Abstract
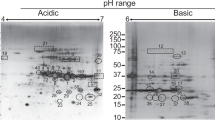
Infection with Brucella causes brucellosis, a chronic disease in humans, which induces abortion and sterility in livestock. Among the different Brucella species, Brucella melitensis is considered the most virulent and is the predominant species associated with outbreaks in China. To date, no safe human vaccine is available against Brucella infection. The currently used live vaccines against Brucella in livestock induce antibodies that interfere with the diagnosis of field infection in vaccinated animals, which is harmful to eradication programs. However, there is as yet no complete profile of immunogenic proteins of B. melitensis. Towards the development of a safer, equally efficacious, and field infection-distinguishable vaccine, we used immunoproteomics to identify novel candidate immunogenic proteins from B. melitensis M5. Eighty-eight immunoreactive protein spots from B. melitensis M5 were identified by Western blotting and were assigned to sixty-one proteins by mass spectrometry, including many new immunoreactive proteins such as elongation factor G, F0F1 ATP synthase subunit beta, and OMP1. These provide many candidate immunoreactive proteins for vaccine development.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Corbel M J. Brucellosis: an overview. Emerg Infect Dis, 1997, 3: 213–221, 9204307, 10.3201/eid0302.970219, 1:STN:280:DyaK2szkvVamtA%3D%3D
Deqiu S, Donglou X, Jiming Y. Epidemiology and control of brucellosis in China. Vet microbiol, 2002, 90: 165–182, 12414142, 10.1016/S0378-1135(02)00252-3
Jahans K L, Foster G, Broughton E S. The characterisation of Brucella strains isolated from marine mammals. Vet Microbiol, 1997, 57: 373–382, 9444074, 10.1016/S0378-1135(97)00118-1, 1:CAS:528:DyaK2sXotVOnt7k%3D
Khan A S, Mujer C V, Alefantis T G, et al. Proteomics and bioinformatics strategies to design countermeasures against infectious threat agents. J Chem Inf Model, 2006, 46: 111–115, 16426046, 10.1021/ci0501138, 1:CAS:528:DC%2BD2MXmtVCls78%3D
Al Dahouk S, Nockler K, Scholz H C, et al. Immunoproteomic characterization of Brucella abortus 1119-3 preparations used for the serodiagnosis of Brucella infections. J Immunol Methods, 2006, 309: 34–47, 16427071, 10.1016/j.jim.2005.11.003
Connolly J P, Comerci D, Alefantis T G, et al. Proteomic analysis of Brucella abortus cell envelope and identification of immunogenic candidate proteins for vaccine development. Proteomics, 2006, 6: 3767–3780, 16739129, 10.1002/pmic.200500730, 1:CAS:528:DC%2BD28XntlCjsLY%3D
DelVecchio V G, Wagner M A, Eschenbrenner M, et al. Brucella proteomes—a review. Vet microbiol, 2002, 90: 593–603, 12414175, 10.1016/S0378-1135(02)00239-0, 1:CAS:528:DC%2BD38XotF2jurw%3D
Pizza M, Scarlato V, Masignani V, et al. Identification of vaccine candidates against serogroup B. meningococcus by whole-genome sequencing. Science, 2000, 287: 1816–1820, 10710308, 10.1126/science.287.5459.1816, 1:CAS:528:DC%2BD3cXhvVWksbk%3D
Rappuoli R. Reverse vaccinology, a genome-based approach to vaccine development. Vaccine, 2001, 19: 2688–2691, 11257410, 10.1016/S0264-410X(00)00554-5, 1:CAS:528:DC%2BD3MXitVOit78%3D
Chain P S, Comerci D J, Tolmasky M E, et al. Whole-genome analyses of speciation events in pathogenic Brucellae. Infect immun, 2005, 73: 8353–8361, 16299333, 10.1128/IAI.73.12.8353-8361.2005, 1:CAS:528:DC%2BD2MXhtlertrzL
Paulsen I T, Seshadri R, Nelson K E, et al. The Brucella suis genome reveals fundamental similarities between animal and plant pathogens and symbionts. Proc Natl Acad Sci USA, 2002, 99: 13148–13153, 12271122, 10.1073/pnas.192319099, 1:CAS:528:DC%2BD38XnvFGhu78%3D
DelVecchio V G, Kapatral V, Redkar R J, et al. The genome sequence of the facultative intracellular pathogen Brucella melitensis. Proc Natl Acad Sci USA, 2002, 99: 443–448, 11756688, 10.1073/pnas.221575398, 1:CAS:528:DC%2BD38Xlt1Crsg%3D%3D
Wagner M A, Eschenbrenner M, Horn T A, et al. Global analysis of the Brucella melitensis proteome: Identification of proteins expressed in laboratory-grown culture. Proteomics, 2002, 2: 1047–1060, 12203900, 10.1002/1615-9861(200208)2:8<1047::AID-PROT1047>3.0.CO;2-8, 1:CAS:528:DC%2BD38Xmslyhs7Y%3D
Eschenbrenner M, Wagner M A, Horn T A, et al. Comparative proteome analysis of Brucella melitensis vaccine strain Rev 1 and a virulent strain, 16M. J Bacteriol, 2002, 184: 4962–4970, 12193611, 10.1128/JB.184.18.4962-4970.2002, 1:CAS:528:DC%2BD38XmslyktL0%3D
Yang Y, Yin J, Guo D, et al. Immunization of mice with recombinant S-adenosyl-L-homocysteine hydrolase protein confers protection against Brucella melitensis infection. FEMS Immunol Med Microbiol, 2011, 61: 159–167, 21166726, 10.1111/j.1574-695X.2010.00758.x, 1:CAS:528:DC%2BC3MXjtFaht7Y%3D
Celli J, Gorvel J P. Organelle robbery: Brucella interactions with the endoplasmic reticulum. Curr Opin Microbiol, 2004, 7: 93–97, 15036147, 10.1016/j.mib.2003.11.001, 1:CAS:528:DC%2BD2cXhtFChurs%3D
Peng X, Ye X, Wang S. Identification of novel immunogenic proteins of Shigella flexneri 2a by proteomic methodologies. Vaccine, 2004, 22: 2750–2756, 15246607, 10.1016/j.vaccine.2004.01.038, 1:CAS:528:DC%2BD2cXls1WktrY%3D
Ying T Y, Wang J J, Wang H L, et al. Immunoproteomics of membrane proteins of Shigella flexneri 2a 2457T. World J Gastroenterol, 2005, 11: 6880–6883, 16425402, 1:CAS:528:DC%2BD28XhsFGgtbg%3D
Delrue R M, Lestrate P, Tibor A, et al. Brucella pathogenesis, genes identified from random large-scale screens. FEMS Microbiol Lett, 2004, 231: 1–12, 14979322, 10.1016/S0378-1097(03)00963-7, 1:CAS:528:DC%2BD2cXhtFWgurc%3D
Patton W F. Proteome analysis. II. Protein subcellular redistribution: linking physiology to genomics via the proteome and separation technologies involved. J Chromatogr, 1999, 722: 203–223, 10.1016/S0378-4347(98)00309-0, 1:CAS:528:DyaK1MXps12jug%3D%3D
Velikovsky C A, Goldbaum F A, Cassataro J, et al. Brucella lumazine synthase elicits a mixed Th1-Th2 immune response and reduces infection in mice challenged with Brucella abortus 544 independently of the adjuvant formulation used. Infect Immun, 2003, 71: 5750–5755, 14500496, 10.1128/IAI.71.10.5750-5755.2003, 1:CAS:528:DC%2BD3sXnslyltLk%3D
Teixeira-Gomes A P, Cloeckaert A, Bezard G, et al. Identification and characterization of Brucella ovis immunogenic proteins using two-dimensional electrophoresis and immunoblotting. Electrophoresis, 1997, 18: 1491–1497, 9298663, 10.1002/elps.1150180824, 1:CAS:528:DyaK2sXlvVChtbo%3D
Rafie-Kolpin M, Essenberg R C, Wyckoff J H 3rd. Identification and comparison of macrophage-induced proteins and proteins induced under various stress conditions in Brucella abortus. Infect Immun, 1996, 64: 5274–5283, 8945577, 1:CAS:528:DyaK28XntFyqsrc%3D
Eschenbrenner M, Horn T A, Wagner M A, et al. Comparative proteome analysis of laboratory grown Brucella abortus 2308 and Brucella melitensis 16M. J Proteome Res, 2006, 5: 1731–1740, 16823981, 10.1021/pr060135p, 1:CAS:528:DC%2BD28XlsFaqsro%3D
Lindler L E, Hadfield T L, Tall B D, et al. Cloning of a Brucella melitensis group 3 antigen gene encoding Omp28, a protein recognized by the humoral immune response during human brucellosis. Infect Immun, 1996, 64: 2490–2499, 8698471, 1:CAS:528:DyaK28XjvFWqtLY%3D
Rossetti O L, Arese A I, Boschiroli M L, et al. Cloning of Brucella abortus gene and characterization of expressed 26-kilodalton periplasmic protein: potential use for diagnosis. J Clin Microbiol, 1996, 34: 165–169, 8748294, 1:CAS:528:DyaK28XksV2hsQ%3D%3D
Sinha S, Kosalai K, Arora S, et al. Immunogenic membrane-associated proteins of Mycobacterium tuberculosis revealed by proteomics. Microbiology, 2005, 151: 2411–2419, 16000731, 10.1099/mic.0.27799-0, 1:CAS:528:DC%2BD2MXmvV2lsrs%3D
Wehmhoner D, Dieterich G, Fischer E, et al. “LaneSpector”, a tool for membrane proteome profiling based on sodium dodecyl sulfate-polyacrylamide gel electrophoresis/liquid chromatography-tandem mass spectrometry analysis: application to Listeria monocytogenes membrane proteins. Electrophoresis, 2005, 26: 2450–2460, 15966022, 10.1002/elps.200410348
Jubier-Maurin V, Boigegrain R A, Cloeckaert A, et al. Major outer membrane protein Omp25 of Brucella suis is involved in inhibition of tumor necrosis factor alpha production during infection of human macrophages. Infect Immun, 2001, 69: 4823–4830, 11447156, 10.1128/IAI.69.8.4823-4830.2001, 1:CAS:528:DC%2BD3MXlsV2iu70%3D
Edmonds M D, Cloeckaert A, Booth N J, et al. Attenuation of a Brucella abortus mutant lacking a major 25 kDa outer membrane protein in cattle. Am J Vet Res, 2001, 62: 1461–1466, 11560278, 10.2460/ajvr.2001.62.1461, 1:STN:280:DC%2BD3Mrht1CgsA%3D%3D
Edmonds M D, Cloeckaert A, Elzer P H. Brucella species lacking the major outer membrane protein Omp25 are attenuated in mice and protect against Brucella melitensis and Brucella ovis. Vet Microbiol, 2002, 88: 205–221, 12151196, 10.1016/S0378-1135(02)00110-4, 1:CAS:528:DC%2BD38XlvVektrk%3D
Edmonds M D, Cloeckaert A, Hagius S D, et al. Pathogenicity and protective activity in pregnant goats of a Brucella melitensis Deltaomp25 deletion mutant. Res Vet Sci, 2002, 72: 235–239, 12076120, 10.1053/rvsc.2002.0555, 1:STN:280:DC%2BD38zjsVCntA%3D%3D
Elzer P H, Phillips R W, Robertson G T, et al. The HtrA stress response protease contributes to resistance of Brucella abortus to killing by murine phagocytes. Infect Immun, 1996, 64: 4838–4841, 8890248, 1:CAS:528:DyaK28XmsVWksLk%3D
Phillips R W, Elzer P H, Robertson G T, et al. A Brucella melitensis high-temperature-requirement A (htrA) deletion mutant is attenuated in goats and protects against abortion. Res Vet Sci, 1997, 63: 165–167, 9429252, 10.1016/S0034-5288(97)90012-6, 1:STN:280:DyaK1c%2FptV2ltA%3D%3D
Kohler S, Ekaza E, Paquet J Y, et al. Induction of dnaK through its native heat shock promoter is necessary for intramacrophagic replication of Brucella suis. Infect Immun, 2002, 70: 1631–1634, 11854256, 10.1128/IAI.70.3.1631-1634.2002, 1:CAS:528:DC%2BD38XhsFSltLw%3D
Valent Q A, Kendall D A, High S, et al. Early events in preprotein recognition in E. coli: interaction of SRP and trigger factor with nascent polypeptides. EMBO J, 1995, 14: 5494–5505, 8521806, 1:CAS:528:DyaK2MXpslamsb8%3D
Yang X, Walters N, Robison A, et al. Nasal immunization with recombinant Brucella melitensis bp26 and trigger factor with cholera toxin reduces B. melitensis colonization. Vaccine, 2007, 25: 2261–2268, 17239499, 10.1016/j.vaccine.2006.12.004, 1:CAS:528:DC%2BD2sXhvVylt7w%3D
Author information
Authors and Affiliations
Corresponding author
Additional information
Contributed equally to this work
This article is published with open access at Springerlink.com
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Zhao, Z., Yan, F., Ji, W. et al. Identification of immunoreactive proteins of Brucella melitensis by immunoproteomics. Sci. China Life Sci. 54, 880–887 (2011). https://doi.org/10.1007/s11427-011-4218-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11427-011-4218-2