks of T. urticae (Kant et al. 2004).
The preference and performance of herbivores are essential components in host-range ecology and evolution, and herbivores are expected to select host plants based on their availability and suitability (Ward 1992; Scheirs et al. 2000). Many studies have shown that induced plant defences play an important role in this: later-arriving herbivores can be positively or negatively affected by changes in plant defences due to previous infestations and this can change host plant preference and herbivore performance (Karban and Carey 1984; Agrawal et al. 2000; Bruessow et al. 2010; Sarmento et al. 2011a; Alba et al. 2015; Kant et al. 2015). Here, we show that a combination of plant-mediated effects via defence induction or suppression and via the production of structural barriers by herbivores alters the attractiveness and suitability of host plants for competing herbivores. Thus, herbivores such as T. urticae can reduce competition with later-arriving herbivores by inducing plant defences, but these defences may also negatively affect their own performance (Kant et al. 2004). Alternatively, herbivores such as T. evansi increase their performance through suppression of plant defences, but this requires investing in other traits to prevent competitors from profiting from the suppressed defences.
Author contributions
CRD, RAS, MCACdS, MV, AP and AJ conceived and designed research. CRD, JM, AMGB, MCACdS and ACC conducted experiments. AJ, CRD and ACC analysed data. AJ, CRD, ACC and MRK wrote the manuscript. All authors read and approved the manuscript.
References
Agrawal AA, Karban R, Colfer RG (2000) How leaf domatia and induced plant resistance affect herbivores, natural enemies and plant performance. Oikos 89:70–80
Alba JM, Allmann S, Glas JJ, et al (2012) Induction and suppression of herbivore-induced indirect defenses. In G. Witzany, F. Baluska (eds) Biocommunication of plants. Signaling and Communication in Plants 14. Springer, pp 197–212
Alba JM, Schimmel BCJ, Glas JJ et al (2015) Spider mites suppress tomato defenses downstream of jasmonate and salicylate independently of hormonal crosstalk. New Phytol 205:828–840. https://doi.org/10.1111/nph.13075
Bates D, Mächler M, Bolker B, Walker S (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67:1–48. https://doi.org/10.18637/jss.v067.i01
Blaazer CJH, Villacis-Perez EA, Chafi R et al (2018) Why do herbivorous mites suppress plant defenses? Front Plant Sci 9:1057. https://doi.org/10.3389/fpls.2018.01057
Bruessow F, Gouhier-Darimont C, Buchala AJ et al (2010) Insect eggs suppress plant defence against chewing herbivores. Plant J 62:876–885. https://doi.org/10.1111/j.1365-313X.2010.04200.x
Clemente SH, Santos I, Ponce R et al (2018) Despite reproductive interference, the net outcome of reproductive interactions among spider mite species is not necessarily costly. Behav Ecol 29:321–327. https://doi.org/10.1093/beheco/arx161
De Lange ES, Laplanche D, Guo H et al (2020) Spodoptera frugiperda caterpillars suppress herbivore-induced volatile emissions in maize. J Chem Ecol 46:344–360. https://doi.org/10.1007/s10886-020-01153-x
de Oliveira EF, Pallini A, Janssen A (2016) Herbivores with similar feeding modes interact through the induction of different plant responses. Oecologia 180:1–10
de Oliveira EF, Pallini A, Janssen A (2019) Herbivore performance and plant defense after sequential attacks by inducing and suppressing herbivores. Insect Sci 26:108–118. https://doi.org/10.1111/1744-7917.12499
Dias CR (2017) Plant defences, spider mites and web affecting arthropod responses to tomato plants. Ph.D. thesis, Federal University of Viçosa
Dicke M, Sabelis MW (1988) How plants obtain predatory mites as bodyguards. Neth J Zool 38:148–165
Duffey SS, Stout MJ (1996) Antinutritive and toxic components of plant defense against insects. Arch Insect Biochem Physiol 32:3–37
Ferragut F, Escudero A (1999) Tetranychus evansi Baker & Pritchard (Acari, Tetranychidae), uma nueava araña roja en los cultivos hortícolas españoles. Bol San Veg Plagas 25:157–164
Ferragut F, Garzón-Luque E, Pekas A (2013) The invasive spider mite Tetranychus evansi (Acari: Tetranychidae) alters community composition and host-plant use of native relatives. Exp Appl Acarol 60:321–341
Gerson U (1985) Webbing. In: Helle W, Sabelis MW (eds) Spider mites: their biology, natural enemies and control. Elsevier, Amsterdam, pp 223–232
Godinho DP, Janssen A, Dias T et al (2016) Down-regulation of plant defence in a resident spider mite species and its effect upon con-and heterospecifics. Oecologia 180:161–167
Godinho DP, Janssen A, Li D et al (2020) The distribution of herbivores between leaves matches their performance only in the absence of competitors. Ecol Evol 10:8405–8415. https://doi.org/10.1002/ece3.6547
Heil M (2008) Indirect defence via tritrophic interactions. New Phytol 178:41–61
Howe GA, Jander G (2008) Plant immunity to insect herbivores. Annu Rev Plant Biol 59:41–66
Inbar M, Gerling D (2008) Plant-mediated interactions between whiteflies, herbivores, and natural enemies. Annu Rev Entomol 53:431–448
Inbar M, Doostdar H, Mayer RT (1999) Effects of sessile whitefly nymphs (Homoptera: Aleyrodidae) on leaf-chewing larvae (Lepidoptera: Noctuidae). Environ Entomol 28:353–357. https://doi.org/10.1093/ee/28.3.353
Janssen A (1999) Plants with spider-mite prey attract more predatory mites than clean plants under greenhouse conditions. Entomol Exp Appl 90:191–198
Kant MR, Ament K, Sabelis MW et al (2004) Differential timing of spider mite-induced direct and indirect defenses in tomato plants. Plant Physiol 135:483–495
Kant MR, Jonckheere W, Knegt B et al (2015) Mechanisms and ecological consequences of plant defence induction and suppression in herbivore communities. Ann Bot 115:1015–1051. https://doi.org/10.1093/aob/mcv054
Karban R, Carey JR (1984) Induced resistance of cotton seedlings to mites. Science 225:53–54
Karban R, Baldwin IT (1997) Induced responses to herbivory. University of Chicago Press, Chicago
Knegt B, Meijer TT, Kant MR et al (2020) Tetranychus evansi spider mite populations suppress tomato defenses to varying degrees. Ecol Evol 10:4375–4390. https://doi.org/10.1002/ece3.6204
Lawrence SD, Novak NG, Ju CJT, Cooke JEK (2008) Potato, Solanum tuberosum, defense against Colorado potato beetle, Leptinotarsa decemlineata (Say): Microarray gene expression profiling of potato by Colorado potato beetle regurgitant treatment of wounded leaves. J Chem Ecol 34:1013–1025
Lenth R (2016) Least-squares means: the R package lsmeans. J Stat Softw 69:1–33. https://doi.org/10.18637/jss.v069.i01
Mayer RT, Inbar M, McKenzie CL et al (2002) Multitrophic interactions of the silverleaf whitefly, host plants, competing herbivores, and phytopathogens. Arch Insect Biochem Physiol 51:151–169
Meng R-X, Janssen A, Nomikou M et al (2006) Previous and present diets of mite predators affect antipredator behaviour of whitefly prey. Exp Appl Acarol 38:113–124
Morimoto K, Furuichi H, Yano S, Osakabe MH (2006) Web-mediated interspecific competition among spider mites. J Econ Entomol 99:678–684
Musser RO, Hum-Musser SM, Eichenseer H et al (2002) Herbivory: caterpillar saliva beats plant defences—a new weapon emerges in the evolutionary arms race between plants and herbivores. Nature 416:599–600
Navajas M, de Moraes GJ, Auger P, Migeon A (2013) Review of the invasion of Tetranychus evansi: biology, colonization pathways, potential expansion and prospects for biological control. Exp Appl Acarol 59:43–65. https://doi.org/10.1007/s10493-012-9590-5
Nombela G, Garzo E, Duque M, Muñiz M (2009) Preinfestations of tomato plants by whiteflies (Bemisia tabaci) or aphids (Macrosiphum euphorbiae) induce variable resistance or susceptibility responses. Bull Entomol Res 99:183–191
Ohgushi T (2005) Indirect interaction webs: herbivore-induced effects through trait change in plants. Ann Rev Ecol Evol Syst 81–105
Oliveira MRV, Henneberry TJ, Anderson P (2001) History, current status, and collaborative research projects for Bemisia tabaci. Crop Prot 20:709–723
Pallini A, Janssen A, Sabelis MW (1997) Odour-mediated responses of phytophagous mites to conspecific and heterospecific competitors. Oecologia 110:179–185
Peñaflor MFGV, Andrade FM, Sales L et al (2019) Interactions between white mealybugs and red spider mites sequentially colonizing coffee plants. J Appl Entomol 143:957–963. https://doi.org/10.1111/jen.12683
Poelman EH, Broekgaarden C, Van Loon JJA, Dicke M (2008) Early season herbivore differentially affects plant defence responses to subsequently colonizing herbivores and their abundance in the field. Mol Ecol 17:3352–3365
Price PW, Bouton CE, Gross P et al (1980) Interactions among three trophic levels: Influence of plants on interactions between insect herbivores and natural enemies. Annu Rev Ecol Syst 11:41–65
R Core Team (2019) R: A language and environment for statistical computing. Version 3.6.0. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org
Rodriguez-Saona C, Chalmers JA, Raj S, Thaler JS (2005) Induced plant responses to multiple damagers: differential effects on an herbivore and its parasitoid. Oecologia 143:566–577
Sabelis MW (1990) How to analyze prey preference when prey density varies? A new method to discriminate between effects of gut fullness and prey type composition. Oecologia 82:289–298
Sabelis MW (1991) Life-history evolution of spider mites. In: Schuster R, Murphy PW (eds) The Acari. Reproduction, development and life-history strategies. Chapman & Hall, London, pp 23–49
Sabelis MW, Bakker FM (1992) How predatory mites cope with the web of their Tetranychid prey—a functional view on dorsal chaetotaxy in the Phytoseiidae. Exp Appl Acarol 16:203–225
Sabelis M, Hanna R, Onzo A et al (2009) Multiple predators, intraguild interactions and biological control of a single spider mite species. IOBCWPRS Bull 50:83–94
Sarmento RA, Lemos F, Bleeker PM et al (2011a) A herbivore that manipulates plant defence. Ecol Lett 14:229–236
Sarmento RA, Lemos F, Dias CR et al (2011b) A herbivorous mite down-regulates plant defence and produces web to exclude competitors. PLoS ONE 6(1–7):e23757
Sato Y, Alba JM, Egas M, Sabelis MW (2016) The role of web sharing, species recognition and host-plant defence in interspecific competition between two herbivorous mite species. Exp Appl Acarol 70:261–274. https://doi.org/10.1007/s10493-016-0079-5
Scheirs J, De Bruyn L, Verhagen R (2000) Optimization of adult performance determines host choice in a grass miner. Proc R Soc Lond B 267:2065–2069
Schimmel BC, Ataide L, Chafi R et al (2017a) Overcompensation of herbivore reproduction through hyper-suppression of plant defenses in response to competition. New Phytol 214:1688–1701
Schimmel BC, Ataide LM, Kant MR (2017b) Spatiotemporal heterogeneity of tomato induced defense responses affects spider mite performance and behavior. Plant Signal Behav 12:1688–1701
Shiojiri K, Takabayashi J, Yano S, Takafuji A (2002) Oviposition preferences of herbivores are affected by tritrophic interaction webs. Ecol Lett 5:186–192
Signorell A (2020) DescTools: tools for descriptive statistics. https://cran.r-project.org/package=DescTools
Takemoto H, Uefune M, Ozawa R et al (2013) Previous infestation of pea aphids Acyrthosiphon pisum on broad bean plants resulted in the increased performance of conspecific nymphs on the plants. J Plant Interact 8:370–374
Tan X-L, Liu T-X (2014) Aphid-induced plant volatiles affect the attractiveness of tomato plants to Bemisia tabaci and associated natural enemies. Entomol Exp Appl 151:259–269
Tan X-L, Wang S, Ridsdill-Smith J, Liu T-X (2014) Direct and indirect impacts of infestation of tomato plant by Myzus persicae (Hemiptera: Aphididae) on Bemisia tabaci (Hemiptera: Aleyrodidae). PLoS ONE 9:1–9. https://doi.org/10.1371/journal.pone.0094310
Turlings T, Tumlinson J, Lewis W (1990) Exploitation of herbivore-induced plant odors by host-seeking parasitic wasps. Science 250:1251–1253
Universidade Federal de Viçosa—UFV (2022) Boletim meteorológico. Departamento de Engenharia Agricola, Estação Climatológica Principal de Viçosa
Villarroel CA, Jonckheere W, Alba JM et al (2016) Salivary proteins of spider mites suppress defenses in Nicotiana benthamiana and promote mite reproduction. Plant J 86:119–131
Walling LL (2009) Adaptive defense responses to pathogens and insects. In: Advances in Botanical Research, vol 51. pp 551–612
Ward SA (1992) Assessing functional explanations of host-specificity. Am Nat 139:883–891
Xu H-X, Qian L-X, Wang X-W et al (2019) A salivary effector enables whitefly to feed on host plants by eliciting salicylic acid-signaling pathway. Proc Natl Acad Sci 116:490. https://doi.org/10.1073/pnas.1714990116
Zarate SI, Kempema LA, Walling LL (2007) Silverleaf whitefly induces salicylic acid defenses and suppresses effectual jasmonic acid defenses. Plant Physiol 143:866–875
Zhang G-F, Li D-C, Liu T-X et al (2011) Interspecific interactions between Bemisia tabaci Biotype B and Trialeurodes vaporariorum (Hemiptera: Aleyrodidae). Environ Entomol 40:140–150. https://doi.org/10.1603/EN10135
Zhang PJ, Zheng SJ, van Loon JJA et al (2009) Whiteflies interfere with indirect plant defense against spider mites in Lima bean. Proc Natl Acad Sci USA 106:21202–21207. https://doi.org/10.1073/pnas.0907890106
Zhang G-F, Lövei GL, Hu M, Wan F-H (2014) Asymmetric consequences of host plant occupation on the competition between the whiteflies Bemisia tabaci cryptic species MEAM1 and Trialeurodes vaporariorum (Hemiptera: Aleyrodidae). Pest Manag Sci 70:1797–1807
Acknowledgements
We thank colleagues from the Laboratory of Acarology of the Federal University of Viçosa for support and constructive comments. The constructive comments of three anonymous reviewers resulted in substantial improvement of the ms.
Funding
CRD was supported by the Foundation for Research Support of the State of Minas Gerais (FAPEMIG) and Coordination for higher Education Staff Development (CAPES, Doctoral Exchange Program). ACC was supported by the National Council for Scientific and Technological Development (CNPq), AMGB by CAPES and AP by CNPq, FAPEMIG and CAPES. MRK was supported by Horizon 2020—Research and Innovation Framework Program (773902-SuperPests) and the Dutch Research Council NWO (ALWOP.283) RAS received a scholarship from the National Council of Technological and Scientific Development, Brazil (CNPq) (Produtividade em Pesquisa-Project: 306652/2018-8). MCACS received a scholarship from Tocantins Research Support Foundation (FAPT). The Coordination of Improvement of Higher Education Personnel (CAPES) provided financial support (PROCAD 2013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest that are relevant to the content of this article.
Ethics approval
Plants and insects were used in this study. All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. This article does not contain any studies with human participants performed by any of the authors.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Communicated by Cesar Rodriguez-Saona.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Dias, C.R., Cardoso, A.C., Kant, M.R. et al. Plant defences and spider-mite web affect host plant choice and performance of the whitefly Bemisia tabaci. J Pest Sci 96, 499–508 (2023). https://doi.org/10.1007/s10340-022-01516-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10340-022-01516-1