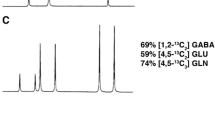
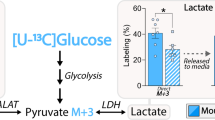
Abstract
Pyruvate carboxylation is of great importance in the brain since it is responsible for adding net carbons to the tricarboxylic acid cycle following removal of carbon backbone for synthesis of the two most abundant neurotransmitters, glutamate and GABA. Despite having such a pivotal role, there is still much uncertainty in the exact metabolic details about where and how this carbon is returned. Pyruvate carboxylation has been studied in various model systems of the brain and 13C magnetic resonance spectroscopy is an excellent tool for doing this. This review will focus on results dealing with the extent and cellular location of pyruvate carboxylation and its role in pathophysiology and concludes that pyruvate carboxylation is an extraordinarily important predominantly astrocytic pathway which plays a pivotal part in a number of diseases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
There are several enzymes involved in carboxylation in the brain (Fig. 1); pyruvate carboxylase (PC), phosphoenolpyruvate carboxykinase (PEPCK), pyruvate kinase and malic enzyme (ME) [1]. Some of these enzymes have a cell specific localization. Only astrocytes have PC and cytosolic ME whereas mitochondrial ME and PEPCK have been found in both neurons and astrocytes [2–9]. The importance of these enzymes is apparent from the severity of disease associated with genetic modifications of these systems. Mitochondrial malic enzyme (ME2) was found to be missing in partially complementing genotypes between lethal deletion alleles at the albino locus in Chromosome 7 of the mouse. Since such partial complementers survive to adulthood, the absence of normal mitochondrial malic enzyme is compatible with life [10]. However, it has been shown that genetic variation in or near the ME2 gene is associated with both psychotic and manic disorders, including schizophrenia and bipolar disorder [11].
Schematic presentation of reactions pertinent to pyruvate carboxylation. This scheme illustrates how the concentrations of pyruvate, oxaloacetate and 2-oxoglutarate, which are normally relatively low, are important in regulating pyruvate carboxylation and how these reactions can restore balance in these compounds (and hence in the TCA cycle). For simplicity, malic enzyme is not included. It can carboxylate malate to pyruvate and can theoretically carry out the reverse reaction. * The bicarbonate pool is fed by all decarboxylation reactions from the tricarboxylic acid cycle. AspAT aspartate amino transferase, AlaAT alanine amino transferase, GDH glutamate dehydrogenase, Lac lactate, LDH lactate dehydrogenase, OAA oxaloacetate, PAG phosphate activated phosphatase, PC pyruvate carboxylase, PEPCK phosphoenolpyruvate carboxykinase, PK pyruvate kinase
Pyruvate carboxylase is a nuclear encoded homotetramer found in most eukaryotic and in many prokaryotic tissues and is a member of the family of biotin-dependent carboxylases [12]. It is also ATP-dependent and catalyses the carboxylation of pyruvate to form oxaloacetate [12]. In mammalian tissues, PC participates in various anabolic pathways including lipogenesis and the synthesis of neurotransmitter substances [12]. Five alternative forms of rat pyruvate carboxylase cDNAs have been identified and these are expressed in a tissue-specific manner in liver, kidney, brain, and adipose tissue [13]. Defects in the expression or biotinylation of pyruvate carboxylase in humans is a rare autosomal recessively inherited disorder of pyruvate metabolism presenting with lactic academia and neurological involvement and almost invariably results in early death or at best a severely debilitating psychomotor retardation [12]. The above clearly reflects the vital role PC plays in intermediary metabolism in many tissues including the brain [14].
13C magnetic resonance spectroscopy (MRS) is an excellent tool for the study of metabolic pathways of glucose and other substrates. Using this method and 13C labeled glucose it could be shown that pyruvate carboxylation occurs in humans, rats and mice in vivo [15–17]. In experiments where slices were superfused with 13C labeled glucose the evidence from MRS analysis of glutamate suggested that pyruvate carboxylation in slices is minimal [18–20]. However, using 13C and 15N labeled alanine it could be shown that carboxylation was taking place in astrocytes in brain slices [21]. Most cell culture studies also showed that carboxylation was a glial pathway [22].
Discussion
It has been shown that there is a moderate efflux of glutamine from the human brain [23, 24] and thus there must be an anaplerotic process that compensates for this since it is essentially a loss of α-ketoglutarate (via glutamate) from the TCA cycle and would lead to a stop of this cycle unless another intermediate is channeled into the cycle. This intermediate is typically oxaloacetate produced by pyruvate carboxylation [1]. To measure the anaplerotic contribution, Mason et al. [15] measured 13C incorporation into glutamate and glutamine in the occipital-parietal region of awake humans while infusing [2-13C]glucose, which labels the C2 and C3 positions of glutamine and glutamate via pyruvate carboxylase (and the pentose phosphate shunt). Metabolic modeling of the labeling data indicated that pyruvate carboxylase flux is used for replacing glutamate lost due to glutamine efflux and therefore can be considered to support neurotransmitter trafficking. Oz et al. [26] have reported a high pyruvate carboxylase rate of approximately 0.14–0.18 μmol/gm/min contributing to the glial TCA cycle flux.
Is Pyruvate Carboxylation an Astrocyte Specific Pathway?
Even though it has been shown that pyruvate carboxylation in the brain is carried out primarily by the glial specific enzyme PC and only to a minor extent by ME and PEPCK [1] there is some debate about the cellular location of pyruvate carboxylation. 13C MRS studies in rats injected with 13C labeled lactate, predominantly a neuronal substrate [27], showed that lactate was metabolized in a compartment without measurable pyruvate carboxylation [28, 29]. This can be interpreted as indicating that carboxylation is taking place in astrocytes. However, Hassel and Brathe [30] concluded from experiments were large amounts of lactate were infused that pyruvate was carboxylated in neurons. The reason for this discrepancy is not known.
In brain tissue slices carboxylation was also detected in astrocytes [31, 32]. Guinea pig cortical tissue slices were incubated with glucose and alanine which was labeled in the C-2 position with 13C and the amino nitrogen was 15N [21]. The 15N coupling could be seen in the 13C MR spectrum [21]. The authors observed [2-13C, 15N]aspartate, but no [2-13C]aspartate was detected [21]. There was very little dilution of the 15N which would have had to be passed to a glutamate intermediate in order to move from alanine to aspartate. There was no detection of [3-13C]aspartate above natural abundance, which would have been found 50/50 with [2-13C]aspartate if the 13C had been incorporated into aspartate via pyruvate deghydrogenase [21]. These data and those of others suggest that alanine is taken up into an astrocytic compartment [21, 33–35]. Furthermore, it is clear from the results with [2-13C, 15N]alanine that aminotransferase and aspartate aminotransferase are in very tight association with a pyruvate carboxylating enzyme (Fig. 1).
Using primary mono cultures of neurons and astrocytes it is possible to obtain information about the cellular location and magnitude of pyruvate carboxylation. Using [1-13C]glucose or [2-13C]acetate as substrates in cultures of cortical astrocytes and GABAergic neurons Sonnewald et al. [36] showed pyruvate carboxylation in astrocytes but not in neurons However, it should be noted that the interpretation of results obtained in these studies is complicated since oxaloacetate can be converted to citrate directly or via several steps to fumarate and subsequent oxaloacetate and citrate (back cycling) [37]. The latter pathway will lead to scrambling of label due to the symmetrical fumarate molecule. Merle et al. [37] suggested incomplete back cycling for astrocytes and complete back cycling for cerebellar neurons. If back cycling in neurons is indeed complete, the study above, utilizing [1-13C]glucose, was not capable of showing carboxylation in neurons. Waagepetersen et al. [22] have performed a study using [U-13C]glucose plus [U-13C]lactate and 3-nitropropionic acid (3-NPA) to specifically block the TCA cycle at the succinate dehydrogenase step [38–40]. In this way multiple cycling of TCA cycle constituents was avoided and carboxylation would be clearly detected in labeling of the C-3 position, regardless of back cycling. Analyzing the labeling patterns in amino acids, pyruvate carboxylation was detected in astrocytes but not in neurons [22]. The authors stated: “Based on the present results it may be safely concluded that neuronal pyruvate carboxylation is unlikely to play an important quantitative role. Therefore it is also unlikely to be able to sustain release of transmitter glutamate in cerebellar granule neurons, a process which is quantitatively demanding.”
What Factors Affect Pyruvate Carboxylation?
Depolarization, depresses transmission in glutamatergic neurons, activates astrocytes but has less effect on GABAergic neurons [41–44]. Using brain slices it was shown that depolarization did not affect carboxylation [25]. This indicates that a mostly astrocytic workload doesn’t involve increased pyruvate carboxylation. Furthermore, the presence of GABAB or GABAC ligands had no effect on pyruvate carboxylation in brain slices [45, 46].
Increased Carboxylation
Zwingman [47] showed that increased ammonia concentration increased pyruvate carboxylation both in astrocytes in culture and in animal models. This carboxylation might be coupled to the increased glutamine formation which is a hallmark of ammonia detoxification [47]. Glutamine formation is the only efficient method of ammonia fixation in brain.
The effect of ketone bodies on pyruvate carboxylation by rat brain mitochondria has been investigated [1]. Melo et al. [48] showed that pyruvate carboxylation increased after rats were fed a ketogenic diet. This increase in carboxylation was coupled to an increased pyruvate recycling.
Activation of the glutamatergic AMPA receptor induces large post-synaptic action potentials [49] and should therefore stimulate release of neurotransmitter glutamate. In experiments with brain tissue slices metabolizing [1-13C]glucose it was possible to show evidence of pyruvate carboxylation in aspartate [50]. Increased carboxylation was detected when the slices were incubated with AMPA or exogenous glutamate [50] suggesting that increased neuronal work increases carboxylation. These results contrast with the above mentioned results using K+ depolarization where no increased carboxylation was detected and where the workload is greater in astrocytes. Interestingly, inhibition of the monocarboxylate transporters MCT1 and MCT2 using AR-C122982 at 10 nM increased carboxylation significantly but increasing the inhibition further by using 100 nM AR-C122982 reduced carboxylation [50].
Decreased Carboxylation
Decreased carboxylation has been reported after pentylenetetrazole (PTZ) injection in rats [51]. PTZ administration is generally used as a seizure model, but when low doses are used it can cause anxiety [51]. Interestingly, the antiepileptic drug carbamazepine (CBZ) decreased biotin concentration in both humans and rats and decreased pyruvate carboxylase activity in rat brain [52]. These results support the use of biotin supplementation as a concurrent strategy during CBZ administration to help maintain pyruvate carboxylation and other important metabolic pathways.
Not surprisingly, ischemia has a profound effect on carboxylation, a pathway dependent on ATP concentration. In rats subjected to 120 min of middle cerebral artery occlusion followed by 120 min of reperfusion it could be shown that use of astrocytic precursors originating from the pyruvate carboxylase pathway was markedly reduced compared to the pyruvate dehydrogenase pathway in glutamate, and completely stopped in GABA in the ischemic core area [53]. Furthermore, in the reperfused penumbra glutamatergic and GABAergic neurons used relatively more astrocytic metabolites derived from the pyruvate carboxylase pathway than in the ischemic core [53]. The pyrrolopyrimidine lipid peroxidation inhibitor, U-101033E was administered to rats after middle cerebral artery occlusion and was shown to specifically decrease mitochondrial pyruvate metabolism via both pyruvate dehydrogenase and pyruvate carboxylase pathways [54]. In the canine cardiac arrest model of global ischemia carboxylation was decreased in the hippocampus in animals that had undergone hyperoxic resuscitation [55]. Scafidi et al. [56], showed that labeling via PC was significantly decreased after traumatic brain injury in immature rats (a model of pediatric traumatic brain injury).
Qu et al. [57] showed that exogenous glutamate had an inhibiting effect on pyruvate carboxylation in astrocytes, presumably by formation of oxaloacetate from 2-oxoglutarate derived from glutamate. Inhibition of pyruvate carboxylation was also observed when glutamine was added to astrocytes [58]. When metabolism of 13C labeled glucose and lactate was investigated in cultured mouse cerebellar astrocytes, it could be shown that carboxylation of pyruvate using glucose as the precursor was more important for biosynthesis of releasable glutamine and citrate, compared with their intracellular pools [59].
Incubation of brain slices with increasing amounts of the solvent dimethylsulphoxide (DMSO) which induces a workload on the mitochondria via an unknown mechanism showed decreased carboxylation of aspartate with increasing amounts of DMSO [60]. This was the case despite increasing flux through the TCA cycle and increased incorporation of label into glutamine via pyruvate dehydrogenase [60]. Thus, where the increased activity was not due to glutamatergic load, there was no increase (indeed there was a decrease) in pyruvate carboxylation.
Conclusion
Taken together, these data support the assumption that anaplerotic activity is increased due to glutamatergic activity but that loss of four carbon molecules due to GABAergic, mitochondrial or astrocytic activity is minimal, due to the reuptake of GABA into the synaptic terminal. It can be concluded that pyruvate carboxylation is an extraordinarily important predominantly astrocytic pathway.
References
Patel MS (1974) The effect of ketone bodies on pyruvate carboxylation by rat brain mitochondria. J Neurochem 23:865–867
Yu AC, Drejer J, Hertz L, Schousboe A (1983) Pyruvate carboxylase activity in primary cultures of astrocytes and neurons. J Neurochem 41:1484–1487
Shank RP, Bennett GS, Freytag SO, Campbell GL (1985) Pyruvate carboxylase: an astrocyte-specific enzyme implicated in the replenishment of amino acid neurotransmitter pools. Brain Res 329:364–367
Cesar M, Hamprecht B (1995) Immunocytochemical examination of neural rat and mouse primary cultures using monoclonal antibodies raised against pyruvate carboxylase. J Neurochem 64:2312–2318
Vogel R, Hamprecht B, Wiesinger H (1998) Malic enzyme isoforms in astrocytes: comparative study on activities in rat brain tissue and astroglia-rich primary cultures. Neurosci Lett 247:123–126
Vogel R, Jennemann G, Seitz J, Wiesinger H, Hamprecht B (1998) Mitochondrial malic enzyme: purification from bovine brain, generation of an antiserum, and immunocytochemical localization in neurons of rat brain. J Neurochem 71:844–852
Kurz GM, Wiesinger H, Hamprecht B (1993) Purification of cytosolic malic enzyme from bovine brain, generation of monoclonal antibodies, and immunocytochemical localization of the enzyme in glial cells of neural primary cultures. J Neurochem 60:1467–1474
McKenna MC, Stevenson JH, Huang X, Tildon JT, Zielke CL, Hopkins IB (2000) Mitochondrial malic enzyme activity is much higher in mitochondria from cortical synaptic terminals compared with mitochondria from primary cultures of cortical neurons or cerebellar granule cells. Neurochem Int 36:451–459
McKenna MC, Tildon JT, Stevenson JH, Huang X, Kingwell KG (1995) Regulation of mitochondrial and cytosolic malic enzymes from cultured rat brain astrocytes. Neurochem Res 20:1491–1501
Eicher EM, Lewis SE, Turchin HA, Gluecksohn-Waelsch S (1978) Absence of mitochondrial malic enzyme in mice carrying two complementing lethal albino alleles. Genet Res 32:1–7
Lee BD, Walss-Bass C, Thompson PM, Dassori A, Montero PA, Medina R, Contreras S, Armas R, Ramirez M, Pereira M, Salazar R, Leach RJ, Quezada P, Raventos H, Escamilla MA (2007) Malic enzyme 2 and susceptibility to psychosis and mania. Psychiatry Res 150:1–11
Wallace JC, Jitrapakdee S, Chapman-Smith A (1998) Pyruvate carboxylase. Int J Biochem Cell Biol 30:1–5
Jitrapakdee S, Gong Q, MacDonald MJ, Wallace JC (1998) Regulation of rat pyruvate carboxylase gene expression by alternate promoters during development, in genetically obese rats and in insulin-secreting cells. Multiple transcripts with 5’-end heterogeneity modulate translation. J Biol Chem 273:34422–34428
Schiff M, Levrat V, Acquaviva C, Vianey-Saban C, Rolland MO, Guffon N (2006) A case of pyruvate carboxylase deficiency with atypical clinical and neuroradiological presentation. Mol Genet Metab 87:175–177
Mason GF, Petersen KF, de Graaf RA, Shulman GI, Rothman DL (2007) Measurements of the anaplerotic rate in the human cerebral cortex using 13C magnetic resonance spectroscopy and [1-13C] and [2-13C] glucose. J Neurochem 100:73–86
Shank RP, Leo GC, Zielke HR (1993) Cerebral metabolic compartmentation as revealed by nuclear magnetic resonance analysis of D-[1-13C]glucose metabolism. J Neurochem 61:315–323
Hassel B, Sonnewald U, Fonnum F (1995) Glial-neuronal interactions as studied by cerebral metabolism of [2-13C]acetate and [1-13C]glucose: an ex vivo 13C NMR spectroscopic study. J Neurochem 64:2773–2782
Lukkarinen J, Oja JME, Turunen M, Kauppinen RA (1997) Quantitative determination of glutamate turnover by H-1-observed, C-13-edited nuclear magnetic resonance spectroscopy in the cerebral cortex ex vivo: Interrelationships with oxygen consumption. Neurochem Int 31:95–104
Badar-Goffer R, Bachelard H, Morris P (1990) Cerebral metabolism of acetate and glucose studied by 13C NMR spectroscopy. Biochem J 266:133–139
Griffin JL, Rae C, Radda GK, Matthews PM (1999) Lactate-induced inhibition of glucose catabolism in guinea pig cortical brain slices. Neurochem Int 35:405–409
Rae C, Hansen JT, Bubb WA, Bröer S, Bröer A (2005) Alanine transport, metabolism and cycling in the brain. Proc Int Soc Magn Reson Med 2481
Waagepetersen HS, Qu H, Schousboe A, Sonnewald U (2001) Elucidation of the quantitative significance of pyruvate carboxylation in cultured cerebellar neurons and astrocytes. J Neurosci Res 66:763–770
Strauss GI, Knudsen GM, Kondrup J, Moller K, Larsen FS (2001) Cerebral metabolism of ammonia and amino acids in patients with fulminant hepatic failure. Gastroenterology 121:1109–1119
Grill V, Bjorkman O, Gutniak M, Lindqvist M (1992) Brain uptake and release of amino acids in nondiabetic and insulin-dependent diabetic subjects: important role of glutamine release for nitrogen balance. Metabolism 41:28–32
Taylor A, McLean M, Morris P, Bachelard H (1996) Approaches to studies on neuronal glial relationships by C-13-MRS analysis. Dev Neurosci 18:434–442
Oz G, Berkich DA, Henry PG, Xu Y, LaNoue K, Hutson SM, Gruetter R (2004) Neuroglial metabolism in the awake rat brain: CO2 fixation increases with brain activity. J Neurosci 24:11273–11279
Waagepetersen HS, Bakken IJ, Larsson OM, Sonnewald U, Schousboe A (1998) Comparison of lactate and glucose metabolism in cultured neocortical neurons and astrocytes using 13C-NMR spectroscopy. Dev Neurosci 20:310–320
Qu H, Haberg A, Haraldseth O, Unsgard G, Sonnewald U (2000) (13)C MR spectroscopy study of lactate as substrate for rat brain. Dev Neurosci 22:429–436
Bouzier-Sore AK, Canioni P, Merle M (2001) Effect of exogenous lactate on rat glioma metabolism. J Neurosci Res 65:543–548
Hassel B, Brathe A (2000) Cerebral metabolism of lactate in vivo: evidence for neuronal pyruvate carboxylation. J Cereb Blood Flow Metab 20:327–336
Griffin JL, Keun H, Moskau D, Rae C, Nicholson JK (2003) Compartmentation of metabolism probed by [2-13C]alanine: Improved 13C NMR sensitivity using a CryoProbe detects evidence of a glial metabolon. Neurochem Int 42:93–99
Griffin JL, Rae C, Dixon RM, Radda GK, Matthews PM (1998) Excitatory amino acid synthesis in hypoxic brain slices: does alanine act as a substrate for glutamate production in hypoxia? J Neurochem 71:2477–2486
Waagepetersen HS, Sonnewald U, Larsson OM, Schousboe A (2000) A possible role of alanine for ammonia transfer between astrocytes and glutamatergic neurons. J Neurochem 75:471–479
Zwingmann C, Richter-Landsberg C, Brand A, Leibfritz D (2000) NMR spectroscopic study on the metabolic fate of [3-13C]alanine in astrocytes, neurons and cocultures: implications for glia-neuron interactions in neurotransmitter metabolism. Glia 32:286–303
Zwingmann C, Richter-Landsberg C, Leibfritz D (2001) 13C isotopomer analysis of glucose and alanine metabolism reveals cytosolic pyruvate compartmentation as part of energy metabolism in astrocytes. Glia 34:200–212
Sonnewald U, Westergaard N, Hassel B, Muller TB, Unsgard G, Fonnum F, Hertz L, Schousboe A, Petersen SB (1993) NMR spectroscopic studies of 13C acetate and 13C glucose metabolism in neocortical astrocytes: evidence for mitochondrial heterogeneity. Dev Neurosci 15:351–358
Merle M, Bouzier-Sore AK, Canioni P (2002) Time-dependence of the contribution of pyruvate carboxylase versus pyruvate dehydrogenase to rat brain glutamine labelling from [1-(13) C]glucose metabolism. J Neurochem 82:47–57
Alston TA, Mela L, Bright HJ (1977) 3-Nitropropionate, the toxic substance of Indigofera, is a suicide inactivator of succinate dehydrogenase. Proc Natl Acad Sci USA 74:3767–3771
Sonnewald U, White LR, Odegard E, Westergaard N, Bakken IJ, Aasly J, Unsgard G, Schousboe A (1996) MRS study of glutamate metabolism in cultured neurons/glia. Neurochem Res 21:987–993
Bakken IJ, Johnsen SF, White LR, Unsgard G, Aasly J, Sonnewald U (1997) NMR spectroscopy study of the effect of 3-nitropropionic acid on glutamate metabolism in cultured astrocytes. J Neurosci Res 47:642–649
Shin DS, Samoilova M, Cotic M, Zhang L, Brotchie JM, Carlen PL (2007) High frequency stimulation or elevated K+ depresses neuronal activity in the rat entopeduncular nucleus. Neuroscience 149:68–86
Meeks JP, Mennerick S (2004) Selective effects of potassium elevations on glutamate signaling and action potential conduction in hippocampus. J Neurosci 24:197–206
Meeks JP, Mennerick S (2007) Astrocyte membrane responses and potassium accumulation during neuronal activity. Hippocampus 17:1100–1108
Kettenmann H, Banati R, Walz W (1993) Electrophysiological behavior of microglia. Glia 7:93–101
Nasrallah F, Griffin JL, Balcar VJ, Rae C (2007) Understanding your inhibitions. Modulation of brain cortical metabolism by GABA-B receptors. J Cereb Blood Flow Metab 27:1510–1520
Rae C, Nasrallah FA, Griffin JL, Balcar VJ (2009) Now I know my ABC. A systems neurochemistry and functional metabolomic approach to understanding the GABAergic system. J Neurochem 109(Suppl 1):109–116
Zwingmann C (2007) The anaplerotic flux and ammonia detoxification in hepatic encephalopathy. Metab Brain Dis 22:235–24948
Melo TM, Nehlig A, Sonnewald U (2006) Neuronal-glial interactions in rats fed a ketogenic diet. Neurochem Int 48:498–507
Rao VR, Finkbeiner S (2007) NMDA and AMPA receptors: old channels, new tricks. Trends Neurosci 30:284–291
Rae C, Nasrallah F, Bröer S (2009) Metabolic effects of blocking lactate transport in brain cortical tissue slices using an inhibitor specific to MCT1 and MCT2. Neurochem Res 34:1783–1791
Eloqayli H, Dahl CB, Gotestam KG, Unsgard G, Sonnewald U (2004) Changes of glial-neuronal interaction and metabolism after a subconvulsive dose of pentylenetetrazole. Neurochem Int 45:739–745
Rathman SC, Gregory JF III, McMahon RJ (2003) Pharmacological biotin supplementation maintains biotin status and function in rats administered dietary carbamazepine. J Nutr 133:2857–2862
Haberg A, Qu H, Sonnewald U (2006) Glutamate and GABA metabolism in transient and permanent middle cerebral artery occlusion in rat: importance of astrocytes for neuronal survival. Neurochem Int 48:531–540
Haberg A, Qu H, Hjelstuen MH, Sonnewald U (2007) Effect of the pyrrolopyrimidine lipid peroxidation inhibitor U-101033E on neuronal and astrocytic metabolism and infarct volume in rats with transient middle cerebral artery occlusion. Neurochem Int 50:932–940
Richards EM, Fiskum G, Rosenthal RE, Hopkins I, McKenna MC (2007) Hyperoxic reperfusion after global ischemia decreases hippocampal energy metabolism. Stroke 38(5):1578–1584
Scafidi S, O’Brien J, Hopkins I, Robertson C, Fiskum G, McKenna M (2009) Delayed cerebral oxidative glucose metabolism after traumatic brain injury in young rats. J Neurochem 109(Suppl 1):189–197
Qu H, Eloqayli H, Unsgard G, Sonnewald U (2001) Glutamate decreases pyruvate carboxylase activity and spares glucose as energy substrate in cultured cerebellar astrocytes. J Neurosci Res 66:1127–1132
Hassel B, Sonnewald U (2002) Effects of potassium and glutamine on metabolism of glucose in astrocytes. Neurochem Res 27:167–171
Waagepetersen HS, Sonnewald U, Larsson OM, Schousboe A (2001) Multiple compartments with different metabolic characteristics are involved in biosynthesis of intracellular and released glutamine and citrate in astrocytes. Glia 35:246–252
Nasrallah F, Garner B, Ball GE, Rae C (2008) Modulation of brain metabolism by very low concentrations of the commonly used drug delivery vehicle dimethyl sulfoxide (DMSO). J Neurosci Res 86:208–214
Acknowledgments
It has been a great pleasure to serve on the editorial board of Neurochemical Research and to be able to honor Dr. Lajtha with this article to celebrate his 35 years as editor of this Journal.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue article in honor of Dr. Abel Lajtha
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Sonnewald, U., Rae, C. Pyruvate Carboxylation in Different Model Systems Studied by 13C MRS. Neurochem Res 35, 1916–1921 (2010). https://doi.org/10.1007/s11064-010-0257-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-010-0257-5