Abstract
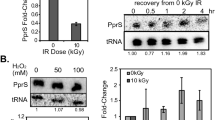
The β-subunit of RNA polymerase, which is involved in rifampin binding, is highly conserved among prokaryotes, and Rifr mutants detected in many bacteria are the result of amino acid changes. Spontaneous rifampin resistance mutations resulting in amino acid replacement (L420R) and deletion (1258–66 9 bp deletion) have been previously isolated in the rpoB gene of Deinococcus radiodurans. In this study, a β-subunit mutation in D. radiodurans resulted in a unique effect on growth rate. We used DNA microarrays and biochemical assays to investigate how the Rifr mutation in the β-subunit led to changes in growth rate via altered regulation of multiple genes. The expression of genes with predicted functions in metabolism, cellular processes and signaling, and information storage and processing were significantly altered in the 9 bp-deletion rpoB mutant. The consensus promoter sequence of up-regulated genes in the 9 bp-deletion rpoB mutant was identified as an AT-rich sequence. Greater levels of reactive oxygen species accumulated in the L420R and 9 bp-deletion rpoB mutants compared with wild type. These results provide insight into the molecular mechanism of how the β-subunit Rifr mutation alters the regulation of multiple genes.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Garibyan L, Huang T, Kim M, et al. Use of the rpoB gene to determine the specificity of base substitution mutations on the Escherichia coli chromosome. DNA Repair (Amst), 2003, 2: 593–608 10.1016/S1568-7864(03)00024-7, 1:CAS:528:DC%2BD3sXjtFWhtrs%3D
Kim M, Wolff E, Huang T, et al. Developing a genetic system in Deinococcus radiodurans for analyzing mutations. Genetics, 2004, 166: 661–668 15020457, 10.1534/genetics.166.2.661, 1:CAS:528:DC%2BD2cXjtlynsbg%3D
Zeibell K, Aguila S, Yan S V, et al. Mutagenesis and repair in Bacillus anthracis: the effect of mutators. J Bacteriol, 2007, 189: 2331–2338 17220233, 10.1128/JB.01656-06, 1:CAS:528:DC%2BD2sXjt1Gksro%3D
Tanaka M, Narumi I, Funayama T, et al. Characterization of pathways dependent on the uvsE, uvrA1, or uvrA2 gene product for UV resistance in Deinococcus radiodurans. J Bacteriol, 2005, 187: 3693–3697 15901692, 10.1128/JB.187.11.3693-3697.2005, 1:CAS:528:DC%2BD2MXks12nt7c%3D
Hua X, Huang L, Tian B, et al. Involvement of recQ in the ultraviolet damage repair pathway in Deinococcus radiodurans. Mutat Res, 2008, 641: 48–53 18343459, 1:CAS:528:DC%2BD1cXlslagtbk%3D
Liu X, Wu J, Zhang W, et al. Resistance of Deinococcus radiodurans to mutagenesis is facilitated by pentose phosphate pathway in the mutS1 mutant background. Curr Microbiol, 2008, 57: 66–71 18389309, 10.1007/s00284-008-9154-4, 1:CAS:528:DC%2BD1cXmtValsbs%3D
Mennecier S, Coste G, Servant P, et al. Mismatch repair ensures fidelity of replication and recombination in the radioresistant organism Deinococcus radiodurans. Mol Genet Genomics, 2004, 272: 460–469 15503140, 10.1007/s00438-004-1077-6, 1:CAS:528:DC%2BD2cXhtVSqs7bK
Wehrli W, Knusel F, Schmid K, et al. Interaction of rifamycin with bacterial RNA polymerase. Proc Nat Acad Sci USA, 1968, 61: 667–673 4879400, 10.1073/pnas.61.2.667, 1:CAS:528:DyaF1MXpsFKr
Jin D J, Gross C A. Characterization of the pleiotropic phenotypes of rifampin-resistant rpoB mutants of Escherichia coli. J Bacteriol, 1989, 171: 5229–5231 2670912, 1:CAS:528:DyaL1MXltlSmsLo%3D
Maughan H, Galeano B, Nicholson W L. Novel rpoB mutations conferring rifampin resistance on Bacillus subtilis: global effects on growth, competence, sporulation, and germination. J Bacteriol, 2004, 186: 2481–2486 15060052, 10.1128/JB.186.8.2481-2486.2004, 1:CAS:528:DC%2BD2cXjsV2isrg%3D
Lai C, Xu J, Tozawa Y, et al. Genetic and physiological characterization of rpoB mutations that activate antibiotic production in Streptomyces lividans. Microbiology, 2002, 148: 3365–3373 12427928, 1:CAS:528:DC%2BD38Xpt1eksbc%3D
Elisabetta C, Clelia P, Salvatore T, et al. Phenotypes and gene expression profiles of Saccharopolyspora erythraea rifampicin-resistant (rif) mutants affected in erythromycin production. Microbial Cell Factories, 2009, 8
Perkins A E, Nicholson W L. Uncovering new metabolic capabilities of Bacillus subtilis using phenotype profiling of rifampin-resistant rpoB mutants. J Bacteriol, 2008, 190: 807–814 17644585, 10.1128/JB.00901-07, 1:CAS:528:DC%2BD1cXhsFShu7o%3D
Hua X T, Wang C, Huang L, et al. Mutation rate and spectrum of spontaneous mutations of Deinococcus radiodurans under rifampin stress. J Nucl Agric Sci, 2010, 24: 1166–1171
Kahnert A, Vermeij P, Wietek C, et al. The ssu locus plays a key role in organosulfur metabolism in Pseudomonas putida S-313. J Bacteriol, 2000, 182: 2869–2878 10781557, 10.1128/JB.182.10.2869-2878.2000, 1:CAS:528:DC%2BD3cXivFKqsLc%3D
Mattimore V, Udupa K S, Berne G A, et al. Genetic characterization of forty ionizing radiation-sensitive strains of Deinococcus radiodurans: linkage information from transformation. J Bacteriol, 1995, 177: 5232–5237 7665511, 1:CAS:528:DyaK2MXotVCjs7g%3D
Chen H, Huang L, Hua X, et al. Pleiotropic effects of recQ in Deinococcus radiodurans. Genomics, 2009, 94: 333–340 19679178, 10.1016/j.ygeno.2009.08.001, 1:CAS:528:DC%2BD1MXht1OhurzF
Rouillard J-M, Zuker M, Gulari E. OligoArray 2.0: design of oligonucleotide probes for DNA microarrays using a thermodynamic approach. Nucleic Acids Res, 2003, 31: 3057–3062 12799432, 10.1093/nar/gkg426, 1:CAS:528:DC%2BD3sXks1WqtLo%3D
Smyth G K. Limma: Linear Models for Microarray Data. Bioinformatics and Computational Biology Solutions Using R and Bioconductor. New York: Springer, 2005. 397–420 10.1007/0-387-29362-0_23
Yang Y H, Dudoit S, Luu P, et al. Normalization for cDNA microarray data: a robust composite method addressing single and multiple slide systematic variation. Nucleic Acids Res, 2002, 30: e15 11842121, 10.1093/nar/30.4.e15
Smyth G K, Michaud J, Scott H S. Use of within-array replicate spots for assessing differential expression in microarray experiments. Bioinformatics, 2005, 21: 2067–2075 15657102, 10.1093/bioinformatics/bti270, 1:CAS:528:DC%2BD2MXjsl2nsLo%3D
Bailey T L, Williams N, Misleh C, et al. MEME: discovering and analyzing DNA and protein sequence motifs. Nucleic Acids Res, 2006, 34: W369–W373 16845028, 10.1093/nar/gkl198, 1:CAS:528:DC%2BD28Xps1yiurc%3D
Crooks G E, Hon G, Chandonia J M, et al. WebLogo: a sequence logo generator. Genome Res, 2004, 14: 1188–1190 15173120, 10.1101/gr.849004, 1:CAS:528:DC%2BD2cXkvFGht7Y%3D
Gupta A, Rosenberger S F, Bowden G T. Increased ROS levels con tribute to elevated transcription factor and MAP kinase activities in malignantly progressed mouse keratinocyte cell lines. Carcinogenesis, 1999, 20: 2063–2073 10545407, 10.1093/carcin/20.11.2063, 1:CAS:528:DyaK1MXns1Kjsb0%3D
Andrews S C, Robinson A K, Rodriguez-Quinones F. Bacterial iron homeostasis. Fems Microbiol Rev, 2003, 27: 215–237 12829269, 10.1016/S0168-6445(03)00055-X, 1:CAS:528:DC%2BD3sXksVOlt70%3D
Katz C, Ron E Z. Dual role of ftsH in regulating lipopolysaccharide biosynthesis in Escherichia coli. J Bacteriol, 2008, 190: 7117–7122 18776015, 10.1128/JB.00871-08, 1:CAS:528:DC%2BD1cXhtlajtLvM
Onishi H R, Pelak B A, Gerckens L S, et al. Antibacterial agents that inhibit lipid a biosynthesis. Science, 1996, 274: 980–982 8875939, 10.1126/science.274.5289.980, 1:CAS:528:DyaK28XmslKmsLw%3D
Gottschalk G. Bacterial Metabolism. 2nd ed. New York: Springer, 1986 10.1007/978-1-4612-1072-6
Taccone-Galluci M, Lubrano R, Trapasso E, et al. Oxidative damage to RBC membranes and pentose phosphate shunt activity in hemodialysis patients after suspension of erythropoietin treatment. ASAIO J, 1994, 40: M663–M666 8555597, 10.1097/00002480-199407000-00081, 1:STN:280:DyaK287ksFCnsw%3D%3D
Pandolfi P P, Sonati F, Rivi R, et al. Targeted disruption of the housekeeping gene encoding glucose 6-phosphate dehydrogenase (G6PD): G6PD is dispensable for pentose synthesis but essential for defense against oxidative stress. EMBO J, 1995, 14: 5209–5215 7489710, 1:CAS:528:DyaK2MXps1Kmsbk%3D
Bjorkman J, Nagaev I, Berg O G, et al. Effects of environment on compensatory mutations to ameliorate costs of antibiotic resistance. Science, 2000, 287: 1479–1482 10688795, 10.1126/science.287.5457.1479, 1:CAS:528:DC%2BD3cXhsV2qt7Y%3D
Barker M M, Gaal T, Gourse R L. Mechanism of regulation of transcription initiation by ppGpp. II. Models for positive control based on properties of RNAP mutants and competition for RNAP. J Mol Biol, 2001, 305: 689–702 11162085, 10.1006/jmbi.2000.4328, 1:CAS:528:DC%2BD3MXjvFylsw%3D%3D
Bramhill D, Kornberg A. A model for initiation at origins of DNA replication. Cell, 1988, 54: 915–918 2843291, 10.1016/0092-8674(88)90102-X, 1:CAS:528:DyaL1cXlvVCgur0%3D
Schmid A K, Howell H A, Battista J R, et al. Global transcriptional and proteomic analysis of the sig1 heat shock regulon of Deinococcus radiodurans. J Bacteriol, 2005, 187: 3339–3351 15866918, 10.1128/JB.187.10.3339-3351.2005, 1:CAS:528:DC%2BD2MXkt1CksrY%3D
Schmid A K, Lidstrom M E. Involvement of two putative alternative sigma factors in stress response of the radioresistant bacterium Deinococcus radiodurans. J Bacteriol, 2002, 184: 6182–6189 12399488, 10.1128/JB.184.22.6182-6189.2002, 1:CAS:528:DC%2BD38XosVartLg%3D
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Hua, X., Wang, H., Wang, C. et al. Global effect of an RNA polymerase β-subunit mutation on gene expression in the radiation-resistant bacterium Deinococcus radiodurans. Sci. China Life Sci. 54, 854–862 (2011). https://doi.org/10.1007/s11427-011-4209-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4209-3