Abstract
The mechanisms by which non-native species impact island-endemic populations are varied and often convoluted. For effective invertebrate conservation where capacity is limited, it is vital to understand both direct and indirect species interactions. Those interactions define specific threats and therefore highlight possible solutions. We quantified several of the non-native species threats faced by an endemic scaly cricket, Discophallus ascension (family: Mogoplistidae), found on Ascension Island’s naturally barren coastlines. We hypothesized that encroaching non-native Mexican thorn Prosopis juliflora (1) directly reduced microclimate suitability, (2) indirectly increased resource competition with non-native species, and (3) indirectly increased non-native predation. Our aim was to assess how rapidly spreading Mexican thorn might reduce endemic cricket populations. Lab trials first confirmed the temperature and diet preferences of D. ascension. Mexican thorn significantly increased ground-level temperature but not above the estimated optimum for D. ascension within key habitat. Observed interspecific resource competition was significantly greater in habitat degraded by Mexican thorn and was driven by non-native ants, house crickets Gryllodes sigillatus and cockroaches Periplaneta americana. Recorded cricket predation was almost entirely by non-native rats Rattus rattus in barren habitat but primarily by ants in Mexican thorn habitat. Threats to D. ascension were therefore modulated by invasive vegetation, but driven by non-native insects and rats at all sites. We conclude that removal of vegetation alone is likely to reduce resource competition but not predation. Managing multiple invasive species is likely necessary for successful species conservation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1. Introduction
Oceanic islands accommodate a disproportionately high number of endemic species (Kier et al. 2009). Despite their importance to global biodiversity, these fragile ecosystems are also subject to disproportionate degradation (Fernández-Palacios et al. 2021). Widespread introduction of non-native plants and animals are closely linked with declines in native populations (Tye et al. 2018) but quantitative analysis is essential in identifying the specific drivers behind such responses (Gurevitch and Padilla 2004). Island climates and habitat structure are, separately, changing rapidly (Russell and Kueffer 2019; Veron et al. 2019). The interactions between non-native vegetation encroachment, animal introductions and microclimate, and resulting impacts on endemic invertebrates, are poorly understood. Such knowledge is crucial in conservation planning on island territories where local management capacity and resources are often limited (Gray et al. 2019).
Ascension is an isolated volcanic island in the South Atlantic which has been subject to a very high number of species introductions (Ashmole and Ashmole 2000; Sharp and Tawatao 2023). It is geologically young (Jicha et al. 2013) with biogeography and simple native ecosystems that lend themselves well to globally applicable studies on invasion biology (Sharp and Tawatao 2023; Sherwood and Sharp 2023; Sherwood et al. 2023). The coastlines are comprised of mainly barren lava flows which accommodate numerous endemic invertebrate species (Duffey 1964; Ashmole and Ashmole 1997). Multiple native invertebrates on Ascension face apparent threats and even extirpation resulting from non-native species encroachment (Ashmole and Ashmole 2000; Wilkins, Knight and Gray 2019).
The coastal lava flows are subject to invasion by multiple species of global significance. Most prominent among those species is the dryland specialist Mexican thorn Prosopis juliflora. This woody shrub species originates from Central/South America and the Caribbean, and was initially introduced near one of Ascension’s settlements to reduce soil erosion in the 1960’s (AIGCFD 2015). It has since spread rapidly and is directly responsible for widespread modification of habitat structure, forming thick carpets and bushes over otherwise barren terrain (Lambdon and Darlow 2008; AIGCFD 2015). Mexican thorn invasion is prevalent in multiple countries and regions, where it is difficult and costly to remove (Shackleton et al. 2014; Dakhil et al. 2021). On Ascension, Mexican thorn is continually removed from most areas of coastline it has already reached. However, the shrub is expected to overwhelm local capacity and inundate coastline around the island in coming decades.
Landscape invasion by such non-native vegetation can have various detrimental impacts on native ecosystems (Renteria et al. 2012), and in particular invertebrates (Knapp et al. 2019). Non-native plants have been shown to modify habitat structure (Pyšek et al. 2012) and alter the local microclimate (Garcia and Clusella-Trullas 2019). Invertebrate distributions are shaped by temperature (Lachenicht et al. 2010; Cook and Talley 2014) and some studies have directly linked declines in native invertebrate diversity to change in microclimate caused by invasive vegetation (Civitello et al. 2008; Magoba and Samways 2010).In addition, Mexican thorn encroachment may facilitate local colonization by non-native predators and resource competitors (Ashmole and Ashmole 2000). Native invertebrates are highly susceptible to predation by introduced animals (Doherty et al. 2016; Anton et al. 2020), and rats (Shiels et al. 2014; Harper and Bunbury 2015) and ants (Suarez et al. 2009) are among the most globally important invasive predators. Both non-native black rats Rattus rattus (AIGCFD 2023) and ants (all species are non-native; Sharp and Tawatao 2023) are abundant on Ascension. Generalist scavengers are also long-established, including the American cockroach Periplaneta americana and tropical house cricket Gryllodes sigillatus (Ashmole and Ashmole 2000). Such introduced species are capable of competing with natives for feeding niche space (Garnas et al. 2014; Fortuna et al. 2022). It is possible, then, that Mexican thorn not only degrades habitat quality directly through altered microclimate, but also indirectly through increased predatory and competitive pressures.
We quantified some of the direct and indirect ecological impacts of Mexican thorn encroachment on one of Ascension’s endemic invertebrate species: Discophallus ascension. This scaly cricket (family: Mogoplistidae) is endemic to the island (Gorochov 2009). Little is known about D. ascension; they have previously been reported only from dead individuals and thus the species’ ecology was almost entirely unknown before this study. The crickets were thought to inhabit only barren coastal areas unaffected by Mexican thorn (AIGCFD 2023), where it has been assumed that they exist as nocturnal generalist scavengers (Ashmole and Ashmole 1997). Understanding and assessing the relative importance of non-native threats against the species will be fundamental in prioritizing methods for sustaining and potentially recovering their populations. This is because coastal spread of Mexican thorn may soon overwhelm removal capacity on the island. Alternatives to costly vegetation removal, e.g. eradication of associated and threatening non-native animals need to be rapidly considered. Such research can also inform management involving non-native species interactions elsewhere.
Discophallus ascension ecology and conservation represents an exemplary case study for assessing impacts of vegetation encroachment because of their presumably simple native ecology. Ascension has no native mammals, amphibians or reptiles (Ashmole and Ashmole 2000) and similarly no native ants (Sharp and Tawatao 2023), so natural competition and predation levels may be low. The case study therefore represents a simplified scenario of non-native vegetation impacts on an endemic invertebrate that may contribute baseline knowledge towards the study of invasions in more complex systems. Even in this simple system, D. ascension might be impacted by temperature (Weckerly 2012), predation by rats (St Clair et al. 2011), or predation by ants (Gasc et al. 2018) in line with prior studies on native crickets. Declines in D. ascension in Mexican thorn-dominated habitat may be resultant of numerous concurrent ecological processes.
We aimed to identify whether D. ascension is impacted, both directly by Mexican thorn (via microclimate modification) and indirectly (via increased interspecific resource competition and predation). We hypothesized that Mexican thorn encroachment (1) directly shifted the nighttime surface temperature away from the optimum for D. ascension, (2) indirectly increased the relative level of resource competition against D. ascension, and (3) indirectly increased the relative level of predation of D. ascension. We intended for our results to influence species management on Ascension, but also to draw broad conclusions of relevance to invasions on other oceanic islands.
2. Methods
Study site
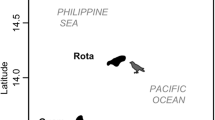
We addressed our hypotheses on Ascension between May and July 2023. The island itself is just 88 km2 in area, is relatively compact in shape, and is isolated from the closest continental land mass (Africa) by approximately 1600 km. We selected three sites which were over 4 km apart: one with extensive Mexican thorn encroachment and two without. Recent surveys had confirmed D. ascension to be present around much of Ascension’s barren coastline (AIGCFD 2023), and so we selected one barren site on each of the northern (Ladies’ Loo, LL) and southern coasts (Waterside, WS) to best represent natural variability in the island coastal ecosystem (Fig. 1). LL comprised barren volcanic lava rock of just a few hundred years in age (Jicha et al. 2013) with sparse ephemeral native grasses. WS comprised similar habitat to LL, but with older rock (~ 0.5 million years; Jicha et al. 2013) moreso smoothed by erosion.
We used the only Mexican thorn site on the island where the shrub was dominant on the coastline but had never been managed (Long Beach, LB). Other coastal sites that are already colonized by Mexican thorn exist, but the invasive shrub is regularly removed and rat poisons are deployed in a subset of sites which are of importance to seabirds. These additional sites were therefore not representative of usual Mexican thorn colonization. While potentially problematic in terms of pseudoreplication that we were only able to study this single Mexican thorn site the urgency in assessing threats against D. ascension warranted immediate study. We attempted to minimize within-site pseudoreplication by collecting data at points separated by substantial distances relative to individual crickets—this is discussed later. LB comprised lava rock of intermediate age where Mexican thorn formed thick bushes covering approximately 25% of the ground, which extended at least 2 km in all directions.
Assessing baseline D. ascension ecology
In order to recognize potential temperature shifts away from the optimum for D. ascension we had to first calculate that optimum. Separately, we had to identify the preferred food resources of D. ascension before quantifying interspecific competition for those resources. Both temperature and feeding preferences were assessed on captured individuals under controlled conditions.
We live-trapped D. ascension at each of the three study sites using pitfall-style traps adapted for rocky terrain. Those traps comprised a 700 ml plastic collection bottle intended for a malaise trap with three rings of 1 cm diameter holes drilled at 5 cm intervals. Each bottle was dug as far as possible into the rocky substrate and surrounded by small rock fragments, so that the drilled entry holes were accessible to crawling invertebrates. A 50 ml bait container was hung inside the bottle and half-filled with a rancid slurry of hotdog meat and water found by prior trials to be highly attractive to D. ascension (Sharp pers. comm.). A lid was added to the bottle to protect any trapped invertebrates from predation or desiccation. Only adults were taken, and juveniles were released. Traps were set between 5 pm and 7 pm and were emptied between 8 am and 9 am the following morning to avoid exposure of trapped individuals to extreme daytime heat. We controlled for the site in which each cricket individual had been collected in our analyses and rereleased all individuals after a maximum of 5 days.
Temperature optimum was assessed via behavioral experiments in purpose-designed enclosures (Treidel et al. 2022). Plastic reptile rearing enclosures of 41.5 × 26.5 × 14.8 cm were divided with sheets of cardboard into four lanes each of approximately 41.5 × 6.5 cm in area (Fig. 2). Those individual lanes were sealed to prevent cricket escape or mixing using tape and fine nylon mesh. A 15-Wheat mat was secured to one end wall of the enclosure, so that one end of each four lanes was heated. The mat was controlled by a thermostat with temperature sensor positioned at the lengthwise midpoint of the enclosure, so that the midpoint temperature of each experimental lane was electronically controlled. As a result, the lane ends furthest from the heat mat were cooler and the ends closest were warmer than defined by thermostat. Ambient temperature was controlled by placing each enclosure in a small temperature-regulated room. Temperatures of the “hot” and “cold” ends were recorded by digital thermometer. By these methods, we were able to create temperature gradients of approximately ± 2 °C around controlled midpoints of 22 °C, 24 °C, 26 °C, 28 °C and 30 °C. This combined gradient of ~ 20–32 °C was chosen to represent values encompassing all possible temperatures encountered by D. ascension in their habitat.
Diagram of the enclosure used to assess the temperature preference of D. ascension. Cardboard sheets divided the enclosure into four lanes of equal dimensions (41.5 × 6.5 cm in area). The heat mat controlled the midpoint temperature via thermostat, creating “hot” (right) and “cold” (left) ends. Temperatures at both ends were recorded by digital thermometers. An egg cup was placed at both ends of each lane to provide tested crickets refugia for rest
We released one D. ascension individual into the center of each lane and recorded their position after a period of movement and acclimatization. Crickets entered the lane between 5 pm and 7 pm and their relative position (“hot” end, “cold” end or middle) were recorded at 7:30 am–8 am the following morning, allowing for one 12 + h period of nocturnal activity. One “cup” from an egg crate was placed upside-down at each end of each lane to provide a refuge for rest, as well as one 1 × 1 cm cube of fresh potato for moisture. Individuals were recorded as being at either “hot” or “cold” end if they were inside or on top of an egg cup, or otherwise within 5 cm of the lane end. Individuals which had not come to rest at either end were recorded as being in the middle of the lane and those trial results were excluded from analysis. Individual crickets were subject to only one evening of temperature trial before rerelease. The enclosures were cleaned thoroughly with a non-toxic disinfectant between trials to ensure that the pheromones of previous individuals did not influence the behavior of tested crickets (Hedrick et al. 2002). We hypothesized that the optimum temperature for D. ascension was that at which 50% of tested individuals would be found at the “hot” and “cold” ends. We performed a total of 292 individual temperature trials.
Optimum temperature was estimated via mixed probit regression. Each data point represented an individual D. ascension cricket and its chosen lane end. The independent variable was the fixed midpoint temperature of the lane (22 °C, 24 °C, 26 °C, 28 °C or 30 °C) and the dependent variable was the chosen “hot”/“cold” position by the individual (values ~ 0 represented the “cold” end and ~ 1 represented the “hot”). Minor variations in the exact temperature recorded at either end were accounted for by reverse-probit transforming the recorded values. This meant that lanes in which the recorded end temperature were further from the midpoint value had a slightly stronger influence on the regression gradient. For example, for an individual which was recorded at a “hot” lane end of + 1 °C compared to the midpoint, the assigned dependent variable was 0.84 (2 d.p.). For an individual recorded at a “cold” lane end of − 1.5 °C from the midpoint, the dependent variable was 0.07 (2 d.p.). The site from which the individual was trapped was included as a random intercept. From the estimated fixed coefficients, the optimum temperature was estimated as the lane midpoint temperature (dependent) where the independent variable was exactly 0.5. We calculated this point as:
where X was the optimum temperature, \({\Phi }^{{{ - }1}} \left( P \right)\) was the inverse of the cumulative probability, \(\beta_{0}\) was the intercept and \(\beta_{1}\) was the temperature coefficient (line gradient).
Live-trapped D. ascension individuals were separately tested for food preference. Individuals from food trials were not used in temperature trials, and the same was true vice versa. A range of food resources was selected for trial: a non-natural sugar-based resource (strawberry jam), a non-natural flesh-based resource (jarred mixed-meat hotdog), and two flesh-based resources occurring naturally in very high abundance on coastlines (Sally lightfoot crab Graspus graspus and black triggerfish Melichthys niger). As there would naturally be no vegetation, even seaweed, on much of the coastal habitat (Ashmole and Ashmole 1997), we assumed that D. ascension cannot be adapted to feeding on either live or dead vegetation and so these potential resources were not tested. We aimed to identify if D. ascension diet was either entirely generalist (all offered resources), protein-based (hotdog, crab or fish), or specialist (either crab or fish only).
Feeding trials comprised paired 200 ml containers: one with a D. ascension individual and one “control” without. Container tops were covered with a fine mesh to allow air circulation and maintain the interior and ambient nighttime temperature. Each pair of containers was loaded with 1 g of the same food resource. Trials were started around 7:30 pm and ended around 7:30 am the following morning to allow a 12 h nocturnal feeding bout in the occupied container. The mass of resource consumed after that period was calculated as 1 minus the remaining test resource mass, altogether minus the remaining mass of the “control” resource (to account for desiccation; Xu et al. 2013). It was possible for estimated consumption to be negative. Resources were considered “consumed” if estimated consumption values were significantly greater than 0. We performed a total of 142 individual food resource trials.
Assessing direct impacts of Mexican thorn on temperature suitability
Once an optimum temperature for D. ascension had been estimated, we compared that value with temperatures recorded at each of the three study sites. We established 4–7 transects at each site (10 between LL and WS, and 7 at LB), spaced a minimum of 50 m apart from one another and leading inland from the coastline. Along every transect, we deployed digital air temperature sensors at 0, 25 and 50 m inland which logged the minimum air temperature and maximum/minimum humidity at surface level over a single nighttime (approximately 6 pm–8 am the following day). All microclimate measurements, a total of 43 for each of air temperature and humidity (after accounting for device failures), were collected within a 1 month period and in randomized order to control for seasonality and weather patterns. We then tested whether temperatures at LB (the Mexican thorn site) were significantly greater than at LL or WS via two-sample T-test, and separately whether air temperatures differed significantly from the optimum for D. ascension via one-sample T-test.
Assessing indirect impacts of Mexican thorn on resource competition
We loosely followed methods by Gray et al. (2018) in quantifying relative competition in different habitat types. We deployed “competition arenas”, comprising 15 × 15 cm white-painted wooden squares representing a controlled area for observing resource competition (Fig. 3). These controlled methods ensured that estimated rates of ecological processes could be equally assessed in habitats of high, low or no local D. ascension population. In the center of each arena, we deposited 10 g of mashed hotdog, which through prior captive trials we had confirmed to be consumed by D. ascension. We used a non-natural food to ensure that any competition for the resource was entirely generalist. The hotdog was also far easier to standardize and therefore reproduce compared with “natural” crab or fish carcasses that we might have been able to collect—we could ensure that the hotdog was consistent in moisture content, odor, and consistency compared with carcasses. To avoid disturbance of any interspecific competitive interactions, and to allow monitoring of multiple arenas in parallel, we recorded species appearance and behavior from recordings. A GoPro Hero 9 Black Edition camera was elevated approximately 15 cm above each arena on a small tripod. Every camera was fitted with a non-brand 15X macro lens to allow close focus on the arena below. The arenas were each illuminated with a battery-operated 350 lm LED light source. Because of this resource detection may have been more rapid than would occur without illumination, however the effect was constant across all sites which allowed valid relative comparison of competition rates. Arenas and cameras were deployed between 5 pm and 6 pm—just before the nocturnal activity of D. ascension. Cameras were operated between 7 pm and 10 pm on “timelapse” settings; photos were taken every 10 s. With the known limited diversity on Ascension’s volcanic coastline (Ashmole and Ashmole 1997), these methods were sufficient to confidently identify most species visiting the resource. We recorded the first taxon to visibly interact with the resource out of the most frequently observed visitors to the arenas: D. ascension, ants, tropical house cricket G. sigillatus, American cockroach P. americana and black rat R. rattus. The identity of the first-arriving taxon was used as a rough proxy for competitive advantage. Five arenas were positioned at 0, 10, 20, 30 and 40 m from the coastline in a single night. We operated three such transects at every site which were positioned a minimum of 50 m apart from each other, totaling 45 individual trials. Logistic regression was used to compare the population-level competitive advantage of each recorded taxon in turn (D. ascension, ants, G. sigillatus, P. americana or R. rattus) between habitat with and without Mexican thorn. The independent variable was Mexican thorn presence (binary) and dependent variable was whether or not the respective taxon has arrived first at the resource (binary also). Logistic regression could not validly be used to quantify per-predator shifts in importance between the two habitat types where that respective predator was only recorded in one. In these cases, a one-sided binomial odds test was employed to determine whether the observed 0 value in one habitat type was to be expected (P < 0.05) under the observed probability ratio from the other.
Assessing indirect impacts of Mexican thorn on predation
We expanded on our methods for quantifying relative competition to quantify relative predation also. Following prior studies which used live prey baits to examine trophic ecology (Birkhofer et al. 2017), we used live baits in “predation arenas”. These comprised very similar equipment as our competition arenas, but a screw was driven into the center of each wooden square and a prey item leashed to the screw by fine fishing line (Fig. 3). D. ascension is not yet protected but we hypothesize that its populations are severely reduced due to the indirect impacts of Mexican thorn. As such, we avoided harm to D. ascension individuals by using a surrogate bait of similar morphology and behavior (Bröder et al. 2023). The introduced cricket G. sigillatus was ideal for being similar size and shape to D. ascension, being of similar generalist ecology, and being already abundant on Ascension. We live-trapped G. sigillatus individuals and attached them to leashes via a small dot of cyanoacrylate superglue on the pronotum, so that they could move freely within a circle of approximately 74 cm2. This movement was intended to stimulate natural predatory responses, although we recognize that the confinement and illumination of prey items likely artificially increased or lowered predation success. Nonetheless the relative predation rate compared between sites remained valid. As before, we deployed arenas in groups of five at 0, 10, 20, 30 and 40 m inland at three locations within each site. Arenas were deployed between 5 and 6 pm, and nocturnal predation activity was observed on timelapse photo between 7 pm and 10 pm. An arena where the prey had been killed before 7 pm (n = 1 only) was removed from analysis as we had no way of identifying the predator. Similarly, arenas where the prey was not killed before 10 pm but was dead or removed on collection the following morning were considered to have not been attacked by predators. We compared the relative predation by the same five taxa between study sites. Logistic regression was used to estimate the relative pressure exerted on D. ascension in both Mexican thorn-dominated and barren habitat, with Mexican thorn presence (binary) being the independent variable and predation occurrence (also binary) being the dependent. As before, binomial odds tests were substituted where logistic regression could not be applied.
Data analysis
All analyses were performed in R version 4.3.0 (R Core Team 2023). Fitting of mixed effect models was via the lme4 package (Bates et al. 2015). We used ggplot2 for data visualization (Wickham 2016). Mapping of study sites was achieved using QGIS version 3.30.2.
Results
Assessing baseline D. ascension ecology
We successfully identified two key requirements of D. ascension, which allowed later analysis of the specific threats faced by the species. Probit regression of D. ascension responses to controlled warming showed that the species generally chose the “cool” end of experimental lanes where the midpoint was warmed to a higher temperature but chose the “hot” end where the midpoint was lesser warmed (line gradient: − 0.12 ± 0.03, P < 0.001; Fig. 4a). From our estimated regression coefficients (where we predict 50% of D. ascension individuals to settle at either end of the lane), we calculated that the optimum nocturnal temperature for the species was 24.0 °C.
Experimental investigation of D. ascension requirements. 4a. Optimum nocturnal temperature of D. ascension. Points (given jitter in the x-direction for clarity) represent individual crickets’ responses to warming, with points towards 0 representing crickets that settled at the “cold” end of experimental lanes and points towards 1 representing crickets that settled at the “hot” end relative to the midpoint temperature (X-axis). The vertical dashed line represents the temperature value (24.0 °C) at which exactly 50% of crickets (horizontal dashed line) settled at “cold” and “hot” ends. 4b. Estimate of D. ascension overnight consumption of four potential food resources Significance of one-sample T-tests (against a mean value of 0) are represented by stars: * for P < 0.05, ** for P < 0.01, and *** for P < 0.001
Mixed modelling of food resource consumption confirmed the protein-based diet of D. ascension. Our model had no intercept term, and so all parameters significantly greater than 0 suggested that the respective food resource had been consumed (Fig. 4b). The black triggerfish (estimate: 0.033 ± 0.009 g, P < 0.01), hotdog (estimate: 0.027 ± 0.009 g, P < 0.05) and Sally lightfoot crab (estimate: 0.060 ± 0.009 g, P < 0.001) were all consumed. The one sweet bait, strawberry jam, was not consumed (estimate: 0.016 ± 0.009 g, P > 0.05). These results suggested that D. ascension was relatively generalist in protein scavenging. Black triggerfish and crab would likely be rapidly located and consumed by the crickets in the natural habitat, but also other less frequently encountered flesh resources. These findings also confirmed the suitability of hotdog for use in the subsequent competition arena trials.
3.2 Assessing direct impacts of Mexican thorn on temperature suitability
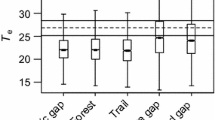
We did not find strong evidence to support the hypothesis that Mexican thorn was directly reducing habitat quality for D. ascension and other coastal-native invertebrates through modification of microclimate (Fig. 5). One-sample T-tests did not report significant differences between the optimum D. ascension temperature, 24.0 °C, in habitat with Mexican thorn at 0 m (estimate: 25.1 °C, 95% CI = 21.7–28.5 °C, P > 0.05), 25 m (estimate: 24.0 °C, 95% CI = 22.0–25.9 °C, P > 0.05) or 50 m inland (estimate: 25.1 °C, 95% CI = 22.9–27.2 °C, P > 0.05). The same was true in barren habitat without Mexican thorn (0 m estimate: 24.2 °C, 95% CI = 21.9–26.6 °C, P > 0.05; 25 m estimate: 24.6 °C, 95% CI = 23.1–26.2 °C, P > 0.5; 50 m estimate: 22.8 °C, 95% CI = 21.5–24.1 °C, P > 0.05). Air temperature at substrate level among dense Mexican thorn coverage was therefore considered suitable for D. ascension habitation. We did, however, identify a significant increase in minimum temperature at 50 m inland with Mexican thorn encroachment (estimate: 2.3 °C, 95% CI = 0.1–4.5 °C, P < 0.05). We found no significant effect of Mexican thorn on minimum humidity at any inland distance (P > 0.05), however we found maximum humidity to be reduced by Mexican thorn at 50 m inland only (estimate: − 12.4%, 95% CI = − 2.6 to − 22.2%, P < 0.05). This suggested that Mexican thorn did in fact impact microclimate inland, but the strong action of oceanic wind and spray on the immediate coastline overrode any measurable effect at 0 m and 25 m.
Minimum overnight temperatures recorded by data loggers in habitat both with and without Mexican thorn encroachment Significant difference (two-sample T-test) between recorded temperatures (Y-axis) at same distance inland (X-axis) are represented by * for P < 0.05. Estimated optimum D. ascension temperature (24.0 °C) is represented by a dashed line
Assessing indirect impacts of Mexican thorn on resource competition
D. ascension was the most frequent first arrival at competition arenas across sites (n = 17), but we found evidence that Mexican thorn encroachment was influencing the relative competition rate between the endemic and non-native species (Fig. 6). D. ascension was less often the first to arrive at competition arenas and interact with the food resource in Mexican thorn habitat compared with barren habitat (estimate: − 2.01 ± 0.89, P < 0.05). Both ants (n = 4, binomial odds test: P < 0.001) and P. americana (n = 5, binomial odds test: P < 0.001) were observed to compete only in this modified habitat. There was no significant difference in the number of first arrivals of G. sigillatus between habitat with and without Mexican thorn (estimate: 0.41 ± 0.84, P > 0.05) but the non-native cricket was a frequent resource competitor (n = 7). The same was true of R. rattus (estimate: − 1.89 ± 1.11, P > 0.05), which was the most frequent non-native first arrival (n = 10). This suggest that Mexican thorn presence facilitated the increased competitive pressure exerted by some, but not all, taxa.
Comparison of rates of successful Resource Competition and Cricket Predation by the five taxa most frequently visiting our experimental arenas. Pairwise comparisons of respective rates with Mexican thorn presence/absence are represented. Symbols above the pairs represent significance: * for P < 0.05, and *** for P < 0.001. All pairwise comparisons are via logistic regresion, besides each case where 100% of observations were in a single habitat type (Resource Competition: Ants and P. americana, and Cricket Predation: Ants and R. rattus) which were binomial odds tests
Assessing indirect impacts of Mexican thorn on predation
We observed predation of our D. ascension surrogate, G. sigillatus, at all sites (Fig. 6). It appears likely that predation rate was elevated across even barren sites, given that 67% of observed predation events at barren sites were by non-native animals. As with estimated relative competition rates, relative predation rates were modulated by Mexican thorn presence. Predation by ants (n = 2, binomial odds test: P < 0.05) was recorded only at sites with Mexican thorn. Conversely, predation by rats (n = 7, binomial odds test: P < 0.05) was recorded only at sites without Mexican thorn. Predation by P. americana was recorded at both sites with (n = 2) and without (n = 1) Mexican thorn. Excluding predation by D. ascension (n = 5), there was no significant difference between the relative predation rate between sites with and without Mexican thorn (estimate: − 0.89 ± 0.77, P > 0.05).
Discussion
We performed a total of 567 individual trials and measurements to define the ecology of D. ascension and examined the specific threats that would be posed by Mexican thorn encroachment. Non-native species of plant, insects and rat all appeared to interact and exert unnatural ecological pressure on our island-endemic case study species, D. ascension. While it would be extremely difficult to calculate a potential population decline in D. ascension, our results strongly suggest that the indirect and direct effects of Mexican thorn encroachment negatively impact the species. However, the complexities of the interspecific interactions represented in our analyses do not indicate that removal of invasive vegetation alone would be sufficient to benefit endemic biodiversity.
In line with previous studies (Civitello et al. 2008; Magoba and Samways 2010; Cook and Talley 2014), our results indicated that invasive vegetation modifies inland microclimate relevant to native invertebrates. The similarity in surface-level microclimate at 0 m and 25 m inland between habitats with and without Mexican thorn suggest that the 50 m inland differences that we detected were caused by presence of the shrub, rather than being cause for the shrub to initially grow there. We assume that our observed increase in minimum nighttime temperature and decrease in maximum humidity among Mexican thorn trees at 50 m inland was resultant of the thick layer of leaf litter and deposition of organic matter by the dense shrubs. That additional layer of material had potential to insulate the rock substrate below, and perhaps slowed overnight cooling and absorbed moisture. Despite this effect, we found no evidence that Mexican thorn directly reduced habitat quality for D. ascension via microclimate modification. This is firstly because the temperature and humidity shifts were only detected away from the immediate coastline, which through previous observation (Ashmole and Ashmole 1997) and apparent feeding preferences (native fish and crab) is likely to be the key D. ascension habitat. Secondly, despite the significant increase in substrate surface temperature with invasive vegetation, the measured increase was not greater than our estimated optimum for D. ascension. However, that optimum is by definition species-specific. The temperature increase might therefore be directly detrimental or even beneficial to Ascension’s other endemic invertebrates, eg Niambia woodlice and Pseudosinella springtails (Ashmole and Ashmole 1997, 2000).
Separately, other microclimate variables such as substrate moisture content (separate from humidity) could not be measured within the scope of this study but are likely to be influenced by non-native vegetation. Substrate moisture is thought to be an important determinant of mogoplistine ovipositing behaviour (Vahed 2020), although we opportunistically observed D. ascension oviposit on both wet and dry substrates while in captivity. Nonetheless, there are likely other microclimatic variables of importance to D. ascension that we did not investigate. Therefore, we cannot reject our first hypothesis, but instead recommend additional research both on D. ascension and other invertebrates to detect potentially influential shifts in microclimate driven by Mexican thorn.
Conversely to our first hypothesis, we accept our second hypothesis. We found strong evidence that non-native resource competition was elevated in habitat degraded by Mexican thorn. The invasive shrub was previously assumed to provide habitat for various non-native animals (Ashmole and Ashmole 2000; AIGCFD 2015), but our results indicate that other non-native animals drove high interspecific competition even in barren habitat. Mexican thorn encroachment did increase competition for the protein resource from ants (of which a subset were identified with relative confidence as being the longhorn crazy ant Paratrechina longicornis) and the American cockroach P. americana. It appears likely that the shrub provides habitat for these species, perhaps through shelter or by modifying the substrate structure. Provision of habitat for non-native animals by non-native vegetation has been previously reported (Rodríguez et al. 2020; Landsman et al. 2021). Competition from the tropical house cricket G. sigillatus and black rat R. rattus were also consistently high in Mexican thorn habitat, and thus it could be derived that the shrub at least is not avoided by those species. It is apparent that while Mexican thorn increased the overall non-native competitive pressure on D. ascension, the effects were again specific to competitor taxa. Nonetheless, these findings suggest that D. ascension numbers may be reduced close to Mexican thorn as a result of competition, and provide plausible explanation in support of other work that found reduced native invertebrate abundances among non-native vegetation (Tanner et al. 2013).
Finally, we reject our third hypothesis: we observed non-native cricket predation both in barren habitat and habitat degraded by Mexican thorn with no significant difference in overall rate. All predation events were either by non-native animals or D. ascension. Our illuminated predation arenas with leashed bait crickets likely increased the relative predation rate across all sites, and so valid comparison could only be made between sites. Even so, it is likely that predation of D. ascension was elevated above the natural level by non-native animals at all studied sites. This finding is of course worrying, as it indicates that removal of invasive vegetation alone is unlikely to reduce island-scale predation rate. Interestingly, the Mexican thorn presence appeared to mediate a near-total shift from rat predation to predation by insects. This implies that reduction of predation by non-native insects achieved through Mexican thorn removal is unlikely to also reduce predation by rats, which we opportunistically observed killing and consuming wild D. ascension for the first time during one of our competition trials. We suggest that rats may be deterred from areas of invasive vegetation encroachment by the presence of ants, which may attack rats nesting among the roots. Ants are often among the dominant taxa in ecosystems (Gray et al. 2018) and it could make sense that invasive rats actively avoid their attacks. Clearly our conclusions could be strengthened by sampling more sites colonized by Mexican thorn, which is presently impossible on Ascension. Despite this limitation, our findings support several important conclusions, not only for Ascension but also on other oceanic islands. We found evidence of various non-native species-interactions on competition and predation, with many indirect implications for future species management. Our results indicate that manual removal of Mexican thorn, a difficult and expensive task in itself (AIGCFD 2015; Eschen et al. 2023), may reduce non-native competitive pressure and predation of endemic invertebrates by other invertebrates but may not influence or may even increase predation of endemic crickets by rats. Any such vegetation clearance should be accompanied by management of rats. On Ascension, bromadiolone rat poisons are already deployed in some areas of conservation importance to seabirds with anecdotal reported success (AIGCFD 2023). Extensive deployment of such poisons may be necessary alongside Mexican thorn clearance to increase D. ascension populations.
Such actions are often unfeasible or unsuccessful for small island nations with limited resources. On Ascension, current active invasive species management is strictly limited by capacity to sites of highest conservation application (AIGCFD 2023). On the 37 km2 Henderson Island, South Pacific, £1.5 million (GBP) was spent on dropping similar poisons by helicopter and failed to eradicate their population of invasive rats (Amos et al. 2016). It is clear that the most efficient way to prevent the ecological damage caused by invasive species is to halt new introductions through heightened biosecurity (Scott et al. 2017; Bodey et al. 2023). Rats are already among the most globally widespread invasive taxa, and there are well-researched best-practices for their effective management (Keitt et al. 2015). Nonetheless, our analyses presented here unfortunately indicate that such species-specific measures should be operated in tandem with invasive vegetation clearance for maximum benefit to endemic invertebrates. This would unavoidably come at significant financial expense.
The findings of this study were surprising and novel in that encroaching invasive vegetation did not appear to be strongly increasing all non-native threats against our endemic study species. Instead, we identified several convoluted interactions that render it impossible to recommend a single-action solution against the threats to biodiversity posed by non-native species. On Ascension, an island with an unusually simple native ecosystem (Ashmole and Ashmole 2000), at least Mexican thorn and black rats would need to be managed together at island-scale for effective invertebrate conservation. For older islands with more complex ecosystems and greater native biodiversity, it seems probable that even more than two non-native species should be managed in parallel for the same benefit. Certainly, understanding direct and indirect species interactions is beneficial in informing management (Scott et al. 2017; Sharp and Tawatao 2023). With island biodiversity at such immediate risk of extinctions resulting from species introductions (Fernández-Palacios et al. 2021), researching and funding control programmes against the most globally important invasive species are of utmost urgency.
Data availability
The data that support the findings of this study are available upon reasonable request.
References
Amos W, Nichols HJ, Churchyard T, Brooke MDL (2016) Rat eradication comes within a whisker! A case study of a failed project from the South Pacific. R Soc Open Sci 3:160110
Anton A, Geraldi NR, Ricciardi A, Dick JTA (2020) Global determinants of prey naiveté to exotic predators. Proc R Soc B 287:20192978
Ascension Island Government Conservation and Fisheries Directorate (2015) Mexican thorn species action plan. In: The Ascension Island Biodiversity Action Plan. Ascension Island Government Conservation Department, Georgetown, Ascension Island
Ascension Island Government Conservation and Fisheries Directorate (2023) Beach nature reserves management plan 2023–2028. Ascension Island Government Conservation Department, Georgetown, Ascension Island
Ashmole NP, Ashmole MJ (1997) The land fauna of Ascension Island: new data from caves and lava flows, and a reconstruction of the prehistoric ecosystem. J Biogeogr 24:549–589
Ashmole, N.P. Ashmole, M.J. (2000) St Helena and Ascension Island: a natural history, Anthony Nelson, Oswestry
Bates D, Mächler M, Bolker BM, Walker SC (2015) Fitting linear mixed-effects models using lme4. J Stat Softw 67(1):1–48
Birkhofer K, Bylund H, Dalin P, Ferlian O, Gagic V, Hambäck PA, Klapwijk M, Mestre L, Roubinet E, Schroeder M, Stenberg JA, Porcel M, Björkman C, Jonsson M (2017) Methods to identify the prey of invertebrate predators in terrestrial field studies. Ecol Evol 7:1942–1953
Bodey TW, Angulo E, Bang A, Bellard C, Fantle-Lepczyk J, Lenzner B, Turbelin A, Watari Y, Courchamp F (2023) The economic cost of protecting islands from invasive alien species. People Nat 37(2):e14034
Bröder L, Tatin L, Hochkirch A (2023) Quantifying predation to insects: an experimental approach. Glob Ecol Conserv 44:e02485
Civitello DJ, Flory SL, Clay K (2008) Exotic grass invasion reduces survival of Amblyomma americanum and Dermacentor variabilis ticks (Acari: Ixodidae). J Med Entomol 45(5):867–872
Cook RW, Talley TS (2014) The invertebrate communities associated with a Chrysanthemum coronarium-invaded coastal sage scrub area in Southern California. Biol Invas 16(2):365–380
Dakhil MA, El-Keblawy A, El-Sheikh MA, Halmy MWA, Ksiksi T, Hassan WA (2021) Global invasion risk assessment of Prosopis juliflora at biome level: does soil matter? Biology 10(3):203
Doherty TS, Glen AS, Nimmo DG, Ritchie EG, Dickman CR (2016) Invasive predators and global biodiversity loss. Proc Natl Acad Sci USA 113(40):11261–11265
Duffey E (1964) The terrestrial ecology of ascension island. J Appl Ecol 1(2):219–251
Eschen R, Bekele K, Jumanne Y, Kibet S, Makale F, Mbwambo JR, Megersa B, Mijay M, Moyo F, Munishi L, Mwihomeke M, Nunda W, Nyangito M, Witt A, Schaffner U (2023) Experimental Prosopis management practices and grassland restoration in three Eastern African countries. CABI Agric Biosci 4:21
Fernández-Palacios JM, Kreft H, Irl SDH, Norder S, Ah-Peng C, Borges PAV, Burns KC, de Nascimento L, Meyer JY, Montes E, Drake DR (2021) Scientists’ warning—the outstanding biodiversity of islands is in peril. Glob Ecol Conserv 31:e01847
Fortuna TM, le Gall P, Mezdour S, Calatayud PA (2022) Impact of invasive insects on native insect communities. Curr Opin Insect Sci 51:100904
Garcia RA, Clusella-Trullas S (2019) Thermal landscape change as a driver of ectotherm responses to plant invasions. Proc R Soc B 286:20191020
Garnas J, Groden E, Drummond FA (2014) Mechanisms of competitive displacement of native ant fauna by invading Myrmica rubra (Hymenoptera: Formicidae) populations. Envir Entomol 43(6):1496–1506
Gasc A, Anso J, Sueur J, Jourdan H, Desutter-Grandcolas L (2018) Cricket calling communities as an indicator of the invasive ant Wasmannia auropunctata in an insular biodiversity hotspot. Biol Invasions 20(5):1099–1111
Gorochov AV (2009) Crickets of the family Mogoplistidae (Orthoptera) from Ascension and Saint Helena islands, Atlantic Ocean. Proc Zool Inst Russ Acad Sci 313(1):23–32
Gray A, Wilkins V, Pryce D, Fowler L, Key RS, Mendel H, Jervois M, Hochkirch A, Cairns-Wicks R, Dutton AJ, Malan L (2019) The status of the invertebrate fauna on the South Atlantic island of St Helena: problems, analysis, and recommendations. Biodivers Conserv 28:275–296
Gray REJ, Ewers RM, Boyle MJW, Chung AYC, Gill RJ (2018) Effect of tropical forest disturbance on the competitive interactions within a diverse ant community. Sci Rep 8:5131
Gurevitch J, Padilla DK (2004) Are invasive species a major cause of extinctions? Trends Ecol Evol 19(9):470–474
Harper GA, Bunbury N (2015) Invasive rats on tropical islands: their population biology and impacts on native species. Glob Ecol Conserv 3:607–627
Hedrick AV, Perez D, Lichti N, Yew J (2002) Temperature preferences of male field crickets (Gryllus integer) alter their mating calls. J Comp Physiol A 188(10):799–805
Jicha BR, Singer BS, Valentine MJ (2013) 40Ar/39 Ar geochronology of subaerial Ascension Island and a Re-evaluation of the temporal progression of basaltic to rhyolitic volcanism. J Petrol 54(12):2581–2596
Keitt B, Griffiths R, Boudjelas S, Broome K, Cranwell S, Millett J, Pitt W, Samaniego-Herrera A (2015) Best practice guidelines for rat eradication on tropical islands. Biol Cons 185:17–26
Kier G, Kreft H, Tien ML, Jetz W, Ibisch PL, Nowicki C, Mutke J, Barthlott W (2009) A global assessment of endemism and species richness across island and mainland regions. Proc Natl Acad Sci USA 106(23):9322–9327
Knapp D, Knapp J, Stahlheber K, Dudley T (2019) A little goes a long way when controlling invasive plants for biodiversity conservation. In: Clout CJWMN, Martin AR, Russell JC (eds) Island invasives: scaling up to meet the challenge. IUCN, Gland, Switzerland
Lachenicht MW, Clusella-Trullas S, Boardman L, Le Roux C, Terblanche JS (2010) Effects of acclimation temperature on thermal tolerance, locomotion performance and respiratory metabolism in Acheta domesticus L. (Orthoptera: Gryllidae). J Insect Physiol 56(7):822–830
Lambdon P, Darlow A (2008) Botanical survey of Ascension Island and St. Helena. A report on the current state of plant invasions, and their implications for conservation and management. Royal Society for the Protection of Birds, Sandy
Landsman AP, Schmit JP, Matthews ER (2021) Invasive plants differentially impact forest invertebrates, providing taxon-specific benefits by enhancing structural complexity. Front Ecol Evol 9:682140
Magoba RN, Samways MJ (2010) Recovery of benthic macroinvertebrate and adult dragonfly assemblages in response to large scale removal of riparian invasive alien trees. J Insect Conserv 14(6):627–636
Pyšek P, Jarošík V, Hulme PE, Pergl J, Hejda M, Schaffner U, Vilà M (2012) A global assessment of invasive plant impacts on resident species, communities and ecosystems: the interaction of impact measures, invading species’ traits and environment. Glob Change Biol 18(5):1725–1737
R Core Team (2023) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Renteria JL, Gardener MR, Panetta FD, Atkinson R, Crawley MJ (2012) Possible impacts of the invasive plant Rubus niveus on the native vegetation of the Scalesia forest in the Galapagos Islands. PLoS ONE 7(10):e48106
Rodríguez J, Novoa A, Cordero-Rivera A, Richardson DM, González L (2020) Biogeographical comparison of terrestrial invertebrates and trophic feeding guilds in the native and invasive ranges of Carpobrotus edulis. NeoBiota 56:49–72
Russell JC, Kueffer C (2019) Island biodiversity in the Anthropocene. Annu Rev Environ Resour 44:31–60
Scott JK, McKirdy SJ, Merwe J, Der Van, Green R, Burbidge AA, Pickles G, Hardie DC, Morris K, Kendrick PG, Thomas ML, Horton KL, O’Connor SM, Downs J, Stoklosa R, Lagdon R, Marks B, Nairn M, Mengersen K (2017) Zero-tolerance biosecurity protects high-conservation-value island nature reserve. Sci Rep 7:772
Shackleton RT, Le Maitre DC, Pasiecznik NM, Richardson DM (2014) Prosopis a global assessment of the biogeography, benefits, impacts and management of one of the world’s worst woody invasive plant taxa. AoB Plants 6:plu027
Sharp A, Tawatao N (2023) Colonization and coexistence of non-native ants on a model Atlantic island. Divers Distrib 29:1278–1288
Sherwood D, Marusik YM, Sharp A, Ashmole P (2023) A survey of Gnaphosidae (Arachnida, Araneae) from Ascension Island with description of a new species of Australoechemus Schmidt & Piepho, 1994. Afr Invertebr 64(3):291–302
Sherwood D, Sharp A (2023) A Caribbean in the South Atlantic: first records of Hentzia antillana Bryant 1940, with notes on other previously reported jumping spider species (Araneae: Salticidae), from Ascension Island. Peckhamia 310(1):1–7
Shiels AB, Pitt WC, Sugihara RT, Witmer GW (2014) Biology and impacts of pacific island invasive species. 11. Rattus rattus, the Black Rat (Rodentia: Muridae). Pacific Sci 68(2):145–184
St Clair JJH, Poncet S, Sheehan DK, Székely T, Hilton GM (2011) Responses of an island endemic invertebrate to rodent invasion and eradication. Anim Conserv 14(1):66–73
Suarez A.V., McGlynn T.P., Tsutsui N.D. (2009) Biogeographic and taxonomic patterns of introduced ants. In: Ant ecology. Oxford University Press, Oxford, pp. 233–244
Tanner RA, Varia S, Eschen R, Wood S, Murphy ST, Gange AC (2013) Impacts of an invasive non-native annual weed, Impatiens glandulifera, on above- and below-ground invertebrate communities in the United Kingdom. PLoS ONE 8(6):e67271
Treidel LA, Huebner C, Roberts KT, Williams CM (2022) Life history strategy dictates thermal preferences across the diel cycle and in response to starvation in variable field crickets, Gryllus Lineaticeps. Curr Res Insect Sci 2:100038
Tye A, Key G, Copsey JA (2018) Threats to islands: invasive species and their impacts. In: Copsey JA, Black SA, Groombridge JJ, Jones CG (eds) Species Conservation: lessons from Islands. Cambridge University Press, pp 83–120
Vahed K (2020) The life cycle of the Atlantic beach-cricket, Pseudomogoplistes vicentae Gorochov, 1996. J Insect Conserv 24(3):473–485
Veron S, Mouchet M, Govaerts R, Haevermans T, Pellens R (2019) Vulnerability to climate change of islands worldwide and its impact on the tree of life. Sci Rep 9(1):14471
Weckerly FW (2012) Cave cricket exit counts: environmental influences and duration of surveys. J Cave Karst Stud 74(1):1–6
Wickham H (2016) ggplot2: elegant graphics for data analysis. Springer-Verlag, New York
Wilkins VL, Knight V, Gray A (2019). Garypus titanius. The IUCN Red List of Threatened Species 2019:e.T135739408A135745815. Accessed on 05 December 2023
Xu Y, Held DW, Hu XP (2013) Dietary choices and their implication for survival and development of omnivorous mole crickets (Orthoptera: Gryllotalpidae). Appl Soil Ecol 71:65–71
Acknowledgements
We would like to extend our gratitude to the staff at AIGCFD for helping to facilitate data collection for this project, as well as Professor Karim Vahed (Buglife, UK) for ongoing advice and support on scaly cricket taxonomy and conservation. We would also like to thank Professor Rob Ewers (Imperial College London) for supporting this project as a thesis project, as well as his research group for their helpful comments on early stages of the manuscript. Three anonymous Reviewers are gratefully acknowledged for their extensive support in improving this article.
Funding
This work was joint-funded by the Indianapolis Zoo Global Center for Species Survival and Darwin Plus Local project DPL00040: Urgent Conservation of a Unique Endemic Genus. The project was hosted by the AIGCFD, which itself was funded by Darwin Plus project DPLUS135: From pseudoscorpions to crickets: securing Ascensions Island’s unique invertebrates.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Chin, W.Y., Wilkins, V. & Sharp, A. Invasive vegetation encroachment modulates dual threats faced by island-endemic scaly crickets. Biol Invasions 26, 2941–2954 (2024). https://doi.org/10.1007/s10530-024-03355-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10530-024-03355-w