Abstract
Background
To investigate the association between abdominal fat distribution represented by the visceral fat area (VFA) to subcutaneous fat area (SFA) ratio, and erosive esophagitis (EE).
Methods
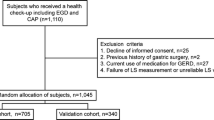
Seven hundred and twenty-eight participants aged >40 years underwent physical examination, blood tests, esophagogastroduodenoscopy, and abdominal computer tomography at Chung-Ang University Hospital from 2007 to 2012.
Results
Of 728 subjects, 65 (8.9%) had EE. The EE patients had higher body mass index, metabolic syndrome prevalence, triglyceride levels, and blood pressure (P < 0.05). The mean VFA/SFA ratio was higher in the EE group than in the non-EE group (1.30 vs. 0.92, P < 0.001). The predominance of EE in the group with higher VFA/SFA ratio was higher than in the group with lower VFA/SFA ratio (P < 0.001). A VFA/SFA ratio ≥1.165 had good accuracy to predict EE (area under the receiver-operating characteristic curve, 0.643). The VFA/SFA ratio and visceral fat volume were positively correlated with the severity of EE (P = 0.002), and a VFA/SFA ratio ≥1.165 was strongly correlated with the severity of EE (P < 0.001).
Conclusion
The high VFA/SFA ratio can be a useful clinical predictor of EE.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Gastroesophageal reflux disease (GERD) is a common disorder. The approximate prevalence of GERD in the Western world is 10–20% [1]. Mucosal damage produced by the abnormal reflux of gastric contents into the esophagus is observed in some cases of GERD [2]. Male gender, hiatal hernia, Helicobacter pylori infection, smoking, and alcohol consumption can increase the risk of erosive esophagitis (EE) [3, 4]. In addition, common features of metabolic syndrome are risk factors for EE [5, 6].
Obesity, a central component of metabolic syndrome, is known to promote gastroesophageal reflux. Of note, the increase in intra-abdominal pressure due to obesity contributes to EE. Some studies have suggested that the risk of GERD or EE increases with increasing body mass index (BMI) [7,8,9]. Most studies have used BMI as an indicator of obesity. However, BMI is an imperfect estimate of adiposity, particularly in men, owing to the greater muscle mass in this group [10]. Moreover, the association between increased BMI and EE is inconsistent and varies with sex, ethnic origin, and other confounding factors [11,12,13].
Therefore, the pattern of obesity may be a better predictor of GERD than BMI [14]. Several studies have suggested that abdominal obesity can be a risk factor for GERD [15]. A previous study indicated that GERD symptoms were positively associated with the abdominal diameter in Caucasian men independently of BMI [16]. Moreover, abdominal adipose tissue consists of subcutaneous fat (SF) and visceral fat (VF), and recent studies revealed the association between abdominal VF and EE [17, 18]. A recent study reported that abdominal VF volume measured by cross-sectional computerized tomography (CT) is a more important risk factor for EE than other factors such as BMI and abdominal circumference in both sexes [17]. However, because of individual differences in the absolute amount of VF, this parameter may not indicate obesity directly; therefore, the relative ratio of VF to SF may indicate obesity more accurately.
We hypothesized that the ratio of visceral fat area (VFA) to subcutaneous fat area (SFA), measured by CT, rather than BMI, might significantly correlate with the risk of EE. Therefore, the aim of this study was to investigate the association between abdominal fat distribution, represented by the VFA/SFA ratio, and the incidence of EE in the general population.
Methods
Study Population
Subjects older than 40 years were enrolled in a routine checkup program at the Health Care Center of Chung-Ang University Hospital from January 2007 to December 2012. A total of 728 participants underwent a physical examination (height, body weight, waist circumference, and blood pressure), blood tests [glucose, triglycerides (TG), and high-density lipoprotein (HDL) cholesterol], esophagogastroduodenoscopy (EGD), and abdominal CT scan. The Institutional Review Board approved the study [Protocol No. C2013001(961)], and all the participants provided a written informed consent.
Measurement of Anthropometric Parameters
Height and body weight measurements were automated (GL-150; G-Tech international Co., Uijungbu City, Korea, Inbody 720; Biospace Co., Chun-An City, Korea), and BMI was calculated as weight divided by height squared (kg/m2) [19]. Waist circumference was measured by trained nurses at the midpoint between the lower borders of the rib cage and upper pole of the iliac crest. After fasting for 12 h, blood samples were taken for measurement of blood lipids and glucose.
Metabolic syndrome was defined on the basis of the criteria established by the National Cholesterol Education Program (NCEP). The NCEP criteria for metabolic syndrome require that at least three of the following conditions be met: waist circumference >40 in. in men and >35 in. in women, plasma TG >150 mg/dl, HDL cholesterol <40 mg/dl in men and <50 mg/dl in women, blood pressure ≥130/85 mmHg, and fasting plasma glucose ≥110 mg/dl [20].
Endoscopy
EGD was performed using a flexible endoscope (CV-260SL; Olympus, Tokyo, Japan) after overnight fasting. We inspected the gastroesophageal junction at the start of the endoscopic examination before inflation of the stomach or at the end of the examination after deflation of the stomach. The severity of EE was graded from A to D according to the Los Angeles (LA) classification system [21]. Hiatal hernia was considered present if the gastroesophageal junction extended at least 2 cm above the diaphragmatic hiatus impression during quiet respiration [22].
Measurement of Abdominal Adipose Tissue Area by CT
The abdominal adipose tissue area was quantified using 64-multidetector CT (Brilliance; Philips medical systems, Cleveland, OH, USA). The fat area was determined by measuring the mean value of the pixels within the range between −175 and −25 Hounsfield units. The total abdominal fat area (TFA), VFA, and SFA were measured by the acquisition of a 10-cm CT slice scan image of the third and fourth lumbar vertebrae during suspended respiration.
The area (cm2) was calculated using Extended Brilliance Workspace software (version 1-4.5.2, Philips Healthcare, Best, Netherland). VFA was calculated by delineating the intra-abdominal cavity bound by the parietal peritoneum or transversalis fascia, and excluding the vertebral column and paraspinal muscles. SFA was calculated by subtracting VFA from TFA.
Statistical Analysis
Categorical variables were evaluated using χ 2 test or Fisher’s exact test. Continuous variables were evaluated using Student’s t test. Continuous variables were expressed as mean ± SD. A p value of <0.05 was considered statistically significant. The software package SPSS version 20.0 was used for analysis (SPSS Inc., Chicago, IL, USA).
Results
Baseline Demographic and Clinical Characteristics of the Subjects
A total of 728 subjects were included in this study. The mean age of the patients was 47.15 years; 629 (86.4%) patients were male, and 99 (13.6%) were female. The mean BMI was 24.66 kg/m2, and 65 (8.9%) patients had metabolic syndrome. On EGD, 65 (8.9%) patients had EE, and 11 (1.5%) patients had hiatal hernias. On CT, the mean VFA, SFA, and VFA/SFA ratio were 2886.97, 3627.27, and 1.02 cm2, respectively (Table 1).
Clinical Characteristics of Subjects With and Without EE
Among the 728 subjects, 65 (8.9%) patients had EE. The patients with EE were predominantly female (Table 2). The mean BMI was higher in the EE group than in the non-EE group (25.80 vs. 24.40, P = 0.002). Metabolic syndrome was more prevalent in the EE group than in the non-EE group (30.8 vs. 13.7%, P < 0.001). Moreover, TG and blood pressure levels were higher in patients with EE. On EGD, hiatal hernia was more prevalent in the EE group than in the non-EE group (9.2 vs. 0.8%, P < 0.001). However, VFA was not significantly different between the two groups (1595.00 vs. 1426.00 cm2, P = 0.927). Conversely, SFA was higher in the non-EE group than in the EE group (1226.92 vs. 1514.46 cm2, P = 0.010). Moreover, the mean VFS/SFA ratio was higher in the EE group than in the non-EE group (1.30 vs. 0.92, P < 0.001).
Risk Factors for EE
The results of the multivariate logistic regression analysis demonstrated that hiatal hernia (P < 0.001), VFA/SFA ratio ≥1.165 (P = 0.010), and high TG level (P = 0.015) were independent risk factors for EE. Hiatal hernia was strongly associated with an increased risk of EE [adjusted odds ratio (OR), 12.90; 95% confidence interval (CI), 3.57–46.65]. Similarly, a VFA/SFA ratio ≥1.165 was a significant risk factor for EE (OR 2.04; 95% CI 1.18–3.51; Table 3).
Association Between EE and VFA/SFA Ratio
The analysis of the correlation between EE and the VFA/SFA ratio indicated that the incidence of EE was higher in the group with the higher VFA/SFA ratio (P < 0.001; Fig. 1). In addition, the receiver-operating characteristic (ROC) curve indicated that a VFA/SFA ratio ≥1.165 had good accuracy to predict EE (area under the ROC curve = 0.643; Fig. 2).
Effect of VFA/SFA Ratio on the Severity of EE
The severity of EE was positively correlated with the VFA/SFA ratio and VF volume (P = 0.002; Table 4). The risk of EE types LA-A, LA-B, and LA-C/LAC-D increased 1.23-fold, 1.27-fold, and 1.56-fold, respectively. Of note, a VFA/SFA ratio ≥1.165 was strongly correlated with the severity of EE (P < 0.001; Table 5).
Discussion
Our study identified a positive correlation between EE and each of the following individual factors: high BMI, metabolic syndrome, high VFA/SFA ratio, hiatal hernia, high TG level, high blood pressure, and female sex. However, multivariate analysis revealed that only high VFA/SFA ratio, hiatal hernia, and high TG level were associated with EE.
In a previous study, multivariate logistic regression analysis revealed that VFA was associated with EE [18]. Previous studies demonstrated that women have lower VF and higher SF than men [17, 23]. In our study, however, VFA did not show a significant association with EE, as a higher proportion of women might have resulted in a lower VFA than expected in the EE group.
In other studies, multivariate analysis found that high BMI, waist circumference, and waist-to-hip ratio were associated with EE [17, 18]. Although these parameters are easily obtainable, these are imperfect measures of adiposity owing to confounding factors such as sex and ethnic origin. While in some Asian populations the prevalence of obesity is lower than that in western countries, health risks associated with obesity occur at a lower BMI in Asian populations, and these populations are predisposed to abdominal obesity [24].
Several hypotheses have been formulated to explain how obesity can cause GERD [25,26,27]. Previous studies have indicated that obesity can lead to EE via dietary, mechanical, and humoral factors [15, 26]. A previous study has shown that the amount and type of dietary intake are responsible for GERD [28]. Moreover, an increase in abdominal adipose tissue leads to increased intra-abdominal and intragastric pressure, increased rate of transit, lower esophageal sphincter relaxation, formation of hiatal hernia, and subsequent esophageal acid reflux [25]. Humoral and hormonal factors are also involved in the correlation between obesity and GERD. A large cohort study found that compared to men with obesity, women with obesity had a greater risk of GERD symptoms, with the risk being highest in premenopausal women and postmenopausal women undergoing estrogen therapy [29]. Furthermore, VF is strongly associated with elevated serum levels of several proinflammatory cytokines, including interleukin-6 and tumor necrosis factor-alpha, which are overexpressed in patients with EE [30, 31].
Several studies have evaluated obesity by measuring BMI and waist circumference. BMI is associated with increased transient lower esophageal sphincter relaxation [32]. Waist circumference and BMI are significantly correlated with intragastric pressure, gastroesophageal pressure gradient, and separation of the gastroesophageal junction pressure components [33, 34]. However, the association between BMI and GERD is affected by many confounding factors [7, 12, 13, 16]. Moreover, anthropometric measurements are less accurate and less reproducible in obesity studies.
Abdominal visceral adipose tissue, which is associated with increased intra-abdominal pressure, may be a better predictor of GERD than other obesity parameters. The most important mechanism involved in the correlation between obesity with GERD is intra-abdominal pressure, which plays a significant role in acid reflux [35]. Moreover, abdominal CT is more accurate and more reproducible than anthropometry because CT allows for the direct assessment of SF and VF compartments, whereas anthropometric measurements do not. VFA is an indicator of abdominal fat and a strong predictor of insulin resistance and coronary artery disease [33, 36]. However, the high cost and radiation exposure should be considered.
In our study, the VFA/SFA ratio was a more significant risk factor for EE than BMI, waist circumference, waist-to-hip ratio, and VFA. Moreover, we found that a VFA/SFA ratio ≥1.165 might be a useful indicator for predicting EE. In addition, the severity of EE was positively correlated with the VFA/SFA ratio.
Our study has several strengths. First, the methods used allowed for the acquisition of high-quality data. Furthermore, EE was objectively evaluated via EGD and categorized using the Los Angeles classification, and minor changes were excluded to increase specificity. Second, abdominal adipose tissue was measured using 64-multidetector CT, which has a high degree of validity and reproducibility [23]. Furthermore, we suggested cutoff values for normal versus abnormal VFA/SFA ratios.
Our study also has the following limitations. First, the study population was based on subjective screening; therefore, there could have been a selection bias. Second, we did not evaluate individual diet or lifestyle factors such as alcohol consumption and smoking status.
Conclusion
The VFA/SFA ratio was positively associated with EE. The pattern of obesity was more important than BMI, and the VFA/SFA ratio was used in the diagnosis of VF-type abdominal obesity. We suggest that a high VFA/SFA ratio is a useful clinical predictor of EE. Therefore, individuals with a high VFA/SFA ratio may need endoscopic evaluation for EE. Further studies are needed to determine the treatment success of EE based on the VFA/SFA ratios.
References
Dent J, El-Serag HB, Wallander MA, Johansson S. Epidemiology of gastro-oesophageal reflux disease: a systematic review. Gut. 2005;54:710–717.
DeVault KR, Castell DO, American College of Gastroenterology. Updated guidelines for the diagnosis and treatment of gastroesophageal reflux disease. Am J Gastroenterol. 2005;100:190–200.
Moki F, Kusano M, Mizuide M, et al. Association between reflux oesophagitis and features of the metabolic syndrome in Japan. Aliment Pharmacol Ther. 2007;26:1069–1075.
Wong BC, Kinoshita Y. Systematic review on epidemiology of gastroesophageal reflux disease in Asia. Clin Gastroenterol Hepatol. 2006;4:398–407.
Watanabe S, Hojo M, Nagahara A. Metabolic syndrome and gastrointestinal diseases. J Gastroenterol. 2007;42:267–274.
Chua CS, Lin YM, Yu FC, et al. Metabolic risk factors associated with erosive esophagitis. J Gastroenterol Hepatol. 2009;24:1375–1379.
Jacobson BC, Somers SC, Fuchs CS, Kelly CP, Camargo CA Jr. Body-mass index and symptoms of gastroesophageal reflux in women. N Engl J Med. 2006;354:2340–2348.
Corley DA, Kubo A. Body mass index and gastroesophageal reflux disease: a systematic review and meta-analysis. Am J Gastroenterol. 2006;101:2619–2628.
Kim N, Lee SW, Cho SI, et al. The prevalence of and risk factors for erosive oesophagitis and non-erosive reflux disease: a nationwide multicentre prospective study in Korea. Aliment Pharmacol Ther. 2008;27:173–185.
von Hafe P, Pina F, Perez A, Tavares M, Barros H. Visceral fat accumulation as a risk factor for prostate cancer. Obes Res. 2004;12:1930–1935.
Nilsson M, Lundegardh G, Carling L, Ye W, Lagergren J. Body mass and reflux oesophagitis: an oestrogen-dependent association? Scand J Gastroenterol. 2002;37:626–630.
Lagergren J, Bergstrom R, Nyren O. No relation between body mass and gastro-oesophageal reflux symptoms in a Swedish population based study. Gut. 2000;47:26–29.
Furukawa N, Iwakiri R, Koyama T, et al. Proportion of reflux esophagitis in 6010 Japanese adults: prospective evaluation by endoscopy. J Gastroenterol. 1999;34:441–444.
Weinsier RL, Hunter GR, Gower BA, Schutz Y, Darnell BE, Zuckerman PA. Body fat distribution in white and black women: different patterns of intraabdominal and subcutaneous abdominal adipose tissue utilization with weight loss. Am J Clin Nutr. 2001;74:631–636.
El-Serag HB, Graham DY, Satia JA, Rabeneck L. Obesity is an independent risk factor for GERD symptoms and erosive esophagitis. Am J Gastroenterol. 2005;100:1243–1250.
Corley DA, Kubo A, Zhao W. Abdominal obesity, ethnicity and gastro-oesophageal reflux symptoms. Gut. 2007;56:756–762.
Nam SY, Choi IJ, Ryu KH, Park BJ, Kim HB, Nam BH. Abdominal visceral adipose tissue volume is associated with increased risk of erosive esophagitis in men and women. Gastroenterology. 2010;139:1902 e1902–1911 e1902.
Lee HL, Eun CS, Lee OY, et al. Association between erosive esophagitis and visceral fat accumulation quantified by abdominal CT scan. J Clin Gastroenterol. 2009;43:240–243.
Willett WC, Dietz WH, Colditz GA. Guidelines for healthy weight. N Engl J Med. 1999;341:427–434.
Miranda PJ, DeFronzo RA, Califf RM, Guyton JR. Metabolic syndrome: definition, pathophysiology, and mechanisms. Am Heart J. 2005;149:33–45.
Armstrong D, Bennett JR, Blum AL, et al. The endoscopic assessment of esophagitis: a progress report on observer agreement. Gastroenterology. 1996;111:85–92.
Zagari RM, Fuccio L, Wallander MA, et al. Gastro-oesophageal reflux symptoms, oesophagitis and Barrett’s oesophagus in the general population: the Loiano–Monghidoro study. Gut. 2008;57:1354–1359.
Maurovich-Horvat P, Massaro J, Fox CS, Moselewski F, O’Donnell CJ, Hoffmann U. Comparison of anthropometric, area- and volume-based assessment of abdominal subcutaneous and visceral adipose tissue volumes using multi-detector computed tomography. Int J Obes. 2007;31:500–506.
WHO Expert Consultation. Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet. 2004;363:157–163.
Wilson LJ, Ma W, Hirschowitz BI. Association of obesity with hiatal hernia and esophagitis. Am J Gastroenterol. 1999;94:2840–2844.
Ruhl CE, Everhart JE. Overweight, but not high dietary fat intake, increases risk of gastroesophageal reflux disease hospitalization: the NHANES I Epidemiologic Followup Study. First National Health and Nutrition Examination Survey. Ann Epidemiol. 1999;9:424–435.
Chang CS, Poon SK, Lien HC, Chen GH. The incidence of reflux esophagitis among the Chinese. Am J Gastroenterol. 1997;92:668–671.
Hampel H, Abraham NS, El-Serag HB. Meta-analysis: obesity and the risk for gastroesophageal reflux disease and its complications. Ann Intern Med. 2005;143:199–211.
Nilsson M, Johnsen R, Ye W, Hveem K, Lagergren J. Obesity and estrogen as risk factors for gastroesophageal reflux symptoms. JAMA. 2003;290:66–72.
Xu H, Barnes GT, Yang Q, et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest. 2003;112:1821–1830.
Cannon JG, Nerad JL, Poutsiaka DD, Dinarello CA. Measuring circulating cytokines. J Appl Physiol. 1985;1993:1897–1902.
Wu JC, Mui LM, Cheung CM, Chan Y, Sung JJ. Obesity is associated with increased transient lower esophageal sphincter relaxation. Gastroenterology. 2007;132:883–889.
Pandolfino JE, El-Serag HB, Zhang Q, Shah N, Ghosh SK, Kahrilas PJ. Obesity: a challenge to esophagogastric junction integrity. Gastroenterology. 2006;130:639–649.
de Vries DR, van Herwaarden MA, Smout AJ, Samsom M. Gastroesophageal pressure gradients in gastroesophageal reflux disease: relations with hiatal hernia, body mass index, and esophageal acid exposure. Am J Gastroenterol. 2008;103:1349–1354.
Locke GR III, Talley NJ, Fett SL, Zinsmeister AR, Melton LJ III. Prevalence and clinical spectrum of gastroesophageal reflux: a population-based study in Olmsted County, Minnesota. Gastroenterology. 1997;112:1448–1456.
Lee SJ, Song CW, Jeen YT, et al. Prevalence of endoscopic reflux esophagitis among Koreans. J Gastroenterol Hepatol. 2001;16:373–376.
Author’s contribution
BJK involved in study concept and design, data were collected by JGK, HK involved in data analysis and interpretation, and EYZ wrote the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License (http://creativecommons.org/licenses/by-nc/4.0/), which permits any noncommercial use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Ze, E.Y., Kim, B.J., Kang, H. et al. Abdominal Visceral to Subcutaneous Adipose Tissue Ratio Is Associated with Increased Risk of Erosive Esophagitis. Dig Dis Sci 62, 1265–1271 (2017). https://doi.org/10.1007/s10620-017-4467-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-017-4467-4